Abstract
Paclitaxel (1a), a well known antitumor agent adopted mainly for the treatment of breast and ovarian cancer, suffers from significant disadvantages such as low solubility, certain toxicity and specific drug-resistance of some tumor cells. To overcome these problems extensive research has been carried out. Among the various proposed strategies, the conjugation of paclitaxel (1a) to a biocompatible polymer, such as hyaluronic acid (HA, 2), has also been considered. Coupling a bioactive compound to a biocompatible polymer offers, in general, many advantages such as better drug solubilization, better stabilization, specific localization and controlled release. Hereafter the design, synthesis and applications of hyaluronic acid-paclitaxel bioconjugates are reviewed. An overview of HA-paclitaxel combinations is also given.
1. Introduction
1.1.Paclitaxel
Paclitaxel (1a), a taxane diterpenoid isolated in 1967 from the bark of Taxus brevifolia (Pacific yew) [], is a well known antitumor agent adopted mainly for the treatment of breast [,,] and ovarian cancer [,,,]. Paclitaxel is a mitotic inhibitor which acts by interfering in the normal microtubule growth during cell division.
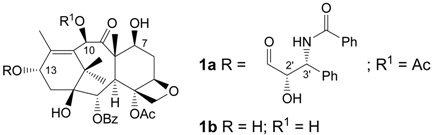
Since the beginning, the difficulty of paclitaxel availability was evident, owing to its scarcity in the bark (10 g of pure material from 1,200 Kg of bark) and to the fact that the use of this tree as the only source of compound 1a would have rapidly caused its disappearance. In addition, owing to the complexity of its structure, paclitaxel can only be obtained in trace amounts by total synthesis. Thus, studies aimed to develop alternative ways of supply were initiated by Green, Guéritte-Voegelein and co-workers [] and Holton []. These authors developed a semisynthetic route to paclitaxel starting from 10-deacetylbaccatin III (1b), extracted in high yield from the leaves of Taxus baccata L. (European yew). The Holton preparation was licensed to Bristol-Myers Squibb enabling the company to produce 1a on a very large scale. Currently paclitaxel production employs plant cell fermentation technology [], but the search for new semisynthetic routes or new culturing media is still in progress, due to the excellent antitumor properties of this drug.
Despite the above valuable therapeutic features, paclitaxel suffers from significant disadvantages among which low water solubility, certain toxicity (which limits the clinically administered dose) and specific drug-resistance of some tumor cells []. Owing to its low water solubility it is generally administered as a castor oil (Cremophor®)/EtOH solution. This type of administration requires hospitalization, since side effects such as hypersensivity, may occur [,,]. In addition it has been reported that Cremophor® reduces the free paclitaxel fraction because of its entrapment in Cremophor® micelles [,].
1.2.Hyaluronic Acid
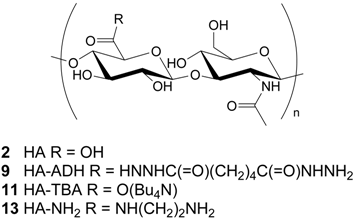
Hyaluronic acid (HA, 2) is a glycosaminoglycan found distributed throughout the connective, epithelial and neural tissues []. It is one of the main components of the extracellular matrix, contributes significantly to cell proliferation and migration and is also involved in the progression of some malignant tumors where it is highly concentrated; besides, it turns out to be an important signal for activating kinase pathways [,] and regulating angiogenesis []. Moreover, since some specific HA receptors (CD44, RHAMM) are overexpressed in various malignant cell types, linking an antitumor drug to 2 might improve targeting to cancerous cells and overcome, if the case, the problem of low drug hydrosolubility. For these reasons, HA has been linked to various antitumor drugs [,].
1.3. Paclitaxel Derivatives
To overcome the disadvantages related to paclitaxel's low water solubility and toxicity, extensive research has been carried out and various strategies have been proposed, such as for instance, changes in its formulation [,,,,] and the preparation of new derivatives, mainly through ester formation at the C-2’ or C-7 positions []. In some of these derivatives, paclitaxel is linked to macromolecules, such as poly(ethyleneglycol) [,,,,,,,,,,,,,,], N-(2-hydroxypropyl)methacrylamide [,,,,,], carboxy-methyldextran [], poly(l-glutamic acid) [,,,,,,,,,,,,,,,,,,,,,,,,,], peptides [,], proteins [,,], dendrimers [,,,] and HA [,,,,,]. Joining a bioactive compound to a biocompatible polymer offers, in general, several advantages like better drug solubilization, stabilization, localization and controlled release [,,]. For derivatives other than those quoted above, see [,,,,,,,,,].
In the present review we shall deal only with HA-paclitaxel bioconjugates. To the best of our knowledge, only four HA-paclitaxel bioconjugates 3-6 (Figure 1) have been described so far in the literature, differing mainly in the anchor chain between HA and paclitaxel.
Prestwich and co-workers, at Stony Brook University in New York and at the University of Utah in Salt Lake City, pioneered these studies and made a major contribution to the field [,]. This work was followed by the work at University of Rome “La Sapienza” of Crescenzi, Marini Bettolo and co-workers [,,], in collaboration with Padua based Fidia Farmaceutici S.p.A., and by that of Tabrizian and co-workers at McGill University in Montreal []. Hereafter we wish to report on the design, synthesis and applications of HA-paclitaxel bioconjugates 3-6, prepared by the above mentioned groups. An overview of HA-paclitaxel combinations is also given.
2. Synthesis and Applications of HA-paclitaxel Bioconjugates 3-4
2.1. Synthesis
Prestwich and co-workers prepared bioconjugate 3 [,] by the dihydrazide method [,,]. This method was developed with the aim of obtaining under mild conditions, necessary to avoid HA degradation, a HA derivative bearing a terminal hydrazido group which would have allowed further coupling still under mild conditions. The spacer should have eased intracellular enzyme degradation or hydrolytic cleavage []. Thus paclitaxel 1a was first reacted with succinic anhydride and pyridine at room temperature for 3 days to give the 2’-hemisuccinate paclitaxel derivative 7, that in turn was transformed, by a reaction with N-hydroxysuccinimido diphenyl phosphate, into 2’-ester 8 containing a N-hydroxysuccinimide moiety. Finally 8 was reacted with adipic dihydrazide modified HA 9 (HA-ADH), prepared from low molecular weight HA 2 and adipic dihydrazide [,,] at pH 6.5 [] in a 3mM phosphate buffer and DMF mixture (Scheme 1).
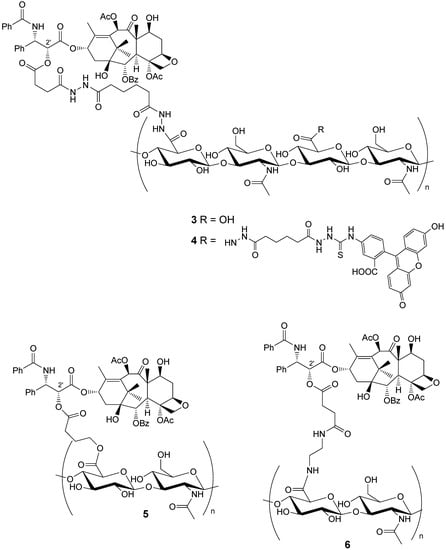
Figure 1.
HA-paclitaxel Bioconjugates 3-6.
The reasons for using low molecular weight HA were: to monitor the biopolymer loading by NMR; to have a readily injectable non viscous solution; besides, once in the plasma, low molecular weight HA is taken up quickly by cells without further degradation and can be expelled from the body via kidneys. Coupling paclitaxel derivative 8 with HA-ADH 9, gave bioconjugate 3 after dialysis and lyophilization. By varying the 8/9 ratio several bioconjugates of type 3 were prepared, whose hydrosolubility depends on ADH and paclitaxel 2’-hemisuccinate loading.
Evaluation of bioconjugate 3 ability to bind to tumor cells and its subsequent internalization by cells, was achieved by converting it with fluorescein isothiocyanate (FITC) in DMF into the fluorescently labeled HA-paclitaxel derivative 4 [].
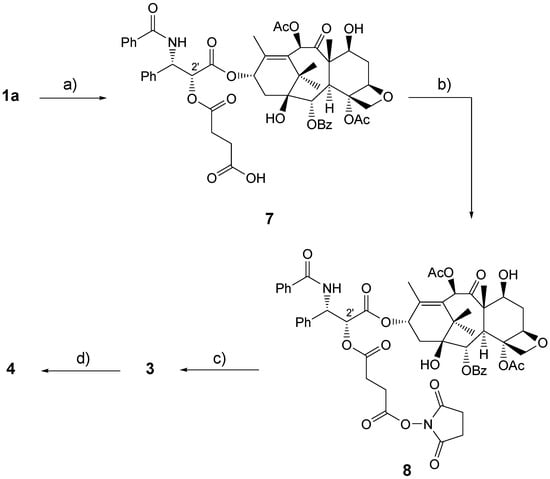
Scheme 1.
Preparation of Bioconjugate 3.
2.2. In Vitro Antitumor Activity of Bioconjugates 3-4
Bioconjugate 3 showed effective in vitro cytotoxicity against HCT-116 (colon tumors), SK-OV-3 (ovarian cancer) and HBL-100 (breast cancer) human cell lines [,]. In contrast, no cytotoxicity was observed against NIH 3-T-3 (nontransformed mouse fibroblast) cell lines. Selective toxicity was attributed to receptor-mediate binding and uptake of HA-paclitaxel bioconjugate owing to overexpression of CD44 receptors by the above human cell lines. Selective targeting due to the receptor characteristic was then confirmed by the possibility of blocking bioconjugate uptake and toxicity with a 100-fold excess of high molecular weight HA and with anti-CD44 mAb; different results were obtained with a 100-fold excess of chondroitin sulfate (a sulfated glycosaminoglycan).
Paclitaxel (1a) release from bioconjugate 3 was evaluated in various media. From these experiments it was found that cleavage occurs at C-2’ ester function, owing to the greater stability of the hydrazide linkage over the ester bond. Thus only free 1a is released. It was also observed that release was dramatically accelerated in the presence of hydrolytic enzymes. Prestwich and co-workers showed also that cytotoxicity of bioconjugate 3 depends on both HA (2) modification and paclitaxel (1a) loading: high loading, in fact, lowers solubility and causes modification in the HA structure masking the HA receptor recognition elements resulting in a citotoxicity decrease.
In vitro cytotoxicity of 3 against the CD44(+) human ovarian tumor cell lines SK-OV-3ip and NMP-1 was also recently evaluated by Klostergaard and co-workers [] at the University of Texas in Houston. Their results turned out to be in good agreement with those previously obtained by Prestwich and co-workers [,], especially as far as “cell targeting” ability is concerned. Klostergaard and co-workers have shown, in fact, that the ability of 3 to reduce cell survival was inhibited by preblocking the HA binding sites with HA 2 (≈40 kDa).
2.3. In Vivo Antitumor Activity of Bioconjugate 3
Better in vivo antitumor efficacy and lower toxicity against CD44(+) human ovarian carcinoma xenografts NMP-1 and SKOV-3ip, as compared to free paclitaxel, was also demonstrated by the same authors for bioconjugate 3 [].
3. Synthesis and Applications of HA-paclitaxel Bioconjugate 5
3.1. Synthesis
The approach chosen by Crescenzi, Marini Bettolo and co-workers, in collaboration with FIDIA Farmaceutici S.p.A., a pharmaceutical company involved for many years in the production and derivatization of HA, is quite simple. Paclitaxel 1a was joined to HA by means of a spacer linked to both paclitaxel 1a and HA 2 via ester functions (Scheme 2) [,,].
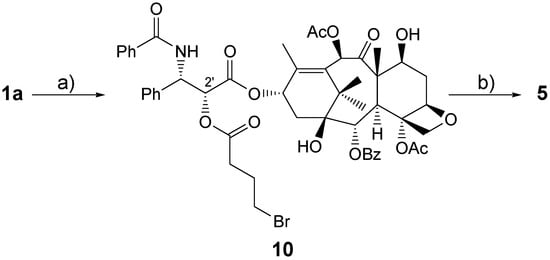
Scheme 2.
Preparation of Bioconjugate 5.
Thus, paclitaxel was treated at room temperature with 4-bromobutanoic acid in the presence of 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide hydrochloride (EDC) and N,N-dimethylpyridine-4-amine (DMAP) to cleanly give in 78 % yield paclitaxel 2’-(4-bromobutanoate) (10, Scheme 2). The authors confirmed the formation of a 2’-O-substituted paclitaxel derivative by the characteristic downfield shifts in the 1H- and 13C-NMR of the signals of H-C(2’) from δ(H) 4.77 [] to 5.50 and of C(2’) from δ(C) 73.2 [] to 74.2, respectively.
Compound 10 was then dissolved in 1-methylpyrrolidin-2-one (NMP) and treated for 7 days at room temperature with HA-TBA 11 (Mw = 185 kDa; 10/11 1:4) to give 5. The ratio of reactants chosen was a good compromise to give hydrosoluble bioconjugate. Bioconjugate 5 was isolated from the reaction mixture after EtOH/NaCl precipitation, extensive dialysis (cut off ca. 2 kDa) against distilled water and finally freeze-dried.
A ca. 25% substitution degree was deduced for 5 by comparing its UV absorbance in EtOH/H2O 7:3 with a UV-absorption calibration plot of paclitaxel 1a and HA-Na. Compound 5 was hydrosoluble. As for the bioconjugate 3 paclitaxel loading can be varied by changing the 10/11 ratio.
3.2. In Vitro Antitumor Activity of Bioconjugate 5
The in vitro antitumor activity of a hydrosoluble bioconjugate 5 with a 20% wt/wt carboxyl esterification, was tested in respect to bladder cancer cells by Rosato and co-workers at Padua University [,]. The in vitro 5 inhibitory activity on the growth of RT-4 and RT-112/84 bladder cancer cells was much stronger than that of paclitaxel (1a). Furthermore, the powerful increase in efficiency of 5 in respect to 1a was attributed, as in the case of bioconjugate 3, to the active intracellular uptake mediated by specific HA receptors (CD44) overexpressed on the tumor cell surface and a subsequent hydrolytic release of the active drug in the intracellular medium only. To prove whether the CD44 HA receptor could directly interact with 5, the CD44 expression was also analyzed at different times after RT-4 and RT112/84 cells were incubated with either the bioconjugate 5 or HA 2. In both cases the authors noted a similar striking up-regulation in the CD44 expression showing therefore a direct interaction of conjugated HA with the receptor. The stability of 5 was tested in human urine and no bioconjugate degradation or paclitaxel release was observed 6 hours after incubation (pH 6.5).
3.3. In Vivo Antitumor Activity of Bioconjugate 5
Rosato and co-workers also studied [] the in vivo antitumor therapeutic activity of bioconjugate 5 by inoculating subcutaneously RT-112/84 TCC bladder tumor cells in mice. A comparison between the antitumor activity of paclitaxel (1a) and bioconjugate 5 showed that the efficacy of 1a and 5 is equal though the former has a slightly stronger cytoreductive activity.
A pharmacokinetic analysis of 5 was also performed in order to exclude the presence of paclitaxel 1a in the blood after administration of bioconjugate 5 to rat bladder. According to the authors, although the stability of 5 had already been tested in vitro, it might be possible that release of paclitaxel takes place in vivo after bladder instillation. The experiments confirmed that paclitaxel concentration in the blood was negligible, since after administration it remained entirely confined in the bladder. Mice were also treated locally with bioconjugate 5 and paclitaxel to determine whether 5 was well tolerated by the urothelial mucosa. From this study it emerged that bioconjugate 5 is very well tolerated and induces only slight morphologic changes in the urothelial epithelium, while paclitaxel produces notable toxic effects on the bladder.
In order to establish its potential therapeutic applications and to evaluate its biodistribution after intravenous, intravesical, oral or intraperitoneal administration in healthy mice, Meléndez-Alafort and co-workers [] at the University of Padua, labeled bioconjugate 5 with 99mTc by treating it with a 99mTc-pertechnetate solution, SnCl2 and sodium gluconate. The solution was stirred and incubated at 65°C for 90 min. The radiopharmaceutical was purified by size exclusion chromatography and the radiochemical purity of the labeled bioconjugate 5 prepared in this way was 100%. The 99mTc labeled bioconjugate 5 was stable for 6 h at 37°C in a phosphate buffer.
From biodistribution studies it emerged that the animals injected intravenously with the 99mTc labeled bioconjugate 5 showed a rapid and high liver and spleen uptake, while those administered intraperitoneally, intravesically and orally showed that it remained at the administration site. Therefore, according to Meléndez-Alafort and co-workers, bioconjugate 5 should be administered intravenously for liver metastasis therapy, orally or intravesically for local treatment of bladder and superficial cancers and intraperitoneally for ovarian cancer or other tumors in the peritoneal cavity.
4. Synthesis and Applications of HA-paclitaxel Bioconjugate 6 and Biomaterial 14
4.1. Synthesis
In the field of drug delivery, great attention has been recently devoted to the possibility of incorporating bioactive molecules into polymeric matrices [,,,,,]. An approach developed some years ago based on alternate deposition of polyanion and polycation layers leading to polyelectrolyte multilayer (PEM) films, constructed by the layer-by-layer (LbL) technique, can be used to build up polymeric matrices in a controlled manner. PEMs may present some advantages in drug delivery such as the possibility of including several drugs. It is, therefore, suitable for complex releasing pharmacokinetics. Tabrizian and co-workers [] reported on the assembling of HA-paclitaxel bioconjugate 6 (Figure 1), previously described by Prestwich and co-workers [] in a PEM system with chitosan 12 (CH) to give biomaterial 14.
The preparation of biomaterial 14 started from known 8 [] (Section 2.1 and Scheme 1). The latter was reacted with 13 (see section 1.2), prepared in turn treating HA (2, Mw = 0.5 MDa) with ethylenediamine hydrochloride (EDA) and EDC in a buffered medium (pH 5) at r.t. for 12 h, purifying the reaction mixture by dialysis against water and freeze-drying the resulting aqueous solution.
Coupling of paclitaxel derivative 8 with 13 was performed at room temperature for 24 h in a DMF and phosphate buffer (pH 6.5). After that time the crude compound was purified by dialysis to give two bioconjugates 6 with 3 and 6 mol % paclitaxel loading respectively; only the former, however, exhibited the hydrosolubility necessary for the PEM preparation.
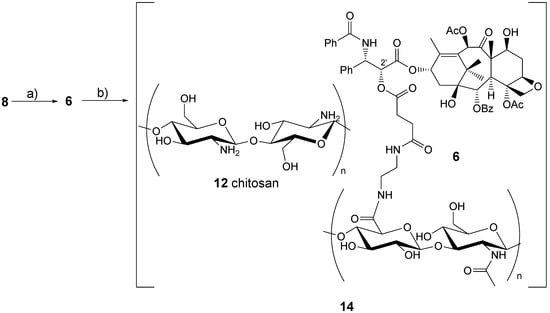
Scheme 2.
Preparation of Biomaterial 14.
The final multilayer was obtained by consecutive adsorption of oppositely charged polyelecrolytes [CH, HA-paclitaxel 6 (3 mol %)]. Paclitaxel 1a release studies were conducted on 10 bilayers of CH/HA-paclitaxel (CH/HA-paclitaxel)10. The latter were kept in contact with water which was then removed at different times. By measuring the UV absorbance of released compound 1a, it was found that the half-life time of 1a was ≈ 3 h. PEM constructed with rhodamine-labeled chitosan (CH-Rho/HA-paclitaxel)10 were subjected to the same release test; contact water UV spectra showed only the paclitaxel absorbance peak, while that of rhodamine was lacking, proving in this way the multilayer stability during release experiments.
4.2. In Vitro Antitumor Activity of Biomaterial 14
In vitro activity of biomaterial 14 was evaluated by means of a cell viability assay. While cells cultured onto (CH/HA-paclitaxel)10 showed a 95% reduction in viability after 4 days, those cultured onto (CH/HA)10 showed no reduction in viability [].
5. HA-paclitaxel Combinations
Although this review is mainly focused on the HA-paclitaxel bioconjugates, for the sake of completeness, we wish to present hereafter a small section on HA-paclitaxel combinations, i.e. species in which no covalent links between HA (2) and paclitaxel (1a) are present, and on their bioactivity.
5.1. Paclitaxel-loaded Crosslinked HA Films for the Prevention of Postsurgical Adhesions
Burt and co-workers at the University of British Columbia [] investigated the use of paclitaxel (1a) as an inhibitor of postsurgical adhesion by loading it into a biocompatible, mucoadhesive film of crosslinked HA to be applied to abraded tissues in order to release the drug over 2-3 days. Since 1a is as a powerful wound healing inhibitor, its permanence on the wound for a limited time might reduce the formation of postsurgical adhesions. After several optimization studies, HA films crosslinked with 2 mM EDAC and 10% glycerol were found to posses suitable flexibility, elasticity and dissolution properties. In these films paclitaxel was present as a solid dispersion. According to Burt and co-workers a possible mechanism for drug release involves the uptake of water, the swelling of the crosslinked HA matrix and the dissolution of dispersed paclitaxel.
An in vivo comparison between the administration of paclitaxel on the abrasion of the rat cecal side wall by repeated intraperitoneal injections in a 1:1 Cremophor EL®:ethanol formulation and by loading it into a crosslinked HA film showed similar, though incomplete, inhibition of adhesion formations in rats.
5.2. In vivo Inhibition of Mice Lewis Lung Carcinoma and U14 Cervical Tumor By Combination of Paclitaxel and HA.
Yuan and co-workers [] at Tianjin University have investigated the effects of combined administration of HA (2) with Cremophor® solutions of paclitaxel (1a) on the control of Lewis lung carcinoma (LLC) migration and ascites formation of U14 cervical tumor. In vivo studies on mice showed that the combined use of HA and paclitaxel is more effective in inhibiting metastasis of LLC and U14 than HA or paclitaxel alone. Thus, Yuan and co-workers hypothesized that the synergic behavior of paclitaxel and HA might be due to an increased host immunity. The increase in the expression of vitamin D3 binding protein (DBP), a macrophage stimulating activator, may be a crucial factor in inhibiting the activity of tumor cells. Yuan and co-workers [] found that DBP expression was increased by administrating the paclitaxel-HA combination. The administration of HA or paclitaxel alone did not have the same effects.
5.3. Polyelectrolyte Multilayers Films Incorporating Paclitaxel
As outlined in Section 4, PEMs have recently become an appropriate substrate coating able to incorporate biological factors for example peptides, proteins, hormones, growth factors or drugs [,,,,,]. The final aim of such approaches is that of controlling the rate and selectivity of cellular adhesion.
In their studies at Louis Pasteur University in Strasbourg Vodouhê, Lavalle and co-workers [] designed a polylysine/hyaluronic acid (PLL/HA) based multilayers surface coating which acts as a reservoir for paclitaxel without the need of chemical modifications both on the PEM and on the drug. The amount of compound 1a embedded in PLL/HA films could be finely tuned. The authors tested the viability of HT29 cell line seeded on (PLL/HA)30 film. Unfortunately these in vitro studies showed these films did not adhere the cells. The HT29 cellular adhesion occurred when the film surface was modified by adding a poly(sodium 4-styrene sulfonate) (PSS) layer on the top of PLL/HA films. As shown previously by Chan and co-workers [], a sulfonate group of PSS chains adsorbed on the surface not only is able to promote cellular adhesion, but also allows modulation of the accessibility of HT29 cells to paclitaxel in terms of delay and/or kinetics by varying its composition. Paclitaxel activity remained constant after embedding in the polyelectrolyte multilayers and cellular viability could be reduced of about 80% 96 h after seeding.
6. Conclusions
HA-paclitaxel hydrosoluble bioconjugates appear promising in cancer therapy. Their cytotoxicity against various cancer cell lines is, in fact, comparable to that of free paclitaxel (1a) and systemic toxicity reduced owing to selective targeting of cancer cells due to HA CD44 receptors overexpression. Besides, as illustrated in the Introduction, the problems connected with the administration of paclitaxel in a castor oil (Cremophor®)/EtOH solution can be avoided. From biodistribution studies it appears recommendable a different way of administration depending on tumor localization. The preparation of nanobiomaterials incorporating HA-paclitaxel bioconjugates offers a further solution for the delivery of this valuable drug. Finally very interesting results were obtained with HA-paclitaxel combinations both for cancer chemotherapy and for better wound healing and prevention of postsurgical adhesion.
Acknowledgements
We are grateful to the Guest Editor and Editor-in-Chief of Molecules for inviting us to present this review which offered us the opportunity to describe the work of the groups operating in the field of HA-paclitaxel bioconjugates and combinations. Financial support by Università degli Studi di Roma “La Sapienza” (Ateneo 60%) is gratefully acknowledged.
References and Notes
- Prof. Vittorio Crescenzi (12-Jan-1932-12-Jun-2007), a leading scientist in the field of macromolecules and biopolymers, taught in several Italian Universities (Bari, Napoli, Trieste, Roma “La Sapienza”). He was a NATO Fellow in the Laboratory of Nobel Laureate P.J. Flory at Stanford University and Visiting Professor at Stevens Institute of Technology in Hoboken, N.J.
- Wani, M.C.; Taylor, H.L.; Wall, M.E.; Coggon, P.; McPhail, A.T. Plant antitumor agents. VI. Isolation and structure of Taxol, a novel antileukemic and antitumor agent from Taxus brevifolia. J. Am. Chem. Soc. 1971, 93, 2325–2327. [Google Scholar] [CrossRef]
- Perez, E.A. Paclitaxel in breast cancer. Oncologist 1998, 3, 373–389. [Google Scholar]
- Gibbs, J.B. Mechanism-based target identification and drug discovery in cancer research. Science 2000, 287, 1969–1973. [Google Scholar] [CrossRef]
- Jordan, M.A.; Wilson, L. Microtubules as a target for anticancer drugs. Nat. Rev. Cancer 2004, 4, 253–265. [Google Scholar] [CrossRef]
- Markman, M. Weekly paclitaxel in the management of ovarian cancer. Semin. Oncol. 2000, 27, 37–40. [Google Scholar]
- Seiden, M.V. Ovarian Cancer. Oncologist 2001, 6, 327–332. [Google Scholar] [CrossRef]
- Singla, A.K.; Garg, A.; Aggarwal, D. Paclitaxel and its formulations. Int. J. Pharm. 2002, 235, 179–192. [Google Scholar] [CrossRef]
- Denis, J.-N.; Greene, A.E.; Guénard, D.; Guéritte-Voegelein, F.; Mangatal, L.; Potier, P. Highly efficient, practical approach to natural Taxol. J. Am. Chem. Soc. 1988, 110, 5917–5919. [Google Scholar]
- Holton, R.A. Method for preparation of Taxol. Eur. Pat. Appl. EP 400971, 1990. [Google Scholar]
- Patel, R.N. Tour de Paclitaxel: biocatalysis for semisynthesis. Annu. Rev. Microbiol. 1998, 52, 361–395. [Google Scholar] [CrossRef]
- Galletti, E.; Magnani, M.; Renzulli, M.L.; Botta, M. Paclitaxel and docetaxel resistance: molecular mechanisms and development of new generation taxanes. ChemMedChem 2007, 2, 920–942. [Google Scholar] [CrossRef]
- Szebeni, J.; Muggia, F.M.; Alving, C.R. Complement activation by Cremophor EL as a possible contributor to hypersensitivity to paclitaxel: an in vitro study. J. Natl. Cancer Inst. 1998, 90, 300–306. [Google Scholar] [CrossRef]
- Thigpen, J.T. Chemotherapy for advanced ovarian cancer: overview of randomized trials. Semin. Oncol. 2000, 27, 11–16. [Google Scholar]
- Knemeyer, I.; Wientjes, M.G.; Au, J.L.-S. Cremophor reduces paclitaxel penetration into bladder wall during intravesical treatment. Cancer Chemother. Pharmacol. 1999, 44, 241–248. [Google Scholar] [CrossRef]
- Gelderblom, H.; Verweij, J.; Van Zomeren, D. M.; Buijs, D.; Ouwens, L.; Nooter, K.; Stoter, G.; Sparreboom, A. Influence of Cremophor EL on the bioavailability of intraperitoneal paclitaxel. Clin. Cancer Res. 2002, 8, 1237–1241. [Google Scholar]
- Laurent, T.C.; Laurent, U.B.; Fraser, J.R. Functions of hyaluronan. Ann. Rheum. Dis. 1995, 54, 429–432. [Google Scholar] [CrossRef]
- Misra, S.; Toole, B.P.; Ghatak, S. Hyaluronan constitutively regulates activation of multiple receptor tyrosine kinases in epithelial and carcinoma cells. J. Biol. Chem. 2006, 281, 34936–34941. [Google Scholar] [CrossRef]
- Ohno, S.; Im, H.-J.; Knudson, C.B.; Knudson, W. Hyaluronic oligosaccharides induce matrix metalloproteinase 13 via transcriptional activation of NFkB and p38 MAP kinase in articular chondrocytes. J. Biol. Chem. 2006, 281, 17952–17960. [Google Scholar]
- Rooney, P.; Kumar, S.; Ponting, J.; Wang, M. The role of hyaluronan in tumour neovascularization (review). Int. J. Cancer 1995, 60, 632–636. [Google Scholar] [CrossRef]
- Akima, K.; Ito, H.; Iwata, Y.; Matsuo, K.; Watari, N.; Yanagi, M.; Hagi, H.; Oshima, K.; Yagita, A.; Atomi, Y.; Tatekawa, I. Evaluation of antitumor activities of hyaluronate binding antitumor drugs: synthesis, characterization and antitumor activity. J. Drug Target. 1996, 4, 1–8. [Google Scholar] [CrossRef]
- Jaracz, S.; Chen, J.; Kuznetsova, L.V.; Ojima, I. Recent advances in tumor-targeting anticancer drug conjugates. Bioorg. Med. Chem. 2005, 13, 5043–5054. [Google Scholar] [CrossRef]
- Pawar, R.; Shikanov, A.; Vaisman, B.; Domb, A.J. Intravenous and regional paclitaxel formulations. Curr. Med. Chem. 2004, 11, 397–402. [Google Scholar] [CrossRef]
- Kim, T.-Y.; Kim, D.-W.; Chung, J.-Y.; Shin, S.G.; Kim, S.-C.; Heo, D.S.; Kim, N.K.; Bang, Y.-J. Phase I and pharmacokinetic study of Genexol-PM, a Cremophor-free, polymeric micelle-formulated paclitaxel, in patients with advanced malignancies. Clin. Cancer Res. 2004, 10, 3708–3716. [Google Scholar] [CrossRef]
- Wang, Y. Tocosol® paclitaxel. Anticancer formulation. Drugs Fut. 2006, 31, 40–42. [Google Scholar] [CrossRef]
- Henderson, I.C.; Bhatia, V. Nab-paclitaxel for breast cancer: a new formulation with an improved safety profile and greater efficacy. Exp. Rev. Anticancer Ther. 2007, 7, 919–943. [Google Scholar] [CrossRef]
- Skwarczynki, M.; Hayashi, Y.; Kiso, Y. Paclitaxel prodrugs: toward smarter delivery anticancer agents. J. Med. Chem. 2006, 49, 7253–7269, and references cited therein. [Google Scholar] [CrossRef]
- Greenwald, R.B.; Pendri, A.; Bolikal, D.; Gilbert, C.W. Highly water soluble taxol derivatives: 2’-polyethylene glycol esters as potential prodrugs. Bioorg. Med. Chem. Lett. 1994, 4, 2465–2470. [Google Scholar]
- Greenwald, R.B.; Pendri, A.; Bolikal, D. Highly water soluble taxol derivatives: 7-polyethylene glycol carbamates and carbonates. J. Org. Chem. 1995, 60, 331–336. [Google Scholar] [CrossRef]
- Li, C.; Yu, D.; Inoue, T.; Yang, D.J.; Milas, L.; Hunter, N.R.; Kim, E.E.; Wallace, S. Synthesis and evaluation of water-soluble polyethylene glycol-paclitaxel conjugate as a paclitaxel prodrug. Anticancer Drugs 1996, 7, 642–648. [Google Scholar] [CrossRef]
- Greenwald, R.B.; Gilbert, C.W.; Pendri, A.; Conover, C.D.; Xia, J.; Martinez, A. Drug delivery systems: water soluble taxol 2’-poly(ethylene glycol) ester prodrugs-design and in vivo effectiveness. J. Med. Chem. 1996, 39, 424–431. [Google Scholar] [CrossRef]
- Pendri, A.; Conover, C.D.; Greenwald, R.B. Antitumor activity of paclitaxel-2’-glycinate conjugate to poly(ethylene glycol): a water soluble prodrug. Anticancer Drug Des. 1998, 13, 387–395. [Google Scholar]
- Ceruti, M.; Crosasso, P.; Brusa, P.; Arpicco, S.; Dosio, F.; Cattel, L. Preparation, characterization, cytotoxicity and pharmacokinetics of liposomes containing water-soluble prodrugs of paclitaxel. J. Controlled Release 2000, 63, 141–153. [Google Scholar] [CrossRef]
- Feng, X.; Yuan, Y.-J.; Wu, J.-C. Synthesis and evaluation of water-soluble paclitaxel prodrugs. Bioorg. Med. Chem. Lett. 2002, 12, 3301–3303. [Google Scholar] [CrossRef]
- Rodrigues, P.C.; Scheuermann, K.; Stockmar, C.; Maier, G.; Fiebig, H.H.; Unger, C.; Mülhaupt, R.; Kratz, F. Synthesis and in vitro efficacy of acid-sensitive poly(ethylene glycol) paclitaxel conjugates. Bioorg. Med. Chem. Lett. 2003, 13, 355–360. [Google Scholar] [CrossRef]
- Zhang, X.; Li, Y.; Chen, X.; Wang, X.; Xu, X.; Liang, Q.; Hu, J.; Jing, X. Synthesis and characterization of the paclitaxel/MPEG-PLA block copolymer conjugate. Biomaterials 2005, 26, 2121–2128. [Google Scholar]
- Khandare, J.J.; Jayant, S.; Singh, A.; Chandna, P.; Wang, Y.; Vorsa, N.; Minko, T. Dendrimer versus linear conjugate: influence of polymeric architecture on the delivery and anticancer effect of paclitaxel. Bioconj. Chem. 2006, 17, 1464–1472. [Google Scholar] [CrossRef]
- Wermeckes, B.; Hess, M.; Dehne, S.; Jo, B.-W.; Sohn, J.-S. Characterization of PEG-modified paclitaxel. Mater. Res. Innov 2006, 10, 364–375. [Google Scholar] [CrossRef]
- Kratz, F.; Abu Ajai, K.; Warnecke, A. Anticancer carrier-linked prodrugs in clinical trials. Expert Opin. Invest. Drugs 2007, 16, 1037–1058. [Google Scholar] [CrossRef]
- Xie, Z.; Lu, T.; Chen, X.; Lu, C.; Zheng, Y.; Jing, X. Triblock poly(lactic acid)-b-poly(ethylene glycol)-b-poly(lactic acid)/paclitaxel conjugates: synthesis, micellization, and cytotoxicity. J. Appl. Polym. Sci. 2007, 105, 2271–2279. [Google Scholar] [CrossRef]
- Xie, Z.; Guan, H.; Chen, X.; Lu, C.; Chen, L.; Hu, X.; Shi, Q.; Jing, X. A novel polymer-paclitaxel conjugate based on amphiphilic triblock copolymer. J. Controlled Release 2007, 117, 210–216. [Google Scholar] [CrossRef]
- Lim, J.; Simanek, E.E. Synthesis of water-soluble dendrimers based on melamine bearing 16 paclitaxel groups. Org. Lett. 2008, 10, 201–204. [Google Scholar] [CrossRef]
- Mendichi, R.; Rizzo, V.; Gigli, M.; Giacometti Schieroni, A. Dilute-solution properties of a polymeric antitumor drug carrier by size-exclusion chromatography, viscometry, and light scattering. J. Appl. Polym. Sci. 1998, 70, 329–338. [Google Scholar] [CrossRef]
- Meerum Terwogt, J.M.; ten Bokkel Huinink, W.W.; Schellens, J.H.M.; Schot, M.; Mandjes, I.A.M.; Zurlo, M.G.; Rocchetti, M.; Rosing, H.; Koopman, F.J.; Beijnen, J.H. Phase I clinical and pharmacokinetic study of PNU166945, a novel water-soluble polymer-conjugated prodrug of paclitaxel. Anti-Cancer Drugs 2001, 12, 315–323. [Google Scholar] [CrossRef]
- Duncan, R.; Gac-Breton, S.; Keane, R.; Musila, R.; Sat, Y.N.; Satchi, R.; Searle, F. Polymer-drug conjugates, PDEPT and PELT: basic principles for design and transfer from the laboratory to clinic. J. Controlled Release 2001, 74, 135–146. [Google Scholar] [CrossRef]
- Rihova, B.; Kubackova, K. Clinical implications of N-(2-hydroxypropyl)methacrylamide copolymers. Curr. Pharm. Biotechnol. 2003, 4, 311–322. [Google Scholar] [CrossRef]
- Haag, R.; Kratz, F. Polymer Therapeutics: concept and applications. Angew. Chem. Int. Ed. 2006, 45, 1198–1215. [Google Scholar] [CrossRef]
- Sugahara, S.; Kajiki, M.; Kuriyama, H.; Kobayashi, T. Paclitaxel delivery systems: the use of amino acid linkers in the conjugation of paclitaxel with carboxymethyldextran to create prodrugs. Biol. Pharm. Bull. 2002, 25, 632–641. [Google Scholar] [CrossRef]
- Multani, A.S.; Li, C.; Ozen, M.; Yadav, M.; Yu, D.-F.; Wallace, S.; Pathak, S. Paclitaxel and water-soluble poly(L-glutamic acid)-paclitaxel induce direct chromosomal abnormalities and cell death in a murine metastatic melanoma cell line. Anticancer Res. 1997, 17, 4269–4274. [Google Scholar]
- Li, C.; Yu, D.-F.; Newman, R.A.; Cabral, F.; Stephens, L.C.; Hunter, N.; Milas, L.; Wallace, S. Complete regression of well-established tumors using a novel water-soluble poly(L-glutamic acid)-paclitaxel conjugate. Cancer Res. 1998, 58, 2404–2409. [Google Scholar]
- Li, C.; Price, J.E.; Milas, L.; Hunter, N.R.; Ke, S.; Yu, D.-F.; Charnsangavej, C.; Wallace, S. Antitumor activity of poly(L-glutamic acid)-paclitaxel on syngeneic and xenografted tumors. Clin. Cancer Res. 1999, 5, 891–897. [Google Scholar]
- Multani, A.S.; Li, C.; Ozen, M.; Imam, A.S.; Wallace, S.; Pathak, S. Cell-killing by paclitaxel in a metastatic murine melanoma cell line is mediated by extensive telomere erosion with no decrease in telomerase activity. Oncol. Rep. 1999, 6, 39–44. [Google Scholar]
- Li, C.; Newman, R.A.; Wu, Q.-P.; Ke, S.; Chen, W.; Hutto, T.; Kan, Z.; Brannan, M.D.; Charnsangavej, C.; Wallace, S. Biodistribution of paclitaxel and poly(L-glutamic acid)-paclitaxel conjugate in mice with ovarian OCa-1 tumor. Cancer Chemother. Pharmacol. 2000, 46, 416–422. [Google Scholar] [CrossRef]
- Li, C.; Ke, S.; Wu, Q.P.; Tansey, W.; Hunter, N.; Buchmiller, L.M.; Milas, L.; Charnsangavej, C.; Wallace, S. Tumor irradiation enhances the tumor-specific distribution of poly(L-glutamic acid)-conjugated paclitaxel and its antitumor efficacy. Clin. Cancer Res. 2000, 6, 2829–2834. [Google Scholar]
- Oldham, E.A.; Li, C.; Ke, S.; Wallace, S.; Huang, P. Comparison of action of paclitaxel and poly(L-glutamic acid)-paclitaxel conjugate in human breast cancer cells. Int. J. Oncol. 2000, 16, 125–132. [Google Scholar]
- Li, C.; Ke, S.; Wu, Q.-P.; Tansey, W.; Hunter, N.; Buchmiller, L.M.; Milas, L.; Charnsangavej, C.; Wallace, S. Potentiation of ovarian OCA-1 tumor radioresponse by poly(L-glutamic acid)-paclitaxel conjugate. Int. J. Radiat. Oncol. Biol. Phys. 2000, 48, 1119–1126. [Google Scholar] [CrossRef]
- Ke, S.; Milas, L.; Charnsangavej, C.; Wallace, S.; Li, C. Potentiation of radioresponse by polymer-drug conjugates. J. Controlled Release 2001, 74, 237–242. [Google Scholar] [CrossRef]
- Auzenne, E.; Donato, N.J.; Li, C.; Leroux, E.; Price, R.E.; Farquhar, D.; Klostergaard, J. Superior therapeutic profile of poly-L-glutamic acid-paclitaxel copolymer compared with Taxol in xenogeneic compartmental models of human ovarian carcinoma. Clin. Cancer Res. 2002, 8, 573–581. [Google Scholar]
- Singer, J.W.; Baker, B.; de Vries, P.; Kumar, A.; Shaffer, S.; Vawter, E.; Bolton, M.; Garzone, P. Poly-(L)-glutamic acid-paclitaxel (CT-2103) [XYOTAX], a biodegradable polymeric drug conjugate: characterization, preclinical pharmacology, and preliminary clinical date. Adv. Exp. Med. Biol. 2003, 519, 81–99. [Google Scholar]
- Milas, L.; Mason, K.A.; Hunter, N.; Li, C.; Wallace, S. Poly(L-glutamic acid)-paclitaxel conjugate is a potent enhancer of tumor radiocurability. Int. J. Radiat. Oncol. Biol. Phys. 2003, 55, 707–712. [Google Scholar] [CrossRef]
- Tishler, R.B. Polymer-conjugated paclitaxel as a radiosensitizing agent-A big step forward for combined modality therapy? Int. J. Radiation Oncology Biol. Phys. 2003, 55, 563–564. [Google Scholar] [CrossRef]
- Zou, Y.; Fu, H.; Ghosh, S.; Farquhar, D.; Klostergaard, J. Antitumor activity of hydrophilic paclitaxel copolymer prodrug using locoregional delivery in human orthotopic non-small cell lung cancer xenograft models. Clin. Cancer Res. 2004, 10, 7382–7391. [Google Scholar] [CrossRef]
- Sabbatini, P.; Aghajanian, C.; Dizon, D.; Anderson, S.; Dupont, J.; Brown, J.V.; Peters, W.A.; Jacobs, A.; Mehdi, A.; Rivkin, S.; Eisenfeld, A.J.; Spriggs, D. Phase II study of CT-2103 in patients with recurrent epithelial ovarian, fallopian tube, or primary peritoneal carcinoma. J. Clin. Oncol. 2004, 22, 4523–4531. [Google Scholar] [CrossRef]
- Singer, J.W.; Shaffer, S.; Baker, B.; Bernareggi, A.; Stromatt, S.; Nienstedt, D.; Besman, M. Paclitaxel poliglumex (XYOTAX; CT-2103): an intracellularly targeted taxane. Anti-Cancer Drugs 2005, 16, 243–254. [Google Scholar] [CrossRef]
- Nemunaitis, J.; Cunningham, C.; Senzer, N.; Gray, M.; Oldham, F.; Pippen, J.; Mennel, R.; Eisenfeld, A. Phase I Study of CT-2103, A polymer-conjugated paclitaxel, and carboplatin in patients with advanced solid tumors. Cancer Invest. 2005, 23, 671–676. [Google Scholar] [CrossRef]
- Singer, J.W. Paclitaxel poliglumex (XYOTAX, CT-2103): a macromolecular taxane. J. Controlled Release 2005, 109, 120–126. [Google Scholar] [CrossRef]
- Chipman, S.D.; Oldham, F.B.; Pezzoni, G.; Singer, J.W. Biological and clinical characterization of paclitaxel poliglumex (PPX, CT-2103), a macromolecular polymer-drug conjugate. Int. J. Nanomed. 2006, 1, 375–383. [Google Scholar] [CrossRef]
- Lee, K.H.; Chung, Y.J.; Kim, Y.C.; Song, S.J. Anti-tumor activity of paclitaxel prodrug conjugated with polyethylene glycol. Bull. Kor. Chem. Soc. 2005, 26, 1079–1082. [Google Scholar] [CrossRef]
- Lee, Y. Preparation and characterization of folic acid linked poly(L-glutamate)nanoparticles for cancer targeting. Macromol. Res. 2006, 14, 387–393. [Google Scholar]
- Dipetrillo, T.; Milas, L.; Evans, D.; Akerman, P.; Ng, T.; Miner, T.; Cruff, D.; Chauhan, B.; Iannitti, D.; Harrington, D.; Safran, H. Paclitaxel poliglumex (PPX-Xyotax) and concurrent radiation for esophageal and gastric cancer: a phase I study. Am. J. Clin. Oncol. 2006, 29, 376–379. [Google Scholar]
- Albain, K.S.; Belani, C.P.; Bonomi, P.; O’Byrne, K.J.; Schiller, J.H.; Socinski, M. Pioneer: a phase III randomized trial of paclitaxel poliglumex versus paclitaxel in chemotherapy-naïve women with advanced-stage non small-cell lung cancer and performance status of 2. Clin. Lung Canc. 2006, 7, 417–419.75. [Google Scholar] [CrossRef]
- Jackson, E.F:; Esparza-Coss, E.; Wen, X.; Ng, C.S.; Daniel, S.L.; Price, R.E.; Rivera, B.; Charnsangavej, C.; Gelovani, J.G.; Li, C. Magnetic resonance imaging of therapy-induced necrosis using gadolinium-chelated polyglutamic acids. Int. J. Radiat. Oncol. Biol. Phys. 2007, 68, 830–838. [Google Scholar] [CrossRef]
- Boddy, A.V. Paclitaxel poliglumex: antimitotic drug oncolytic. Drugs Fut. 2007, 32, 776–780. [Google Scholar] [CrossRef]
- Bonomi, P. Paclitaxel poliglumex (PPX, CT-2103): macromolecular medicine for advanced non-small-cell lung cancer. Expert Rev. Anticancer Ther. 2007, 7, 415–422. [Google Scholar] [CrossRef]
- Papas, S.; Akoumianaki, T.; Kalogiros, C.; Hadjiarapoglou, L.; Theodoropoulos, P.A.; Tsikaris, V. Synthesis and antitumor activity of peptide-paclitaxel conjugates. J. Pept. Sci. 2007, 13, 662–671. [Google Scholar]
- Kumar, S.K.; Williams, S.A.; Isaacs, J.T.; Denmeade, S.R.; Khan, S.R. Modulating paclitaxel bioavailability for targeting prostate cancer. Bioorg. Med. Chem. 2007, 15, 4973–4984. [Google Scholar]
- Dosio, F.; Brusa, P.; Crosasso, P.; Arpicco, S.; Cattel, L. Preparation, characterization and properties in vitro and in vivo of a paclitaxel-albumin conjugate. J. Controlled Release 1997, 47, 293–304. [Google Scholar] [CrossRef]
- Bicamumpaka, C.; Page, M. In vitro cytotoxicity of paclitaxel-transferrin conjugate on H69 cells. Oncol. Rep. 1998, 5, 1381–1383. [Google Scholar]
- Dosio, F.; Arpicco, S.; Brusa, P.; Stella, B.; Cattel, L. Poly(ethylene glycol)-human serum albumin-paclitaxel conjugates: preparation, characterization and pharmacokinetics. J. Controlled Release 2001, 76, 107–117. [Google Scholar] [CrossRef]
- de Groot, F.M.H.; Albrecht, C.; Koekkoek, R.; Beusker, P.H.; Scheeren, H.W. "Cascade-release dendrimers" liberate all end groups upon a single triggering event in the dendritic core. Angew. Chem. Int. Edit. Engl. 2003, 42, 4490–4494. [Google Scholar] [CrossRef]
- Majoros, I.J.; Myc, A.; Thomas, T.; Mehta, C.B.; Baker, J.R. PAMAM dendrimer-based multifunctional conjugate for cancer therapy: synthesis, characterization, and functionality. Biomacromolecules 2006, 7, 572–579. [Google Scholar]
- Luo, Y.; Prestwich, G.D. Synthesis and selective cytotoxicity of a hyaluronic acid-antitumor bioconjugate. Bioconj. Chem. 1999, 10, 755–763. [Google Scholar] [CrossRef]
- Luo, Y.; Ziebell, M.R.; Prestwich, G.D. A Hyaluronic acid-taxol antitumor bioconjugate targeted to cancer cells. Biomacromol. 2000, 1, 208–218. [Google Scholar] [CrossRef]
- Marini Bettolo, R.; Migneco, L.M.; De Luca, G. Taxane covalently bounded to hyaluronic acid or hyaluronic acid derivatives. PCT Int. Appl. No. WO2004035629, 2004. [Google Scholar]
- Capodilupo, A.L.; Crescenzi, V.; Francescangeli, A.; Joudioux, R.; Leonelli, F.; Marini Bettolo, R.; Migneco, L.M.; Quagliariello, M.; Bellato, P.; De Luca, G.; Galbiati, E.; Renier, D.; Banzato, A.; Rosato, A. Hydrosoluble, Metabolically Fragile Bioconjugates By Coupling Tetrabutyl-ammonium Hyaluronan with 2’Paclitaxel-4-Bromobutyrate: Synthesis and Antitumor Properties. In “Hyaluronan 2003” Hyaluronan Structure, Metabolism, Biological Activities, Therapeutic Applications; Balazs, E.A., Hascall, V.C., Eds.; Matrix Biology Institute: New Jersey, 2005; Vol. I, pp. 391–395. [Google Scholar]
- Leonelli, F.; La Bella, A.; Francescangeli, A.; Joudioux, R.; Capodilupo, A.-L.; Quagliariello, M.; Migneco, L.M.; Marini Bettolo, R.; Crescenzi, V.; De Luca, G.; Renier, D. A new and simply available class of hydrosoluble bioconjugates by coupling paclitaxel to hyaluronic acid through a 4-hydroxybutanoic acid derived linker. Helv. Chim. Acta. 2005, 88, 154–159. [Google Scholar]
- Thierry, B.; Kujawa, P.; Tkaczyk, C.; Winnik, F.M.; Bilodeau, L.; Tabrizian, M. Delivery platform for hydrophobic drugs: prodrug approach combined with self assembled multilayers. J. Am. Chem. Soc. 2005, 127, 1626–1627. [Google Scholar]
- Maeda, H.; Seymour, L.W.; Miyamoto, Y. Conjugates of anticancer agents and polymers: advantages of macromolecular therapeutics in vivo. Bioconjug. Chem. 1992, 3, 351–362. [Google Scholar]
- Vincent, M.J.; Duncan, R. Polymer conjugates: nanosized medicines for treating cancer. Trends Biotechnol. 2006, 24, 39–47. [Google Scholar]
- Luo, Y.; Prestwich, G.D. Cancer targeted polymeric drugs. Curr. Cancer Drug Targets 2002, 2, 209–226. [Google Scholar]
- Mellado, W.; Magri, N.F.; Kingstone, D.G.I.; Garcia-Arenas, R.; Orr, G.A.; Horwitz, S.B. Preparation and biological activity of taxol acetates. Biochem. Biophys. Res. Commun. 1984, 124, 329–336. [Google Scholar]
- Magri, N.F.; Kingston, D.G.I. Modified taxols, 4. Synthesis and biological activity of taxols modified in the side chain. J. Nat. Prod. 1988, 51, 298–306. [Google Scholar] [CrossRef]
- Souto, A.A.; Acuna, A.U.; Andreu, J.M.; Barasoain, I.; Abal, M.; Amat-Guerri, F. New fluorescent water-soluble taxol derivatives. Angew. Chem. Int. Ed. Engl. 1996, 34, 2710–2712. [Google Scholar]
- Rose, W.C.; Clark, J.L.; Lee, F.Y.F.; Casazza, A.M. Preclinical antitumor activity of water-soluble paclitaxel derivatives. Cancer Chemother. Pharmacol. 1997, 39, 486–492. [Google Scholar]
- Harada, N.; Ozaki, K.; Yamaguchi, T.; Hiroaki, A.; Ando, A.; Oda, K.; Nakanishi, N.; Ohashi, M.; Hashiyama, T.; Tsujihara, K. Synthesis of taxoids II. Synthesis and antitumor activity of water-soluble taxoids. Heterocycles 1997, 46, 241–258. [Google Scholar] [CrossRef]
- Rao, C.S.; Chu, J.-J.; Liu, R.S.; Lai, Y.-K. ynthesis and evaluation of [14C]-labelled and fluorescent-tagged paclitaxel derivatives as new biological probes. Bioorg. Med. Chem. 1998, 6, 2193–2204. [Google Scholar]
- Bhat, L.; Liu, Y.; Victory, S.F.; Himes, R.H.; Georg, G.I. Synthesis and evaluation of paclitaxel C7 derivatives: solution phase synthesis of combinatorial libraries. Bioorg. Med. Chem. Lett. 1998, 8, 3181–3186. [Google Scholar]
- Jagtap, P.G.; Baloglu, E.; Barron, D.M.; Bane, S.; Kingston, D.G.I. Design and synthesis of a combinatorial chemistry library of 7-acyl, 10-acyl, and 7,10-diacyl analogues of paclitaxel (taxol) using solid phase synthesis. J. Nat. Prod. 2002, 65, 1136–1142. [Google Scholar] [CrossRef]
- El Alaoui, A.; Schmidt, F.; Monneret, C.; Florent, J.-C. Protecting groups for glucuronic acid: application to the synthesis of new paclitaxel (taxol) derivatives. J. Org. Chem. 2006, 71, 9628–9636. [Google Scholar]
- Forrest, M.L.; Yanez, J.A.; Remsberg, C.M.; Ohgami, Y.; Kwon, G.S.; Davies, NM. Paclitaxel prodrugs with sustained release and high solubility in poly(ethylene glycol)-b-poly(ε-caprolactone) micelle nanocarriers: pharmacokinetics disposition, tolerability, and cytotoxicity. Pharm. Res. 2008, 25, 194–206. [Google Scholar]
- Pouyani, T.; Prestwich, G.D. Functionalized derivatives of hyaluronic acid oligosaccharides-drug carriers and novel biomaterials. Bioconj. Chem. 1994, 5, 339–347. [Google Scholar] [CrossRef]
- Pouyani, T.; Harbison, G.S.; Prestwich, G.D. Novel hydrogels of hyaluronic acid: synthesis, surface morphology, and solid state NMR. J. Am. Chem. Soc. 1994, 116, 7515–7522. [Google Scholar] [CrossRef]
- Vercruysse, K.P.; Marecak, D.M.; Marecek, J.F.; Prestwich, G.D. Synthesis and in vitro degradation of new polyvalent hydrazide cross-linked hydrogels of hyaluronic acid. Bioconjug. Chem. 1997, 8, 686–694. [Google Scholar]
- Klostergaard and co-workers [] prepared bioconjugate 3 basically according to Prestwich and co-workers method [,], but using pH 8.5 in final coupling, which gives a better yield.
- Auzenne, E.; Ghosh, S.C.; Khodadadian, M.; Rivera, B.; Farquhar, D.; Price, R.E.; Ravoori, M.; Kundra, V.; Freedman, R.S.; Klostergaard, J. Hyaluronic acid-paclitaxel: antitumor efficacy against CD44(+) human ovarian carcinoma xenografts. Neoplasia 2007, 9, 479–486. [Google Scholar] [CrossRef]
- Rosato, A.; Banzato, A.; De Luca, G.; Renier, D.; Bettella, F.; Pagano, C.; Esposito, G.; Zanovello, P.; Bassi, P. HYTAD1-p20: A new paclitaxel-hyaluronic acid hydrosoluble bioconjugate for treatment of superficial bladder cancer. Urol. Oncol.-Semin. Ori. 2006, 24, 207–215. [Google Scholar] [CrossRef]
- Meléndez-Alafort, L.; Riondato, M.; Nadali, A.; Banzato, A.; Camporese, D.; Boccaccio, P.; Uzunov, N.; Rosato, A.; Mazzi, U. Bioavailability of 99mTc-Ha-paclitaxel complex [99mTc-ONCOFID-P] in mice using four different administration routes. J. Labelled Compd. Radiopharm. 2006, 49, 939–950. [Google Scholar]
- Sukhishvili, S.A. Layered, erasable polymer multilayers formed by hydrogen-bonded sequential self-assembly. Macromolecules 2002, 35, 301–310. [Google Scholar]
- Chung, A.J.; Rubner, M.F. Methods of loading and releasing low molecular weight cationic molecules in weak polyelecrolyte multilayer films. Langmuir 2002, 18, 1176–1183. [Google Scholar]
- Vázquez, E.; Dewitt, D.M.; Hammond, P.T.; Lynn, D.M. Construction of hydrolytically-degradable thin films via layer-by-layer deposition of degradable polyelectrolytes. J. Am. Chem. Soc. 2002, 124, 13992–13993. [Google Scholar]
- Hiller, J.; Rubner, M.F. Reversible molecular memory and pH-switchable swelling transitions in polyelectrolyte multilayers. Macromolecules 2003, 36, 4078–4083. [Google Scholar]
- Shchukin, D.G.; Patel, A.A.; Sukhorukov, G.B.; Lvov, Y.M. Nanoassembly of biodegradable microcapsules for DNA encasing. J. Am. Chem. Soc. 2004, 126, 3374–3375. [Google Scholar]
- Peyratout, C.S.; Dähne, L. Tailor-made polyelectrolyte microcapsules: from multilayers to smart containers. Angew. Chem. Int. Ed. 2004, 43, 3762–3783. [Google Scholar]
- Jackson, J.K.; Skinner, K.C.; Burgess, L.; Sun, T.; Hunter, W.L.; Burt, H.M. Paclitaxel-loaded crosslinked hyaluronic acid films for the prevention of postsurgical adhesions. Pharm. Res. 2002, 19, 411–417. [Google Scholar]
- Yin, D.; Ge, Z.; Yang, W.; Liu, C.; Yuan, Y. Inhibition of tumor metastasis in vivo by combination of paclitaxel and hyaluronic acid. Cancer Lett. 2006, 243, 71–79. [Google Scholar]
- Tezcaner, A.; Hicks, D.; Boulmedais, F.; Sahel, J.; Schaaf, P.; Voegel, J.-C.; Lavalle, P. Polyelectrolyte multilayer films as substrates for photoreceptor cells. Biomacromolecules 2006, 7, 86–94. [Google Scholar] [CrossRef]
- Vodouhê, C.; Schmittbuhl, M; Boulmedais, F.; Bagnard, D.; Vautier, D.; Schaaf, P.; Egles, C.; Voegel, J.-C.; Ogier, J. Effect of functionalization of multilayered polyelectrolyte films on motoneuron growth. Biomaterials 2005, 26, 545–554. [Google Scholar] [CrossRef]
- Jessel, N.; Atalar, F.; Lavalle, P.; Mutterer, J.; Decher, G.; Schaaf, P.; Voegel, J.-C.; Ogier, J. Bioactive coatings based on polyelectrolyte multilayer architecture functionalized by embedded proteins. Adv. Mater. 2003, 15, 692–695. [Google Scholar]
- Chluba, J.; Voegel, J.-C.; Decher, G.; Erbacher, P.; Schaaf, P.; Ogier, J. Peptide hormone covalently bound to polyelectrolytes and embedded into multilayer architectures conserving full biological activity. Biomacromolecules 2001, 2, 800–805. [Google Scholar]
- Caruso, F.; Schüler, C. Enzyme multilayers on colloid particles: assembly, stability, and enzymatic activity. Langmuir 2000, 16, 9595–9603. [Google Scholar]
- Caruso, F.; Niikura, K.; Furlong, D.N.; Okahata, Y. Assembly of alternating polyelectrolyte and protein multilayer films for immunosensing. Langmuir 1997, 13, 3427–3433. [Google Scholar] [CrossRef]
- Vodouhê, C.; Le Guen, E.; Garza, J.M.; Francius, G.; Déjugnat, C.; Ogier, J.; Schaaf, P.; Voegel, J.-C.; Lavalle, P. Control of drug accessibility on functional polyelectrolyte multilayer films. Biomaterials 2006, 27, 4149–4156. [Google Scholar]
- Kidambi, S.; Lee, I.; Chan, C. Controlling primary hepatocyte adhesion and spreading on protein-free polyelectrolyte multilayer films. J. Am. Chem. Soc. 2004, 126, 16286–16287. [Google Scholar]
© 2008 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.