Target Therapies for Uterine Carcinosarcomas: Current Evidence and Future Perspectives
Abstract
:1. Introduction
2. Results
2.1. Genetic Landscape
2.2. Human Epidermal Growth Factor Family of Receptors (ErbB Family)
2.3. HER2
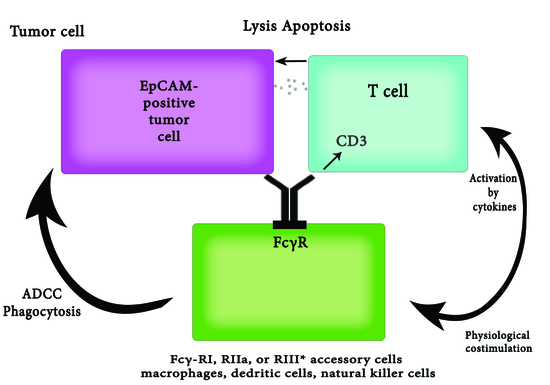
2.4. Epithelial Cell Adhesion Molecule-1 (EpCAM)
3. Discussion
4. Materials and Methods
4.1. Data Sources
4.2. Screening of Abstracts for Eligibility
4.3. Study Selection and Eligibility Criteria
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ADCC | Antibody-Dependent Cell-Mediated Cytotoxicity |
| AT | Adjuvant Therapy |
| CS | Carcinosarcoma |
| CSCs | Cancer Stem Cells |
| DM1 | Maytansinoid Cytotoxin |
| EMA | Epithelial Membrane Antigen |
| ErbB family | Human Epidermal Growth Factor Family of Receptors |
| FDA | Food and Drug Administration |
| FIGO | International Federation of Gynecology and Obstetrics |
| HER2 | Human Epidermal Growth Factor Receptor 2 |
| MMT | Malignant Mixed Müllerian Tumor |
| CD10 | Cluster of Differentiation 10 |
| EpCAM | Epithelial Cell Adhesion Molecule-1 |
| SEM | Standard Error of the Mean |
| T | Trastuzumab |
| T-DM1 | Trastuzumab Emtansine |
| VEGF | Vascular Endothelial Growth Factor |
| μM | Micromole |
| NK | Natural Killer |
| DCs | Dendritic Cells |
| TACE | Tumor Necrosis Factor-Alpha Converting Enzyme |
| EpICD | Epithelial Cell Intra-Cellular Domain |
| FHL2 | Four and a Half LIM Domain 2 |
| HLA-DR | Human Leukocyte Antigens D Related |
| TICs | Tumor Infiltrating Cells |
References
- D’Angelo, E.; Prat, J. Pathology of mixed Müllerian tumours. Best Pract. Res. Clin. Obstet. Gynaecol. 2011, 25, 705–718. [Google Scholar] [CrossRef] [PubMed]
- Nicoletti, R.; Lopez, S.; Bellone, S.; Cocco, E.; Schwab, C.L.; Black, J.D.; Centritto, F.; Zhu, L.; Bonazzoli, E.; Buza, N.; et al. T-DM1, a novel antibody-drug conjugate, is highly effective against uterine and ovarian carcinosarcomas overexpressing HER2. Clin. Exp. Metastasis 2015, 32, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Kounelis, S.; Jones, M.W.; Papadaki, H.; Bakker, A.; Swalsky, P.; Finkelstein, S.D. Carcinosarcomas (malignant mixed mullerian tumors) of the female genital tract: Comparative molecular analysis of epithelial and mesenchymal components. Hum. Pathol. 1998, 29, 82–87. [Google Scholar] [CrossRef]
- Brooks, S.E.; Zhan, M.; Cote, T.; Baquet, C.R. Surveillance, epidemiology, and end results analysis of 2677 cases of uterine sarcoma 1989–1999. Gynecol. Oncol. 2004, 93, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, M.; Villena-Heinsen, C.; Mink, D.; Bonkhoff, H.; Schmidt, W. Carcinosarcoma, endometrial intraepithelial carcinoma and endometriosis after tamoxifen therapy in breast cancer. Eur. J. Obstet. Gynecol. Reprod. Biol. 1999, 82, 85–87. [Google Scholar] [CrossRef]
- Evans, M.J.; Langlois, N.E.; Kitchener, H.C.; Miller, I.D. Is there an association between long-term tamoxifen treatment and the development of carcinosarcoma (malignant mixed Müllerian tumor) of the uterus? Int. J. Gynecol. Cancer 1995, 5, 310–313. [Google Scholar] [CrossRef] [PubMed]
- Wickerham, D.L.; Fisher, B.; Wolmark, N.; Bryant, J.; Costantino, J.; Bernstein, L.; Runowicz, C.D. Association of tamoxifen and uterine sarcoma. J. Clin. Oncol. 2002, 20, 2758–2760. [Google Scholar] [CrossRef] [PubMed]
- Pothuri, B.; Ramondetta, L.; Eifel, P.; Deavers, M.T.; Wilton, A.; Alektiar, K.; Barakat, R.; Soslow, R.A. Radiation-associated endometrial cancers are prognostically unfavorable tumors: A clinicopathologic comparison with 527 sporadic endometrial cancers. Gynecol. Oncol. 2006, 103, 948–951. [Google Scholar] [CrossRef] [PubMed]
- Hagiwara, T.; Mori, T.; Kaku, T. Development of endometrial cancer following radiation therapy for cervical carcinoma. Eur. J. Gynaecol. Oncol. 2005, 26, 191–195. [Google Scholar] [PubMed]
- Callister, M.; Ramondetta, L.M.; Jhingran, A.; Burke, T.W.; Eifel, P.J. Malignant mixed Müllerian tumors of the uterus: Analysis of patterns of failure, prognostic factors, and treatment outcome. Int. J. Radiat. Oncol. Biol. Phys. 2004, 58, 786–796. [Google Scholar] [CrossRef]
- Salazar, O.M.; Bonfiglio, T.A.; Patten, S.F.; Keller, B.E.; Feldstein, M.; Dunne, M.E.; Rudolph, J. Uterine sarcomas: Natural history, treatment and prognosis. Cancer 1978, 42, 1152–1160. [Google Scholar] [CrossRef]
- NCCN Guidelines Version 3.2012—Endometrial Carcinoma. Available online: http://www.nccn.org/professionals/physician_gls/pdf/uterine.pdf (accessed on 19 September 2012).
- Prat, J. FIGO staging for uterine sarcomas. Int. J. Gynaecol. Obstet. 2009, 104, 177–178. [Google Scholar] [CrossRef] [PubMed]
- Galaal, K.; Godfrey, K.; Naik, R.; Kucukmetin, A.; Bryant, A. Adjuvant radiotherapy and/or chemotherapy after surgery for uterine carcinosarcoma. Cochrane Database Syst. Rev. 2011, 1. [Google Scholar] [CrossRef]
- Garg, G.; Shah, J.P.; Kumar, S.; Bryant, C.S.; Munkarah, A.; Morris, R.T. Ovarian and uterine carcinosarcomas: A comparative analysis of prognostic variables and survival outcomes. Int. J. Gynecol. Cancer 2010, 20, 888–894. [Google Scholar] [CrossRef] [PubMed]
- Cantrell, L.A.; van Le, L. Carcinosarcoma of the ovary a review. Obstet. Gynecol. Surv. 2009, 64, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Inthasorn, P.; Beale, P.; Dalrymple, C.; Carter, J. Malignant mixed Mullerian tumour of the ovary: Prognostic factor and response of adjuvant platinum-based chemotherapy. Aust. N. Z. J. Obstet. Gynaecol. 2003, 43, 61–64. [Google Scholar] [CrossRef] [PubMed]
- McCluggage, W.G. Uterine carcinosarcomas (malignant mixed Mullerian tumors) are metaplastic carcinomas. Int. J. Gynecol. Cancer 2002, 12, 687–690. [Google Scholar] [CrossRef] [PubMed]
- McCluggage, W.G. Malignant biphasic uterine tumours: Carcinosarcomas or metaplastic carcinomas? J. Clin. Pathol. 2002, 55, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Vitale, S.G.; Padula, F.; Gulino, F.A. Management of uterine fibroids in pregnancy: Recent trends. Curr. Opin. Obstet. Gynecol. 2015, 27, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Vitale, S.G.; Rossetti, D.; Tropea, A.; Biondi, A.; Laganà, A.S. Fertility sparing surgery for stage IA type I and G2 endometrial cancer in reproductive-aged patients: Evidence-based approach and future perspectives. Updates Surg. 2017, 69, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Cignini, P.; Vitale, S.G.; Laganà, A.S.; Biondi, A.; La Rosa, V.L.; Cutillo, G. Preoperative work-up for definition of lymph node risk involvement in early stage endometrial cancer: 5-Year follow-up. Updates Surg. 2017, 69, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Cianci, S.; Vitale, S.G.; Tozzi, R.; Cignini, P.; Padula, F.; D’Emidio, L.; Mangiafico, L.; Giorlandino, C.; Frigerio, L.; Rossetti, D.; et al. Tubal primary metastatic choriocarcinoma coexistent with a viable early pregnancy: A case report. J. Prenat. Med. 2014, 8, 47–49. [Google Scholar] [PubMed]
- Rossetti, D.; Bogani, G.; Carnelli, M.; Vitale, S.G.; Grosso, G.; Frigerio, L. Efficacy of IVF following conservative management of endometrial cancer. Gynecol. Endocrinol. 2014, 30, 280–281. [Google Scholar] [CrossRef] [PubMed]
- Carnero, A.; Lleonart, M. The hypoxic microenvironment: A determinant of cancer stem cell evolution. Bioessays 2016, 38, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, D.; Dick, J.E. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat. Med. 1997, 3, 730–737. [Google Scholar] [CrossRef] [PubMed]
- Basu-Roy, U.; Basilico, C.; Mansukhani, A. Perspectives on cancer stem cells in osteosarcoma. Cancer Lett. 2013, 338, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Bellone, S.; Lopez, S.; Thakral, D.; Schwab, C.; English, D.P.; Black, J.; Cocco, E.; Choi, J.; Zammataro, L.; et al. Mutational landscape of uterine and ovarian carcinosarcomas implicates histone genes in epithelial-mesenchymal transition. Proc. Natl. Acad. Sci. USA 2016, 113, 12238–12243. [Google Scholar] [CrossRef] [PubMed]
- Halldorsson, A.; Brooks, S.; Montgomery, S.; Graham, S. Lung metastasis 21 years after initial diagnosis of osteosarcoma: A case report. J. Med. Case Rep. 2009, 3, 9298. [Google Scholar] [CrossRef] [PubMed]
- Basu-Roy, U.; Bayin, N.S.; Rattanakorn, K.; Han, E.; Placantonakis, D.G.; Mansukhani, A.; Basilico, C. Sox2 antagonizes the Hippo pathway to maintain stemness in cancer cells. Nat. Commun. 2015, 6, 6411. [Google Scholar] [CrossRef] [PubMed]
- Shiozawa, Y.; Berry, J.E.; Eber, M.R.; Jung, Y.; Yumoto, K.; Cackowski, F.C.; Yoon, H.J.; Parsana, P.; Mehra, R.; Wang, J.; et al. The marrow niche controls the cancer stem cell phenotype of disseminated prostate cancer. Oncotarget 2016, 7, 41217–41232. [Google Scholar] [CrossRef] [PubMed]
- Heymann, D.; Rédini, F. Bone sarcomas: Pathogenesis and new therapeutic approaches. IBMS BoneKEy 2011, 8, 402–414. [Google Scholar] [CrossRef]
- Ferrari, F.; Bellone, S.; Black, J.; Schwab, C.L.; Lopez, S.; Cocco, E.; Bonazzoli, E.; Predolini, F.; Menderes, G.; Litkouhi, B.; et al. Solitomab, an EpCAM/CD3 bispecific antibody construct (BiTE®), is highly active against primary uterine and ovarian carcinosarcoma cell lines in vitro. J. Exp. Clin. Cancer Res. 2015, 34, 123. [Google Scholar] [CrossRef] [PubMed]
- Livasy, C.A.; Reading, F.C.; Moore, D.T.; Boggess, J.F.; Lininger, R.A. EGFR expression and HER2/neu overexpression/amplification in endometrial carcinosarcoma. Gynecol. Oncol. 2006, 100, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Amant, F.; Vloeberghs, V.; Woestenborghs, H.; Debiec-Rychter, M.; Verbist, L.; Moerman, P.; Vergote, I. ERBB-2 gene overexpression and amplification in uterine sarcomas. Gynecol. Oncol. 2004, 95, 583–587. [Google Scholar] [CrossRef] [PubMed]
- Gabos, Z.; Sinha, R.; Hanson, J.; Chauhan, N.; Hugh, J.; Mackey, J.R.; Abdulkarim, B. Prognostic significance of human epidermal growth factor receptor positivity for the development of brain metastasis after newly diagnosed breast cancer. J. Clin. Oncol. 2006, 24, 5658–5663. [Google Scholar] [CrossRef] [PubMed]
- Cobleigh, M.A.; Vogel, C.L.; Tripathy, D.; Robert, N.J.; Scholl, S.; Fehrenbacher, L.; Wolter, J.M.; Paton, V.; Shak, S.; Lieberman, G.; et al. Multinational study of the efficacy and safety of humanized anti-HER2 monoclonal antibody in women who have HER2-overexpressing metastatic breast cancer that has progressed after chemotherapy for metastatic disease. J. Clin. Oncol. 1999, 17, 2639–2648. [Google Scholar] [CrossRef] [PubMed]
- Boyraz, B.; Sendur, M.A.; Aksoy, S.; Babacan, T.; Roach, E.C.; Kizilarslanoglu, M.C.; Petekkaya, I.; Altundag, K. Trastuzumab emtansine (T-DM1) for HER2-positive breast cancer. Curr. Med. Res. Opin. 2013, 29, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Lewis Phillips, G.D.; Li, G.; Dugger, D.L.; Crocker, L.M.; Parsons, K.L.; Mai, E.; Blättler, W.A.; Lambert, J.M.; Chari, R.V.; Lutz, R.J.; et al. Targeting HER2-positive breast cancer with trastuzumab-DM1, an antibody-cytotoxic drug conjugate. Cancer Res. 2008, 68, 9280–9290. [Google Scholar] [CrossRef] [PubMed]
- Guzzo, F.; Bellone, S.; Buza, N.; Hui, P.; Carrara, L.; Varughese, J.; Cocco, E.; Betti, M.; Todeschini, P.; Gasparrini, S.; et al. HER2/neu as a potential target for immunotherapy in gynecologic carcinosarcomas. Int. J. Gynecol. Pathol. 2012, 31, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Fabi, A.; Malaguti, P.; Vari, S.; Cognetti, F. First-line therapy in HER2 positive metastatic breast cancer: Is the mosaic fully completed or are we missing additional pieces? J. Exp. Clin. Cancer Res. 2016, 35, 104. [Google Scholar] [CrossRef] [PubMed]
- Preston, C.C.; Goode, E.L.; Hartmann, L.C.; Kalli, K.R.; Knutson, K.L. Immunity and immune suppression in human ovarian cancer. Immunotherapy 2011, 3, 539–556. [Google Scholar] [CrossRef] [PubMed]
- Boku, N. HER2-positive gastric cancer. Gastric Cancer 2014, 17, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Raspollini, M.R.; Susini, T.; Amunni, G.; Paglierani, M.; Taddei, A.; Marchionni, M.; Scarselli, G.; Taddei, G.L. COX-2, c-KIT and HER-2/neu expression in uterine carcinosarcomas: Prognostic factors or potential markers for targeted therapies? Gynecol. Oncol. 2005, 96, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Sawada, M.; Tsuda, H.; Kimura, M.; Okamoto, S.; Kita, T.; Kasamatsu, T.; Yamada, T.; Kikuchi, Y.; Honjo, H.; Matsubara, O. Different expression patterns of KIT, EGFR, and HER-2 (c-erbB-2) oncoproteins between epithelial and mesenchymal components in uterine carcinosarcoma. Cancer Sci. 2003, 94, 986–991. [Google Scholar] [CrossRef] [PubMed]
- Park, J.W.; Neve, R.M.; Szollosi, J.; Benz, C.C. Unraveling the biologic and clinical complexities of HER2. Clin. Breast Cancer 2008, 8, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Tai, W.; Mahato, R.; Cheng, K. The role of HER2 in cancer therapy and targeted drug delivery. J. Control Release 2010, 146, 264–275. [Google Scholar] [CrossRef] [PubMed]
- Yan, M.; Parker, B.A.; Schwab, R.; Kurzrock, R. HER2 aberrations in cancer: Implications for therapy. Cancer Treat Rev. 2014, 40, 770–780. [Google Scholar] [CrossRef] [PubMed]
- Santin, A.D.; Bellone, S.; van Stedum, S.; Bushen, W.; Palmieri, M.; Siegel, E.R.; de Las Casas, L.E.; Roman, J.J.; Burnett, A.; Pecorelli, S. Amplification of c-erbB2 oncogene: A major prognostic indicator in uterine serous papillary carcinoma. Cancer 2005, 104, 1391–1397. [Google Scholar] [CrossRef] [PubMed]
- El-Sahwi, K.; Bellone, S.; Cocco, E.; Cargnelutti, M.; Casagrande, F.; Bellone, M.; Abu-Khalaf, M.; Buza, N.; Tavassoli, F.A.; Hui, P.; et al. In vitro activity of pertuzumab in combination with trastuzumab in uterine serous papillary adenocarcinoma. Br. J. Cancer 2010, 102, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Cimbaluk, D.; Rotmensch, J.; Scudiere, J.; Gown, A.; Bitterman, P. Uterine carcinosarcoma: Immunohistochemical studies on tissue microarrays with focus on potential therapeutic targets. Gynecol. Oncol. 2007, 105, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Schwab, C.L.; English, D.P.; Roque, D.M.; Bellone, S.; Lopez, S.; Cocco, E.; Nicoletti, R.; Rutherford, T.J.; Schwartz, P.E.; Santin, A.D. Neratinib shows efficacy in the treatment of HER2/neu amplified uterine serous carcinoma in vitro and in vivo. Gynecol. Oncol. 2014, 135, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Choijamts, B.; Jimi, S.; Kondo, T.; Naganuma, Y.; Matsumoto, T.; Kuroki, M.; Iwasaki, H.; Emoto, M. CD133+ cancer stem cell-like cells derived from uterine carcinosarcoma (malignant mixed Müllerian tumor). Stem Cells 2011, 29, 1485–1495. [Google Scholar] [CrossRef] [PubMed]
- Herlyn, M.; Steplewski, Z.; Herlyn, D.; Koprowski, H. Colorectal carcinoma-specific antigen: Detection by means of monoclonal antibodies. Proc. Natl. Acad. Sci. USA 1979, 76, 1438–1442. [Google Scholar] [CrossRef] [PubMed]
- Riethmüller, G.; Holz, E.; Schlimok, G.; Schmiegel, W.; Raab, R.; Höffken, K.; Gruber, R.; Funke, I.; Pichlmaier, H.; Hirche, H.; et al. Monoclonal antibody therapy for resected Dukes’ C colorectal cancer: Seven-year outcome of a multicenter randomized trial. J. Clin. Oncol. 1998, 16, 1788–1794. [Google Scholar] [CrossRef] [PubMed]
- Riethmüller, G.; Schneider-Gädicke, E.; Schlimok, G.; Schmiegel, W.; Raab, R.; Höffken, K.; Gruber, R.; Pichlmaier, H.; Hirche, H.; Pichlmayr, R.; et al. Randomised trial of monoclonal antibody for adjuvant therapy of resected Dukes’ C colorectal carcinoma. Lancet 1994, 343, 1177–1183. [Google Scholar] [CrossRef]
- Litvinov, S.V.; Balzar, M.; Winter, M.J.; Bakker, H.A.; Briaire-de Bruijn, I.H.; Prins, F.; Fleuren, G.J.; Warnaar, S.O. Epithelial cell adhesion molecule (Ep-CAM) modulates cell-cell interactions mediated by classic cadherins. J. Cell Biol. 1997, 139, 1337–1348. [Google Scholar] [CrossRef] [PubMed]
- Litvinov, S.V.; van Driel, W.; van Rhijn, C.M.; Bakker, H.A.; van Krieken, H.; Fleuren, G.J.; Warnaar, S.O. Expression of Ep-CAM in cervical squamous epithelia correlates with an increased proliferation and the disappearance of markers for terminal differentiation. Am. J. Pathol. 1996, 148, 865–875. [Google Scholar] [PubMed]
- Litvinov, S.V.; Velders, M.P.; Bakker, H.A.; Fleuren, G.J.; Warnaar, S.O. Ep-CAM: A human epithelial antigen is a homophilic cell–cell adhesion molecule. J. Cell Biol. 1994, 125, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Fields, A.L.; Keller, A.; Schwartzberg, L.; Bernard, S.; Kardinal, C.; Cohen, A.; Schulz, J.; Eisenberg, P.; Forster, J.; Wissel, P. Adjuvant therapy with the monoclonal antibody Edrecolomab plus fluorouracil-based therapy does not improve overall survival of patients with stage III colon cancer. J. Clin. Oncol. 2009, 27, 1941–1947. [Google Scholar] [CrossRef] [PubMed]
- Schmoll, H.J.; Arnold, D. When wishful thinking leads to a misty-eyed appraisal: The story of the adjuvant colon cancer trials with edrecolomab. J. Clin. Oncol. 2009, 27, 1926–1929. [Google Scholar] [CrossRef] [PubMed]
- Gires, O.; Baeuerle, P.A. EpCAM as a target in cancer therapy. J. Clin. Oncol. 2010, 28, e239–e240. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Scheulen, M.E.; Dittrich, C.; Obrist, P.; Marschner, N.; Dirix, L.; Schmidt, M.; Rüttinger, D.; Schuler, M.; Reinhardt, C.; et al. An open-label, randomized phase II study of adecatumumab, a fully human anti-EpCAM antibody, as monotherapy in patients with metastatic breast cancer. Ann. Oncol. 2010, 21, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Münz, M.; Murr, A.; Kvesic, M.; Rau, D.; Mangold, S.; Pflanz, S.; Lumsden, J.; Volkland, J.; Fagerberg, J.; Riethmüller, G.; et al. Side-by-side analysis of five clinically tested anti-EpCAM monoclonal antibodies. Cancer Cell Int. 2010, 10, 44. [Google Scholar] [CrossRef] [PubMed]
- Spizzo, G.; Went, P.; Dirnhofer, S.; Obrist, P.; Moch, H.; Baeuerle, P.A.; Mueller-Holzner, E.; Marth, C.; Gastl, G.; Zeimet, A.G. Overexpression of epithelial cell adhesion molecule (Ep-CAM) is an independent prognostic marker for reduced survival of patients with epithelial ovarian cancer. Gynecol. Oncol. 2006, 103, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Spizzo, G.; Went, P.; Dirnhofer, S.; Obrist, P.; Simon, R.; Spichtin, H.; Maurer, R.; Metzger, U.; von Castelberg, B.; Bart, R.; et al. High Ep-CAM expression is associated with poor prognosis in node-positive breast cancer. Breast Cancer Res. Treat. 2004, 86, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Benko, G.; Spajić, B.; Krušlin, B.; Tomas, D. Impact of the EpCAM expression on biochemical recurrence-free survival in clinically localized prostate cancer. Urol. Oncol. 2013, 31, 468–474. [Google Scholar] [CrossRef] [PubMed]
- Akita, H.; Nagano, H.; Takeda, Y.; Eguchi, H.; Wada, H.; Kobayashi, S.; Marubashi, S.; Tanemura, M.; Takahashi, H.; Ohigashi, H.; et al. Ep-CAM is a significant prognostic factor in pancreatic cancer patients by suppressing cell activity. Oncogene 2011, 30, 3468–3476. [Google Scholar] [CrossRef] [PubMed]
- Pietzner, K.; Woopen, H.; Richter, R.; Joens, T.; Braicu, E.I.; Dimitrova, D.; Mellstedt, H.; Darb-Esfahani, S.; Denkert, C.; Lindhofer, H.; et al. Expression of epithelial cell adhesion molecule in paired tumor samples of patients with primary and recurrent serous ovarian cancer. Int. J. Gynecol. Cancer 2013, 23, 797–802. [Google Scholar] [CrossRef] [PubMed]
- Munz, M.; Baeuerle, P.A.; Gires, O. The emerging role of EpCAM in cancer and stem cell signaling. Cancer Res. 2009, 69, 5627–5629. [Google Scholar] [CrossRef] [PubMed]
- Seimetz, D.; Lindhofer, H.; Bokemeyer, C. Development and approval of the trifunctional antibody catumaxomab (anti-EpCAM × anti-CD3) as a targeted cancer immunotherapy. Cancer Treat. Rev. 2010, 36, 458–467. [Google Scholar] [CrossRef] [PubMed]
- Ott, M.G.; Marmé, F.; Moldenhauer, G.; Lindhofer, H.; Hennig, M.; Spannagl, R.; Essing, M.M.; Linke, R.; Seimetz, D. Humoral response to catumaxomab correlates with clinical outcome: Results of the pivotal phase II/III study in patients with malignant ascites. Int. J. Cancer 2012, 130, 2195–2203. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, I.; Baeuerle, P.A.; Friedrich, M.; Murr, A.; Filusch, S.; Rüttinger, D.; Majdoub, M.W.; Sharma, S.; Kufer, P.; Raum, T.; et al. Highly efficient elimination of colorectal tumor-initiating cells by an EpCAM/CD3-bispecific antibody engaging human T cells. PLoS ONE 2010, 5, e13474. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, A.; Eck, S.L. EpCAM: A new therapeutic target for an old cancer antigen. Cancer Biol. Ther. 2003, 2, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Pauli, C.; Münz, M.; Kieu, C.; Mack, B.; Breinl, P.; Wollenberg, B.; Lang, S.; Zeidler, R.; Gires, O. Tumor-specific glycosylation of the carcinoma-associated epithelial cell adhesion molecule EpCAM in head and neck carcinomas. Cancer Lett. 2003, 193, 25–32. [Google Scholar] [CrossRef]
- Malanchi, I.; Huelsken, J. Cancer stem cells: Never Wnt away from the niche. Curr. Opin. Oncol. 2009, 21, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Shigdar, S.; Lin, J.; Yu, Y.; Pastuovic, M.; Wei, M.; Duan, W. RNA aptamer against a cancer stem cell marker epithelial cell adhesion molecule. Cancer Sci. 2011, 102, 991–998. [Google Scholar] [CrossRef] [PubMed]
- Brischwein, K.; Schlereth, B.; Guller, B.; Steiger, C.; Wolf, A.; Lutterbuese, R.; Offner, S.; Locher, M.; Urbig, T.; Raum, T.; et al. MT110: A novel bispecific single-chain antibody construct with high efficacy in eradicating established tumors. Mol. Immunol. 2006, 43, 1129–1143. [Google Scholar] [CrossRef] [PubMed]
- Schipf, A.; Mayr, D.; Kirchner, T.; Diebold, J. Molecular genetic aberrations of ovarian and uterine carcinosarcomas—A CGH and FISH study. Virchows Arch. 2008, 452, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Laganà, A.S.; Colonese, F.; Colonese, E.; Sofo, V.; Salmeri, F.M.; Granese, R.; Chiofalo, B.; Ciancimino, L.; Triolo, O. Cytogenetic analysis of epithelial ovarian cancer’s stem cells: An overview on new diagnostic and therapeutic perspectives. Eur. J. Gynaecol. Oncol. 2015, 36, 495–505. [Google Scholar] [PubMed]
- Laganà, A.S.; Sofo, V.; Vitale, S.G.; Triolo, O. Epithelial ovarian cancer inherent resistance: May the pleiotropic interaction between reduced immunosurveillance and drug-resistant cells play a key role? Gynecol. Oncol. Rep. 2016, 18, 57–58. [Google Scholar] [CrossRef] [PubMed]
- Bellia, A.; Vitale, S.G.; Laganà, A.S.; Cannone, F.; Houvenaeghel, G.; Rua, S.; Ladaique, A.; Jauffret, C.; Ettore, G.; Lambaudie, E. Feasibility and surgical outcomes of conventional and robot-assisted laparoscopy for early-stage ovarian cancer: A retrospective, multicenter analysis. Arch. Gynecol. Obstet. 2016, 294, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Vitale, S.G.; Marilli, I.; Lodato, M.; Tropea, A.; Cianci, A. The role of cytoreductive surgery in advanced-stage ovarian cancer: A systematic review. Updates Surg. 2013, 65, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Rossetti, D.; Vitale, S.G.; Gulino, F.A.; Rapisarda, A.M.C.; Valenti, G.; Zigarelli, M.; Sarpietro, G.; Frigerio, L. Laparoendoscopic single-site surgery for the assessment of peritoneal carcinomatosis resectability in patients with advanced ovarian cancer. Eur. J. Gynaecol. Oncol. 2016, 37, 671–673. [Google Scholar]
- Vitale, S.G.; La Rosa, V.L.; Rapisarda, A.M.; Laganà, A.S. Comment on: “Anxiety and depression in patients with advanced ovarian cancer: A prospective study”. J. Psychosom. Obstet. Gynaecol. 2017, 38, 83–84. [Google Scholar] [CrossRef] [PubMed]
- Vitale, S.G.; Valenti, G.; Gulino, F.A.; Cignini, P.; Biondi, A. Surgical treatment of high stage endometrial cancer: Current perspectives. Updates Surg. 2016, 68, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Rossetti, D.; Vitale, S.G.; Gulino, F.A.; Cignini, P.; Rapisarda, A.M.C.; Biondi, A.; Frigerio, L. Concomitant chemoradiation treatment in selected stage I endometrioid endometrial cancers. Eur. J. Gynaecol. Oncol. 2016, 37, 657–661. [Google Scholar]
- Bentivegna, E.; Gouy, S.; Maulard, A.; Chargari, C.; Leary, A.; Morice, P. Oncological outcomes after fertility-sparing surgery for cervical cancer: A systematic review. Lancet Oncol. 2016, 17, 240–253. [Google Scholar] [CrossRef]
- Laganà, A.S.; Vitale, S.G.; La Rosa, V.L.; Rapisarda, A.M. Comment on: “Needs and priorities of women with endometrial and cervical cancer”. J. Psychosom. Obstet. Gynaecol. 2017, 38, 85–86. [Google Scholar] [CrossRef] [PubMed]
- De Hullu, J.A.; van der Avoort, I.A.; Oonk, M.H.; van der Zee, A.G. Management of vulvar cancers. Eur. J. Surg. Oncol. 2006, 32, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Vitale, S.G.; Valenti, G.; Biondi, A.; Rossetti, D.; Frigerio, L. Recent trends in surgical and reconstructive management of vulvar cancer: Review of literature. Updates Surg. 2015, 67, 367–371. [Google Scholar] [CrossRef] [PubMed]
- JAMA. Julius Cohnheim (1839–1884) experimental pathologist. JAMA 1968, 206, 1561–1562. [Google Scholar]
- Gupta, P.B.; Chaffer, C.L.; Weinberg, R.A. Cancer stem cells: Mirage or reality? Nat. Med. 2009, 15, 1010–1012. [Google Scholar] [CrossRef] [PubMed]
- Gorai, I.; Yanagibashi, T.; Taki, A.; Udagawa, K.; Miyagi, E.; Nakazawa, T.; Hirahara, F.; Nagashima, Y.; Minaguchi, H. Uterine carcinosarcoma is derived from a single stem cell: An in vitro study. Int. J. Cancer 1997, 72, 821–827. [Google Scholar] [CrossRef]
- Al-Hajj, M.; Wicha, M.S.; Benito-Hernandez, A.; Morrison, S.J.; Clarke, M.F. Prospective identification of tumorigenic breast cancer cells. Proc. Natl. Acad. Sci. USA 2003, 100, 3983–3988. [Google Scholar] [CrossRef] [PubMed]
- Rutella, S.; Bonanno, G.; Procoli, A.; Mariotti, A.; Corallo, M.; Prisco, M.G.; Eramo, A.; Napoletano, C.; Gallo, D.; Perillo, A.; et al. Cells with characteristics of cancer stem/progenitor cells express the CD133 antigen in human endometrial tumors. Clin. Cancer Res. 2009, 15, 4299–4311. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Maruyama, T.; Masuda, H.; Kajitani, T.; Nagashima, T.; Arase, T.; Ito, M.; Ohta, K.; Uchida, H.; Asada, H.; et al. Side population in human uterine myometrium displays phenotypic and functional characteristics of myometrial stem cells. Proc. Natl. Acad. Sci. USA 2007, 104, 18700–18705. [Google Scholar] [CrossRef] [PubMed]
- Chaffer, C.L.; Weinberg, R.A. A perspective on cancer cell metastasis. Science 2011, 331, 1559–1564. [Google Scholar] [CrossRef] [PubMed]
- He, Y.C.; Zhou, F.L.; Shen, Y.; Liao, D.F.; Cao, D. Apoptotic death of cancer stem cells for cancer therapy. Int. J. Mol. Sci. 2014, 15, 8335–8351. [Google Scholar] [CrossRef] [PubMed]
- Dawood, S.; Austin, L.; Cristofanilli, M. Cancer stem cells: Implications for cancer therapy. Oncology (Williston Park) 2014, 28, 1101–1107. [Google Scholar] [PubMed]
- Reya, T.; Morrison, S.J.; Clarke, M.F.; Weissman, I.L. Stem cells, cancer, and cancer stem cells. Nature 2001, 414, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Visvader, J.E.; Lindeman, G.J. Cancer stem cells in solid tumours: Accumulating evidence and unresolved questions. Nat. Rev. Cancer 2008, 8, 755–768. [Google Scholar] [CrossRef] [PubMed]
- Akunuru, S.; James Zhai, Q.; Zheng, Y. Non-small cell lung cancer stem/progenitor cells are enriched in multiple distinct phenotypic subpopulations and exhibit plasticity. Cell Death Dis. 2012, 3, e352. [Google Scholar] [CrossRef] [PubMed]
- Vermeulen, L.; Felipe de Sousa, E.M.; van der Heijden, M.; Cameron, K.; de Jong, J.H.; Borovski, T.; Tuynman, J.B.; Todaro, M.; Merz, C.; Rodermond, H.; et al. Wnt activity defines colon cancer stem cells and is regulated by the microenvironment. Nat. Cell Biol. 2010, 12, 468–476. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.J.; Li, B.; Winer, J.; Armanini, M.; Gillett, N.; Phillips, H.S.; Ferrara, N. Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumour growth in vivo. Nature 1993, 362, 841–844. [Google Scholar] [CrossRef] [PubMed]
- Hurwitz, H.; Fehrenbacher, L.; Novotny, W.; Cartwright, T.; Hainsworth, J.; Heim, W.; Berlin, J.; Baron, A.; Griffing, S.; Holmgren, E.; et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N. Engl. J. Med. 2004, 350, 2335–2342. [Google Scholar] [CrossRef] [PubMed]
- Sandler, A.; Gray, R.; Perry, M.C.; Brahmer, J.; Schiller, J.H.; Dowlati, A.; Lilenbaum, R.; Johnson, D.H. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N. Engl. J. Med. 2006, 355, 2542–2550. [Google Scholar] [CrossRef] [PubMed]

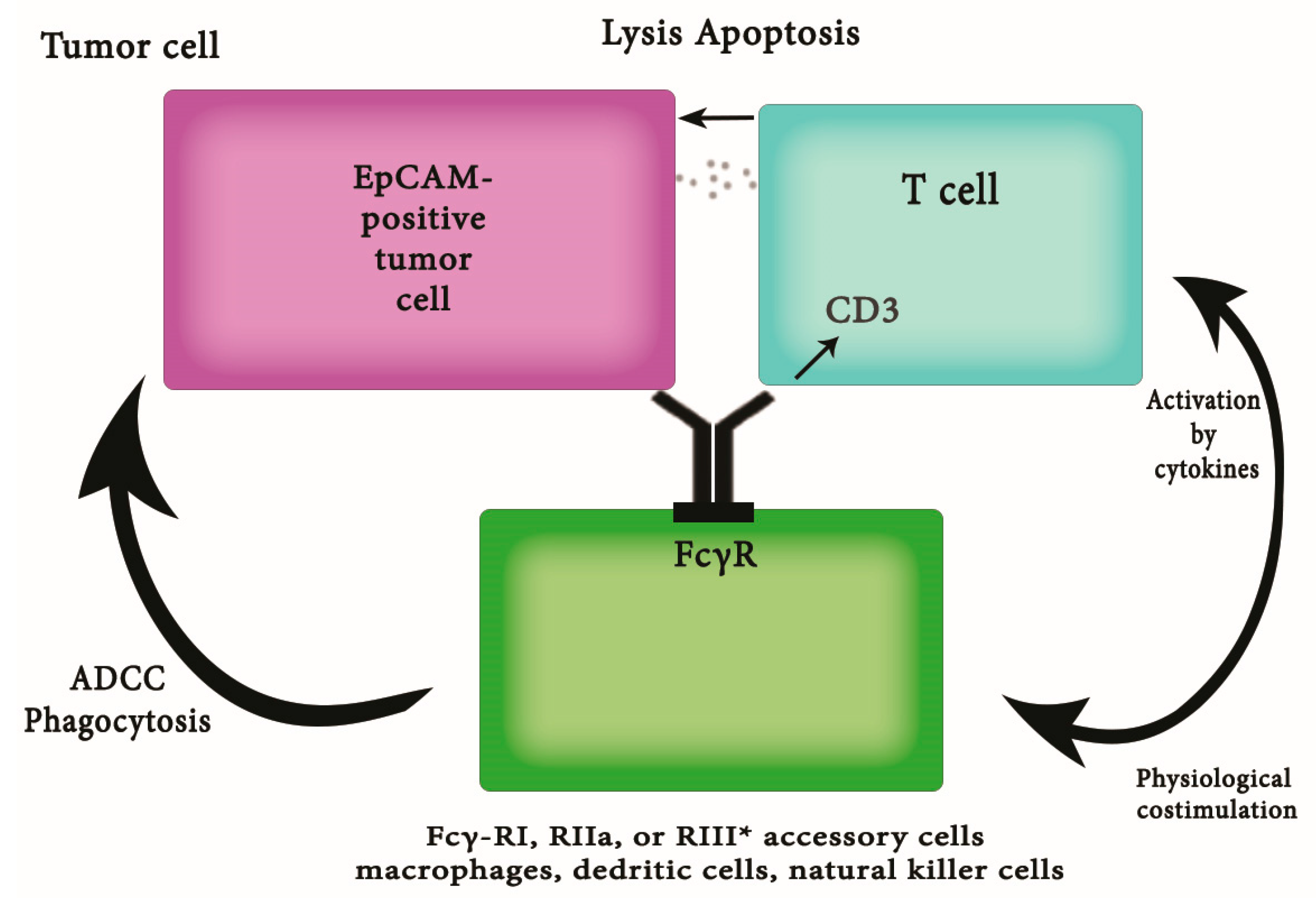
| 30 Potentially Citations Identified and Screened for Retrieval | |
| Primary survey: excluded | three studies about other cancer types |
| two studies about benign disease or healthy women | |
| 25 potentially eligible studies retrieved for more detailed evaluation | |
| Secondary survey: excluded | two studies available only in non-English language |
| 23 eligible studies identified | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vitale, S.G.; Laganà, A.S.; Capriglione, S.; Angioli, R.; La Rosa, V.L.; Lopez, S.; Valenti, G.; Sapia, F.; Sarpietro, G.; Butticè, S.; et al. Target Therapies for Uterine Carcinosarcomas: Current Evidence and Future Perspectives. Int. J. Mol. Sci. 2017, 18, 1100. https://doi.org/10.3390/ijms18051100
Vitale SG, Laganà AS, Capriglione S, Angioli R, La Rosa VL, Lopez S, Valenti G, Sapia F, Sarpietro G, Butticè S, et al. Target Therapies for Uterine Carcinosarcomas: Current Evidence and Future Perspectives. International Journal of Molecular Sciences. 2017; 18(5):1100. https://doi.org/10.3390/ijms18051100
Chicago/Turabian StyleVitale, Salvatore Giovanni, Antonio Simone Laganà, Stella Capriglione, Roberto Angioli, Valentina Lucia La Rosa, Salvatore Lopez, Gaetano Valenti, Fabrizio Sapia, Giuseppe Sarpietro, Salvatore Butticè, and et al. 2017. "Target Therapies for Uterine Carcinosarcomas: Current Evidence and Future Perspectives" International Journal of Molecular Sciences 18, no. 5: 1100. https://doi.org/10.3390/ijms18051100