Effectiveness and Temporal Variation of a Full-Scale Horizontal Constructed Wetland in Reducing Nitrogen and Phosphorus from Domestic Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Sampling and Physiochemical Analysis
2.3. Statistical Analysis
3. Results and Discussion
3.1. Nitrogen Removal
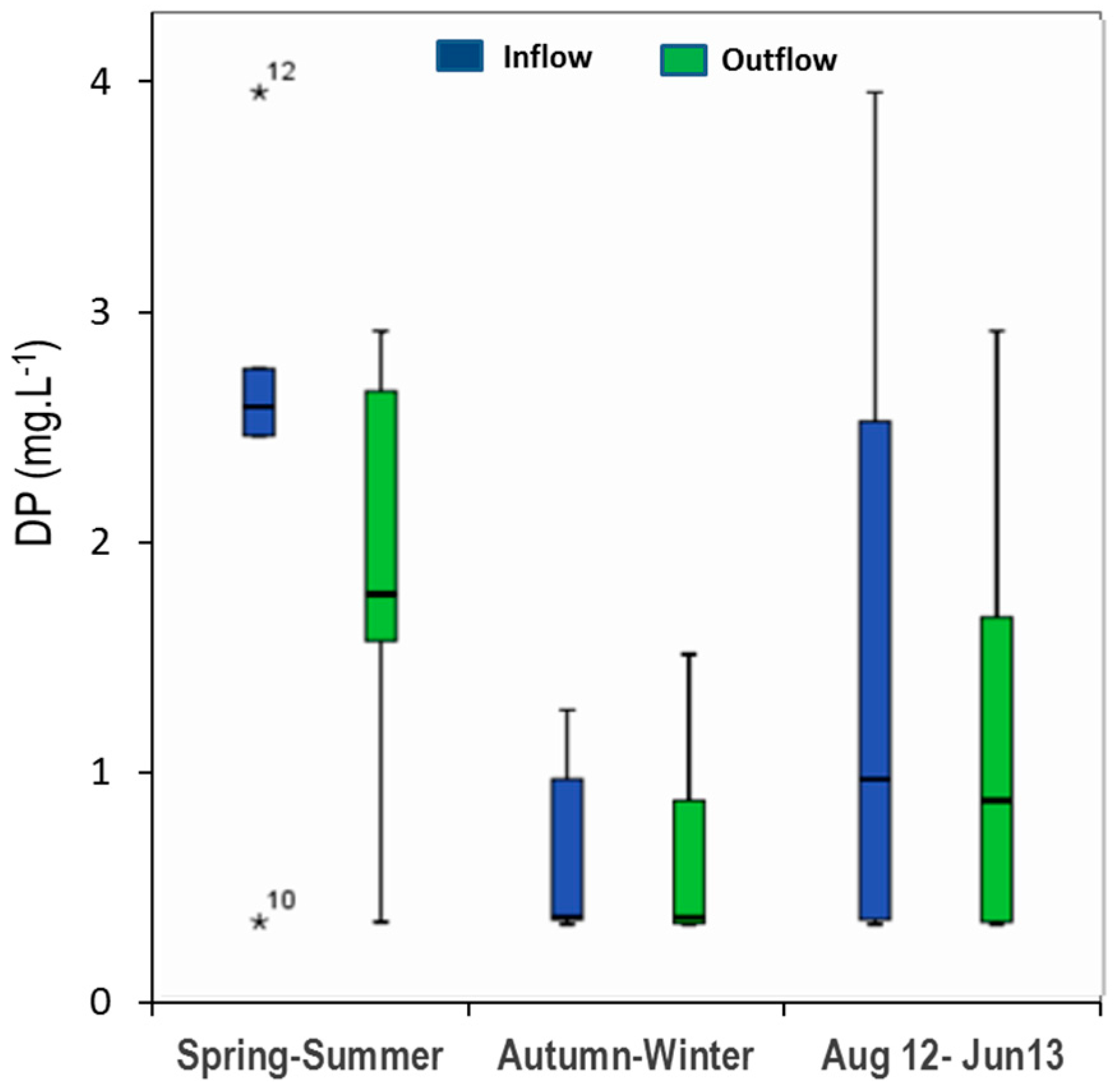
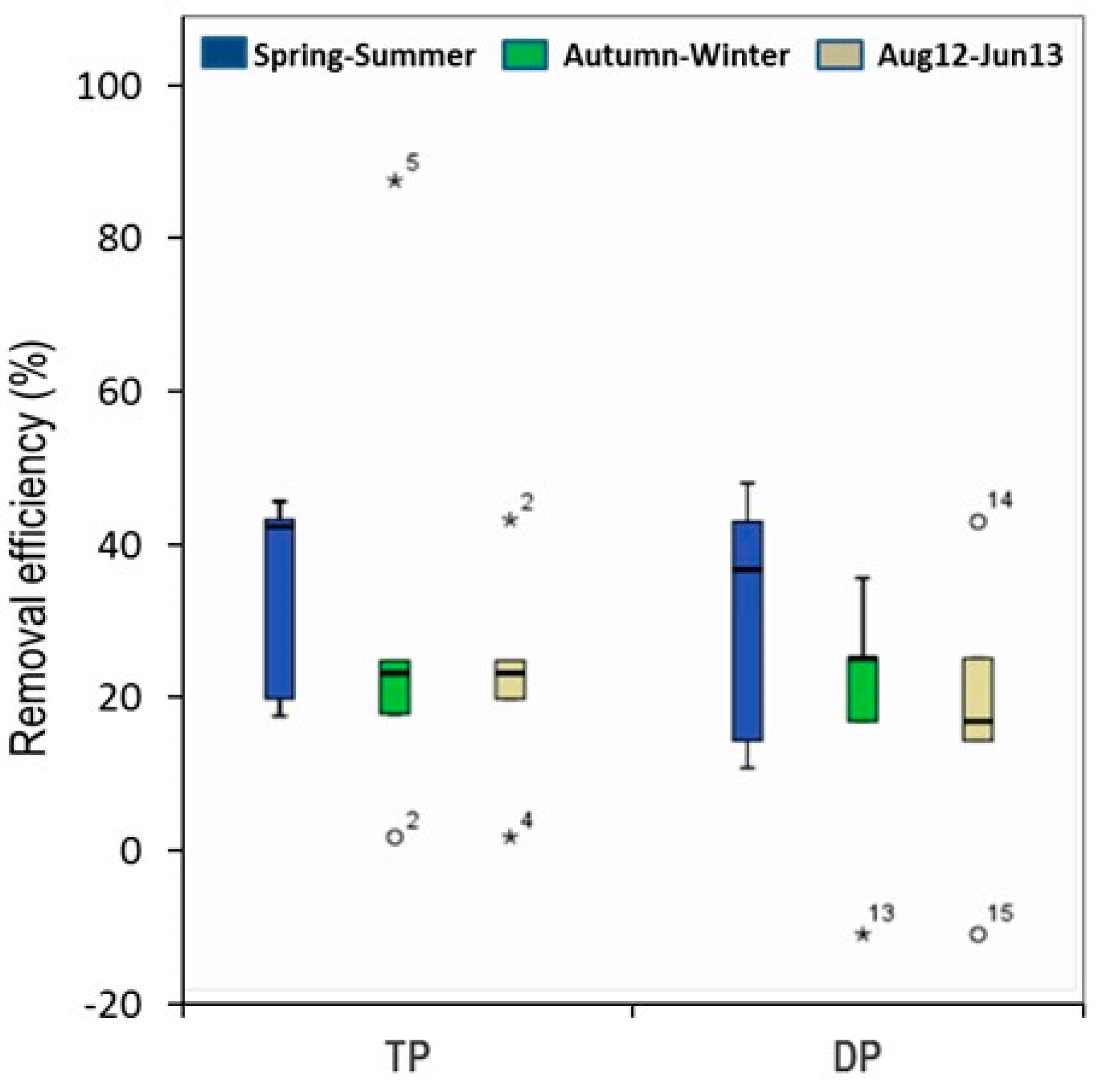
3.2. Phosphorous Removal
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Meng, P.; Pei, H.; Hu, W.; Shao, Y.; Li, Z. How to increase microbial degradation in constructed wetlands: Influencing factors and improvement measures. Bioresour. Technol. 2014, 157, 316–326. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Zhang, S.; Wang, Y.; Li, Y.; Xiao, R.; Li, H. Nitrogen removal and mass balance in newly-formed Myriophyllum aquaticum mesocosm during a single 28-day incubation with swine wastewater treatment. J. Environ. Manag. 2016, 166, 596–604. [Google Scholar] [CrossRef] [PubMed]
- Coban, O.; Kuschk, P.; Wells, N.S.; Strauch, G.; Knoeller, K. Microbial nitrogen transformation in constructed wetlands treating contaminated groundwater. Environ. Sci. Pollut. 2015, 22, 12829–12839. [Google Scholar] [CrossRef] [PubMed]
- Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a Framework for Community Action in the Field of Water Policy. Available online: http://eur-lex.europa.eu/resource.html?uri=cellar:5c835afb-2ec6-4577-bdf8-756d3d694eeb.0004.02/DOC_1&format=PDF (accessed on 28 October 2017).
- Ko, C.H.; Lee, T.M.; Chang, F.C.; Liao, S.P. The correlations between system treatment efficiencies and aboveground emergent macrophyte nutrient removal for the Hsin-Hai Bridge phase II constructed wetland. Bioresour. Technol. 2011, 102, 5431–5437. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Sample, D.J.; Day, S.D.; Grizzard, T.J. Floating treatment wetland nutrient removal through vegetation harvest and observations from a field study. Ecol. Eng. 2015, 78, 15–26. [Google Scholar] [CrossRef]
- Butterworth, E.; Richards, A.; Jones, M.; Mansi, G.; Ranieri, E.; Dotro, G.; Jefferson, B. Performance of four full-scale artificially aerated horizontal flow constructed wetlands for domestic wastewater treatment. Water 2016, 8, 365. [Google Scholar] [CrossRef]
- Verhofstada, M.J.J.M.; Poelenb, M.D.M.; van Kempenc, M.M.L.; Bakker, E.S.; Smolders, A.J.P. Finding the harvesting frequency to maximize nutrient removal in a constructed wetland dominated by submerged aquatic plants. Ecol. Eng. 2017, 106, 423–430. [Google Scholar] [CrossRef]
- Vymazal, J. The use constructed wetlands for nitrogen removal from agricultural drainage: A Review. Sci. Agric. Bohem. 2017, 2, 82–91. [Google Scholar] [CrossRef]
- Dzakpasu, M.; Wang, X.; Zheng, Y.; Ge, Y.; Xiong, J.; Zhao, Y. Characteristics of nitrogen and phosphorus removal by a surface-flow constructed wetland for polluted river water treatment. Water Sci. Technol. 2015, 71, 904–912. [Google Scholar] [CrossRef] [PubMed]
- Vymazal, J. Removal of nutrients in various types of constructed wetlands. Sci. Total Environ. 2007, 380, 48–65. [Google Scholar] [CrossRef] [PubMed]
- Tanner, C.C.; Sukias, J.P.S. Multi-year nutrient removal performance of three constructed wetlands intercepting drainage flows from intensively grazed pastures. J. Environ. Qual. 2011, 40, 620–633. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Zhang, H.; Li, W.; Ke, F. Full-Scale Experiment on Domestic Wastewater Treatment by Combining Artificial Aeration Vertical- and Horizontal-Flow Constructed Wetlands System. Water Air Soil Pollut. 2012, 223, 5673–5683. [Google Scholar] [CrossRef]
- Weerakoon, G.M.P.R.; Jinadasa, K.B.S.N.; Herath, G.B.B.; Mowjood, M.I.M.; van Bruggen, J.J.A. Impact of the hydraulic loading rate on pollutants removal in tropical horizontal subsurface flow constructed wetlands. Ecol. Eng. 2013, 61, 154–160. [Google Scholar] [CrossRef]
- Ding, Y.; Wang, W.; Song, X.; Wang, G.; Wang, Y. Effect of spray aeration on organics and nitrogen removal in vertical subsurface flow constructed wetland. Chemosphere 2014, 117, 502–505. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, A.; Arendacz, M.; Gajewska, M.; Obarska-Pempkowiak, H.; Randerson, P.; Kowalik, P. Removal of organic matter and nitrogen in a HSSF constructed wetland under transient loads. Water Sci. Technol. 2009, 60, 1677–1682. [Google Scholar] [CrossRef] [PubMed]
- Galvão, A.F. Comportamento Hidráulico e Ambiental de Zonas Húmidas Construídas Para o Tratamento de Águas Residuais. Ph.D. Thesis, Instituto Superior Técnico-Universidade de Lisboa, Lisboa, Portugal, 2009. (In Portuguese). [Google Scholar]
- Marecos do Monte, H.; Albuquerque, A. Analysis of constructed wetland performance for irrigation reuse. Water Sci. Technol. 2010, 61, 1699–1705. [Google Scholar] [CrossRef] [PubMed]
- Botequilha, J.R.M. Avaliação do Desempenho das Zonas Húmidas Construídas: Estudo de Caso Barroca D’Alva. Master’s Thesis, Instituto Superior de Agronomia-Universidade de Lisboa, Lisboa, Portugal, 2013. (In Portuguese). [Google Scholar]
- Yu, G.; Tan, M.; Chong, Y.; Long, X. Spatial Variation of Phosphorous Retention Capacity in Subsurface Flow Constructed Wetlands: Effect of Wetland Type and Inflow Loading. PLoS ONE 2015, 10, e0134010. [Google Scholar] [CrossRef] [PubMed]
- Zhai, J.; Xiao, J.; Rahaman, H.M.; John, Y.; Xiao, J. Seasonal variation of nutrient removal in a full-scale artificial aerated hybrid constructed wetland. Water 2016, 8, 551. [Google Scholar] [CrossRef]
- Instituto Português do Mar e da Atmosfera (IPMA) Normais Climatológicas 1971–2000 em Castelo Branco. Available online: http://www.ipma.pt/pt/oclima/normais.clima/1971-2000/005/ (accessed on 13 August 2013).
- Rousseau, D.P.L.; Vanrolleghem, P.A.; De Pauw, N. Model-based design of horizontal subsurface flow constructed treatment wetlands: A review. Water Res. 2004, 38, 1484–1493. [Google Scholar] [CrossRef] [PubMed]
- Vymazal, J. Constructed wetlands with horizontal sub-surface flow and hybrid systems for wastewater treatment. Ecol. Eng. 2005, 25, 478–490. [Google Scholar] [CrossRef]
- Kadlec, R.H.; Wallace, S.D. Treatment Wetlands, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2009; ISBN 978-1-56670-526-4. [Google Scholar]
- Pascoal, S.F.; Sousa, H.P. Tratamento de Águas Residuais Domésticas em Leitos de Macrófitas. Indústria e Ambiente 2013, 80, 29–34. [Google Scholar]
- Duarte, A.A.L.S.; Seco, T.; Peres, J.A.; Bentes, I.; Pinto, J. Sustainability indicators of subsurface flow constructed wetlands in Portuguese small communities. WSEAS Trans. Environ. Dev. 2010, 9, 625–634. [Google Scholar]
- Relvão, A. Sistemas de Tratamento de Efluentes em Aglomerados Urbanos por Leitos de Macrófitas Emergentes; Comissão Coordenação Desenvolvimento Regional do Centro: Coimbra, Portugal, 1999. [Google Scholar]
- International Water Association (IWA). Constructed Wetland for Pollution Control: Processes, Performance, Design and Operation; Scientific and Technical Report 8; IWA Publishing: London, UK, 2000; p. 156. [Google Scholar]
- Vymazal, J.; Kröpfelová, L. Wastewater Treatment in Constructed Wetlands with Horizontal Sub-Surface Flow; Series of Environmental Pollution 14; Springer: Berlin, Germany, 2008; p. 566. ISBN 978-1-4020-8580-2. [Google Scholar]
- APHA-AWWA-WEF Standard Methods for the Examination of Water and Wastewater, 21th ed.; American Public Health Association/American Water Works Association/Water Environment Federation: Washington, DC, USA, 2005; ISBN 9780875530475.
- Vymazal, J. Constructed wetlands for wastewater treatment. Water 2010, 2, 530–549. [Google Scholar] [CrossRef]
- Vera, I.; García, J.; Sáez, K. Performance evaluation of eight years’ experience of constructed wetland systems in Catalonia as alternative treatment for small communities. Ecol. Eng. 2011, 37, 364–371. [Google Scholar] [CrossRef]
- Puigagut, J.; Villasenor, J.; Salas, J.J.; Becares, E.; Garcia, J. Subsurface-flow constructed wetlands in Spain for the sanitation of small communities: A comparative study. Ecol. Eng. 2007, 30, 312–319. [Google Scholar] [CrossRef]
- Mietto, A.; Borin, M. Performance of two small subsurface flow constructed wetlands treating domestic wastewaters in Italy. Environ. Technol. 2013, 34, 1085–1095. [Google Scholar] [CrossRef] [PubMed]
- Simões, M. Avaliação da Influência do Tipo de Enchimento no Rendimento de Leitos com Escoamento Sub-Superficial e Horizontal. Master’s Thesis, Universidade da Beira Interior, Covilhã, Portugal, 2009. (In Portuguese). [Google Scholar]
- Oliveira, J.C. Diagnóstico e Otimização do Tratamento de Águas Residuais em Leitos de Macrófitas. Master’s Thesis, Faculdade de Engenharia, Universidade do Porto, Porto, Portugal, 2007. (In Portuguese). [Google Scholar]
- Hijosa-Valsero, M.; Sidrach-Cardona, R.; Bécares, E. Comparison of interannual removal variation of various constructed wetland types. Sci. Total Environ. 2012, 430, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Akratos, C.; Tsihrintzis, V. Effect of temperature, HRT, vegetation and porous media on removal efficiency of pilot-scale horizontal subsurface flow constructed wetlands. Ecol. Eng. 2007, 29, 173–191. [Google Scholar] [CrossRef]
- Silveira, D.D.; Filho, P.B.; Philippi, L.S.; Kim, B.; Molle, P. Influence of partial saturation on total nitrogen removal in a single-stage French constructed wetland treating raw domestic wastewater. Ecol. Eng. 2015, 77, 257–264. [Google Scholar] [CrossRef]
- Burchell, M.R.; Skaggs, R.W.; Lee, C.R.; Broome, S.; Chescheir, G.M.; Osborne, J. Substrate organic matter to improve nitrate removal in surface-flow constructed wetlands. J. Environ. Qual. 2007, 36, 194–207. [Google Scholar] [CrossRef] [PubMed]
- Shao, X.; Wu, M.; Gu, B.; Chen, Y.; Liang, X. Nutrient Retention in Plant Biomass and Sediments from the Salt Marsh in Hangzhou Bay Estuary, China. Environ. Sci. Pollut. Res. 2013, 20, 6382–6391. [Google Scholar] [CrossRef] [PubMed]
- Dan, T.H.; Quang, L.N.; Chiem, N.H.; Brix, H. Treatment of high-strength wastewater in tropical constructed wetlands planted with Sesbania sesban: Horizontal subsurface flow versus vertical downflow. Ecol. Eng. 2011, 37, 711–720. [Google Scholar] [CrossRef]
- Cui, L.; Li, W.; Zhang, Y.; Wei, J.; Lei, Y.; Zhang, M.; Pan, X.; Zhao, X.; Li, K.; Ma, W. Nitrogen removal in a horizontal subsurface flow constructed wetland estimated using the first-order kinetic model. Water 2016, 8, 514. [Google Scholar] [CrossRef]
- Zhang, D.Q.; Jinadasa, K.B.S.N.; Gersberg, R.M.; Liu, Y.; Tan, S.K.; Jern, W. Application of constructed wetlands for wastewater treatment in tropical and subtropical regions (2000–2013). J. Environ. Sci. 2015, 30, 30–46. [Google Scholar] [CrossRef] [PubMed]
- Tunçsiper, B. Removal of nutrient and bacteria in pilot-scale constructed wetlands. J. Environ. Sci. Health 2007, 42, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Wallace, S.D.; Knight, R.L. Small-Scale Constructed Wetland Treatment Systems—Feasibility, Design Criteria, and O&M Requirements; Water Environment Research Federation and IWA Publishing: London, UK, 2006; ISBN 1-84339-728-5. [Google Scholar]
- García, J.; Rousseau, D.; Morató, J.; Lesage, E.; Matamoros, V.; Bayona, J. Contaminant removal processes in subsurface-flow constructed wetlands: A Review. Environ. Sci. Technol. 2010, 40, 561–661. [Google Scholar] [CrossRef]
- Gagnon, V.; Chazarenc, F.; Kõiv, M.; Brisson, J. Effect of plant species on water quality at the outlet of a sludge treatment wetland. Water Res. 2012, 46, 5305–5315. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.K.; Takashi, I.; Kato, K.; Ietsugu, H.; Tomita, K.; Nagasawa, T. Seasonal efficiency of a hybrid sub-surface flow constructed wetland system in treating milking parlor wastewater at northern Hokkaido. Ecol. Eng. 2013, 53, 257–266. [Google Scholar] [CrossRef]
- Dong, Y.; Wiliński, P.R.; Dzakpasu, M.; Scholz, M. Impact of hydraulic loading rate and season on water contaminant reductions within integrated constructed wetlands. Wetlands 2011, 31, 499–509. [Google Scholar] [CrossRef]
- Rai, U.N.; Upadhyay, A.K.; Singh, N.K.; Dwivedi, S.; Tripathi, R.D. Seasonal applicability of horizontal sub-surface flow constructed wetland for trace elements and nutrient removal from urban wastes to conserve Ganga River water quality at Haridwar, India. Ecol. Eng. 2015, 81, 115–122. [Google Scholar] [CrossRef]
- Crites, R.W. Design Criteria and Practice for Constructed Wetlands. Water Sci. Technol. 1994, 29, 1–6. [Google Scholar]
- Maltais-Landry, G.; Chazarenc, F.; Comeau, Y.; Troesch, S.; Brisson, J. Effects of artificial aeration, macrophyte species, and loading rate on removal efficiency in constructed wetland mesocosms treating fish farm wastewater. J. Environ. Eng. Sci. 2007, 6, 409–414. [Google Scholar] [CrossRef]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mesquita, C.; Albuquerque, A.; Amaral, L.; Nogueira, R. Effectiveness and Temporal Variation of a Full-Scale Horizontal Constructed Wetland in Reducing Nitrogen and Phosphorus from Domestic Wastewater. ChemEngineering 2018, 2, 3. https://doi.org/10.3390/chemengineering2010003
Mesquita C, Albuquerque A, Amaral L, Nogueira R. Effectiveness and Temporal Variation of a Full-Scale Horizontal Constructed Wetland in Reducing Nitrogen and Phosphorus from Domestic Wastewater. ChemEngineering. 2018; 2(1):3. https://doi.org/10.3390/chemengineering2010003
Chicago/Turabian StyleMesquita, Conceição, António Albuquerque, Leonor Amaral, and Regina Nogueira. 2018. "Effectiveness and Temporal Variation of a Full-Scale Horizontal Constructed Wetland in Reducing Nitrogen and Phosphorus from Domestic Wastewater" ChemEngineering 2, no. 1: 3. https://doi.org/10.3390/chemengineering2010003
APA StyleMesquita, C., Albuquerque, A., Amaral, L., & Nogueira, R. (2018). Effectiveness and Temporal Variation of a Full-Scale Horizontal Constructed Wetland in Reducing Nitrogen and Phosphorus from Domestic Wastewater. ChemEngineering, 2(1), 3. https://doi.org/10.3390/chemengineering2010003






