Molecules 2012, 17(11), 12821-12835; https://doi.org/10.3390/molecules171112821 - 1 Nov 2012
Cited by 284 | Viewed by 19348
Abstract
The Kabachnik–Fields (phospha-Mannich) reaction involving the condensation of primary or secondary amines, oxo compounds (aldehydes and ketones) and >P(O)H species, especially dialkyl phosphites, represents a good choice for the synthesis of α-aminophosphonates that are of significant importance due to their biological activity. In
[...] Read more.
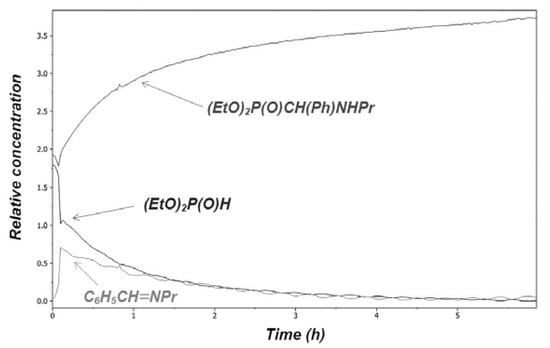
The Kabachnik–Fields (phospha-Mannich) reaction involving the condensation of primary or secondary amines, oxo compounds (aldehydes and ketones) and >P(O)H species, especially dialkyl phosphites, represents a good choice for the synthesis of α-aminophosphonates that are of significant importance due to their biological activity. In general, these three-component reactions may take place via an imine or an α-hydroxy-phosphonate intermediate. The monitoring of a few Kabachnik–Fields reactions by in situ Fourier transform IR spectroscopy has indicated the involvement of the imine intermediate that was also justified by theoretical calculations. The Kabachnik–Fields reaction was extended to >P(O)H species, comprising cyclic phosphites, acyclic and cyclic H-phosphinates, as well as secondary phosphine oxides. On the other hand, heterocyclic amines were also used to prepare new α-amino phosphonic, phosphinic and phosphine oxide derivatives. In most cases, the synthesis under solvent-free microwave (MW) conditions is the method of choice. It was proved that, in the cases studied by us, there was no need for the use of any catalyst. Moreover, it can be said that sophisticated and environmentally unfriendly catalysts suggested are completely unnecessary under MW conditions. Finally, the double Kabachnik–Fields reaction has made available bis(phosphonomethyl)amines, bis(phosphinoxidomethyl)amines and related species. The bis(phosphinoxidomethyl)amines serve as precursors for bisphosphines that furnish ring platinum complexes on reaction with dichlorodibenzonitriloplatinum.
Full article
(This article belongs to the Special Issue Organophosphorus Chemistry)
►
Show Figures