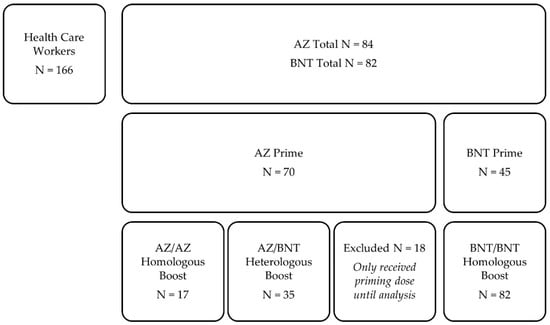
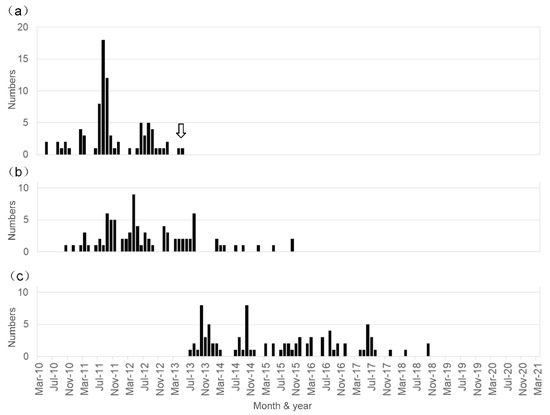
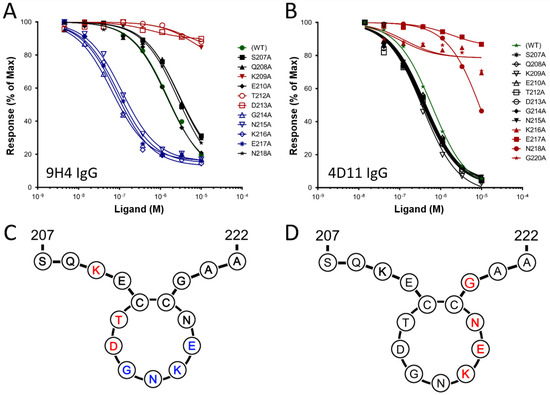
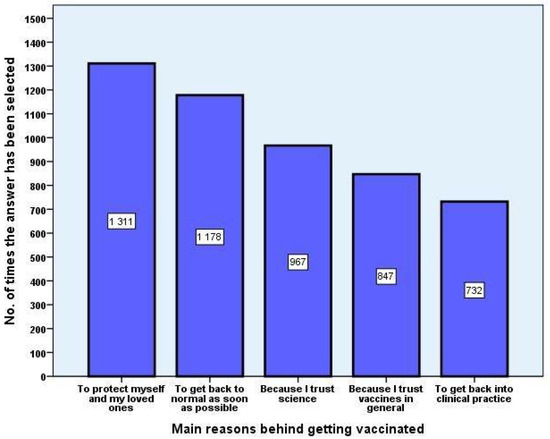
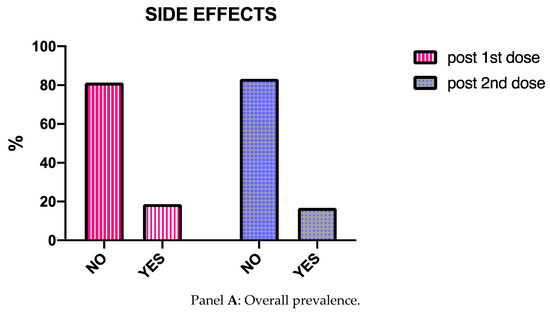
Vaccines 2021, 9(8), 864; https://doi.org/10.3390/vaccines9080864 - 5 Aug 2021
Cited by 62 | Viewed by 8427
Abstract
We examined the intention and predictors of accepting the COVID-19 vaccine in Saudi Arabia. We conducted a nation-wide, cross-sectional online survey between February and March 2021. A total of 1387 people (≥18 years) participated. Only 27.3% adults had a definite and 30.2% had
[...] Read more.
We examined the intention and predictors of accepting the COVID-19 vaccine in Saudi Arabia. We conducted a nation-wide, cross-sectional online survey between February and March 2021. A total of 1387 people (≥18 years) participated. Only 27.3% adults had a definite and 30.2% had a probable vaccination intent; 26.8% and 15.6% had a probable and definite negative vaccination intent. Older people (≥50 years) (p < 0.01), healthcare workers/professionals (p < 0.001), and those who received flu vaccine (p < 0.001) were more likely to have a positive intent. People from Riyadh were less likely to receive the vaccine (p < 0.05). Among the health belief model constructs, perceived susceptibility to and severity of COVID-19 (p < 0.001), and perceived benefit of the vaccine (p < 0.001) were positively associated with vaccination intent, whereas perceived barriers had a negative association (p < 0.001). Individuals were more likely to receive the vaccine after obtaining complete information (p < 0.001) and when the vaccine uptake would be more common amongst the public (p < 0.001).
Full article