Preventing and Mitigating Alcohol Toxicity: A Review on Protective Substances
Abstract
:1. Introduction
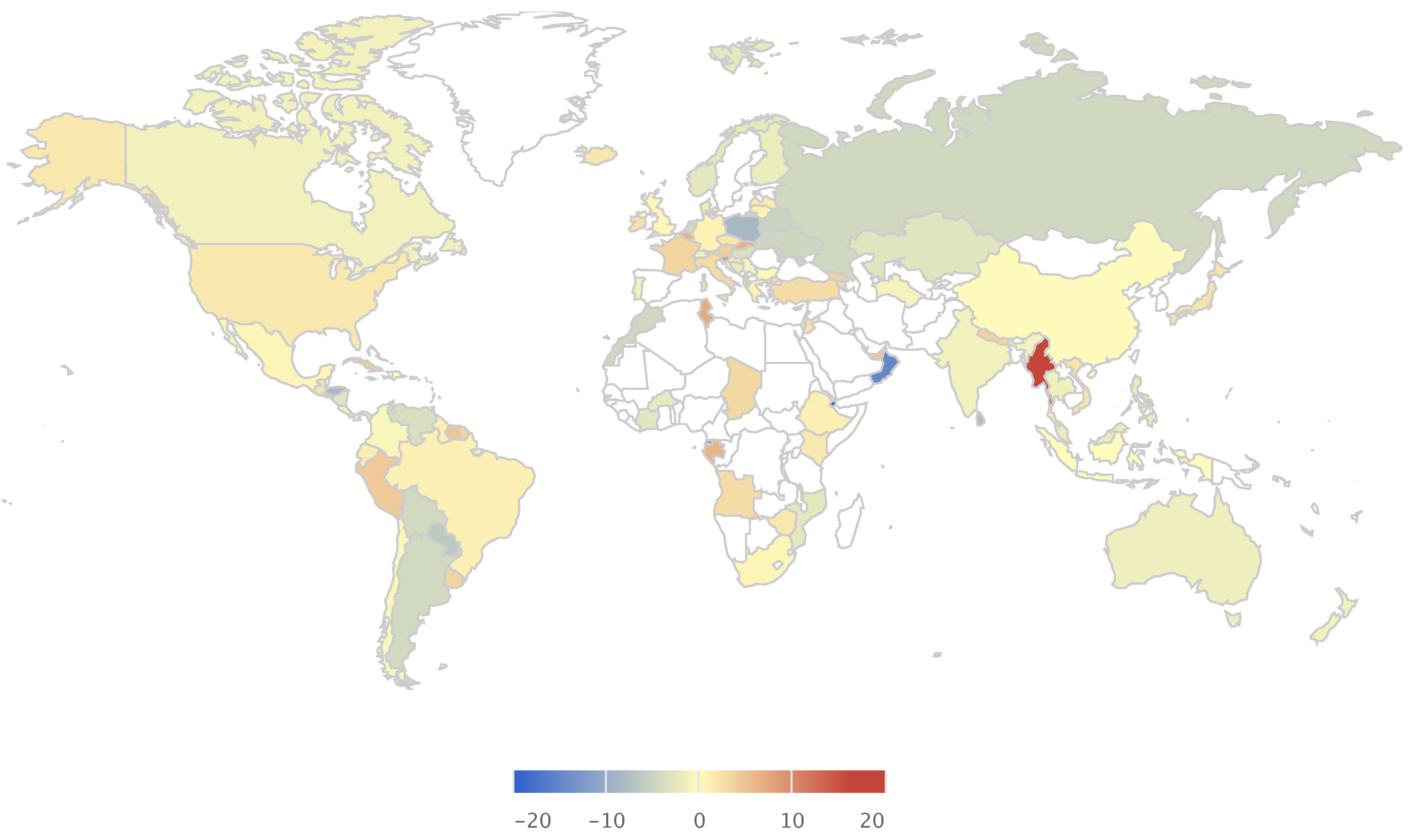
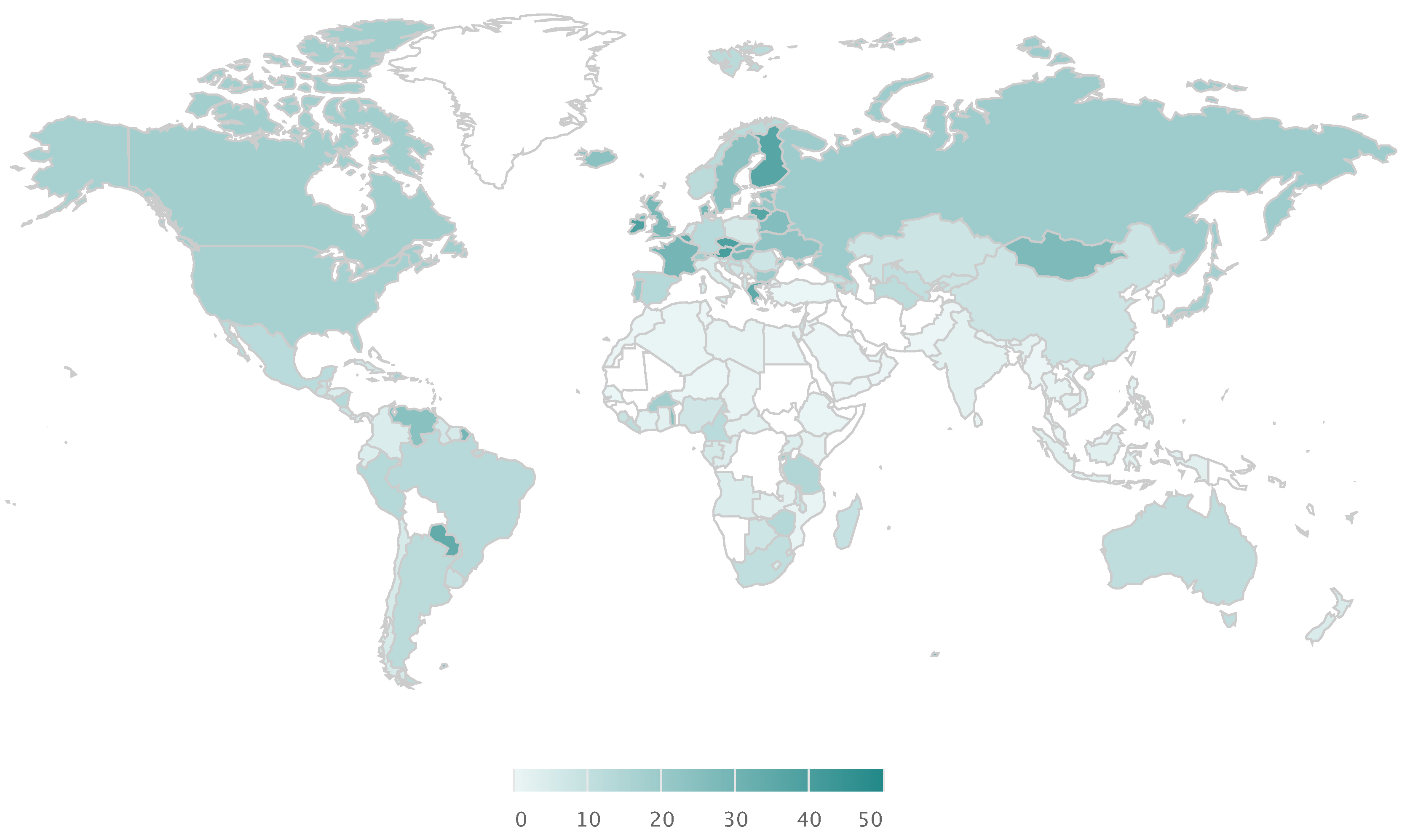
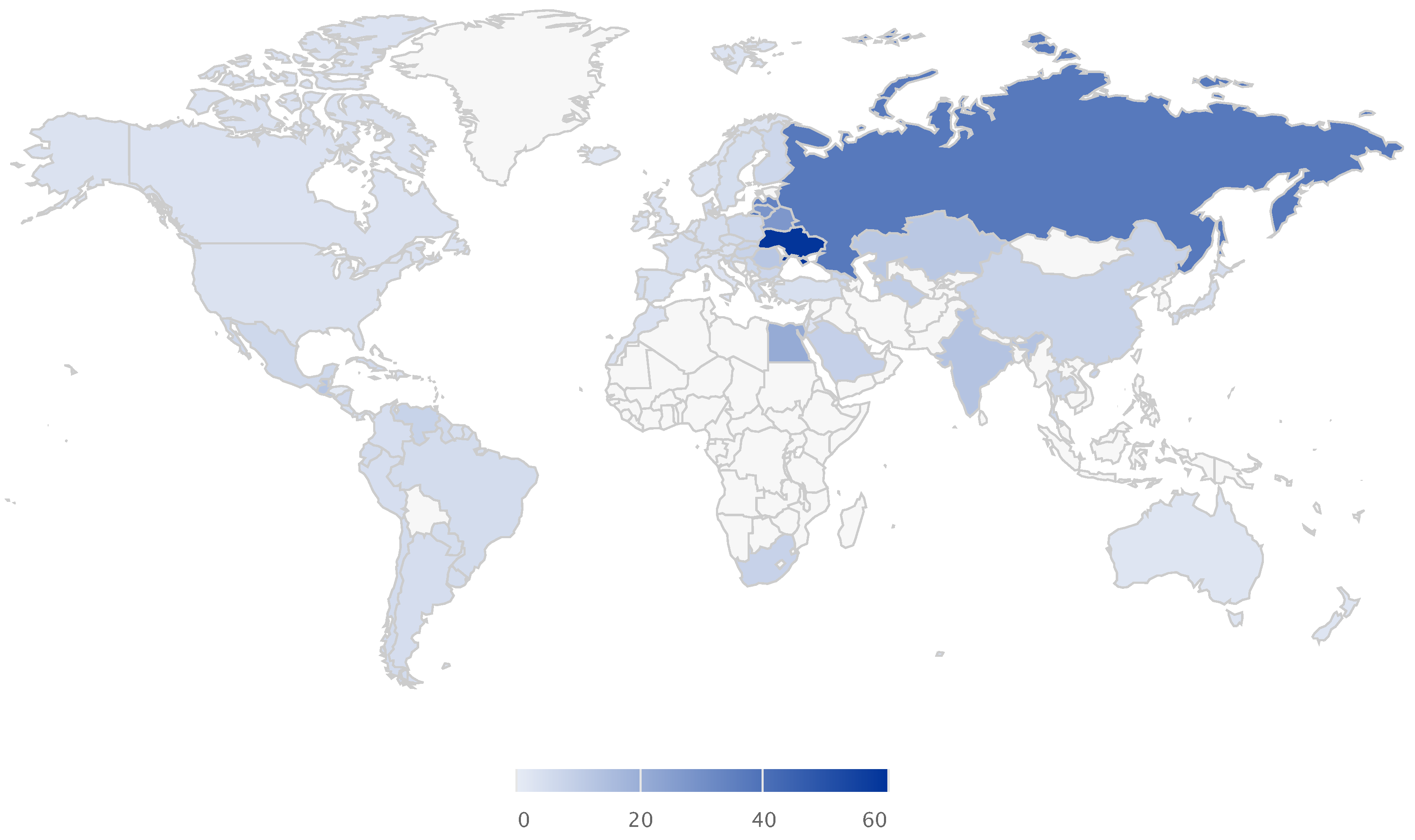
Global Alcohol Consumption
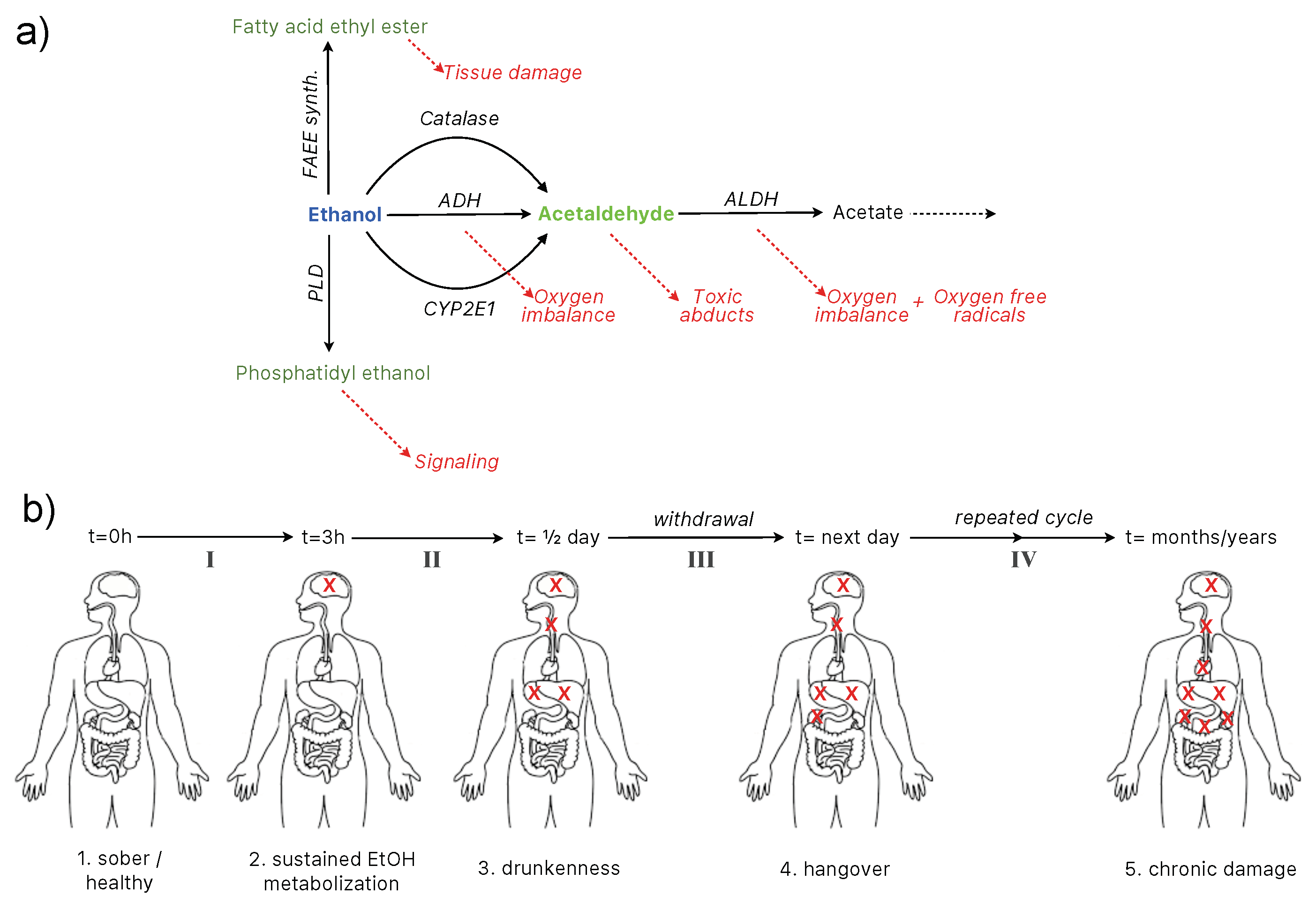
2. Alcohol Metabolism and Toxicity
2.1. Key Organs, Pathways, and Metabolic Processes
2.2. Adverse Effects of Alcohol Consumption in Human Health
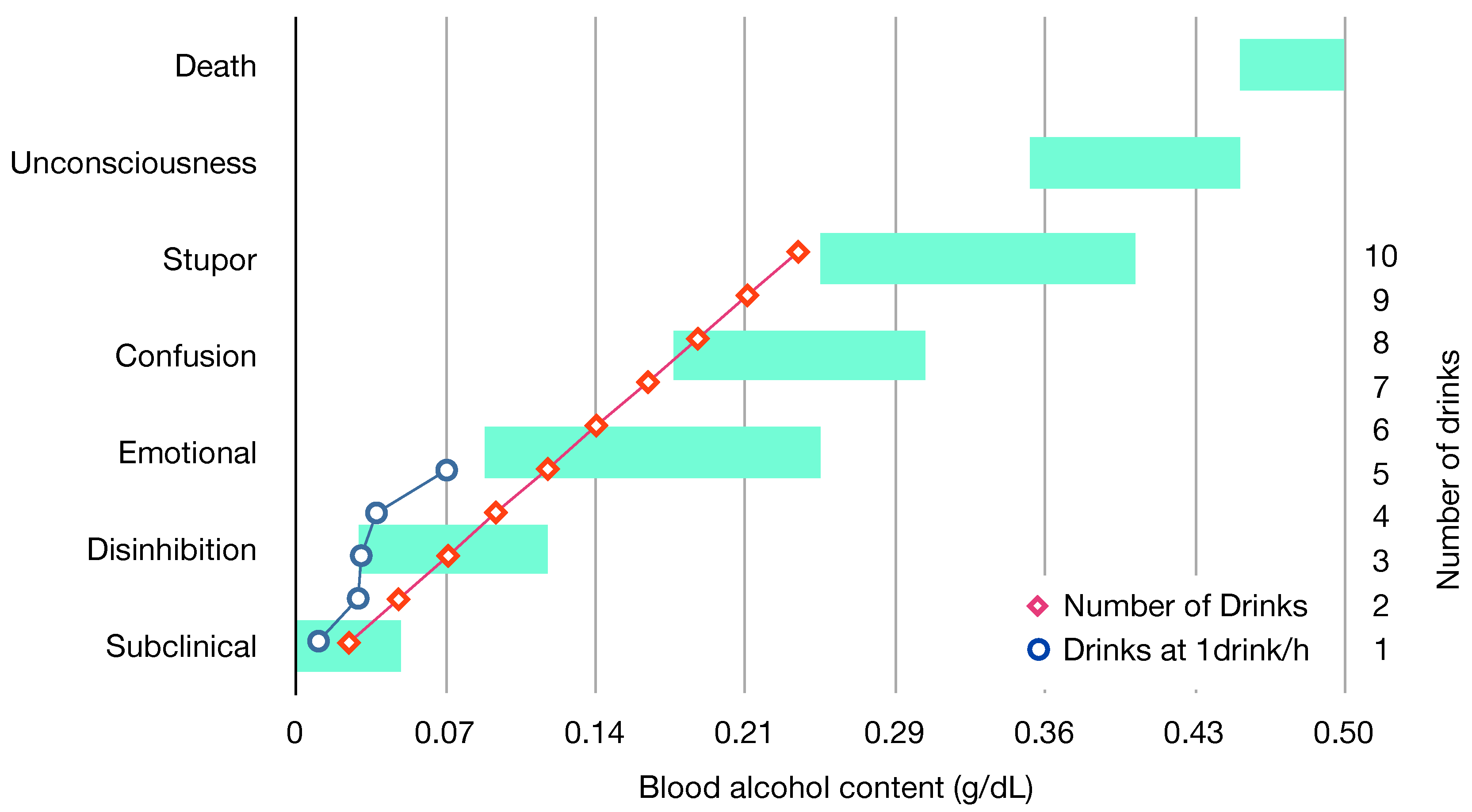
2.2.1. Drunkenness
2.2.2. Hangover
2.2.3. Intoxication and Metabolic Long-Term Damage
2.3. Molecular and Physiological Pathways on a Time-Scale Basis
- Level I: from sober to sustained metabolization (1–2),
- Level II: from sustained metabolization to drunkenness,
- Level III: from drunkenness to hangover (withdrawal),
- Level IV: chronic intoxication.
3. Alcohol Toxicity Prevention and Relief
3.1. Drunkenness Prevention and Relief
3.2. Hangover Prevention and Relief
4. Known Compounds for Alcohol Toxicity Mitigation
4.1. Review Strategy Methodology
4.2. Plant Extracts and Plant Concoctions
Characterised Plant Bioactive Compounds
4.3. Fungi, Bacterial, and Animal Extracts
4.4. Synthetic Drugs and Other Chemicals
4.5. Complex Formulations
5. Key Substances and Key Blockage Points for Mitigation
Timely Mitigation and Preventive Formulations
6. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Teaford, M.F.; Ungar, P.S. Diet and the evolution of the earliest human ancestors. Proc. Natl. Acad. Sci. USA 2000, 97, 13506–13511. [Google Scholar] [CrossRef] [PubMed]
- Phillips, R. Alcohol: A History; UNC Press Books: Chapel Hill, NC, USA, 2014. [Google Scholar]
- McGovern, P.E. Uncorking the Past: The Quest for Wine, Beer, and Other Alcoholic Beverages; Univ of California Press: Oakland, CA, USA, 2009. [Google Scholar]
- McGovern, P.E.; Zhang, J.; Tang, J.; Zhang, Z.; Hall, G.R.; Moreau, R.A.; Nuñez, A.; Butrym, E.D.; Richards, M.P.; Wang, C.S.; et al. Fermented beverages of pre-and proto-historic China. Proc. Natl. Acad. Sci. USA 2004, 101, 17593–17598. [Google Scholar] [CrossRef] [PubMed]
- Forbes, R.J. A Short History of the Art of Distillation: From the Beginnings up to the Death of Cellier Blumenthal; Brill: Leiden, The Netherlands, 1970. [Google Scholar]
- World Health Organization (WHO). Global Status Report on Alcohol and Health 2014; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Scholz, H.; Franz, M.; Heberlein, U. The hangover gene defines a stress pathway required for ethanol tolerance development. Nature 2005, 436, 845–847. [Google Scholar] [CrossRef] [PubMed]
- Thomasson, H.R.; Crabb, D.W.; Edenberg, H.J.; Li, T.K. Alcohol and aldehyde dehydrogenase polymorphisms and alcoholism. Behav. Genet. 1993, 23, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Zakhari, S. Overview: How is alcohol metabolized by the body? Alcoh. Res. Health 2006, 29, 245–255. [Google Scholar]
- Bennion, L.J.; Li, T.K. Alcohol metabolism in American Indians and whites: Lack of racial differences in metabolic rate and liver alcohol dehydrogenase. N. Engl. J. Med. 1976, 294, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Kopun, M.; Propping, P. The kinetics of ethanol absorption and elimination in twins and supplementary repetitive experiments in singleton subjects. Eur. J. Clin. Pharmacol. 1977, 11, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Crabb, D.W.; Edenberg, H.J.; Bosron, W.F.; Li, T.K. Genotypes for aldehyde dehydrogenase deficiency and alcohol sensitivity. The inactive ALDH2 (2) allele is dominant. J. Clin.l Investig. 1989, 83, 314–316. [Google Scholar] [CrossRef] [PubMed]
- Borinskaya, S.; Gasemianrodsari, F.; Kalyina, N.; Sokolova, M.; Yankovsky, N. Polymorphism of alcohol dehydrogenase gene ADH1B in Eastern Slavic and Iranian-speaking populations. Russ. J. Genet. 2005, 41, 1291–1294. [Google Scholar] [CrossRef]
- Saunders, J.B.; Stockley, C. The Biology of Intoxication. In Expressions of Drunkenness (Four Hundred Rabbits); Routledge: Abingdon-on-Thames, UK, 2011; pp. 31–70. [Google Scholar]
- Sleet, D.A.; Mercer, S.L.; Hopkins Cole, K.; Shults, R.A.; Elder, R.W.; Nichols, J.L. Scientific evidence and policy change: Lowering the legal blood alcohol limit for drivers to 0.08% in the USA. Glob. Health Promot. 2011, 18, 23–26. [Google Scholar] [CrossRef] [PubMed]
- Dubowski, K.M. Stages of Acute Alcoholic Influence/intoxication; The University of Oklahoma, Department of Medicine: Oklahoma City, OK, USA, 2006. [Google Scholar]
- Miller, W.; Munoz, R. How to Control Your Drinking: A Practical Guide to Responsible Drinking, Revised Edition; University of New Mexico Press: Albuquerque, New Mexico, 1982. [Google Scholar]
- Liu, Y.; Hunt, W.A. The Drunken Synapse: Studies of Alcohol-Related Disorders; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Swift, R.; Davidson, D. Alcohol hangover. Alcohol. Health Res. World 1998, 22, 54–60. [Google Scholar] [PubMed]
- Surawicz, B. Relationship between electrocardiogram and electrolytes. Am. Heart J. 1967, 73, 814–834. [Google Scholar] [CrossRef]
- Wieloch, T. Hypoglycemia-induced neuronal damage prevented by an N-methyl-D-aspartate antagonist. Science 1985, 230, 681–683. [Google Scholar] [CrossRef] [PubMed]
- Gauvin, D.V.; Briscoe, R.J.; Baird, T.J.; Vallett, M.; Carl, K.L.; Holloway, F.A. Cross-generalization of an EtOH "hangover" cue to endogenously and exogenously induced stimuli. Pharmacol. Biochem. Behav. 1997, 57, 199–206. [Google Scholar] [CrossRef]
- Seitz, H.K.; Becker, P. Alcohol metabolism and cancer risk. Alcohol. Res. Health 2007, 30, 38. [Google Scholar] [PubMed]
- Wu, D.; Cederbaum, A.I. Ethanol-Induced Apoptosis to Stable HepG2 Cell Lines Expressing Human Cytochrome P-4502E1. Alcohol. Clin. Exp. Res. 1999, 23, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Arteel, G.E.; Raleigh, J.A.; Bradford, B.U.; Thurman, R.G. Acute alcohol produces hypoxia directly in rat liver tissue in vivo: Role of Kupffer cells. Am. J. Physiol. 1996, 271, G494–G500. [Google Scholar] [CrossRef] [PubMed]
- Bordone, L.; Guarente, L. Calorie restriction, SIRT1 and metabolism: Understanding longevity. Nat. Rev. Mol. Cell Biol. 2005, 6, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Nagy, L.E. Molecular aspects of alcohol metabolism: Transcription factors involved in early ethanol-induced liver injury. Annu. Rev. Nutr. 2004, 24, 55–78. [Google Scholar] [CrossRef] [PubMed]
- Bleich, S.; Degner, D.; Sperling, W.; Bönsch, D.; Thürauf, N.; Kornhuber, J. Homocysteine as a neurotoxin in chronic alcoholism. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2004, 28, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Henderson, G.I.; Chen, J.; Schenker, S. Ethanol, oxidative stress, reactive aldehydes, and the fetus. Front. Biosci. 1999, 4, D541–D550. [Google Scholar] [PubMed]
- Cohen-Kerem, R.; Koren, G. Antioxidants and fetal protection against ethanol teratogenicity: I. Review of the experimental data and implications to humans. Neurotoxicol. Teratol. 2003, 25, 1–9. [Google Scholar] [CrossRef]
- Seitz, H.K.; Stickel, F.; Homann, N. Pathogenetic mechanisms of upper aerodigestive tract cancer in alcoholics. Int. J. Cancer 2004, 108, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Stickel, F.; Schuppan, D.; Hahn, E.; Seitz, H. Cocarcinogenic effects of alcohol in hepatocarcinogenesis. Gut 2002, 51, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Muñiz-Hernández, S.; Velázquez-Fernández, J.; Díaz-Chávez, J.; López-Sánchez, R.; Hernández, J. Alcoholism: Common and oxidative damage biomarkers. J. Clin. Toxicol. S 2014, 7, S7-006. [Google Scholar] [CrossRef]
- Sean, O. A Unified Theory of Optimal Booze Drinking. Journapocalypse 2011, 32, 4195–4203. [Google Scholar]
- El-Sayed, M.S.; Ali, N.; Ali, Z.E.S. Interaction between alcohol and exercise. Sports Med. 2005, 35, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Nutt, D.J. Alcohol alternatives–a goal for psychopharmacology? J. Psychopharmacol. 2006, 20, 318–320. [Google Scholar] [CrossRef] [PubMed]
- Penetar, D.M.; Toto, L.H.; Lee, D.Y.W.; Lukas, S.E. A single dose of kudzu extract reduces alcohol consumption in a binge drinking paradigm. Drug Alcohol. Depend. 2015, 153, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Carai, M.A.; Agabio, R.; Bombardelli, E.; Bourov, I.; Gessa, G.L.; Lobina, C.; Morazzoni, P.; Pani, M.; Reali, R.; Vacca, G.; et al. Potential use of medicinal plants in the treatment of alcoholism. Fitoterapia 2000, 71, S38–S42. [Google Scholar] [CrossRef]
- Rezvani, A.H.; Overstreet, D.H.; Perfumi, M.; Massi, M. Plant derivatives in the treatment of alcohol dependency. Pharmacol. Biochem. Behav. 2003, 75, 593–606. [Google Scholar] [CrossRef]
- Lukas, S.E.; Penetar, D.; Berko, J.; Vicens, L.; Palmer, C.; Mallya, G.; Macklin, E.A.; Lee, D.Y.W. An extract of the Chinese herbal root kudzu reduces alcohol drinking by heavy drinkers in a naturalistic setting. Alcohol. Clin. Exp. Res. 2005, 29, 756–762. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.C.; Li, T.K. Effects of isoflavones on alcohol pharmacokinetics and alcohol-drinking behavior in rats. Am. J. Clin. Nutr. 1998, 68, 1512S–1515S. [Google Scholar] [CrossRef] [PubMed]
- McGregor, N.R. Pueraria lobata (Kudzu root) hangover remedies and acetaldehyde-associated neoplasm risk. Alcohol 2007, 41, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.S.; Hong, S.G.; Choi, S.M.; Kim, B.N.; Sung, H.J. Effect of an Oriental Herbal Composition, Jang Baek Union (JBU), on Alcohol-Induced Hangover and CCl4-Induced Liver Injury in Rats. J. Korean Soc. Food Sci. Nutr. 2004, 33, 1. [Google Scholar]
- Chapman, L.F. Experimental induction of hangover. Q. J. Stud. Alcohol. Suppl. 1970, 5, 67–68. [Google Scholar]
- Pawan, G. Alcoholic drinks and hangover effects. Proc. Nutr. Soc. 1973, 32, 15A. [Google Scholar] [PubMed]
- Seppälä, T.; Leino, T.; Linnoila, M.; Huttunen, M.; YIikahri, R. Effects of hangover on psychomotor skills related to driving: Modification by fructose and glucose. Acta Pharmacol. Toxicol. 1976, 38, 209–218. [Google Scholar] [CrossRef]
- Kaivola, S.; Parantainen, J.; Österman, T.; Timonen, H. Hangover headache and prostaglandins: Prophylactic treatment with tolfenamic acid. Cephalalgia 1983, 3, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Pautasso, M. Ten simple rules for writing a literature review. PLoS Comput. Biol. 2013, 9, e1003149. [Google Scholar] [CrossRef] [PubMed]
- Haddaway, N.R.; Collins, A.M.; Coughlin, D.; Kirk, S. The role of Google Scholar in evidence reviews and its applicability to grey literature searching. PLoS ONE 2015, 10, e0138237. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.H.; Kwak, J.H.; Jeon, G.; Lee, J.W.; Seo, J.H.; Lee, H.S.; Lee, J.H. Red ginseng relieves the effects of alcohol consumption and hangover symptoms in healthy men: A randomized crossover study. Food Funct. 2014, 5, 528–534. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Zhu, P.; Jiang, C.; Ma, L.; Zhang, Z.; Zeng, X. Preliminary characterization, antioxidant activity in vitro and hepatoprotective effect on acute alcohol-induced liver injury in mice of polysaccharides from the peduncles of Hovenia dulcis. Food Chem. Toxicol. 2012, 50, 2964–2970. [Google Scholar] [CrossRef] [PubMed]
- Foxman, E. Hangover Relief Composition. U.S. Patent 7,037,532, 2 May 2006. [Google Scholar]
- Lee, F.Y.; Lee, S.D.; Tsai, Y.T.; Lai, K.H.; Chao, Y.; Lin, H.C.; Wang, S.S.; Lo, K.J. A randomized controlled trial of quinidine in the treatment of cirrhotic patients with muscle cramps. J. Hepatol. 1991, 12, 236–240. [Google Scholar] [CrossRef]
- Arumugam, S.; Watanabe, K. Japanese Kampo Medicines for the Treatment of Common Diseases: Focus on Inflammation; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- Haranaka, R.; Okada, N.; Kosoto, H.; Ohwada, S.; Nakagawa, S.; Kobayashi, M. Effects of Goreisan and Inchingoreisan on Alcohol Metabolism in Mice Liver. Jpn. J. Pharmacogn. 1984, 38, 243–248. [Google Scholar]
- Suzuki, S.; Ojima, T.; Takayama, S.; Nakano, Y.; Nawa, D.; Saito, N.; Arita, R.; Kaneko, S.; Kamiya, T.; Mikami, N.; et al. Preliminary study of the effects of orengedokuto on breath alcohol concentration. Tradit. Kampo Med. 2016, 3, 167–169. [Google Scholar] [CrossRef]
- Wakita, H.; Miyamoto, K. Effects of Oren-gedoku-to and its constituents on the cardiovascular system: Investigation of its efficacy in hotflush. J. Tradit. Med. 2002, 19, 230–237. [Google Scholar]
- Koizumi, K.; Iijima, K.; Nohmi, M.; Nakayama, S.; Oguchi, K. Effects of Byakushi and Ogon on the hepatic drug metabolizing enzymes in rats. Folia Pharmacol. Jpn. 1994, 104, 413–419. [Google Scholar] [CrossRef]
- Wang, H.; Li, T.; Xiang, H.; Zhang, X.; Fang, K.; Wu, G.; Yan, M.; Xue, N.; Chen, M.; Xie, T.; et al. Origin and Formation Mechanism Investigation of Compound Precipitation from the Traditional Chinese Prescription Huang-Lian-Jie-Du-Tang by Isothermal Titration Calorimetry. Molecules 2017, 22, 1456. [Google Scholar] [CrossRef] [PubMed]
- Yamakawa, J.I.; Motoo, Y.; Moriya, J.; Ogawa, M.; Uenishi, H.; Akazawa, S.; Sasagawa, T.; Nishio, M.; Kobayashi, J. Significance of Kampo, traditional Japanese medicine, in supportive care of cancer patients. Evid.-Based Complement. Altern. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Panda, V.; Ashar, H.; Srinath, S. Antioxidant and hepatoprotective effect of Garcinia indica fruit rind in ethanolinduced hepatic damage in rodents. Interdiscip. Toxicol. 2012, 5, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Padhye, S.; Ahmad, A.; Oswal, N.; Sarkar, F.H. Emerging role of Garcinol, the antioxidant chalcone from Garcinia indica Choisy and its synthetic analogs. J. Hematol. Oncol. 2009, 2, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, R.; Rajendra Prasad, N.; Pugalendi, K. Effect of Piper beetle leaf extract on alcoholic toxicity in the rat brain. J. Med. Food 2003, 6, 261–265. [Google Scholar] [CrossRef] [PubMed]
- Rimando, A.M.; Han, B.H.; Park, J.H.; Cantoria, M.C. Studies on the constituents of PhilippinePiper beetle leaves. Arch. Pharm. Res. 1986, 9, 93–97. [Google Scholar] [CrossRef]
- Foo, L.W.; Salleh, E.; Mamat, S.N.H. Extraction and qualitative analysis of piper betle leaves for antimicrobial Activities. Int. J. Eng. Technol. Sci. Res. 2015, 2, 1–8. [Google Scholar]
- Choi, N.E.; Ro, J.Y.; Lee, J.Y.; Ryu, J.H.; Cho, H.J. Effects of Cudrania tricuspidata Root Extract (CTE) on Ethanol-Induced Hangover via Modulating Alcohol Metabolizing Enzyme Activities and Blood Gas Levels in Rats. J. Korea Acad.-Ind. 2017, 18, 218–225. [Google Scholar]
- An, R.B.; Sohn, D.H.; Kim, Y.C. Hepatoprotective compounds of the roots of Cudrania tricuspidata on tacrine-induced cytotoxicity in Hep G2 cells. Biol. Pharm. Bull. 2006, 29, 838–840. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.; Khaja, M.N.; Aara, A.; Khan, A.A.; Habeeb, M.A.; Devi, Y.P.; Narasu, M.L.; Habibullah, C.M. Hepatoprotective activity of Sapindus mukorossi and Rheum emodi extracts: In vitro and in vivo studies. World J. Gastroenterol. 2008, 14, 2566. [Google Scholar] [CrossRef] [PubMed]
- Yadav, J.; Arya, V.; Yadav, S.; Panghal, M.; Kumar, S.; Dhankhar, S. Cassia occidentalis L.: A review on its ethnobotany, phytochemical and pharmacological profile. Fitoterapia 2010, 81, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Jafri, M.; Subhani, M.J.; Javed, K.; Singh, S. Hepatoprotective activity of leaves of Cassia occidentalis against paracetamol and ethyl alcohol intoxication in rats. J. Ethnopharmacol. 1999, 66, 355–361. [Google Scholar] [CrossRef]
- Lanhers, M.C.; Joyeux, M.; Soulimani, R.; Fleurentin, J.; Sayag, M.; Mortier, F.; Younos, C.; Pelt, J.M. Hepatoprotective and anti-inflammatory effects of a traditional medicinal plant of Chile, Peumus boldus. Planta Med. 1991, 57, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Pramyothin, P.; Ngamtin, C.; Poungshompoo, S.; Chaichantipyuth, C. Hepatoprotective activity of Phyllanthus amarus Schum. et. Thonn. extract in ethanol treated rats: In vitro and in vivo studies. J. Ethnopharmacol. 2007, 114, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Kaviarasan, S.; Ramamurty, N.; Gunasekaran, P.; Varalakshmi, E.; Anuradha, C.V. Fenugreek (Trigonella foenum graecum) seed extract prevents ethanol-induced toxicity and apoptosis in Chang liver cells. Alcohol Alcohol. 2006, 41, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Thirunavukkarasu, V.; Anuradha, C.; Viswanathan, P. Protective effect of fenugreek (Trigonella foenum graecum) seeds in experimental ethanol toxicity. Phytother. Res. 2003, 17, 737–743. [Google Scholar] [CrossRef] [PubMed]
- Benayad, Z.; Gómez-Cordovés, C.; Es-Safi, N.E. Characterization of flavonoid glycosides from fenugreek (Trigonella foenum-graecum) crude seeds by HPLC–DAD–ESI/MS analysis. Int. J. Mol. Sci. 2014, 15, 20668–20685. [Google Scholar] [CrossRef] [PubMed]
- Miquel, J.; Bernd, A.; Sempere, J.; Dıaz-Alperi, J.; Ramırez, A. The curcuma antioxidants: Pharmacological effects and prospects for future clinical use. A review. Arch. Gerontol. Geriatr. 2002, 34, 37–46. [Google Scholar] [CrossRef]
- Sheeba, M.; Asha, V. Effect of Cardiospermum halicacabum on ethanol-induced gastric ulcers in rats. J. Ethnopharmacol. 2006, 106, 105–110. [Google Scholar] [CrossRef] [PubMed]
- You, Y.; Yoo, S.; Yoon, H.G.; Park, J.; Lee, Y.H.; Kim, S.; Oh, K.T.; Lee, J.; Cho, H.Y.; Jun, W. In vitro and in vivo hepatoprotective effects of the aqueous extract from Taraxacum officinale (dandelion) root against alcohol-induced oxidative stress. Food Chem. Toxicol. 2010, 48, 1632–1637. [Google Scholar] [CrossRef] [PubMed]
- Cortés, N.; Mora, C.; Muñoz, K.; Díaz, J.; Serna, R.; Castro, D.; Osorio, E. Microscopical descriptions and chemical analysis by HPTLC of Taraxacum officinale in comparison to Hypochaeris radicata: A solution for mis-identification. Rev. Bras. Farmacogn. 2014, 24, 381–388. [Google Scholar] [CrossRef]
- Shanmugasundaram, P.; Venkataraman, S. Hepatoprotective and antioxidant effects of Hygrophila auriculata (K. Schum) Heine Acanthaceae root extract. J. Ethnopharmacol. 2006, 104, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Kshirsagar, A.; Ingale, K.; Vyawahare, N.; Thorve, V. Hygrophila spinosa: A comprehensive review. Pharmacogn. Rev. 2010, 4, 167. [Google Scholar] [CrossRef] [PubMed]
- Bang, J.S.; Chung, Y.H.; Chung, S.J.; Lee, H.S.; Song, E.H.; Shin, Y.K.; Lee, Y.J.; Kim, H.C.; Nam, Y.; Yeong, J.H. Clinical effect of a polysaccharide-rich extract of Acanthopanax senticosus on alcohol hangover. Die Pharm. 2015, 70, 269–273. [Google Scholar]
- El-Ashmawy, I.M.; Saleh, A.; Salama, O.M. Effects of marjoram volatile oil and grape seed extract on ethanol toxicity in male rats. Basic Clin. Pharmacol. Toxicol. 2007, 101, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Dogan, A.; Celik, I. Hepatoprotective and antioxidant activities of grapeseeds against ethanol-induced oxidative stress in rats. Br. J. Nutr. 2012, 107, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Katalinic, V.; Mozina, S.S.; Generalic, I.; Skroza, D.; Ljubenkov, I.; Klancnik, A. Phenolic profile, antioxidant capacity, and antimicrobial activity of leaf extracts from six Vitis vinifera L. varieties. Int. J. Food Prop. 2013, 16, 45–60. [Google Scholar] [CrossRef]
- Sandoval, M.; Lazarte, K.; Arnao, I. Hepatoprotección Antioxidante de la Cáscara y Semilla de Vitis vinifera L.(uva); Anales de la Facultad de Medicina; UNMSM, Facultad de Medicina: Lima, Peru, 2008; Volume 69, pp. 250–259. [Google Scholar]
- Chen, S.; Teoh, N.C.; Chitturi, S.; Farrell, G.C. Coffee and non-alcoholic fatty liver disease: Brewing evidence for hepatoprotection? J. Gastroenterol. Hepatol. 2014, 29, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Pari, L.; Karthikesan, K. Protective role of caffeic acid against alcohol-induced biochemical changes in rats. Fund. Clin. Pharmacol. 2007, 21, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.H.; Kim, M.Y.; Son, C.W.; Cheong, S.H.; Kim, M.R. Effect of black garlic on acute alcohol-induced hangover and chronic alcohol-induced liver injury in rats. FASEB J. 2009, 23, 111–116. [Google Scholar]
- Wiese, J.; McPherson, S.; Odden, M.C.; Shlipak, M.G. Effect of Opuntia ficus indica on symptoms of the alcohol hangover. Arch. Intern. Med. 2004, 164, 1334–1340. [Google Scholar] [CrossRef] [PubMed]
- Alimi, H.; Hfaeidh, N.; Mbarki, S.; Bouoni, Z.; Sakly, M.; Ben, R.K. Evaluation of Opuntia ficus indica f. inermis fruit juice hepatoprotective effect upon ethanol toxicity in rats. Gen. Physiol. Biophys. 2012, 31, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Madrigal-Santillán, E.; García-Melo, F.; Morales-González, J.A.; Vázquez-Alvarado, P.; Muñoz-Juárez, S.; Zuñiga-Pérez, C.; Sumaya-Martínez, M.T.; Madrigal-Bujaidar, E.; Hernández-Ceruelos, A. Antioxidant and anticlastogenic capacity of prickly pear juice. Nutrients 2013, 5, 4145–4158. [Google Scholar] [CrossRef] [PubMed]
- Fernández-López, J.A.; Almela, L.; Obón, J.M.; Castellar, R. Determination of antioxidant constituents in cactus pear fruits. Plant Foods Hum. Nutr. 2010, 65, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Kuti, J.O. Antioxidant compounds from four Opuntia cactus pear fruit varieties. Food Chem. 2004, 85, 527–533. [Google Scholar] [CrossRef]
- Kim, S.Y.; Cho, E.A.; Yoo, G.J.; Yoo, J.M.; Son, S.M.; In, M.J.; Kim, D.C.; Chae, H.J. Physiological activity of astaxanthin and its inclusion complex with cyclodextrin. KSBB J. 2009, 24, 570–578. [Google Scholar]
- Yan, T.; Zhao, Y.; Zhang, X.; Lin, X. Astaxanthin Inhibits Acetaldehyde-Induced Cytotoxicity in SH-SY5Y Cells by Modulating Akt/CREB and p38MAPK/ERK Signaling Pathways. Mar. Drugs 2016, 14, 56. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.; Li, Y.; He, L.; Tang, Y.; Li, X.; Shen, Q.; Yin, D.; Peng, Y. The protective effect of astaxanthin on fetal alcohol spectrum disorder in mice. Neuropharmacology 2014, 84, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Moldowan, M.J.; Moldowan, C. Composition and Method for Reducing Hangover. U.S. Patent 4,496,548, 29 January 1985. [Google Scholar]
- Shen, Y.; Lindemeyer, A.K.; Gonzalez, C.; Shao, X.M.; Spigelman, I.; Olsen, R.W.; Liang, J. Dihydromyricetin as a novel anti-alcohol intoxication medication. J. Neurosci. 2012, 32, 390–401. [Google Scholar] [CrossRef] [PubMed]
- Kiso, Y.; Ishikawa, T. The Liver Protective Effect of Sesamin. In Novel Compounds from Natural Products in the New Millennium: Potential and Challenges; World Scientific Publisher: Singapore, 2004; p. 139. [Google Scholar]
- Nakai, M.; Harada, M.; Nakahara, K.; Akimoto, K.; Shibata, H.; Miki, W.; Kiso, Y. Novel antioxidative metabolites in rat liver with ingested sesamin. J. Agric. Food Chem. 2003, 51, 1666–1670. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, A.; Lagos, C.; Schmidt, K.; Videla, L.A. Silymarin protection against hepatic lipid peroxidation induced by acute ethanol intoxication in the rat. Biochem. Pharmacol. 1985, 34, 2209–2212. [Google Scholar] [CrossRef]
- Song, Z.; Deaciuc, I.; Song, M.; Lee, D.Y.W.; Liu, Y.; Ji, X.; McClain, C. Silymarin Protects Against Acute Ethanol-Induced Hepatotoxicity in Mice. Alcohol. Clin. Exp. Res. 2006, 30, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Schönfeld, J.v.; Weisbrod, B.; Müller, M. Silibinin, a plant extract with antioxidant and membrane stabilizing properties, protects exocrine pancreas from cyclosporin A toxicity. Cell. Mol. Life Sci. 1997, 53, 917–920. [Google Scholar] [CrossRef]
- Saller, R.; Meier, R.; Brignoli, R. The use of silymarin in the treatment of liver diseases. Drugs 2001, 61, 2035–2063. [Google Scholar] [CrossRef] [PubMed]
- Tseng, T.H.; Chu, C.Y.; Huang, J.M.; Shiow, S.J.; Wang, C.J. Crocetin protects against oxidative damage in rat primary hepatocytes. Cancer Lett. 1995, 97, 61–67. [Google Scholar] [CrossRef]
- Lin, Y.R.; Chen, H.H.; Ko, C.H.; Chan, M.H. Neuroprotective activity of honokiol and magnolol in cerebellar granule cell damage. Eur. J. Pharmacol. 2006, 537, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, A.; Antarkar, D.; Doshi, J.; Bhatt, A.; Ramesh, V.; Vora, P.; Perissond, D.; Baxi, A.; Kale, P. Picrorhiza kurroa (Kutaki) Royle ex Benth as a hepatoprotective agent—Experimental & clinical studies. J. Postgrad. Med. 1996, 42, 105. [Google Scholar] [PubMed]
- Rao, G.M.M.; Rao, C.V.; Pushpangadan, P.; Shirwaikar, A. Hepatoprotective effects of rubiadin, a major constituent of Rubia cordifolia Linn. J. Ethnopharmacol. 2006, 103, 484–490. [Google Scholar] [CrossRef] [PubMed]
- McKay, D.L.; Blumberg, J.B. A review of the bioactivity and potential health benefits of peppermint tea (Mentha piperita L.). Phytother. Res. 2006, 20, 619–633. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Zheng, L.; Xu, L.; Yin, L.; Qi, Y.; Xu, Y.; Han, X.; Peng, J. Protective effects of dioscin against alcohol-induced liver injury. Arch. Toxicol. 2014, 88, 739–753. [Google Scholar] [CrossRef] [PubMed]
- Sumi, H.; Yatagai, C.; Wada, H.; Yoshida, E.; Maruyama, M. Effect of Bacillus natto-fermented product (BIOZYME) on blood alcohol, aldehyde concentrations after whisky drinking in human volunteers, and acute toxicity of acetaldehyde in mice. Jpn. J. Alcohol Stud. Drug Depend. 1995, 30, 69–79. [Google Scholar]
- Lee, H.S.; Song, J.; Kim, T.M.; Joo, S.S.; Park, D.; Jeon, J.H.; Shin, S.; Park, H.K.; Lee, W.K.; Ly, S.Y.; et al. Effects of a preparation of combined glutathione-enriched yeast and rice embryo/soybean extracts on ethanol hangover. J. Med. Food 2009, 12, 1359–1367. [Google Scholar] [CrossRef] [PubMed]
- McCarty, M.F. Nutraceutical strategies for ameliorating the toxic effects of alcohol. Med. Hypotheses 2013, 80, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Pillai, K.; Husain, S.; Giri, D.K. Protective role of propolis against alcohol carbon tetrachloride-induced hepatotoxicity in rats. Indian J. Pharmacol. 1997, 29, 76. [Google Scholar]
- Lin, S.C.; Lin, Y.H.; Chen, C.F.; Chung, C.Y.; Hsu, S.H. The hepatoprotective and therapeutic effects of propolis ethanol extract on chronic alcohol-induced liver injuries. Am. J. Chin. Med. 1997, 25, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Marcucci, M.C. Propolis: Chemical composition, biological properties and therapeutic activity. Apidologie 1995, 26, 83–99. [Google Scholar] [CrossRef]
- Hata, J.; Tsuji, T.; Yanagimoto, K. Alcoholic Injury Mitigating Agent. U.S. Patent 9,061,035, 8 April 2015. [Google Scholar]
- Ulven, S.M.; Kirkhus, B.; Lamglait, A.; Basu, S.; Elind, E.; Haider, T.; Berge, K.; Vik, H.; Pedersen, J.I. Metabolic effects of krill oil are essentially similar to those of fish oil but at lower dose of EPA and DHA, in healthy volunteers. Lipids 2011, 46, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Wu, Y.; Wei, W.; Li, Z.; Wang, P.; Zhu, S.; Dong, W. Protective effects of recombinant human cytoglobin against chronic alcohol-induced liver disease in vivo and in vitro. Sci. Rep. 2017, 7, 41647. [Google Scholar] [CrossRef] [PubMed]
- Min, J.A.; Lee, K.; Kim, D.J. The application of minerals in managing alcohol hangover: A preliminary review. Curr. Drug Abuse Rev. 2010, 3, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Gan, L.Q.; Li, S.K.; Zheng, J.C.; Xu, D.P.; Li, H.B. Effects of herbal infusions, tea and carbonated beverages on alcohol dehydrogenase and aldehyde dehydrogenase activity. Food Funct. 2014, 5, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Devi, S.L.; Viswanathan, P.; Anuradha, C.V. Taurine enhances the metabolism and detoxification of ethanol and prevents hepatic fibrosis in rats treated with iron and alcohol. Environ. Toxicol. Pharmacol. 2009, 27, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Jensen, K.; Krogh, H. Alcohol-induced hangover: A double-blind comparison of pyritinol and placebo in preventing hangover symptoms. Q. J. Stud. Alcohol 1973, 34, 1195–1201. [Google Scholar] [PubMed]
- Jung, T.W.; Lee, J.Y.; Shim, W.S.; Kang, E.S.; Kim, S.K.; Ahn, C.W.; Lee, H.C.; Cha, B.S. Rosiglitazone relieves acute ethanol-induced hangover in Sprague—Dawley rats. Alcohol Alcohol. 2006, 41, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Brimberg, B.J.; Kamath, B.L. Therapeutic Composition and Method of Using Same for Treatment of Hangover. U.S. Patent 5,296,241, 22 March 1994. [Google Scholar]
- Jacobs, M.M. Compositions and Methods for the Prevention and Treatment of Alcohol-Induced Hangover Syndrome. U.S. Patent 14,539,776, 14 May 2014. [Google Scholar]
- Hausheer, F.H. Method of Treating Hangover. U.S. Patent 6,077,838, 20 June 2000. [Google Scholar]
- Halamicek, W.A., III. Compositions for Treating Alcohol Hangover. U.S. Patent 8,377,907, 19 February 2013. [Google Scholar]
- Hollander, D.; Tarnawski, A.; Krause, W.J.; Gergely, H. Protective effect of sucralfate against alcohol-induced gastric mucosal injury in the rat: Macroscopic, histologic, ultrastructural, and functional time sequence analysis. Gastroenterology 1985, 88, 366–374. [Google Scholar] [CrossRef]
- Zhao, J.; Chen, H.; Li, Y. Protective effect of bicyclol on acute alcohol-induced liver injury in mice. Eur. J. Pharmacol. 2008, 586, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wang, L.; Zhang, H.; Zhang, D.; Zhang, Z.; Zhang, J. Protective effect of artemisinin on chronic alcohol induced-liver damage in mice. Environ. Toxicol. Pharmacol. 2017, 52, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Kema, V.H.; Khan, I.; Jamal, R.; Vishwakarma, S.K.; Lakki Reddy, C.; Parwani, K.; Patel, F.; Patel, D.; Khan, A.A.; Mandal, P. Protective effects of diallyl sulphide against ethanol-induced injury in rat adipose tissue and primary human adipocytes. Alcohol. Clin. Exp. Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Arafa, H.M.; Sayed-Ahmed, M.M. Protective role of carnitine esters against alcohol-induced gastric lesions in rats. Pharmacol. Res. 2003, 48, 285–290. [Google Scholar] [CrossRef]
- Yuan, Z.Q.; Gao, J.J.; Ou, Y.X.; Zou, Y.G. Observation on therapeutic efficacy of composite Salvia miltiorrhiza injection and Naloxone in acute severe alcoholism. Pract. Clin. Med. 2001, 10, 10–15. [Google Scholar]
- Kang, Y.J.; Zhou, Z. Zinc prevention and treatment of alcoholic liver disease. Mol. Aspects Med. 2005, 26, 391–404. [Google Scholar] [CrossRef] [PubMed]
- Cochrane, P. Composition for Prevention or Treatment of an Alcohol Hangover. U.S. Patent 11,146,747, 8 December 2005. [Google Scholar]
- Sreeraj, G.; George, R.; Thankachen, R.; Sriraam, V.; Shakthi, A.; et al. Studies on the effectiveness and safety of anti hangover drink (Oh! K) in reducing cocktail (alcohol) induced hangover symptoms in adult male social drinkers. Int. J. Herb. Med. 2014, 2, 115–117. [Google Scholar]
- Yoon, T.J.; Jo, S.Y.; Lee, S.J.; Kim, E.Y.; Shin, K.S.; Suh, H.J. Effect of herbal composition, DTS20 on alcohol degradation and anti-inflammatory activity. KSBB J. 2011, 26, 433–438. [Google Scholar] [CrossRef]
- Zheng, W.; Zhang, M.; Zhao, Y.; Wang, Y.; Miao, K.; Wei, Z. Accumulation of antioxidant phenolic constituents in submerged cultures of Inonotus obliquus. Bioresour. Technol. 2009, 100, 1327–1335. [Google Scholar] [CrossRef] [PubMed]
- Twardziok, M.; Kleinsimon, S.; Rolff, J.; Jäger, S.; Eggert, A.; Seifert, G.; Delebinski, C.I. Multiple active compounds from Viscum album L. synergistically converge to promote apoptosis in Ewing sarcoma. PLoS ONE 2016, 11, e0159749. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.h.; Ren, K.; Lu, S.j.; Yang, S.y.; Sun, D.z. Progress of research on Inonotus obliquus. Chin. J. Integr. Med. 2009, 15, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, Y.; Tanaka, M.; Murai, R.; Kuribayashi, K.; Kobayashi, D.; Yanagihara, N.; Watanabe, N. Prophylactic and Therapeutic Effects of Acanthopanax senticosus Harms Extract on Murine Collagen-induced Arthritis. Phytother. Res. 2014, 28, 1513–1519. [Google Scholar] [CrossRef] [PubMed]
- Qian, J.Y.; Liu, D.; Huang, A.G. The efficiency of flavonoids in polar extracts of Lycium chinense Mill fruits as free radical scavenger. Food Chem. 2004, 87, 283–288. [Google Scholar] [CrossRef]
- Kang, B.; Bang, C.Y.; Choung, S.Y.; Choi, K. Oral Intake of Anti-Hangover Substance Increases Metabolizing Capacity of Aldehyde Dehydrogenase 2 in Rat Model: New Therapeutic Potentials for Chronic Itch? J. Allergy Clin. Immunol. 2016, 137, AB254. [Google Scholar] [CrossRef]
- Bang, C.Y.; Kang, B.; Choung, S.Y.; Choi, K. Oral intake of anti-hangover substance increases aldehyde dehydrogenase activity: New preventive and therapeutic potentials for oxidative neuronal injury? Parkinsonism Relat. Disord. 2016, 22, e190. [Google Scholar] [CrossRef]
- Liu, Y.; Du, J.; Yan, M.; Lau, M.Y.; Hu, J.; Han, H.; Yang, O.O.; Liang, S.; Wei, W.; Wang, H.; et al. Biomimetic enzyme nanocomplexes and their use as antidotes and preventive measures for alcohol intoxication. Nat. Nanotechnol. 2013, 8, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Kinjo, T.; Tian, H.; Hamasaki, T.; Teruya, K.; Kabayama, S.; Shirahata, S. Mechanism of the lifespan extension of Caenorhabditis elegans by electrolyzed reduced water?participation of PT nanoparticles. Biosci. Biotechnol. Biochem. 2011, 75, 1295–1299. [Google Scholar] [CrossRef] [PubMed]
| Mitigation Level/Model | Active Component | Production Sources | Refs. |
|---|---|---|---|
| I/h,m,i | isoflavonoids, puerarin, daidzin, daidzein, ibogaine | Extracts of Pueraria lobata, Tabernanthe iboga, Panax ginseng, Salvia miltiorrhiza and Hypericum perforatum | [37,38,39,40,41] |
| I/m,i | Isoflavones, puerarin, daidzein, daidzin, mirificin, and salvianolic acid | Extracts with Kudzu: Hovenia dulcis, Artemisia capillaris, Pueraria thunbergiana, Atractylodes macrocephala, Polyporus umbellatus, Poria cocos, Citrus unshiu, Lycium chinense | [39,41,42] |
| II/m | Flavonoids and antioxidants, artemisin, capillarisin, puerarin, hoventins, ampelopsin, laricetrin | JBU extracts: Artemisia capillaris, Pueraria thunbergiana, Hovenia dulcis, Artractylodes macrocephala, Polyporus umbellatus, Poria cocos, Citrus unshiu, Lycium chinense, Panax ginseng, and Silybum maximum | [43] |
| II/i,c,m | Viscotoxins, flavonoids, triterpene acids, mistletoe lectins, melanins (lanoline alkane triterpenes), polyphenols, lignans, and coumarins, syringic acid, chlorogenic acid, eleutheroside, syringaresinol- di-glucoside, isofraxidin | Extracts: Viscum album L., Inonotus obliquus, Acanthopanax senticosus H. | [139,140,141,142,143] |
| II/i | Viscotoxins, flavonoids, triterpene acids, mistletoe lectins, melanins (lanoline alkane triterpenes), polyphenols, lignans, and coumarins, rutin, chlorogenic acid, protocatechuic acid and citric acid | Extracts: Lycium chinense L., Viscum album L., Inonotus obliquus | [139,144] |
| II/h,m,i | Coptisine, baicalin, palmatine, berberine, and wogonoside | Orengedokotu: Scutellaria root, Coptis rhizome, Gardenia fruit, and Phellodendron bark | [56,57,58,59] |
| II/m | Cinnamic acid, alisol A, and atractyloside III | Goreisan: Aslisma rhizome, A. lancea rhizome, Polyporus sclerotium, Artemisa capilaris flower and Poria sclerotium | [55] |
| II/m,i | Benzoic acid, hydroxychavicol, eugenol, and chavibetol | Extracts: Piper beetle leaf | [63,64,65] |
| II/m,c | Xanthones and prenyl-flavones | Extracts: Cudrania tricuspidata | [66,67] |
| II/h | Dihydromyricetin | Hovenia dulcis | [99] |
| II/i,m | Sesamin | Sesame seeds | [100,101] |
| II/c,i | Magnolol and honokiol | Bark of Magnolia officinalis | [54,107] |
| II/i | Apigenin and alpha-bisabolol | Matricaria recutita | [110] |
| II/h,m | Enzyme cocktail: lipases, amilases, dehydrogenases | Biozyme: Bacillus subtilis natto fermented vegetable broth | [112] |
| II/h,c | Gluthathione, S-adenosyl-l-methionine, vitamin E | Glutathione enriched yeast and rice embryo | [24,113] |
| II/h,i | Phospholipids, omega-3 polyunsaturated fatty acids, eicosapentaenoic acid, docosahexaenoic acid | Krill and Fish oil extracts | [118,119] |
| II/m | Sucralfate | Synthetic | [130] |
| II/h | Salvinorin A + Naloxene | Salvia miltiorrhiza extracts + synthetic | [135] |
| II/m | Enzymes cocktail + ALDH | Enzyme rich soybeans + microbial ALDH | [145,146] |
| II/m | Oxidase, catalase nanocomplexes | Microbial and synthesized | [147] |
| II/m,i | Taurine, pantethine, and lipoic acid | Synthetic | [114,123] |
| II/c,m | Astaxanthin | Green algae Haematococcus pluvialis, flowers of Tagetes erecta, yeast Xanthophyllomyces dendrorhous, and shrimp | [95,96,97] |
| III/m | Flavonoids and antioxidants, artemisin, capillarisin, puerarin, hoventins, ampelopsin, laricetrin | JBU extracts ** | [43] |
| III/h | Complex carbohydrates | Fruits, fruit juices, and bread. | [19,46] |
| III/h | Electrolytes | Fruit juices, soft drinks, electrolyte solutions. | [46] |
| III/h | Tolfenamic acid | Synthetic | [47] |
| III/h,m | Ginsenosides | Panax ginseng | [50] |
| III/m | Heteropolysaccharides, rhamnose, arabinose, galactose and galacturonic acid | Hovenia dulcis | [51,95] |
| III/m | Cinnamic acid, alisol A, and atractyloside III | Goreisan ** | [55] |
| III/h,m,i | Coptisine, baicalin, palmatine, berberine, and wogonoside | Orengendokotu ** | [56,57,58,59] |
| III/h | Glycyrrhizin, liquiritin, baicalin, berberine, and glucuronides | Hangeshashinto: Pinellia tuber, Scutellaria root, processed ginger, Glycyrrhiza, Jujube, Ginseng, and Coptis rhizome | [60] |
| III/h,m,i | Vitamin C and E, polyphenols, carotenoids, flavonoid such as kaempferol, quercetin, and isorhamnetin, taurine, and betalains: betacyanin and betaxanthin | Tuna: Opuntia ficus indica | [90,91,92,93,94] |
| III/m,i | Taraxinic acid, cycloartenol, taraxasterol, inulin, eudesmanolides, tetrahydroridentin, guaianolides, dihydrolactucin, ixerin, glucopyranoside, and various triterpenes | Dandelion root extract: Taraxacum officinale | [78,79] |
| III/h | Polysaccharides | Acanthopanax senticosus | [82] |
| III/c,m | Astaxanthin ** | Green algae Haematococcus pluvialis, flowers of Tagetes erecta, yeast Xanthophyllomyces dendrorhous, and shrimp | [95,96,97] |
| III/h | Rutin, hesperidin, and hesperidin-methyl-chalcone | Citrus fruits | [98] |
| III/h | Dihydromyricetin | Hovenia dulcis ** | [99] |
| III/c,i | Magnolol and honokiol | Bark of Magnolia officinalis ** | [54,107] |
| III/m | Rosiglitazone | Synthetic | [125] |
| III/h | Acetaminophen or aspirin + dextrose + caffeine + magnesium | Synthetic cocktail | [126] |
| III/h | Naproxen sodium + loratadine, cetirizine, ranitidine or famotidine, + B complex + magnesium | Synthetic cocktail | [127] |
| III/h | Mesna and dimesna (sodium ethane sulfonates) | Synthetic | [128] |
| III/h | B complex + amino acids + lecithin glycerophospholipids | synthetic and plant extracts | [129] |
| III/h | l-glutamine, l-cysteine, fumaric acid, succinic acid, vitamin B12, vitamin B1 | Young barley grass juice powder | [137,138] |
| III/i,m | Taurine + sodium citrate + flavour compounds + sugars | Soft carbonated citrus and ginger drinks | [122,123] |
| III/h | Vitamin B6 | Synthetic | [124] |
| IV/m | Heteropolysaccharides, rhamnose, arabinose, galactose and galacturonic acid | Hovenia dulcis ** | [51,95] |
| IV/h | Zinc + plant extracts | Cinchona oficinalis, Lobelia inflata, Nux vomica, Quercus glandium spiritus, Ranunculus bulbosus, and Rhododendron crysanthum | [52] |
| IV/h | Quinidine | Cinchona pubescens | [53] |
| IV/i,c,m | Viscotoxins, flavonoids, triterpene acids, mistletoe lectins, melanins (lanoline alkane triterpenes), polyphenols, lignans, and coumarins, syringic acid, chlorogenic acid, eleutheroside, syringaresinol-di-glucoside, isofraxidin | Extracts **: Viscum album L. + Inonotus obliquus + Acanthopanax senticosus H. | [139,140,141,142,143] |
| IV/i | Viscotoxins, flavonoids, triterpene acids, mistletoe lectins, melanins (lanoline alkane triterpenes), polyphenols, lignans, and coumarins, rutin, chlorogenic acid, protocatechuic acid and citric acid | Extracts **: Lycium chinense L. + Viscum album L. + Inonotus obliquus | [139,144] |
| IV/m,i | Hydroxycitric acid , garcinol, iso-garcinol, hydroxycitric acid lactone, citric acid, oxalic acid, xanthochymol, iso-xanthochymol, and benzhophenone | Kokum: Gracinia indica | [61,62] |
| IV/m | Phenolic terpenoids, flavonoids, tannins, phenolic glycosides, sitosterol, and pro-anthocyanidins oligomers | Volatile oil Origanum majorana L. and grape seed extract Vitis vinifera L. | [83] |
| IV/m,i | Catechins, epicatechins, anthocyanidins, proanthocyanidins, and resveratrol | Grape juice | [84] |
| IV/m | Caffeic acid, polyphenols and melanoidins | Coffee | [87,88] |
| IV/i | Allyl sulfides, allyl cysteines, furtosyl aminoacids, allixinm, selenium, thb-carbolines | Black garlic, Allium sativum | [89] |
| IV/i | Alkaloids: Boldine | Peumus boldus | [71] |
| IV/m,i | Saponins and anthraquinones | Sapindus mukorossi and Rheum emodi | [68] |
| IV/m,i | Achrosin, emodin, anthraquinones, anthrones, apigenin, aurantiobtusin, campesterol, cassiollin chryso-obtusin, chrysophanic acid, chrysarobin, chrysophanol, chrysoeriol | Kasondi extracts: Cassia occidentalis L. | [69,70] |
| IV/c | Trolox | Synthetic | [24] |
| IV/m | Phyllanthin, hypophyllanthin, triacontanal and tricontanol | Phyllanthus amarus Schum. et. Thonn | [72] |
| IV/c,i | Apigenin, kaempferol, luteolin | Extract: Fenugreek seed Trigonella foenum graecum | [73,74,75] |
| IV/i,m | Curcumins | Curcumin: rhizome of Curcuma longa | [76] |
| IV/m | Luteloin, myo-inositol, chrysoeriol, epidermin, quebrachitol | Cardiospermum halicacabum L. | [77] |
| IV/m,i | Taraxinic acid, cycloartenol, taraxasterol, inulin, eudesmanolides, tetrahydroridentin, guaianolides, dihydrolactucin, ixerin, taraxinic acid, glucopyranoside, and various triterpenes | Dandelion root extract: Taraxacum officinale ** | [78,79] |
| IV/m | Lupeol, betulin, sitosterol, and stigmasterol | Hygrophila auriculata root | [80,81] |
| IV/i,m | Sesamin | Sesame seeds ** | [100,101] |
| IV/h,m,i | Sylymarin | Silybum marianum | [102,103,104,105] |
| IV/m,i | Crocetin | Gardenia jaminoides E. | [106] |
| IV/h | Picroside I & II | Kutaki: Picrorhiza kurroa | [108] |
| IV/c | Rubiadin | Rubia cordifolia Linn | [109] |
| IV/m | Apigenin and alpha-bisabolol | Matricaria recutita** | [110] |
| IV/m,i | Crocetin | Gardenia jaminoides E. | [106] |
| IV/h | Picroside I & II | Kutaki: Picrorhiza kurroa | [108] |
| IV/c | Rubiadin | Rubia cordifolia Linn | [109] |
| IV/m | Apigenin and alpha-bisabolol | Matricaria recutita ** | [110] |
| IV/m,i | Dioscin | Rhizomes of Tamus communis and Dioscorea communis | [111] |
| IV/h,m,i | Flavonoids, chalcones and terpenoids, acacetin, apigenin, quercetin, naringenin, or pinobanksin | Red propolis | [115,116,117] |
| IV/m,c | Cytoglobin | Human recombinant expressed in E. coli | [120] |
| IV/h | Antioxidant precursors: Selenium, zinc, copper, vanadium, iron, and magnesium | Synthetic | [121] |
| IV/m | Bicyclol | Synthetic | [131] |
| IV/m | Artemisinin | Artemisia annua | [132] |
| IV/m | Diallyl sulfide | Synthesized and Allium sp. plants | [133] |
| IV/m | Propionyl l-carnitine and acetyl l-carnitine | Synthetized | [134] |
| IV/h,i | Zinc | Various sources (in combination with plant extracts) | [52,136] |
| IV/i | Phycocyanobilin | Spirulina | [114] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martinez-Hurtado, J.L.; Calo-Fernandez, B.; Vazquez-Padin, J. Preventing and Mitigating Alcohol Toxicity: A Review on Protective Substances. Beverages 2018, 4, 39. https://doi.org/10.3390/beverages4020039
Martinez-Hurtado JL, Calo-Fernandez B, Vazquez-Padin J. Preventing and Mitigating Alcohol Toxicity: A Review on Protective Substances. Beverages. 2018; 4(2):39. https://doi.org/10.3390/beverages4020039
Chicago/Turabian StyleMartinez-Hurtado, Juan Leonardo, Bruno Calo-Fernandez, and Jose Vazquez-Padin. 2018. "Preventing and Mitigating Alcohol Toxicity: A Review on Protective Substances" Beverages 4, no. 2: 39. https://doi.org/10.3390/beverages4020039