Emulsions, Foams, and Suspensions: The Microscience of the Beverage Industry
Abstract
:1. Introduction
2. The Colloidal State
- -
- Crystalloid: readily pass through animal and vegetable membranes;
- -
- Colloids: diffused very slowly and cannot pass through membranes.
3. Classification of Emulsions, Foams, and Suspensions
3.1. Emulsions
3.2. Foams
3.3. Suspensions
4. Recent Developments in Beverage Emulsions
4.1. Cream Liqueurs
4.2. Soft Drinks
4.2.1. Carbonated Soft Drinks
4.2.2. Low-Calorie and Mid-Calorie Soft Drinks
4.3. Milk-Coffee and Rich-Milk-Coffee Beverages
4.4. Beverage Concentrates
4.5. Functional Beverages
4.5.1. Dairy-Based Beverages
4.5.2. Milk-Like Beverages
4.5.3. Sport and Energy Drinks
4.6. Bottled Water
4.7. Fruit Drinks
4.8. Sparkling Wine and Beer
5. Final Remarks
Conflicts of Interest
References
- Schramm, L.L. Emulsions, Foams, and Suspensions: Fundamentals and Applications; WILEY-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2005; ISBN 3-527-30743-5. [Google Scholar]
- Green, A.J.; Littlejohn, K.A.; Hooley, P.; Cox, P.W. Formation and stability of food foams and aerated emulsions: Hydrophobins as novel functional ingredients. Curr. Opin. Colloid Interface Sci. 2013, 18, 292–301. [Google Scholar] [CrossRef]
- McClements, D.J.; Decker, E.A.; Weiss, J. Emulsion-based delivery systems for lipophilic bioactive components. J. Food Sci. 2007, 72, 109–124. [Google Scholar] [CrossRef] [PubMed]
- Given, P.S. Encapsulation of Flavors in Emulsions for Beverages. Curr. Opin. Colloid Interface Sci. 2009, 14, 43–47. [Google Scholar] [CrossRef]
- Dickinson, E. Interfacial structure and stability of food emulsions as affected by protein-polysaccharide interactions. Soft Matter 2008, 4, 932–942. [Google Scholar] [CrossRef]
- European Food Emulsifiers Manufacturers Association (EFEMA). Available online: http://www.emulsifiers.org/ViewDocument.asp?ItemId=9 (accessed on 6 December 2017).
- Florence, A.T.; Attwood, D. Physicochemical Principles of Pharmacy: In Manufacture, Formulation and Clinical Use, 6th ed.; Pharmaceutical Press: London, UK, 2016; ISBN 978 0 85711 174 6. [Google Scholar]
- Eagland, D. The colloidal state. Contemp. Phys. 1973, 14, 119–148. [Google Scholar] [CrossRef]
- Petrucci, R.; William, H.; Herring, F.; Madura, J. General Chemistry: Principles and Modern Applications, 9th ed.; Pearson Prentice Hall: Upper Saddle River, NJ, USA, 2007; ISBN-13: 978-0132388269. [Google Scholar]
- Urbina-Villaba, G.; Toro-Mendoza, J.; Lozsan, A.; Garcia-Sucre, M. Brownian Dynamics simulation of emulsions stability. In Emulsions: Structure, Stability and Interactions; Elsevier Academic Press: Amsterdam, The Netherland, 2004; ISBN 9780080472652. [Google Scholar]
- Smoluchowski, M. Mathematical Theory of the Kinetics of the Coagulation of Colloidal Solutions. Z. Phys. Chem. 1917, 19, 129–135. [Google Scholar]
- Lagasse, P.; Columbia University. Colloid. In The Columbia Encyclopedia, 7th ed.; Columbia University Press: New York, NY, USA, 2017. [Google Scholar]
- Shaw, D.J. Introduction to Colloid and Surface Chemistry, 4th ed.; Butterworth-Heinemann: Boston, MA, USA, 1992; pp. 1–20. ISBN 0 7506 1182 0. [Google Scholar]
- Zumdahl, S.S.; Zumdahl, S.A. Chemistry, 7th ed.; Houghton Mifflin Company: Boston, MA, USA, 2007; ISBN-13: 978-0618528448. [Google Scholar]
- Esquena, J. Water-in-water (W/W) emulsions. Curr. Opin. Colloid Interface Sci. 2016, 25, 109–119. [Google Scholar] [CrossRef]
- Nicolai, T.; Murray, B. Particle stabilized water in water emulsions. Food Hydrocoll. 2017, 68, 157–163. [Google Scholar] [CrossRef]
- Calvert, J.B. Colloids. 2002. Available online: https://mysite.du.edu/~jcalvert/phys/colloid.htm#Emul (accessed on 22 December 2017).
- Tarko, T.; Tuszyński, T. Influence of Selected Additives on Colloid Stability of Alcoholic Emulsion Creams. Pol. J. Food Nutr. Sci. 2007, 57, 17–24. [Google Scholar]
- Nowicki, Z.T. Liquors fashionable again. Rynki Alkoholowe 1997, 5, 18–20. [Google Scholar]
- Dalgleish, D.G. Food Emulsions in Emulsions and Emulsion Stability; Sjoblom, J., Ed.; Marcel Dekker: New York, NY, USA, 1996; pp. 287–325. [Google Scholar]
- Miner, P.E. Emulsion Rheology: Creams and Lotions in Rheological Properties of Cosmetics and Toiletries; Laba, D., Ed.; Marcel Dekker: New York, NY, USA, 1993; pp. 313–370. [Google Scholar]
- Banks, W.; Muir, D.D. Effect of alcohol content on emulsion stability of cream liqueurs. Food Chem. 1985, 18, 139–152. [Google Scholar] [CrossRef]
- Dickinson, E.; Narhan, S.K.; Stainsby, G. Stability of Cream Liqueurs Containing Low-Molecular-Weight Surfactants. J. Food Sci. 1989, 54, 77–81. [Google Scholar] [CrossRef]
- Sen, D.J. Fizzy Yummy Liquid: The Most Hankering Item in Age Eight to Eighty. Int. J. Pharm. Res. Bio-Sci. 2014, 3, 532–559. [Google Scholar]
- Stryer, L.; Berg, J.M.; Tymoczko, J.L. Biochemistry, 5th ed.; W. H. Freeman: San Francisco, CA, USA, 2002; ISBN 0-7167-3051-0. [Google Scholar]
- Woodward-Lopez, G.; Kao, J.; Ritchie, L. To what extent have sweetened beverages contributed to the obesity epidemic? Public Health Nutr. 2010, 14, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Schulze, M.B.; Manson, J.A.E.; Ludwig, D.S.; Colditz, G.A.; Stampfer, M.J.; Willet, W.C.; Hu, F.B. Sugar-sweetened beverages, weight gain, and incidence of type 2 diabetes in young and middle-aged women. J. Am. Med. Assoc. 2004, 292, 927–934. [Google Scholar] [CrossRef] [PubMed]
- Vartanian, L.R.; Schwartz, M.B.; Brownell, K.D. Effects of soft drink consumption on nutrition and health: A systematic review and meta-analysis. Am. J. Public Health 2007, 97, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Bhupathiraju, S.N.; Pan, A.; Malik, V.S.; Manson, J.E.; Willett, W.C.; van Dam, R.M.; Hu, F.B. Caffeinated and caffeine-free beverages and risk of type 2 diabetes. Am. J. Clin. Nutr. 2013, 97, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Ashurst, P. Soft Drink and Fruit Juice Problems Solved; Woodhead Publishing Limited: Oxford, UK, 2009; ISBN 978-1-84569-326-8. [Google Scholar]
- Taylor, B. Ingredients, and formulation of carbonated soft drinks. In Carbonated Soft Drinks: Formulation and Manufacture; Steen, D.P., Ashurst, P.R., Eds.; Blackwell Publishing: Oxford, UK, 2006; pp. 48–86. [Google Scholar]
- Tan, C.T. Physical Chemistry in Flavor Products Preparation in Flavor Technology, Physical Chemistry, Modification, and Process; Ho, C.-T., Tan, C.-T., Tong, C.-H., Eds.; American Chemical Society: Washington, DC, USA, 1995; pp. 1–17. [Google Scholar]
- Slavin, J. Beverages and body weight: Challenges in the evidence-based review process of the Carbohydrate Subcommittee from the 2010 Dietary Guidelines Advisory Committee. Nutr. Rev. 2012, 70, S111–S120. [Google Scholar] [CrossRef] [PubMed]
- Francis, N.; Marron, D.; Rueben, K. The Pros, and Cons of Taxing Sweetened Beverages Based on Sugar Content; Research Report; Urban Institute: Washington, DC, USA, 2016; 30p. [Google Scholar]
- McClements, D.J. Food Emulsions: Principles, Practices and Techniques, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2005; ISBN 9781498726689. [Google Scholar]
- Dickinson, E.; Lorient, D. Food Macromolecules, and Colloids; The Royal Society of Chemistry: Cambridge, UK, 1995; ISBN-10: 085404700X. [Google Scholar]
- Kong, Y.; Nikolov, A.; Wasan, D.; Ogawa, A. Emulsion Texture, and Stability: Role of Surfactant Micellar Interactions in the Presence of Proteins. Ind. Eng. Chem. Res. 2008, 47, 9108–9114. [Google Scholar] [CrossRef]
- Ogawa, A.; Cho, H. Role of food emulsifiers in milk coffee beverages. J. Colloid Interface Sci. 2015, 449, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Klein, M.; Aserin, A.; Svitov, I.; Garti, N. Enhanced stabilization of cloudy emulsions with gum Arabic and whey protein isolate. Colloids Surf. B Biointerfaces 2010, 77, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Piorkowski, D.T.; McClements, D.J. Beverage emulsions: Recent developments in formulation, production, and applications. Food Hydrocoll. 2014, 42, 5–41. [Google Scholar] [CrossRef]
- Gürakan, G.C.; Cebeci, A.; Özer, B. Probiotic dairy beverages: Microbiology and technology. In Development and Manufacture of Yogurt and Other Functional Dairy Products; Yildiz, F., Ed.; Taylor & Francis Group: Florence, SC, USA, 2010; pp. 165–195. [Google Scholar]
- Saarela, M. Probiotics as ingredients in functional beverages. In Functional and Speciality Beverage Technology; Paquin, P., Ed.; Woodhead Publishing Limited: Cambridge, UK; CRC Press LLC: Boca Raton, FL, USA, 2009; pp. 55–70. [Google Scholar]
- Talwalkar, A.; Kailasapathy, K. A review of oxygen toxicity in probiotic yogurts: Influence on the survival of probiotic bacteria and protective techniques. Compr. Rev. Food Sci. Food Saf. 2004, 3, 117–124. [Google Scholar] [CrossRef]
- Özer, B.; Kirmaci, H.Y. Functional milks and dairy beverages. Int. J. Dairy Technol. 2010, 63, 1–15. [Google Scholar] [CrossRef]
- Alemany-Costa, L.; González-Larena, M.; García-Llatas, G.; Alegría, A.; Barberá, R.; Sánchez-Siles, L.M.; Lagarda, M.J. Sterol stability in functional fruit beverages enriched with different plant sterol sources. Food Res. Int. 2012, 48, 265–270. [Google Scholar] [CrossRef]
- Prado, F.C.; Parada, J.L.; Pandey, A.; Soccol, C.R. Trends in non-dairy probiotic beverages. Food Res. Int. 2008, 41, 111–123. [Google Scholar] [CrossRef]
- Davoodi, H.; Esmaeili, S.; Mortazavian, A.M. Effects of milk and milk products consumption on cancer: A review. Compr. Rev. Food Sci. Food Saf. 2013, 12, 249–264. [Google Scholar] [CrossRef]
- Corbo, M.R.; Bevilacqua, A.; Petruzzi, L.; Casanova, F.P.; Sinigaglia, M. Functional Beverages: The Emerging Side of Functional Foods. Compr. Rev. Food Sci. Food Saf. 2014, 13, 1192–1206. [Google Scholar] [CrossRef]
- Nyanzi, R.; Jooste, P.J. Cereal-based functional foods. In Probiotics; Rigobelo, E.C., Ed.; InTech: Rijeka, Croatia, 2012; pp. 161–196. [Google Scholar]
- Luana, N.; Rossana, C.; Curiel, J.A.; Kaisa, P.; Marco, G.; Rizzello, C.G. Manufacture and characterization of a yogurt-like beverage made with oat flakes fermented by selected lactic acid bacteria. Int. J. Food Microbiol. 2014, 185, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Gaanappriya, M.; Guhankumar, P.; Kiruththica, V.; Santhiya, N.; Anita, S. Probiotication of fruit juices by Lactobacillus acidophilus. Int. J. Adv. Biotechnol. Res. 2013, 4, 72–77. [Google Scholar]
- Heckman, M.A.; Sherry, K.; Gonzalez de Mejia, E. Energy drinks: An assessment of their market size, consumer demographics, ingredient profile, functionality, and regulations in the United States. Compr. Rev. Food Sci. Food Saf. 2010, 9, 303–317. [Google Scholar] [CrossRef]
- Dikici, S.; Saritas, A.; Kilinc, S.; Guneysu, S.; Gunes, H. Does an energy drink cause a transient ischemic attack? Am. J. Emerg. Med. 2015, 33, 129. [Google Scholar] [CrossRef] [PubMed]
- Gunja, N.; Brown, J.A. Energy drinks: Health risks and toxicity. Res. MJA 2012, 196, 46–49. [Google Scholar] [CrossRef]
- Higgins, J.P.; Tuttle, T.D.; Higgins, C.L. Energy beverages: Content and safety. Mayo Clin. Proc. 2010, 85, 1033–1041. [Google Scholar] [CrossRef] [PubMed]
- Azagba, S.; Langille, D.; Asbridge, M. An emerging adolescent health risk: Caffeinated energy drink consumption patterns among high school students. Prev. Med. 2014, 62, 54–59. [Google Scholar] [CrossRef] [PubMed]
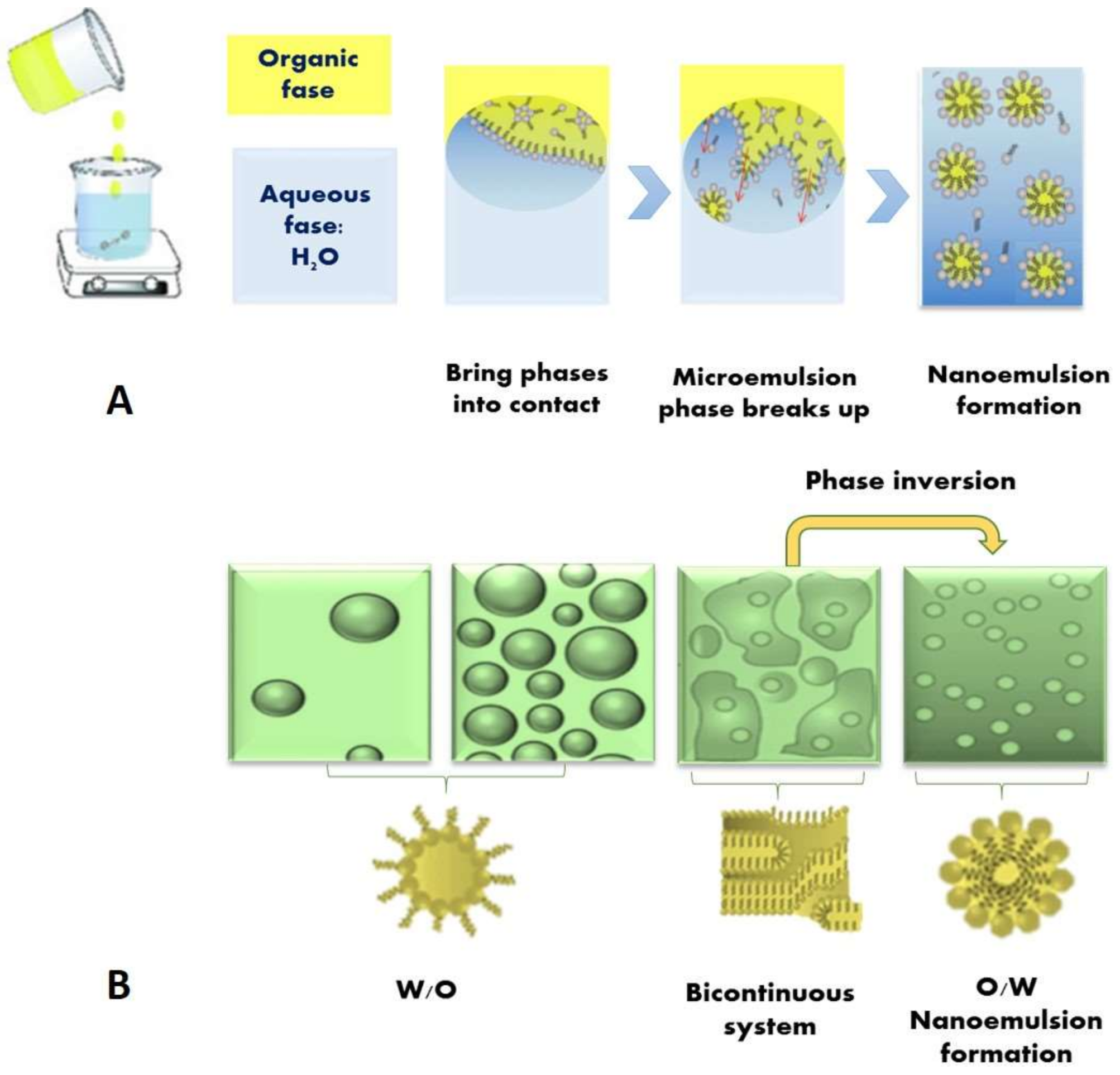
- Komaiko, J.S.; McClements, D.J. Formation of Food-Grade Nanoemulsions Using Low-Energy Preparation Methods: A Review of Available Methods. Compr. Rev. Food Sci. Food Saf. 2016, 15, 331–352. [Google Scholar] [CrossRef]
- Spernath, A.; Aserin, A. Microemulsions as carriers for drugs and nutraceuticals. Adv. Colloid Interface Sci. 2006, 128–130, 47–64. [Google Scholar] [CrossRef] [PubMed]
- Gebhardt, D. All-Natural Cloudy Beverage Emulsions. 2017. Available online: https://sensientfoodcolors.com/en-ap/research-development/natural-cloudy-beverage-emulsions/ (accessed on 24 January 2018).
- Akkarachaneeyakorn, S.; Tinrat, S. Effects of types and amounts of stabilizers on physical and sensory characteristics of cloudy ready-to-drink mulberry fruit juice. Food Sci. Nutr. 2015, 3, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Zecher, D.; Van Coillie, R. Cellulose derivatives. In Thickening and Gelling Agents for Food; Imeson, A., Ed.; Blackie Academic & Professional: London, UK, 1992; pp. 40–65. [Google Scholar]
- Codex Alimentarius Commission. Sodium Carboxymethyl Cellulose (Cellulose gum); GFSA Online; FAO: Rome, Italy, 2016. [Google Scholar]
- Andres-Lacueva, C.; Gaillard, M.; Lopez-Tamames, E.; Lamuela-Raventos, R.M. Influence of variety and aging on foaming properties of sparkling wine (Cava). J. Agric. Food Chem. 1996, 44, 3826–3829. [Google Scholar] [CrossRef]
- Andres-Lacueva, C.; Lamuela-Raventos, R.M.; Buxaderas, S.; Torre-Boronat, M.C. Influence of variety and aging on foaming properties of cava (sparkling wine). J. Agric. Food Chem. 1997, 45, 2520–2525. [Google Scholar] [CrossRef]
- Martínez-Lapuente, L.; Guadalupe, Z.; Ayestarán, B.; Pérez-Magariño, S. Role of major wine constituents in the foam properties of white and rosé sparkling wines. Food Chem. 2015, 174, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Vincenzi, S.; Crapisi, A.; Curioni, A. Foamability of Prosecco wine: Cooperative effects of high molecular weight glycocompounds and wine PR-proteins. Food Hydrocoll. 2014, 34, 202–207. [Google Scholar] [CrossRef]
- Perkowitz, S. Gloriously bubbly. New Sci. 2000, 164, 58–61. [Google Scholar]
- Evans, D.E.; Robinson, L.H.; Sheehan, M.C.; Hill, A.; Skerritt, J.S.; Barr, A.R. Application of immunological methods to differentiate between foam-positive and haze-active proteins originating from malt. J. Am. Soc. Brew. Chem. 2003, 61, 55–62. [Google Scholar]
- Ferreira, M.P.L.V.O.; Jorge, K.; Nogueira, L.C.; Silva, F.; Trugo, L.C. Effects of the combination of hydrophobic polypeptides, ISO-alpha acids, and maltooligosaccharides on beer foam stability. J. Agric. Food Chem. 2005, 53, 4976–4981. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.E.; Surrel, A.; Sheehy, M.; Stewart, D.C.; Robinson, L.H. Comparison of foam quality and the influence of hop alpha-acids and proteins using five foam analysis methods. J. Am. Soc. Brew. Chem. 2008, 66, 1–10. [Google Scholar] [CrossRef]
- Kunimune, T.; Shellhammer, T.H. Foam-Stabilizing Effects and Cling Formation Patterns of ISO-α-acids and Reduced ISO-α-acids in Lager Beer. J. Agric. Food Chem. 2008, 56, 8629–8634. [Google Scholar] [CrossRef] [PubMed]
- Kordialik-Bogacka, E.; Antczak, N. Prediction of Beer Foam Stability from Malt Components. Czech J. Food Sci. 2011, 29, 243–249. [Google Scholar] [CrossRef]
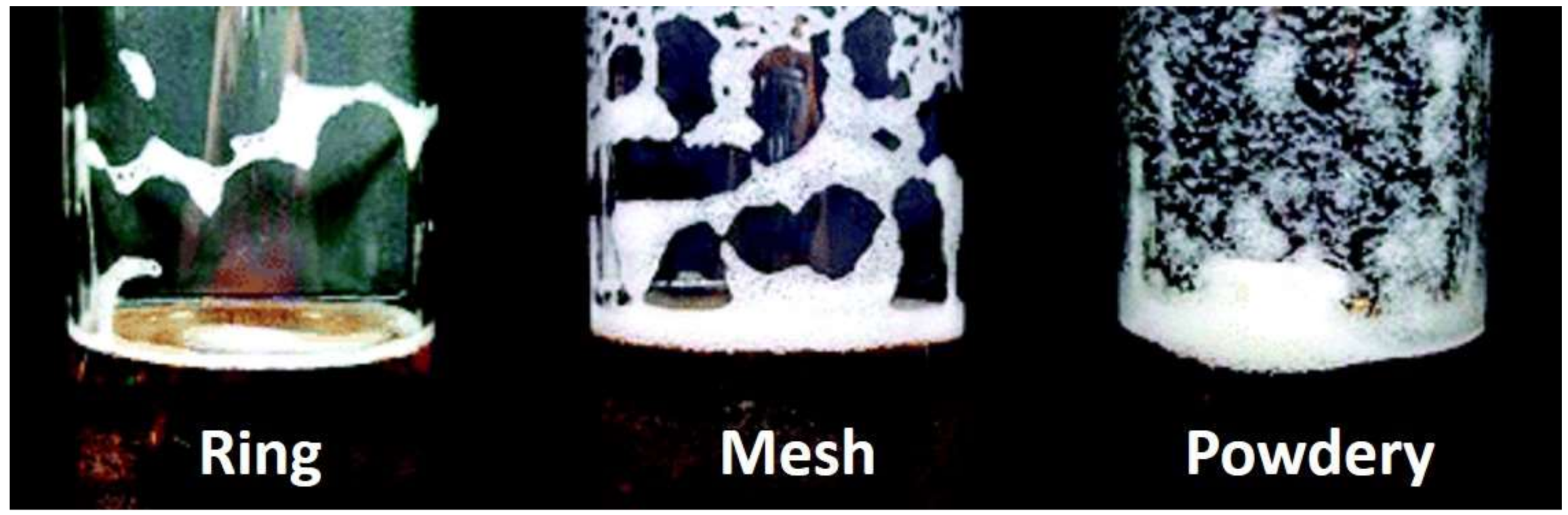
- Evans, D.E.; Sheehan, M.C. Don’t be fobbed off: The substance of beer foam—A review. J. Am. Soc. Brew. Chem. 2002, 60, 47–57. [Google Scholar] [CrossRef]
- Government of Canada. List of Permitted Emulsifying, Gelling, Stabilizing or Thickening Agents [2017-09-25] Document Reference Number: NOM/ADM-0101; NOM/ADM-0088; NOM/ADM-0085; NOM/ADM-0083; NOM/ADM-0069; NOM/ADM-0060; NOM/ADM-0048; NOM/ADM-0044; NOM/ADM-0040; NOM/ADM-0036; NOM/ADM-0015, NOM/ADM-0014, NOM/ADM-0005. Available online: https://www.canada.ca/en/health-canada/services/food-nutrition/food-safety/food-additives/lists-permitted/4-emulsifying-gelling-stabilizing-thickening-agents-2015-09-28.html (accessed on 5 March 2018).



| Dispersing Medium | Dispersed Phase | Name | Examples |
|---|---|---|---|
| Gas | Liquid | Aerosol | Fog, sprays, clouds, mists |
| Gas | Solid | Smoke, dust, airborne bacteria | |
| Liquid | Gas | Foam | Whipped cream, soap suds |
| Liquid | Liquid | Emulsion | Milk, mayonnaise |
| Liquid | Solid | Sol | Paint, ink, starch in water, clays |
| Solid | Gas | Solid foam | Marshmallow, pumice |
| Solid | Liquid | Solid emulsion | Butter, cheese, gels |
| Solid | Solid | Solid sol | Pearl, opals, ruby glass |
| Additive | Permitted in or Upon |
|---|---|
| Acacia Gum/Arabic Gum | Flavored skim milk; ice milk mix. |
| Acacia Gum modified with octenyl succinicanhydride (OSA) | Unstandardized beverages. |
| Agar | Partly skimmed milk; partly skimmed milk with added milk solids; flavored skim milk. |
| Algin/Potassium Alginate | Ale; beer; flavored skim milk. |
| Ammonium Carrageenan | Ale; beer; light beer; malt liquor; flavored milk; flavored skim milk. |
| Ammonium Salt of Phosphorylated Glyceride | Flavored milk; flavored skim milk. |
| Arabino-galactan | Unstandardized beverage bases; Unstandardized beverage mixes. |
| Calcium Alginate | Ale; beer; flavored skim milk. |
| Calcium Carrageenan | Ale; beer; light beer; malt liquor; flavored milk; flavored skim milk; stout. |
| Carboxymethyl Cellulose/Sodium Carboxymethyl Cellulose/Cellulose Gum/Sodium Cellulose Glycolate | Flavored milk; flavored skim milk. |
| Carob Bean Gum | Flavored milk; flavored skim milk. |
| Carrageenan | Ale; beer; light beer; malt liquor; flavored milk; flavored skim milk. |
| Furcelleran/Potassium Furcelleran/Sodium Furcelleran | Ale; beer; light beer; malt liquor; porter; stout. |
| Gelatin | Flavored milk; flavored skim milk. |
| Gellan Gum | Unstandardized beverages. |
| Guar Gum | Flavored milk; flavored skim milk. |
| Hydroxypropyl Methylcellulose/Propylene Glycol Ether of Methylcellulose | Flavored milk; flavored skim milk. |
| Irish Moss Gelose | Ale; beer; flavored milk; flavored skim milk; Stout. |
| Karaya Gum | Flavored milk; flavored skim milk. |
| Lecithin | Flavored milk; flavored skim milk. |
| Methylcellulose | Ale; beer; light beer; malt liquor; porter; Stout. |
| Pectin | Flavored milk; flavored skim milk. |
| Polyoxyethylene (20) Sorbitan Monooleate (Polysorbate 80)/Polyoxyethylene (20) Sorbitan Monostearate (Polysorbate 60) | Unstandardized beverage bases; Unstandardized beverage mixes. |
| Polyoxyethylene (20) Sorbitan Tristearate (Polysorbate 65) | Flavored milk; flavored skim milk. |
| Potassium Carrageenan | Ale; beer; light beer; malt liquor; flavored milk. |
| Propylene Glycol Alginate | Ale; beer; light beer; malt liquor; porter; Stout. |
| Quillaia extract (Type 2) | Oil-based coloring formulations or unstandardized oil-based ingredients for use in non-carbonated water-based flavored and sweetened beverages, or in unstandardized flavored alcoholic beverages or in caffeinated energy drinks, sports drinks or beverages with vitamin and mineral nutrients added, except beverages with vitamins added in accordance with Part D of the Food and Drug Regulations. |
| Sodium Carrageenan | Ale; beer; light beer; malt liquor; flavored milk; flavored skim milk; Stout. |
| Sodium Phosphate, dibasic | Flavored milk; flavored skim milk. |
| Sorbitan Monostearate | Unstandardized beverage bases; Unstandardized beverage mixes. |
| Sucrose Monoesters of Lauric, Palmitic or Stearic Acid | Unstandardized beverages; Unstandardized beverage concentrates; Unstandardized beverage mixes. |
| Xanthan Gum | Fruit-flavored drinks. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vilela, A.; Cosme, F.; Pinto, T. Emulsions, Foams, and Suspensions: The Microscience of the Beverage Industry. Beverages 2018, 4, 25. https://doi.org/10.3390/beverages4020025
Vilela A, Cosme F, Pinto T. Emulsions, Foams, and Suspensions: The Microscience of the Beverage Industry. Beverages. 2018; 4(2):25. https://doi.org/10.3390/beverages4020025
Chicago/Turabian StyleVilela, Alice, Fernanda Cosme, and Teresa Pinto. 2018. "Emulsions, Foams, and Suspensions: The Microscience of the Beverage Industry" Beverages 4, no. 2: 25. https://doi.org/10.3390/beverages4020025









