Targeting Cell Entry of Enveloped Viruses as an Antiviral Strategy
Abstract
:Introduction

Three Classes of Viral Fusion Proteins


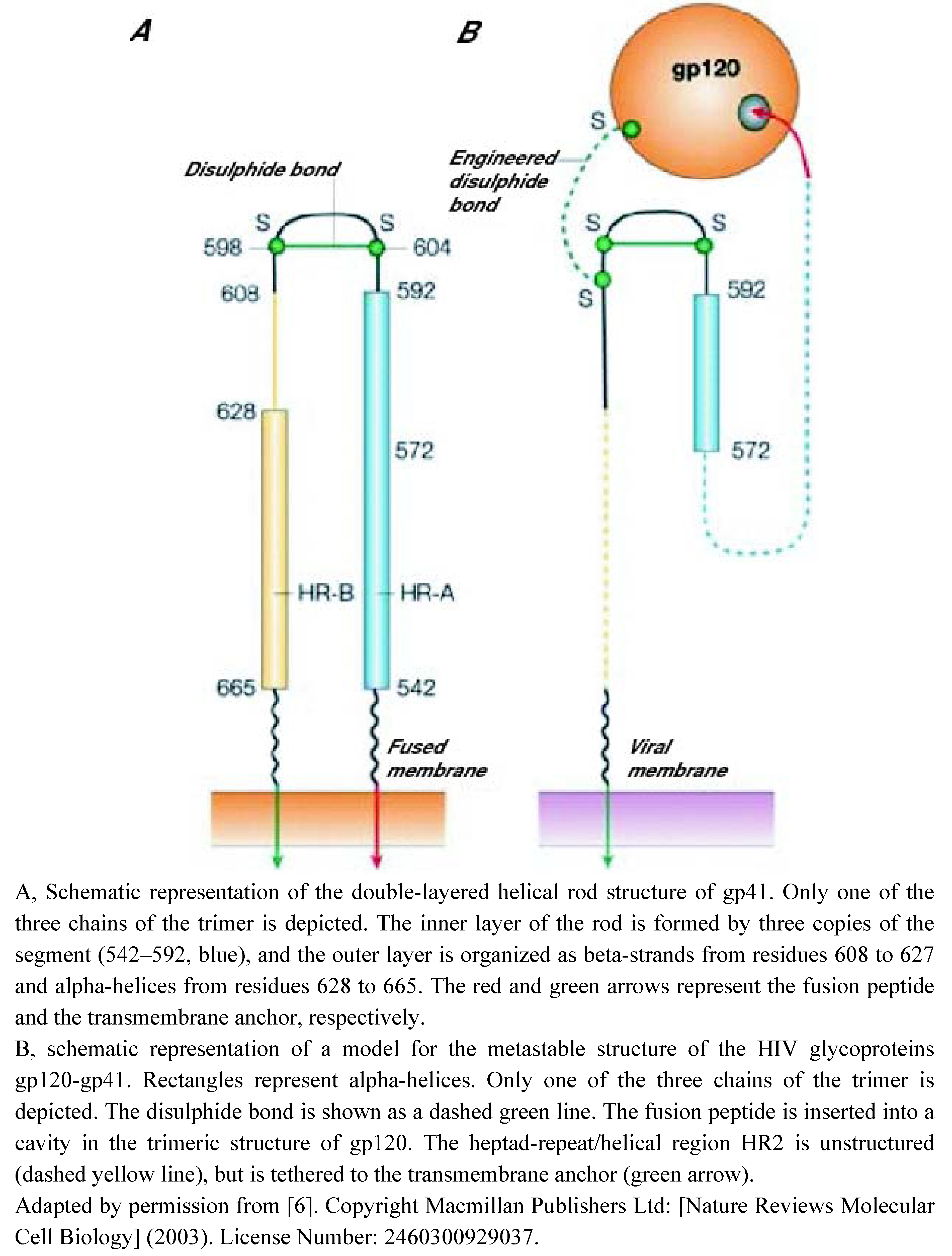
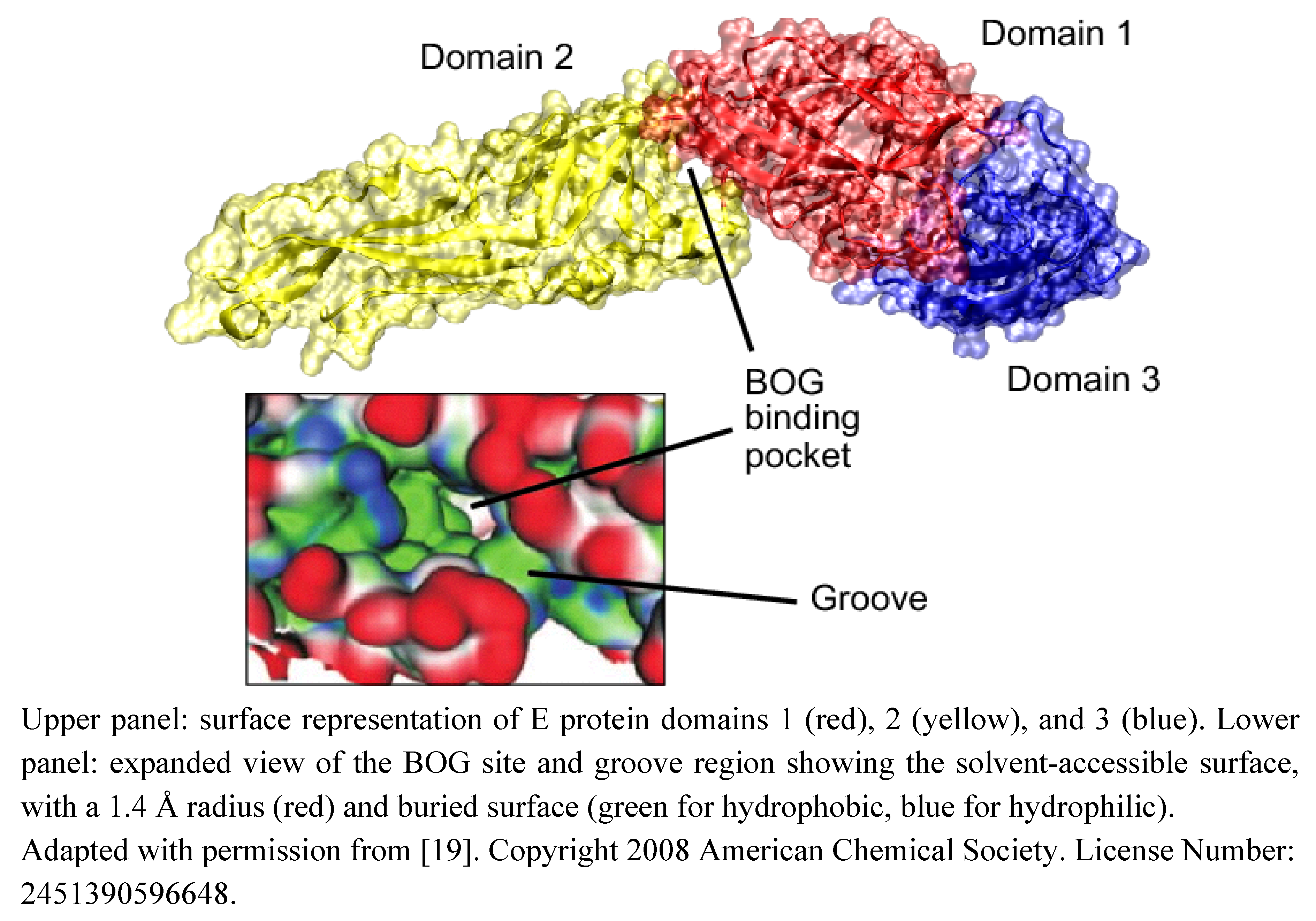
Inhibitors Preventing Virus Attachment to Target Cells
Antibodies
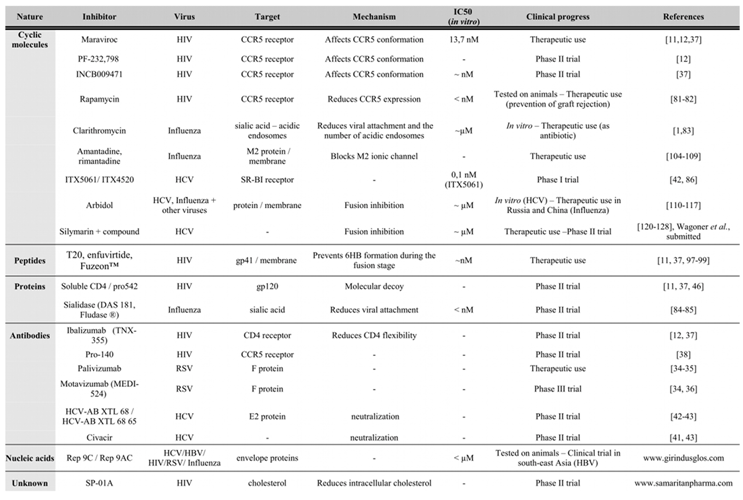 |
Other attachment inhibitors
Inhibitors targeting envelope proteins
Inhibitors targeting host receptors
Viral Envelope Proteins as Targets for Entry Inhibitors
Blocking Cellular Proteins to Inhibit Virus Entry
Target: Membranes and Proteins
HCV Inhibitors Targeting Unknown Targets
Conclusions
References
- Thorley, J.A.; McKeating, J.A.; Rappoport, J.Z. Mechanisms of viral entry: Sneaking in the front door. Protoplasma 2010, 244, 15–24. [Google Scholar] [CrossRef]
- White, J.M.; Delos, S.E.; Brecher, M.; Schornberg, K. Structures and mechanisms of viral membrane fusion proteins: multiple variations on a common theme. Crit. Rev. Biochem. Mol. Biol 2008, 43, 189–219. [Google Scholar] [CrossRef]
- Harrison, S.C. Viral membrane fusion. Nat. Struct. Mol. Biol 2008, 15, 690–698. [Google Scholar] [CrossRef]
- Kielian, M. Class II virus membrane fusion proteins. Virology 2006, 344, 38–47. [Google Scholar] [CrossRef]
- Kielian, M.; Rey, F.A. Virus membrane-fusion proteins: More than one way to make a hairpin. Nat. Rev. Microbiol 2006, 4, 67–76. [Google Scholar] [CrossRef]
- Colman, P.M.; Lawrence, M.C. The structural biology of type I viral membrane fusion. Nat. Rev. Mol. Cell Biol. 2003, 4, 309–319. [Google Scholar]
- Chan, D.C.; Kim, P.S. HIV entry and its inhibition. Cell 1998, 93, 681–684. [Google Scholar] [CrossRef]
- Roymans, D.; De Bondt, H.L.; Arnoult, E.; Geluykens, P.; Gevers, T.; Van Ginderen, M.; Verheyen, N.; Kim, H.; Willebrords, R.; Bonfanti, J.; Bruinzeel, W.; Cummings, M.D.; van Vlijmen, H.; Andries, K. Binding of a potent small-molecule inhibitor of six-helix bundle formation requires interactions with both heptad-repeats of the RSV fusion protein. Proc. Natl. Acad. Sci. USA 2010, 107, 308–313. [Google Scholar]
- Porotto, M.; Yokoyama, C.; Palermo, L.M.; Mungall, B.; Aljofan, M.; Cortese, R.; Pessi, A.; Moscona, A. Viral entry inhibitors targeted to the membrane site of action. J. Virol. 2010, 84, 6760–6768. [Google Scholar] [CrossRef]
- Zhu, P.; Liu, J.; Bess, J.; Chertova, E.; Lifson, J.D.; Grisé, H.; Ofek, G.A.; Taylor, K.A.; Roux, K.H. Distribution and three-dimensional structure of AIDS virus envelope spikes. Nature 2006, 44, 847–852. [Google Scholar]
- Melby, T.; Heilek, G.; Cammack, N.; Greenberg, M.L. Resistance to Enfuvirtide and Other HIV Entry Inhibitors. In Antimicrobial Drug Resistance; Mayers, D.L., Ed.; Humana Press: New York, NY, USA, 2009; pp. 493–506. [Google Scholar]
- Melby, T.; Westby, M. Inhibitors of Viral Entry. In Antiviral Strategies; Krausslich, R., Bartenschlager, R., Eds.; Springer-Verlag: Berlin, Heidelberg, Germany, 2009; pp. 177–202. [Google Scholar]
- Teissier, E.; Pécheur, E.I. Lipids as modulators of membrane fusion mediated by viral fusion proteins. Eur. Biophys. J. 2007, 36, 887–899. [Google Scholar] [CrossRef]
- Lamb, R.A.; Jardetzky, T.S. Structural basis of viral invasion: Lessons from paramyxovirus F. Curr. Opin. Struct. Biol. 2007, 17, 427–436. [Google Scholar] [CrossRef]
- Smith, E.C.; Popa, A.; Chang, A.; Masante, C.; Dutch, R.E. Viral entry mechanisms: The increasing diversity of paramyxovirus entry. FEBS J. 2009, 276, 7217–7227. [Google Scholar] [CrossRef]
- Rust, M.J.; Lakadamyali, M.; Zhang, F.; Zhuang, X. Assembly of endocytic machinery around individual influenza viruses during viral entry. Nat. Struct. Mol. Biol. 2004, 11, 567–573. [Google Scholar]
- Samji, T. Influenza A: Understanding the viral life cycle. Yale J. Biol. Med. 2009, 82, 153–159. [Google Scholar]
- Pielak, R.M.; Chou, J.J. Influenza M2 proton channels. Biochim. Biophys. Acta 2011, 1808, 522–529. [Google Scholar]
- Zhou, Z.; Khaliq, M.; Suk, J.; Patkar, C.; Li, L.; Kuhn, R.J.; Post, C.B. Antiviral compounds discovered by virtual screening of small-molecule libraries against dengue virus E protein. ACS Chem. Biol. 2008, 3, 765–775. [Google Scholar] [CrossRef]
- Modis, Y.; Ogata, S.; Clements, D.; Harrison, S.C. A ligand-binding pocket in the dengue virus envelope glycoprotein. Proc. Natl. Acad. Sci. USA 2003, 100, 6986–6991. [Google Scholar]
- Bressanelli, S.; Stiasny, K.; Allison, S.L.; Stura, E.A.; Duquerroy, S.; Lescar, J.; Heinz, F.X.; Rey, F.A. Structure of a flavivirus envelope glycoprotein in its low-pH-induced membrane fusion conformation. EMBO J. 2004, 23, 728–738. [Google Scholar] [CrossRef]
- Krey, T.; d'Alayer, J.; Kikuti, C.M.; Saulnier, A.; Damier-Piolle, L.; Petitpas, I.; Johansson, D.X.; Tawar, R.G.; Baron, B.; Robert, B.; England, P.; Persson, M.A.A.; Martin, A.; Rey, F.A. The disulfide bonds in glycoprotein E2 of hepatitis C virus reveal the tertiary organization of the molecule. PLoS Pathog. 2010, 6, e1000762:1–e1000762:11. [Google Scholar]
- Bartosch, B.; Dubuisson, J.; Cosset, F.L. Infectious hepatitis C virus pseudo-particles containing functional E1-E2 envelope protein complexes. J. Exp. Med. 2003, 197, 633–642. [Google Scholar] [CrossRef]
- Wakita, T.; Pietschmann, T.; Kato, T.; Date, T.; Miyamoto, M.; Zhao, Z.; Murthy, K.; Habermann, A.; Kräusslich, H.; Mizokami, M.; Bartenschlager, R.; Liang, T.J. Production of infectious hepatitis C virus in tissue culture from a cloned viral genome. Nat. Med. 2005, 11, 791–796. [Google Scholar]
- Evans, M.J.; von Hahn, T.; Tscherne, D.M.; Syder, A.J.; Panis, M.; Wölk, B.; Hatziioannou, T.; McKeating, J.A.; Bieniasz, P.D.; Rice, C.M. Claudin-1 is a hepatitis C virus co-receptor required for a late step in entry. Nature 2007, 446, 801–805. [Google Scholar]
- Ploss, A.; Evans, M.J.; Gaysinskaya, V.A.; Panis, M.; You, H.; de Jong, Y.P.; Rice, C.M. Human occludin is a hepatitis C virus entry factor required for infection of mouse cells. Nature 2009, 457, 882–886. [Google Scholar]
- Bartosch, B.; Cosset, F.L. Cell entry of hepatitis C virus. Virology 2006, 348, 1–12. [Google Scholar] [CrossRef]
- Moradpour, D.; Penin, F.; Rice, C.M. Replication of hepatitis C virus. Nat. Rev. Microbiol. 2007, 5, 453–463. [Google Scholar] [CrossRef]
- Helle, F.; Dubuisson, J. Hepatitis C virus entry into host cells. Cell. Mol. Life Sci. 2008, 65, 100–112. [Google Scholar] [CrossRef]
- Perrault, M.; Pécheur, E.I. The hepatitis C virus and its hepatic environment: A toxic but finely tuned partnership. Biochem. J. 2009, 423, 303–314. [Google Scholar] [CrossRef]
- Blanchard, E.; Belouzard, S.; Goueslain, L.; Wakita, T.; Dubuisson, J.; Wychowski, C.; Rouillé, Y. Hepatitis C virus entry depends on clathrin-mediated endocytosis. J. Virol. 2006, 80, 6964–6972. [Google Scholar]
- Lavillette, D.; Bartosch, B.; Nourrisson, D.; Verney, G.; Cosset, F.; Penin, F.; Pécheur, E.I. Hepatitis C virus glycoproteins mediate low pH-dependent membrane fusion with liposomes. J. Biol. Chem. 2006, 281, 3909–3917. [Google Scholar]
- Backovic, M.; Jardetzky, T.S. Class III viral membrane fusion proteins. Curr. Opin. Struct. Biol. 2009, 19, 189–196. [Google Scholar] [CrossRef]
- Ramilo, O. Evolution of prophylaxis: MoAb, siRNA, vaccine, and small molecules. Paediatr. Respir. Rev. 2009, 10 (Suppl. 1), 23–25. [Google Scholar] [CrossRef]
- Hampp, C.; Saidi, A.; Winterstein, A.G. Palivizumab Utilization and Compliance: Trends in Respiratory Syncytial Virus Prophylaxis in Florida. J. Pediatr. 2010, 156, 953–959. [Google Scholar] [CrossRef]
- Huang, K.; Incognito, L.; Cheng, X.; Ulbrandt, N.D.; Wu, H. Respiratory Syncytial Virus-Neutralizing Monoclonal Antibodies Motavizumab and Palivizumab Inhibit Fusion. J. Virol. 2010, 84, 8132–8140. [Google Scholar] [CrossRef]
- Kuritzkes, D.R. HIV-1 entry inhibitors: An overview. Curr. Opin. HIV AIDS 2009, 4, 82–87. [Google Scholar] [CrossRef]
- Jacobson, J.M.; Thompson, M.A.; Lalezari, J.P.; Saag, M.S.; Zingman, B.S.; D'Ambrosio, P.; Stambler, N.; Rotshteyn, Y.; Marozsan, A.J.; Maddon, P.J.; Morris, S.A.; Olson, W.C. Anti-HIV-1 activity of weekly or biweekly treatment with subcutaneous PRO 140, a CCR5 monoclonal antibody. J. Infect. Dis. 2010, 201, 1481–1487. [Google Scholar] [CrossRef]
- Meuleman, P.; Hesselgesser, J.; Paulson, M.; Vanwolleghem, T.; Desombere, I.; Reiser, H.; Leroux-Roels, G. Anti-CD81 antibodies can prevent a hepatitis C virus infection in vivo. Hepatology 2008, 48, 1761–1768. [Google Scholar] [CrossRef]
- Fofana, I.; Krieger, S.E.; Grunert, F.; Glauben, S.; Xiao, F.; Fafi-Kremer, S.; Soulier, E.; Royer, C.; Thumann, C.; Mee, C.J.; McKeating, J.A.; Dragic, T.; Pessaux, P.; Stoll-Keller, F.; Schuster, C.; Thompson, J.; Baumert, T.F. Monoclonal anti-claudin 1 antibodies prevent hepatitis C virus infection of primary human hepatocytes. Gastroenterology 2010, 139, 953–964. [Google Scholar]
- Timpe, J.M.; Stamataki, Z.; Jennings, A.; Hu, K.; Farquhar, M.J.; Harris, H.J.; Schwarz, A.; Desombere, I.; Roels, G.L.; Balfe, P.; McKeating, J.A. Hepatitis C virus cell-cell transmission in hepatoma cells in the presence of neutralizing antibodies. Hepatology 2008, 47, 17–24. [Google Scholar]
- Donia, M.; Cacopardo, B.; Libra, M.; Scalia, G.; McCubrey, J.A.; Nicoletti, F. New Perspectives in HCV Therapy: Entry Inhibitors. Recent Pat. Antiinfect. Drug Discov. 2010, 5, 181–194. [Google Scholar] [CrossRef]
- Eren, R.; Landstein, D.; Terkieltaub, D.; Nussbaum, O.; Zauberman, A.; Ben-Porath, J.; Gopher, J.; Buchnick, R.; Kovjazin, R.; Rosenthal-Galili, Z.; Aviel, S.; Ilan, E.; Shoshany, Y.; Neville, L.; Waisman, T.; Ben-Moshe, O.; Kischitsky, A.; Foung, S.K.H.; Keck, Z.; Pappo, O.; Eid, A.; Jurim, O.; Zamir, G.; Galun, E.; Dagan, S. Preclinical evaluation of two neutralizing human monoclonal antibodies against hepatitis C virus (HCV): A potential treatment to prevent HCV reinfection in liver transplant patients. J. Virol. 2006, 80, 2654–2664. [Google Scholar]
- Davis, G.L.; Nelson, D.R.; Terrault, N.; Pruett, T.L.; Schiano, T.D.; Fletcher, C.V.; Sapan, C.V.; Riser, L.N.; Li, Y.; Whitley, R.J.; Gnann, J.W. A randomized, open-label study to evaluate the safety and pharmacokinetics of human hepatitis C immune globulin (Civacir) in liver transplant recipients. Liver Transpl. 2005, 11, 941–949. [Google Scholar] [CrossRef]
- Beltramello, M.; Williams, K.L.; Simmons, C.P.; Macagno, A.; Simonelli, L.; Quyen, N.T.H.; Sukupolvi-Petty, S.; Navarro-Sanchez, E.; Young, P.R.; de Silva, A.M.; Rey, F.A.; Varani, L.; Whitehead, S.S.; Diamond, M.S.; Harris, E.; Lanzavecchia, A.; Sallusto, F. The human immune response to Dengue virus is dominated by highly cross-reactive antibodies endowed with neutralizing and enhancing activity. Cell Host Microbe 2010, 8, 271–283. [Google Scholar] [CrossRef]
- Jacobson, J.M.; Thompson, M.A.; Lalezari, J.P.; Saag, M.S.; Zingman, B.S.; D'Ambrosio, P.; Stambler, N.; Rotshteyn, Y.; Marozsan, A.J.; Maddon, P.J.; Morris, S.A.; Olson, W.C. Anti-HIV-1 activity of weekly or biweekly treatment with subcutaneous PRO 140, a CCR5 monoclonal antibody. J. Infect. Dis. 2010, 201, 1481–1487. [Google Scholar] [CrossRef]
- Wang, T.; Zhang, Z.; Wallace, O.B.; Deshpande, M.; Fang, H.; Yang, Z.; Zadjura, L.M.; Tweedie, D. L.; Huang, S.; Zhao, F.; Ranadive, S.; Robinson, B.S.; Gong, Y.; Ricarrdi, K.; Spicer, T.P.; Deminie, C.; Rose, R.; Wang, H.H.; Blair, W.S.; Shi, P.; Lin, P.; Colonno, R.J.; Meanwell, N.A. Discovery of 4-benzoyl-1-[(4-methoxy-1H-pyrrolo[2,3-b]pyridin-3-yl)oxoacetyl]-2-(R)-methylpiperazine (BMS-378806): a novel HIV-1 attachment inhibitor that interferes with CD4-gp120 interactions. J. Med. Chem. 2003, 46, 4236–4239. [Google Scholar]
- Guo, Q.; Ho, H.; Dicker, I.; Fan, L.; Zhou, N.; Friborg, J.; Wang, T.; McAuliffe, B.V.; Wang, H.H.; Rose, R.E.; Fang, H.; Scarnati, H.T.; Langley, D.R.; Meanwell, N.A.; Abraham, R.; Colonno, R.J.; Lin, P. Biochemical and genetic characterizations of a novel human immunodeficiency virus type 1 inhibitor that blocks gp120-CD4 interactions. J. Virol. 2003, 77, 10528–10536. [Google Scholar]
- Jones, J.C.; Turpin, E.A.; Bultmann, H.; Brandt, C.R.; Schultz-Cherry, S. Inhibition of influenza virus infection by a novel antiviral peptide that targets viral attachment to cells. J. Virol. 2006, 80, 11960–11967. [Google Scholar] [CrossRef]
- Rajik, M.; Jahanshiri, F.; Omar, A. R.; Ideris, A.; Hassan, S.S.; Yusoff, K. Identification and characterisation of a novel anti-viral peptide against avian influenza virus H9N2. Virol. J. 2009, 6, 74:1–74:12. [Google Scholar]
- Helle, F.; Wychowski, C.; Vu-Dac, N.; Gustafson, K.R.; Voisset, C.; Dubuisson, J. Cyanovirin-N inhibits hepatitis C virus entry by binding to envelope protein glycans. J. Biol. Chem. 2006, 281, 25177–25183. [Google Scholar]
- Boyd, M.R.; Gustafson, K.R.; McMahon, J.B.; Shoemaker, R.H.; O'Keefe, B.R.; Mori, T.; Gulakowski, R.J.; Wu, L.; Rivera, M.I.; Laurencot, C.M.; Currens, M.J.; Cardellina, J.H.; Buckheit, R.W.; Nara, P.L.; Pannell, L.K.; Sowder, R.C.; Henderson, L.E. Discovery of cyanovirin-N, a novel human immunodeficiency virus-inactivating protein that binds viral surface envelope glycoprotein gp120: Potential applications to microbicide development. Antimicrob. Agents Chemother. 1997, 41, 1521–1530. [Google Scholar]
- O'Keefe, B.R.; Smee, D.F.; Turpin, J.A.; Saucedo, C.J.; Gustafson, K.R.; Mori, T.; Blakeslee, D.; Buckheit, R.; Boyd, M.R. Potent anti-influenza activity of cyanovirin-N and interactions with viral hemagglutinin. Antimicrob. Agents Chemother. 2003, 47, 2518–2525. [Google Scholar]
- Tanaka, H.; Chiba, H.; Inokoshi, J.; Kuno, A.; Sugai, T.; Takahashi, A.; Ito, Y.; Tsunoda, M.; Suzuki, K.; Takénaka, A.; Sekiguchi, T.; Umeyama, H.; Hirabayashi, J.; Omura, S. Mechanism by which the lectin actinohivin blocks HIV infection of target cells. Proc. Natl. Acad. Sci. USA 2009, 106, 15633–15638. [Google Scholar]
- Hoorelbeke, B.; Huskens, D.; Férir, G.; François, K.O.; Takahashi, A.; Van Laethem, K.; Schols, D.; Tanaka, H.; Balzarini, J. Actinohivin, a broadly neutralizing prokaryotic lectin, inhibits HIV-1 infection by specifically targeting high-mannose-type glycans on the gp120 envelope. Antimicrob. Agents Chemother. 2010, 54, 3287–3301. [Google Scholar]
- Matoba, N.; Husk, A.S.; Barnett, B.W.; Pickel, M.M.; Arntzen, C.J.; Montefiori, D.C.; Takahashi, A.; Tanno, K.; Omura, S.; Cao, H.; Mooney, J.P.; Hanson, C.V.; Tanaka, H. HIV-1 neutralization profile and plant-based recombinant expression of actinohivin, an Env glycan-specific lectin devoid of T-cell mitogenic activity. PLoS One 2010, 5, e11143. [Google Scholar]
- Chiba, H.; Inokoshi, J.; Okamoto, M.; Asanuma, S.; Matsuzaki, K.; Iwama, M.; Mizumoto, K.; Tanaka, H.; Oheda, M.; Fujita, K.; Nakashima, H.; Shinose, M.; Takahashi, Y.; Omura, S. Actinohivin, a novel anti-HIV protein from an actinomycete that inhibits syncytium formation: isolation, characterization, and biological activities. Biochem. Biophys. Res. Commun. 2001, 282, 595–601. [Google Scholar] [CrossRef]
- Chiba, H.; Inokoshi, J.; Nakashima, H.; Omura, S.; Tanaka, H. Actinohivin, a novel anti-human immunodeficiency virus protein from an actinomycete, inhibits viral entry to cells by binding high-mannose type sugar chains of gp120. Biochem. Biophys. Res. Commun. 2004, 316, 203–210. [Google Scholar] [CrossRef]
- Takahashi, A.; Inokoshi, J.; Chiba, H.; Omura, S.; Tanaka, H. Essential regions for antiviral activities of actinohivin, a sugar-binding anti-human immunodeficiency virus protein from an actinomycete. Arch. Biochem. Biophys. 2005, 437, 233–240. [Google Scholar] [CrossRef]
- Takahashi, A.; Inokoshi, J.; Tsunoda, M.; Suzuki, K.; Takenaka, A.; Sekiguchi, T.; Omura, S.; Tanaka, H. Actinohivin: specific amino acid residues essential for anti-HIV activity. J Antibiot. (Tokyo) 2010, 63, 661–66. [Google Scholar] [CrossRef]
- Bertaux, C.; Daelemans, D.; Meertens, L.; Cormier, E.G.; Reinus, J.F.; Peumans, W.J.; Van Damme, E.J.M.; Igarashi, Y.; Oki, T.; Schols, D.; Dragic, T.; Balzarini, J. Entry of hepatitis C virus and human immunodeficiency virus is selectively inhibited by carbohydrate-binding agents but not by polyanions. Virology 2007, 366, 40–50. [Google Scholar] [CrossRef]
- Kato, D.; Era, S.; Watanabe, I.; Arihara, M.; Sugiura, N.; Kimata, K.; Suzuki, Y.; Morita, K.; Hidari, K.I.P.J.; Suzuki, T. Antiviral activity of chondroitin sulphate E targeting dengue virus envelope protein. Antiviral Res. 2010, 88, 236–243. [Google Scholar] [CrossRef]
- Rocha-Perugini, V.; Montpellier, C.; Delgrange, D.; Wychowski, C.; Helle, F.; Pillez, A.; Drobecq, H.; Le Naour, F.; Charrin, S.; Levy, S.; Rubinstein, E.; Dubuisson, J.; Cocquerel, L. The CD81 partner EWI-2wint inhibits hepatitis C virus entry. PLoS One 2008, 3, e1866:1–e1866:12. [Google Scholar]
- Schuster, C.; Baumert, T.F. EWI-2wint--a host cell factor inhibiting hepatitis C virus entry. J. Hepatol. 2009, 50, 222–224. [Google Scholar] [CrossRef]
- Holzer, M.; Ziegler, S.; Albrecht, B.; Kronenberger, B.; Kaul, A.; Bartenschlager, R.; Kattner, L.; Klein, C.D.; Hartmann, R.W. Identification of terfenadine as an inhibitor of human CD81-receptor HCV-E2 interaction: synthesis and structure optimization. Molecules 2008, 13, 1081–1110. [Google Scholar] [CrossRef]
- Cianci, C.; Yu, K.; Combrink, K.; Sin, N.; Pearce, B.; Wang, A.; Civiello, R.; Voss, S.; Luo, G.; Kadow, K.; Genovesi, E.V.; Venables, B.; Gulgeze, H.; Trehan, A.; James, J.; Lamb, L.; Medina, I.; Roach, J.; Yang, Z.; Zadjura, L.; Colonno, R.; Clark, J.; Meanwell, N.; Krystal, M. Orally Active Fusion Inhibitor of Respiratory Syncytial Virus. Antimicrob. Agents Chemother. 2004, 48, 413–422. [Google Scholar]
- Cianci, C.; Langley, D.R.; Dischino, D.D.; Sun, Y.; Yu, K.; Stanley, A.; Roach, J.; Li, Z.; Dalterio, R.; Colonno, R.; Meanwell, N.A.; Krystal, M. Targeting a binding pocket within the trimer-of-hairpins: small-molecule inhibition of viral fusion. Proc. Natl. Acad. Sci. USA 2004, 101, 15046–15051. [Google Scholar]
- Vanderlinden, E.; Göktas, F.; Cesur, Z.; Froeyen, M.; Reed, M.L.; Russell, C.J.; Cesur, N.; Naesens, L. Novel inhibitors of influenza virus fusion: Structure-activity relationship and interaction with the viral hemagglutinin. J. Virol. 2010, 84, 4277–4288. [Google Scholar]
- Russell, R.J.; Kerry, P.S.; Stevens, D.J.; Steinhauer, D.A.; Martin, S.R.; Gamblin, S.J.; Skehel, J.J. Structure of influenza hemagglutinin in complex with an inhibitor of membrane fusion. Proc. Natl. Acad. Sci. USA 2008, 105, 17736–17741. [Google Scholar]
- Poh, M.K.; Yip, A.; Zhang, S.; Priestle, J.P.; Ma, N.L.; Smit, J.M.; Wilschut, J.; Shi, P.; Wenk, M.R.; Schul, W. A small molecule fusion inhibitor of dengue virus. Antiviral Res. 2009, 84, 260–266. [Google Scholar] [CrossRef]
- Wang, Q.; Patel, S.J.; Vangrevelinghe, E.; Xu, H.Y.; Rao, R.; Jaber, D.; Schul, W.; Gu, F.; Heudi, O.; Ma, N.L.; Poh, M.K.; Phong, W.Y.; Keller, T.H.; Jacoby, E.; Vasudevan, S.G. A small-molecule dengue virus entry inhibitor. Antimicrob. Agents Chemother. 2009, 53, 1823–1831. [Google Scholar]
- Li, Z.; Khaliq, M.; Zhou, Z.; Post, C. B.; Kuhn, R.J.; Cushman, M. Design, synthesis, and biological evaluation of antiviral agents targeting flavivirus envelope proteins. J. Med. Chem. 2008, 51, 4660–4671. [Google Scholar]
- Hrobowski, Y.M.; Garry, R.F.; Michael, S.F. Peptide inhibitors of dengue virus and West Nile virus infectivity. Virol. J. 2005, 2, 49–1. [Google Scholar]
- Schmidt, A.G.; Yang, P.L.; Harrison, S.C. Peptide inhibitors of dengue-virus entry target a late-stage fusion intermediate. PLoS Pathog. 2010, 6, e1000851. [Google Scholar] [CrossRef]
- Liao, M.; Kielian, M. Domain III from class II fusion proteins functions as a dominant-negative inhibitor of virus membrane fusion. J. Cell Biol. 2005, 171, 111–120. [Google Scholar] [CrossRef]
- Chu, J.J.H.; Rajamanonmani, R.; Li, J.; Bhuvanakantham, R.; Lescar, J.; Ng, M. Inhibition of West Nile virus entry by using a recombinant domain III from the envelope glycoprotein. J. Gen. Virol. 2005, 86, 405–412. [Google Scholar] [CrossRef]
- Vaillant, A.; Juteau, J.; Lu, H.; Liu, S.; Lackman-Smith, C.; Ptak, R.; Jiang, S. Phosphorothioate oligonucleotides inhibit human immunodeficiency virus type 1 fusion by blocking gp41 core formation. Antimicrob. Agents Chemother. 2006, 50, 1393–1401. [Google Scholar] [CrossRef]
- Matsumura, T.; Hu, Z.; Kato, T.; Dreux, M.; Zhang, Y.; Imamura, M.; Hiraga, N.; Juteau, J.; Cosset, F.; Chayama, K.; Vaillant, A.; Liang, T.J. Amphipathic DNA polymers inhibit hepatitis C virus infection by blocking viral entry. Gastroenterology 2009, 137, 673–681. [Google Scholar]
- Counihan, N.A.; Lindenbach, B.D. Gumming up the works: DNA polymers as HCV entry inhibitors. Gastroenterology 2009, 137, 427–430. [Google Scholar] [CrossRef]
- Baldick, C.J.; Wichroski, M.J.; Pendri, A.; Walsh, A.W.; Fang, J.; Mazzucco, C.E.; Pokornowski, K.A.; Rose, R.E.; Eggers, B.J.; Hsu, M.; Zhai, W.; Zhai, G.; Gerritz, S.W.; Poss, M.A.; Meanwell, N.A.; Cockett, M.I.; Tenney, D.J. A novel small molecule inhibitor of hepatitis C virus entry. PLoS Pathog. 2010, 6, e1001086. [Google Scholar]
- Gilliam, B.L.; Heredia, A.; Devico, A.; Le, N.; Bamba, D.; Bryant, J.L.; Pauza, C.D.; Redfield, R.R. Rapamycin reduces CCR5 mRNA levels in macaques: Potential applications in HIV-1 prevention and treatment. AIDS 2007, 21, 2108–2110, Gilliam, B.L.; Heredia, A.; Devico, A.; Le, N.; Bamba, D.; Bryant, J.L.; Pauza, C.D.; Redfield, R.R. Rapamycin reduces CCR5 mRNA levels in macaques: Potential applications in HIV-1 prevention and treatment. AIDS 2007, 21, 2108-2110 . [Google Scholar] [CrossRef]
- Nicoletti, F.; Lapenta, C.; Lamenta, C.; Donati, S.; Spada, M.; Ranazzi, A.; Cacopardo, B.; Mangano, K.; Belardelli, F.; Perno, C.; Aquaro, S. Inhibition of human immunodeficiency virus (HIV-1) infection in human peripheral blood leucocytes-SCID reconstituted mice by rapamycin. Clin. Exp. Immunol. 2009, 155, 28–34. [Google Scholar] [CrossRef]
- Yamaya, M.; Shinya, K.; Hatachi, Y.; Kubo, H.; Asada, M.; Yasuda, H.; Nishimura, H.; Nagatomi, R. Clarithromycin inhibits type a seasonal influenza virus infection in human airway epithelial cells. J. Pharmacol. Exp. Ther. 2010, 333, 81–90. [Google Scholar] [CrossRef]
- Malakhov, M.P.; Aschenbrenner, L.M.; Smee, D.F.; Wandersee, M.K.; Sidwell, R.W.; Gubareva, L.V.; Mishin, V.P.; Hayden, F.G.; Kim, D.H.; Ing, A.; Campbell, E.R.; Yu, M.; Fang, F. Sialidase fusion protein as a novel broad-spectrum inhibitor of influenza virus infection. Antimicrob. Agents Chemother. 2006, 50, 1470–1479. [Google Scholar]
- Triana-Baltzer, G.B.; Gubareva, L.V.; Klimov, A.I.; Wurtman, D.F.; Moss, R.B.; Hedlund, M.; Larson, J.L.; Belshe, G.B.; Fang, F. Inhibition of neuraminidase inhibitor-resistant influenza virus by DAS181, a novel sialidase fusion protein. PLoS One 2009, 4, e7838:1–e7838:9. [Google Scholar]
- Lemon, S.M.; McKeating, J.A.; Pietschmann, T.; Frick, D.N.; Glenn, J.S.; Tellinghuisen, T.L.; Symons, J.; and Furman, P.A. Development of novel therapies for hepatitis C. Antiviral Res. 2010, 86, 79–92. [Google Scholar] [CrossRef]
- Shah, P.P; Wang, T.; Kaletsky, R.L.; Myers, M.C.; Purvis, J.E.; Jing, H.; Huryn, D.M.; Greenbaum, D.C.; Smith, A.B.; Bates, P.; Diamond, S.L. A small-molecule oxocarbazate inhibitor of human cathepsin L blocks severe acute respiratory syndrome and ebola pseudotype virus infection into human embryonic kidney 293T cells. Mol. Pharmacol. 2010, 78, 319–324. [Google Scholar] [CrossRef]
- Rojek, J.M.; Pasqual, G.; Sanchez, A.B.; Nguyen, N.; de la Torre, J.; Kunz, S. Targeting the proteolytic processing of the viral glycoprotein precursor is a promising novel antiviral strategy against arenaviruses. J. Virol. 2010, 84, 573–584. [Google Scholar]
- Kido, H.; Okumura, Y.; Takahashi, E.; Pan, H.; Wang, S.; Chida, J.; Le, T.Q.; Yano, M. Host envelope glycoprotein processing proteases are indispensable for entry into human cells by seasonal and highly pathogenic avian influenza viruses. J. Mol. Genet. Med. 2008, 3, 167–175. [Google Scholar]
- Böttcher-Friebertshäuser, E.; Freuer, C.; Sielaff, F.; Schmidt, S.; Eickmann, M.; Uhlendorff, J.; Steinmetzer, T.; Klenk, H.; Garten, W. Cleavage of influenza virus hemagglutinin by airway proteases TMPRSS2 and HAT differs in subcellular localization and susceptibility to protease inhibitors. J. Virol. 2010, 84, 5605–5614. [Google Scholar]
- Cheng, G.; Montero, A.; Gastaminza, P.; Whitten-Bauer, C.; Wieland, S.F.; Isogawa, M.; Fredericksen, B.; Selvarajah, S.; Gallay, P.A.; Ghadiri, M.R.; Chisari, F.V. A virocidal amphipathic {alpha}-helical peptide that inhibits hepatitis C virus infection in vitro. Proc. Natl. Acad. Sci. USA 2008, 105, 3088–3093. [Google Scholar]
- Brass, V.; Bieck, E.; Montserret, R.; Wölk, B.; Hellings, J.A.; Blum, H.E.; Penin, F.; Moradpour, D. An amino-terminal amphipathic alpha-helix mediates membrane association of the hepatitis C virus nonstructural protein 5A. J. Biol. Chem. 2002, 277, 8130–8139. [Google Scholar]
- Penin, F.; Brass, V.; Appel, N.; Ramboarina, S.; Montserret, R.; Ficheux, D.; Blum, H.E.; Bartenschlager, R.; Moradpour, D. Structure and function of the membrane anchor domain of hepatitis C virus nonstructural protein 5A. J. Biol. Chem. 2004, 279, 40835–40843. [Google Scholar]
- Bobardt, M.D.; Cheng, G.; de Witte, L.; Selvarajah, S.; Chatterji, U.; Sanders-Beer, B.E.; Geijtenbeek, T.B.H.; Chisari, F.V.; Gallay, P. A. Hepatitis C virus NS5A anchor peptide disrupts human immunodeficiency virus. Proc. Natl. Acad. Sci. USA 2008, 105, 5525–5530. [Google Scholar]
- St Vincent, M.R.; Colpitts, C.C.; Ustinov, A.V.; Muqadas, M.; Joyce, M.A.; Barsby, N.L.; Epand, R.F.; Epand, R.M.; Khramyshev, S.A.; Valueva, O.A.; Korshun, V.A.; Tyrrell, D.L.J.; Schang, L.M. Rigid amphipathic fusion inhibitors, small molecule antiviral compounds against enveloped viruses. Proc. Natl. Acad. Sci. USA 2010, 107, 17339–17344. [Google Scholar]
- Wolf, M.C.; Freiberg, A.N.; Zhang, T.; Akyol-Ataman, Z.; Grock, A.; Hong, P.W.; Li, J.; Watson, N.F.; Fang, A.Q.; Aguilar, H.C.; Porotto, M.; Honko, A.N.; Damoiseaux, R.; Miller, J.P.; Woodson, S.E.; Chantasirivisal, S.; Fontanes, V.; Negrete, O.A.; Krogstad, P.; Dasgupta, A.; Moscona, A.; Hensley, L.E.; Whelan, S.P.; Faull, K.F.; Holbrook, M.R.; Jung, M.E.; Lee, B. A broad-spectrum antiviral targeting entry of enveloped viruses. Proc. Natl. Acad. Sci. USA 2010, 107, 3157–3162. [Google Scholar]
- Kliger, Y.; Gallo, S.A.; Peisajovich, S.G.; Munoz-Barroso, I.; Avkin, S.; Blumenthal, R.; Shai, Y. Mode of action of an antiviral peptide from HIV-1. Inhibition at a post-lipid mixing stage. J. Biol. Chem. 2001, 276, 1391–1397. [Google Scholar]
- Veiga, S.; Henriques, S.; Santos, N.C.; Castanho, M. Putative role of membranes in the HIV fusion inhibitor enfuvirtide mode of action at the molecular level. Biochem. J. 2004, 377, 107–110. [Google Scholar] [CrossRef]
- Matos, P.M.; Castanho, M.A.R.B.; and Santos, N.C. HIV-1 fusion inhibitor peptides enfuvirtide and T-1249 interact with erythrocyte and lymphocyte membranes. PLoS One 2010, 5, e9830–1. [Google Scholar]
- Ingallinella, P.; Bianchi, E.; Ladwa, N. A.; Wang, Y.; Hrin, R.; Veneziano, M.; Bonelli, F.; Ketas, T.J.; Moore, J.P.; Miller, M.D.; Pessi, A. Addition of a cholesterol group to an HIV-1 peptide fusion inhibitor dramatically increases its antiviral potency. Proc. Natl. Acad. Sci. USA 2009, 106, 5801–5806. [Google Scholar]
- Petersen, J.; Dandri, M.; Mier, W.; Lütgehetmann, M.; Volz, T.; von Weizsäcker, F.; Haberkorn, U.; Fischer, L.; Pollok, J.; Erbes, B.; Seitz, S.; Urban, S. Prevention of hepatitis B virus infection in vivo by entry inhibitors derived from the large envelope protein. Nat. Biotechnol. 2008, 26, 335–341. [Google Scholar] [CrossRef]
- Glebe, D.; Urban, S.; Knoop, E.V.; Cag, N.; Krass, P.; Grün, S.; Bulavaite, A.; Sasnauskas, K.; Gerlich, W. H. Mapping of the hepatitis B virus attachment site by use of infection-inhibiting preS1 lipopeptides and tupaia hepatocytes. Gastroenterology 2005, 129, 234–245. [Google Scholar] [CrossRef]
- Gripon, P.; Cannie, I.; Urban, S. Efficient inhibition of hepatitis B virus infection by acylated peptides derived from the large viral surface protein. J. Virol. 2005, 79, 1613–1622. [Google Scholar] [CrossRef]
- Wang, K.; Xie, S.; Sun, B. Viral proteins function as ion channels. Biochim. Biophys. Acta 2010. [epub ahead of print]. [Google Scholar]
- Hay, A.J.; Wolstenholme, A.J.; Skehel, J.J.; Smith, M.H. The molecular basis of the specific anti-influenza action of amantadine. EMBO J. 1985, 4, 3021–3024. [Google Scholar]
- Rezak, M. Current pharmacotherapeutic treatment options in Parkinson's disease. Dis. Mon. 2007, 53, 214–222. [Google Scholar] [CrossRef]
- Chew, C.F.; Guy, A.; Biggin, P.C. Distribution and dynamics of adamantanes in a lipid bilayer. Biophys. J. 2008, 95, 5627–5636. [Google Scholar] [CrossRef]
- Bright, R.A.; Shay, D.K.; Shu, B.; Cox, N.J.; Klimov, A.I. Adamantane resistance among influenza A viruses isolated early during the 2005-2006 influenza season in the United States. JAMA 2006, 295, 891–894. [Google Scholar]
- Weinstock, D.M.; Zuccotti, G. Adamantane resistance in influenza A. JAMA 2006, 295, 934–936. [Google Scholar] [CrossRef]
- Boriskin, Y.S.; Leneva, I.A.; Pécheur, E.I.; Polyak, S.J. Arbidol: A broad-spectrum antiviral compound that blocks viral fusion. Curr. Med. Chem. 2008, 15, 997–1005. [Google Scholar] [CrossRef]
- Leneva, I.A.; Russell, R. J.; Boriskin, Y. S.; Hay, A. J. Characteristics of arbidol-resistant mutants of influenza virus: Implications for the mechanism of anti-influenza action of arbidol. Antiviral Res. 2009, 81, 132–140. [Google Scholar] [CrossRef]
- Brooks, M.J.; Sasadeusz, J.J.; and Tannock, G.A. Antiviral chemotherapeutic agents against respiratory viruses: Where are we now and what's in the pipeline? Curr. Opin. Pulm. Med. 2004, 10, 197–203. [Google Scholar] [CrossRef]
- Liu, M.; Wang, S.; Yao, W.; Wu, H.; Meng, S.; Wei, M. Pharmacokinetic properties and bioequivalence of two formulations of arbidol: An open-label, single-dose, randomized-sequence, two-period crossover study in healthy Chinese male volunteers. Clin. Ther. 2009, 31, 784–792. [Google Scholar] [CrossRef]
- Pécheur, E.I.; Lavillette, D.; Alcaras, F.; Molle, J.; Boriskin, Y.S.; Roberts, M.; Cosset, F.L.; Polyak, S.J. Biochemical mechanism of hepatitis C virus inhibition by the broad-spectrum antiviral arbidol. Biochemistry 2007, 46, 6050–6059. [Google Scholar]
- Haid, S.; Pietschmann, T.; Pécheur, E.I. Low pH-dependent hepatitis C virus membrane fusion depends on E2 integrity, target lipid composition, and density of virus particles. J. Biol. Chem. 2009, 284, 17657–17667. [Google Scholar]
- Boriskin, Y.S.; Pécheur, E.I.; Polyak, S.J. Arbidol: A broad-spectrum antiviral that inhibits acute and chronic HCV infection. Virol. J. 2006, 3, 56–1. [Google Scholar]
- Teissier, E.; Zandomeneghi, G.; Loquet, A.; Lavillette, D.; Lavergne, J.P.; Montserret, R.; Cosset, F.L.; Böckmann, A.; Meier, B.H.; Penin, F. Pécheur EI: Mechanism of inhibition of enveloped virus membrane fusion by the antiviral drug arbidol. PLoS One 2011, in press. [Google Scholar]
- Liu, R.; Tewari, M.; Kong, R.; Zhang, R.; Ingravallo, P.; Ralston, R. A peptide derived from hepatitis C virus E2 envelope protein inhibits a post-binding step in HCV entry. Antiviral Res. 2010, 86, 172–179. [Google Scholar] [CrossRef]
- Zhang, H.; Rothwangl, K.; Mesecar, A.D.; Sabahi, A.; Rong, L.; Fong, H.H.S. Lamiridosins, hepatitis C virus entry inhibitors from Lamium album. J. Nat. Prod. 2009, 72, 2158–2162. [Google Scholar] [CrossRef]
- Kroll, D.J.; Shaw, H.S.; Oberlies, N.H. Milk thistle nomenclature: Why it matters in cancer research and pharmacokinetic studies. Integr. Cancer Ther. 2007, 6, 110–119. [Google Scholar] [CrossRef]
- Stickel, F.; Schuppan, D. Herbal medicine in the treatment of liver diseases. Dig. Liver Dis. 2007, 39, 293–304. [Google Scholar] [CrossRef]
- Wu, J.; Lin, L.; Tsai, T. Drug-drug interactions of silymarin on the perspective of pharmacokinetics. J. Ethnopharmacol. 2009, 121, 185–193. [Google Scholar] [CrossRef]
- Polyak, S.J.; Morishima, C.; Lohmann, V.; Pal, S.; Lee, D.Y.W.; Liu, Y.; Graf, T.N.; Oberlies, N.H. Identification of hepatoprotective flavonolignans from silymarin. Proc. Natl. Acad. Sci. USA 2010, 107, 5995–5999. [Google Scholar]
- Seeff, L.B.; Curto, T.M.; Szabo, G.; Everson, G.T.; Bonkovsky, H.L.; Dienstag, J.L.; Shiffman, M.L.; Lindsay, K.L.; Lok, A.S.F.; Di Bisceglie, A.M.; Lee, W.M.; Ghany, M.G. Herbal product use by persons enrolled in the hepatitis C Antiviral Long-Term Treatment Against Cirrhosis (HALT-C) Trial. Hepatology 2008, 47, 605–612. [Google Scholar]
- Ferenci, P.; Scherzer, T.; Kerschner, H.; Rutter, K.; Beinhardt, S.; Hofer, H.; Schöniger-Hekele, M.; Holzmann, H.; Steindl-Munda, P. Silibinin is a potent antiviral agent in patients with chronic hepatitis C not responding to pegylated interferon/ribavirin therapy. Gastroenterology 2008, 135, 1561–1567. [Google Scholar] [CrossRef]
- Neumann, U.P.; Biermer, M.; Eurich, D.; Neuhaus, P.; Berg, T. Successful prevention of hepatitis C virus (HCV) liver graft reinfection by silibinin mono-therapy. J. Hepatol. 2010, 52, 951–952. [Google Scholar] [CrossRef]
- Ahmed-Belkacem, A.; Ahnou, N.; Barbotte, L.; Wychowski, C.; Pallier, C.; Brillet, R.; Pohl, R.; Pawlotsky, J. Silibinin and related compounds are direct inhibitors of hepatitis C virus RNA-dependent RNA polymerase. Gastroenterology 2010, 138, 1112–1122. [Google Scholar]
- Wagoner, J.; Negash, A.; Kane, O.J.; Martinez, L.E.; Nahmias, Y.; Bourne, N.; Owen, D.M.; Grove, J.; Brimacombe, C.; McKeating, J.A.; Pécheur, E.I.; Graf, T.N.; Oberlies, N.H.; Lohmann, V.; Cao, F.; Tavis, J.E.; Polyak, S.J. Multiple effects of silymarin on the hepatitis C virus lifecycle. Hepatology 2010, 51, 1912–1921. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Teissier, E.; Penin, F.; Pécheur, E.-I. Targeting Cell Entry of Enveloped Viruses as an Antiviral Strategy. Molecules 2011, 16, 221-250. https://doi.org/10.3390/molecules16010221
Teissier E, Penin F, Pécheur E-I. Targeting Cell Entry of Enveloped Viruses as an Antiviral Strategy. Molecules. 2011; 16(1):221-250. https://doi.org/10.3390/molecules16010221
Chicago/Turabian StyleTeissier, Elodie, François Penin, and Eve-Isabelle Pécheur. 2011. "Targeting Cell Entry of Enveloped Viruses as an Antiviral Strategy" Molecules 16, no. 1: 221-250. https://doi.org/10.3390/molecules16010221
APA StyleTeissier, E., Penin, F., & Pécheur, E.-I. (2011). Targeting Cell Entry of Enveloped Viruses as an Antiviral Strategy. Molecules, 16(1), 221-250. https://doi.org/10.3390/molecules16010221




