Biodiversity and Enological Potential of Non-Saccharomyces Yeasts from Nemean Vineyards
Abstract
:1. Introduction
2. Materials and Methods
2.1. Isolation of Non-Saccharomyces (NS) Yeasts
2.2. Molecular Identification of Yeasts
2.3. Pure-Culture Fermentations
2.4. Mixed-Culture Fermentations
2.5. Analytical Determinations
2.6. Statistical Analysis
3. Results and Discussion
3.1. Heterogeneity of Non-Saccharomyces Yeasts in Fermented Musts
3.2. Technological Characterization of NS Yeast Strains
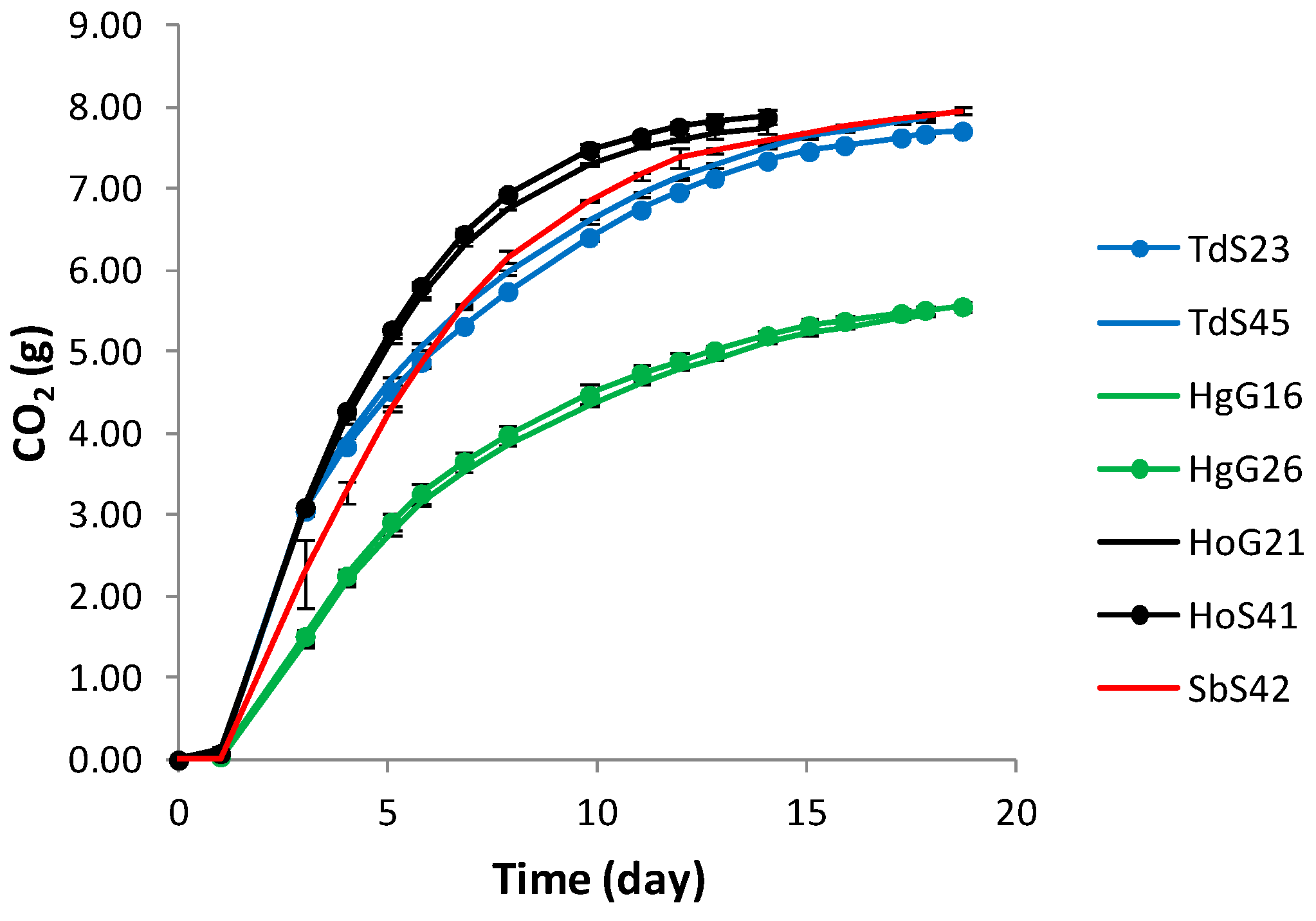
3.3. Fermentation Kinetics and Yeast Population Dynamics in Mixed-Culture Fermentations
3.4. Chemical Profiles of Wines Produced in Mixed-Culture Fermentations
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Fleet, G.H. Yeast interactions and wine flavour. Int. J. Food Microbiol. 2003, 86, 11–22. [Google Scholar] [CrossRef]
- Combina, M.; Elía, A.; Mercado, L.; Catania, C.; Ganga, A.; Martinez, C. Dynamics of indigenous yeast populations during spontaneous fermentation of wines from Mendoza, Argentina. Int. J. Food Microbiol. 2005, 99, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Nisiotou, A.A.; Nychas, G.J.E. Yeast populations residing on healthy or botrytis-infected grapes from a vineyard in Attica, Greece. Appl. Environ. Microbiol. 2007, 73, 2765–2768. [Google Scholar] [CrossRef] [PubMed]
- Nisiotou, A.A.; Spiropoulos, A.E.; Nychas, G.J.E. Yeast community structures and dynamics in healthy and botrytis-affected grape must fermentations. Appl. Environ. Microbiol. 2007, 73, 6705–6713. [Google Scholar] [CrossRef] [PubMed]
- Renouf, V.; Claisse, O.; Lonvaud-Funel, A. Inventory and monitoring of wine microbial consortia. Appl. Microbiol. Biotechnol. 2007, 75, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Fleet, G.H. Wine yeasts for the future. FEMS Yeast Res. 2008, 8, 979–995. [Google Scholar] [CrossRef] [PubMed]
- Ciani, M.; Comitini, F.; Mannazzu, I.; Domizio, P. Controlled mixed culture fermentation: A new perspective on the use of non-Saccharomyces yeasts in winemaking. FEMS Yeast Res. 2010, 10, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Fleet, G.H. The microorganisms of winemaking—Isolation enumeration and identification. In Wine Microbiology and Biotechnology; Fleet, G.H., Ed.; Harwood Academic Publishers: Chur, Switzerland, 1993; pp. 1–27. [Google Scholar]
- Mortimer, R.K.; Romano, P.; Suzzi, G.; Polsinelli, M. Genome Renewal: A new phenomenon revealed from a genetic study of 43 strains of Saccharomyces cerevisiae derived from natural fermentation of grape musts. Yeast 1994, 10, 1543–1552. [Google Scholar] [CrossRef] [PubMed]
- Torija, M.J.; Rozès, N.; Poblet, M.; Guillamón, J.M.; Mas, A. Yeast population dynamics in spontaneous fermentations: Comparison between two different wine-producing areas over a period of three years. Anton. Leeuw. 2001, 79, 345–352. [Google Scholar] [CrossRef]
- Di Maro, E.; Ercolini, D.; Coppola, S. Yeast dynamics during spontaneous wine fermentation of the Catalanesca grape. Int. J. Food Microbiol. 2007, 117, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Ravasio, D.; Carlin, S.; Boekhout, T.; Groenewald, M.; Vrhovsek, U.; Walther, A.; Wendland, J. Adding flavor to beverages with non-conventional yeasts. Fermentation 2018, 4, 15. [Google Scholar] [CrossRef]
- Jolly, N.P.; Varela, C.; Pretorius, I.S. Not your ordinary yeast: Non-Saccharomyces yeasts in wine production uncovered. FEMS Yeast Res. 2014, 14, 215–237. [Google Scholar] [CrossRef] [PubMed]
- Petruzzi, L.; Capozzi, V.; Berbegal, C.; Corbo, M.R.; Bevilacqua, A.; Spano, G.; Sinigaglia, M. Microbial resources and enological significance: Opportunities and benefits. Front. Microbiol. 2017, 8, 995. [Google Scholar] [CrossRef] [PubMed]
- Padilla, B.; Gil, J.V.; Manzanares, P. Past and future of non-Saccharomyces Yeasts: From spoilage microorganisms to biotechnological tools for improving wine aroma complexity. Front. Microbiol. 2016, 7, 411. [Google Scholar] [CrossRef] [PubMed]
- Capozzi, V.; Fragasso, M.; Romaniello, R.; Berbegal, C.; Russo, P.; Spano, G. Spontaneous Food fermentations and potential risks for human health. Fermentation 2017, 3, 49. [Google Scholar] [CrossRef]
- Esteve-Zarzoso, B.; Belloch, C.; Uruburu, F.; Querol, A. Identification of yeasts by RFLP analysis of the 5.8S rRNA gene and the two ribosomal internal transcribed spacers. Int. J. Syst. Bacteriol. 1999, 49, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Barquet, M.; Martín, V.; Medina, K.; Pérez, G.; Carrau, F.; Gaggero, C. Tandem repeat-tRNA (TRtRNA) PCR method for the molecular typing of non-Saccharomyces subspecies. Appl. Microbiol. Biotechnol. 2012, 93, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Paffetti, D.; Barberio, C.; Casalone, E.; Cavalieri, D.; Fani, R.; Fia, G.; Mori, E.; Polsinelli, M. DNA fingerprinting by random amplified polymorphic DNA and restriction fragment length polymorphism is useful for yeast typing. Res. Microbiol. 1995, 146, 587–594. [Google Scholar] [CrossRef]
- Martín, B.; Garriga, M.; Hugas, M.; Bover-Cid, S.; Veciana-Nogués, M.T.; Aymerich, T. Molecular, technological and safety characterization of gram-positive catalase-positive cocci from slightly fermented sausages. Int. J. Food Microbiol. 2006, 107, 148–158. [Google Scholar] [CrossRef] [PubMed]
- International Organization of Vines and Wines (OIV). Compendium of International Methods of Wine and Must Analysis—Volume 1. In Bulletin de L’organisation Internationale de la Vigne et du Vin; OIV: Paris, France, 2018. [Google Scholar]
- Gump, B.H.; Zoecklein, B.W.; Fugelsang, K.C.; Whiton, R.S. Comparison of analytical methods for prediction of prefermentation nutritional status of grape juice. Am. J. Enol. Vitic. 2002, 53, 325–329. [Google Scholar]
- European Commission. Commission Regulation (EC) no. 2870/2000 of 19 December 2000 on laying down the community reference methods for the analysis of spirits drinks. Off. J. Eur. Communities 2000, L 333, 20–46. [Google Scholar]
- Pinto, C.; Pinho, D.; Cardoso, R.; Custódio, V.; Fernandes, J.; Sousa, S.; Pinheiro, M.; Egas, C.; Gomes, A.C. Wine fermentation microbiome: A landscape from different portuguese wine appellations. Front. Microbiol. 2015, 6, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Romano, P.; Marchese, R.; Laurita, C.; Saleano, G.; Turbanti, L. Biotechnological suitability of Saccharomycodes ludwigii for fermented beverages. World J. Microbiol. Biotechnol. 1999, 15, 451–454. [Google Scholar] [CrossRef]
- Jolly, N.P.; Augustyn, O.P.H.; Pretorius, I.S. The role and use of non-Saccharomyces yeasts in wine production. S. Afr. J. Enol. Vitic. 2006, 27, 15–39. [Google Scholar] [CrossRef]
- Loureiro, V.; Malfeito-Ferreira, M. Spoilage yeasts in the wine industry. Int. J. Food Microbiol. 2003, 86, 23–50. [Google Scholar] [CrossRef]
- Capozzi, V.; Garofalo, C.; Chiriatti, M.A.; Grieco, F.; Spano, G. Microbial terroir and food innovation: The case of yeast biodiversity in wine. Microbiol. Res. 2015, 181, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Bely, M.; Stoeckle, P.; Masneuf-Pomarède, I.; Dubourdieu, D. Impact of mixed Torulaspora delbrueckii-Saccharomyces cerevisiae culture on high-sugar fermentation. Int. J. Food Microbiol. 2008, 122, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Escribano, R.; González-Arenzana, L.; Portu, J.; Garijo, P.; López-Alfaro, I.; López, R.; Santamaría, P.; Gutiérrez, A. Wine aromatic compound production and fermentative behaviour within different non-Saccharomyces species and clones. J. Appl. Microbiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Viana, F.; Gil, J.V.; Genovés, S.; Vallés, S.; Manzanares, P. Rational selection of non-Saccharomyces wine yeasts for mixed starters based on ester formation and enological traits. Food Microbiol. 2008, 25, 778–785. [Google Scholar] [CrossRef] [PubMed]
- Sadoudi, M.; Tourdot-Maréchal, R.; Rousseaux, S.; Steyer, D.; Gallardo-Chacón, J.J.; Ballester, J.; Vichi, S.; Guérin-Schneider, R.; Caixach, J.; Alexandre, H. Yeast-yeast interactions revealed by aromatic profile analysis of Sauvignon Blanc wine fermented by single or co-culture of non-Saccharomyces and Saccharomyces yeasts. Food Microbiol. 2012, 32, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Granchi, L.; Ganucci, D.; Messini, A.; Vincenzini, M. Oenological properties of Hanseniaspora osmophila and Kloeckera corticis from wines produced by spontaneous fermentations of normal and dried grapes. FEMS Yeast Res. 2002, 2, 403–407. [Google Scholar] [PubMed]
- Englezos, V.; Rantsiou, K.; Torchio, F.; Rolle, L.; Gerbi, V.; Cocolin, L. Exploitation of the non-Saccharomyces yeast Starmerella Bacillaris (synonym Candida zemplinina) in wine fermentation: Physiological and molecular characterizations. Int. J. Food Microbiol. 2015, 199, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Ciani, M.; Fatichenti, F. Selective sugar consumption by apiculate yeasts. Lett. Appl. Microbiol. 1999, 28, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Comitini, F.; Gobbi, M.; Domizio, P.; Romani, C.; Lencioni, L.; Mannazzu, I.; Ciani, M. selected non-Saccharomyces wine yeasts in controlled multistarter fermentations with Saccharomyces cerevisiae. Food Microbiol. 2011, 28, 873–882. [Google Scholar] [CrossRef] [PubMed]
- Andorrà, I.; Berradre, M.; Mas, A.; Esteve-Zarzoso, B.; Guillamón, J.M. Effect of mixed culture fermentations on yeast populations and aroma profile. LWT-Food Sci. Technol. 2012, 49, 8–13. [Google Scholar] [CrossRef]
- Ciani, M.; Morales, P.; Comitini, F.; Tronchoni, J.; Canonico, L.; Curiel, J.A.; Oro, L.; Rodrigues, A.J.; Gonzalez, R. Non-conventional yeast species for lowering ethanol content of wines. Front. Microbiol. 2016, 7, 642. [Google Scholar] [CrossRef] [PubMed]
- Cus, F.; Jenko, M. Influence of yeast on quality of Gewürztraminer wine. Food Technol. Biotechnol. 2013, 51, 547–553. [Google Scholar]
- Belda, I.; Ruiz, J.; Beisert, B.; Navascués, E.; Marquina, D.; Calderón, F.; Rauhut, D.; Benito, S.; Santos, A. Influence of Torulaspora delbrueckii in varietal thiol (3-SH and 4-MSP) release in wine sequential fermentations. Int. J. Food Microbiol. 2017, 257, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Puertas, B.; Jiménez, M.J.; Cantos-Villar, E.; Cantoral, J.M.; Rodríguez, M.E. Use of Torulaspora delbrueckii and Saccharomyces cerevisiae in semi-industrial sequential inoculation to improve quality of Palomino and Chardonnay wines in warm climates. J. Appl. Microbiol. 2017, 122, 733–746. [Google Scholar] [CrossRef] [PubMed]
- Belda, I.; Navascués, E.; Marquina, D.; Santos, A.; Calderon, F.; Benito, S. Dynamic analysis of physiological properties of Torulaspora delbrueckii in wine fermentations and its incidence on wine Quality. Appl. Microbiol. Biotechnol. 2014, 99, 1911–1922. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Renault, P.; Coulon, J.; de Revel, G.; Barbe, J.C.; Bely, M. Increase of fruity aroma during mixed T. delbrueckii/S. cerevisiae wine fermentation is linked to specific esters enhancement. Int. J. Food Microbiol. 2015, 207, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Escott, C.; Loira, I.; del Fresno, J.M.; Morata, A.; Tesfaye, W.; Calderon, F.; Suárez-Lepe, J.A.; Han, S.; Benito, S. Use of non-Saccharomyces yeasts and oenological tannin in red winemaking: Influence on colour, aroma and sensorial properties of young wines. Food Microbiol. 2018, 69, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, R.; Quiros, M.; Morales, P. Yeast respiration of sugars by non-Saccharomyces yeast species: A promising and barely explored approach to lowering alcohol content of wines. Trends Food Sci. Technol. 2013, 29, 55–61. [Google Scholar] [CrossRef]



| Yeast Species | Ferment | ||||
|---|---|---|---|---|---|
| NAG1 | NAG2 | NAG3 | NAG4 | NAG5 | |
| Hanseniaspora guilliermondii | 64 | 25 | 25 | ||
| Hanseniaspora osmophila | 37 | 6 | |||
| Lachancea thermotolerans | 9 | 13 | 25 | ||
| Torulaspora delbrueckii | 25 | 25 | 19 | ||
| Saccharomycodes ludwigii | 27 | ||||
| Starmerella bacillaris | 25 | 75 | |||
| Zygosaccharomyces bailii | 100 | ||||
| Enological Parameter | Yeast Strain | ||||||
|---|---|---|---|---|---|---|---|
| TdS45 | TdS23 | HoS41 | HoG21 | HgG26 | HgG16 | SbS42 | |
| Fermentation rate (gCO2/day, time period 3 days) | 1.05 ± 0.01 a | 1.03 ± 0.02 a | 1.05 ± 0.00 a | 1.05 ± 0.01 a | 0.51 ± 0.03 c | 0.47 ± 0.01 c | 0.75 ± 0.13 b |
| Fermentation vigour (gCO2 /100 mL at d 7) | 22.34 ± 0.18 a | 21.63 ± 0.11 a | 24.93 ± 0.16 a | 24.39 ± 0.11 a | 13.62 ± 0.49 d | 12.98 ± 0.12 d | 20.25 ± 0.55 a |
| CO2 production (g/flask) | 7.86 ± 0.03 a | 7.67 ± 0.01 a | 7.88 ± 0.09 a | 7.74 ± 0.06 a | 5.44 ± 0.01 b | 5.56 ± 0.05 b | 7.95 ± 0.04 a |
| Total acidity (as tartaric acid g/L) | 5.7 ± 0.1 a | 5.5 ± 0.0 b | 5.1 ± 0.0 c | 4.9 ± 0.1 d | 4.9 ± 0.0 d | 4.9 ± 0.0 d | 5.5 ± 0.0 b |
| pH | 3.60 ± 0.01 f | 3.65 ± 0.00 e | 3.75 ± 0.00 c | 3.80 ± 0.01 a | 3.79 ± 0.1 a,b | 3.78 ± 0.00 b | 3.70 ± 0.00 d |
| Volatile acidity (as acetic acid g/L) | 0.33 ± 0.01 d | 0.66 ± 0.01 c | 0.72 ± 0.01 b | 0.67 ± 0.00 c | 0.76 ± 0.0 a | 0.73 ± 0.01 b | 0.76 ± 0.02 a |
| Malic acid (g/L) | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 |
| Lactic acid (g/L) | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 |
| Succinic acid (g/L) | 1.4 ± 0.0 a | 1.1 ± 0.0 b | 0.7 ± 0.0 c | 0.7 ± 0.0 c | 0.7 ± 0.0 c | 0.8 ± 0.0 c | 0.5 ± 0.0 d |
| Glucose (g/L) | 46.4 ± 0.8 e | 48.2 ± 0.2 d | 41.8 ± 1.3 g | 43.2 ± 0.3 f | 103.4 ± 0.8 c | 105.4 ± 0.0 b | 122.6 ± 0.4 a |
| Fructose (g/L) | 77.5 ± 0.6 d | 80.4 ± 0.0 c | 82.0 ± 1.0 b | 87.3 ± 0.4 a | 64.2 ± 0.7 f | 66.1 ± 0.1 e | 0.9 ± 0.0 g |
| Glycerol (g/L) | 6.0 ± 0.1 d | 6.8 ± 0.0 c | 5.7 ± 0.4 d,e | 5.5 ± 0.0 e | 8.6 ± 0.1 b | 8.7 ± 0.0 b | 14.1 ± 0.1 a |
| Acetic acid (mg/L) | 290 ± 5 f | 628 ± 12 e | 664 ± 6 c,d | 661 ± 9 d | 682 ± 1 c | 712 ± 9 b | 853 ± 11 a |
| Ethanol (g/L) | 78.8 ± 0.5 c | 78.5 ± 0.3 c | 80.8 ± 0.8 a | 80.5 ± 0.4 a,b | 55.3 ± 0.7 d | 53.9 ± 0.6 e | 79.3 ± 0.4 b,c |
| Compound | Inoculation Protocol | ||
|---|---|---|---|
| SSc | CoSb | CoTd | |
| Total acidity (as tartaric acid g/L) | 8.1 ± 0.1 a | 8.0 ± 0.1 a | 8.1 ± 0.0 b |
| pH | 3.51 ± 0.01 a,b | 3.53 ± 0.01 a | 3.50 ± 0.02 b |
| Volatile acidity (as acetic acid g/L) | 0.22 ± 0.02 a | 0.25 ± 0.01 a | 0.23 ± 0.01 a |
| Free SO2 (mg/L) | 6 ± 2 a | 6 ± 1 a | 6 ± 1 a |
| Total SO2 (mg/L) | 16 ± 1 a | 16 ± 2 a | 15 ± 2 a |
| Citric acid (mg/L) | 897 ± 16 a | 855 ± 1 b | 843 ± 18 b |
| Tartaric acid (g/L) | 1.9 ± 0.1 a | 1.7 ± 0.1 b | 1.8 ± 0.0 a,b |
| Malic acid (g/L) | 2.3 ± 0.1 a | 2.2 ± 0.0 b | 2.2 ± 0.1 b |
| Succinic acid (g/L) | 1.2 ± 0.0 a | 1.2 ± 0.0 a | 1.2 ± 0.1 a |
| Acetic acid (mg/L) | 205 ± 5 b | 221 ± 2 a | 178 ± 5 c |
| Glucose (g/L) | 1.0 ± 0.1 a | 0.9 ± 0.0 a | 0.9 ± 0.0 a |
| Glycerol (g/L) | 10.4 ± 0.2 a | 10.4 ± 0.1 a | 9.9 ± 0.3 b |
| Ethanol (g/L) | 113.6 ± 1.6 a | 109.2 ± 0.6 b | 107.0 ± 2.2 b |
| Acetaldehyde (mg/L) | 5.7 ± 0.9 a | 7.8 ± 1.3 a | 5.9 ± 1.3 a |
| Ethyl acetate (mg/L) | 31.8 ± 0.4 b | 33.5 ± 0.5 ab | 34.4 ± 1.6 a |
| Methanol (mg/L) | 38.4 ± 1.3 a | 38.8 ± 0.4 a | 37.3 ± 2.3 a |
| Propanol (mg/L) | 26.6 ± 0.8 b | 31.5 ± 1.8 a | 28.2 ± 2.1 a,b |
| Isobutanol (mg/L) | 68.3 ± 0.9 b | 76 ± 0.9 a | 70.8 ± 2.2 b |
| 2-Methyl-1-butanol (mg/L) | 74.9 ± 1.0 a | 70.7 ± 0.2 b | 74.5 ± 2.9 a |
| 3-Methyl-1-butanol (mg/L) | 340.4 ± 5.7 a | 335.1 ± 2.5 a | 341.7 ± 12.6 a |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sgouros, G.; Chalvantzi, I.; Mallouchos, A.; Paraskevopoulos, Y.; Banilas, G.; Nisiotou, A. Biodiversity and Enological Potential of Non-Saccharomyces Yeasts from Nemean Vineyards. Fermentation 2018, 4, 32. https://doi.org/10.3390/fermentation4020032
Sgouros G, Chalvantzi I, Mallouchos A, Paraskevopoulos Y, Banilas G, Nisiotou A. Biodiversity and Enological Potential of Non-Saccharomyces Yeasts from Nemean Vineyards. Fermentation. 2018; 4(2):32. https://doi.org/10.3390/fermentation4020032
Chicago/Turabian StyleSgouros, Georgios, Ioanna Chalvantzi, Athanasios Mallouchos, Yiannis Paraskevopoulos, Georgios Banilas, and Aspasia Nisiotou. 2018. "Biodiversity and Enological Potential of Non-Saccharomyces Yeasts from Nemean Vineyards" Fermentation 4, no. 2: 32. https://doi.org/10.3390/fermentation4020032





