Genotoxicity of Silver Nanoparticles in Lung Cells of Sprague Dawley Rats after 12 Weeks of Inhalation Exposure
Abstract
:1. Introduction
2. Results and Discussion
2.1. Animal Observations: Effect on Body Weight and Lung Weight
| Summary of body weights | ||||||||
|---|---|---|---|---|---|---|---|---|
| GROUP: (mean ± S.E) | Control | N | Low | N | Middle | N | High | N |
| Day 0 | 171.06 ± 1.61 | 17 | 171.04 ± 1.61 | 17 | 171.14 ± 1.61 | 17 | 171.26 ± 1.64 | 17 |
| Day 1 | 192.76 ± 1.91 | 17 | 191.29 ± 1.81 | 17 | 190.39 ± 1.64 | 17 | 193.09 ± 1.83 | 17 |
| Week 1 | 219.44 ± 2.24 | 17 | 218.03 ± 2.07 | 17 | 214.26 ± 1.88 | 17 | 220.14 ± 2.21 | 17 |
| Week 2 | 265.39 ± 2.91 | 17 | 263.12 ± 2.91 | 17 | 258.41 ± 2.66 | 17 | 263.82 ± 3.23 | 17 |
| Week 3 | 297.72 ± 3.66 | 17 | 290.87 ± 4.05 | 17 | 288.19 ± 3.31 | 17 | 296.90 ± 4.40 | 17 |
| Week 4 | 324.52 ± 4.50 | 17 | 315.15 ± 5.61 | 17 | 310.48 ± 4.33 | 17 | 320.94 ± 5.60 | 17 |
| Week 5 | 342.62 ± 5.18 | 17 | 334.66 ± 6.57 | 17 | 330.17 ± 4.71 | 17 | 342.92 ± 6.24 | 17 |
| Week 6 | 352.05 ± 5.69 | 17 | 337.49 ± 6.81 | 17 | 339.65 ± 4.60 | 17 | 349.72 ± 7.33 | 17 |
| Week 7 | 369.36 ± 6.06 | 17 | 362.93 ± 7.20 | 17 | 358.58 ± 5.20 | 17 | 369.94 ± 7.40 | 17 |
| Week 8 | 382.38 ± 6.22 | 17 | 375.19 ± 8.06 | 17 | 371.60 ± 5.57 | 17 | 380.96 ± 7.68 | 17 |
| Week 9 | 395.49 ± 6.58 | 17 | 389.68 ± 8.67 | 17 | 383.28 ± 5.51 | 17 | 393.93 ± 8.10 | 17 |
| Week 10 | 405.93 ± 6.89 | 17 | 400.85 ± 8.93 | 17 | 394.81 ± 5.88 | 17 | 402.02 ± 8.40 | 17 |
| Week 11 | 416.25 ± 7.23 | 17 | 410.86 ± 9.21 | 17 | 404.68 ± 5.87 | 17 | 410.87 ± 9.03 | 17 |
| Week 12 | 419.33 ± 7.22 | 17 | 413.90 ± 9.20 | 17 | 407.70 ± 5.86 | 17 | 413.95 ± 9.03 | 17 |
| Sacrifice (12 weeks exposure) | 397.05 ± 7.23 | 9 | 402.10 ± 13.47 | 9 | 397.16 ± 6.69 | 9 | 394.96 ± 11.72 | 9 |
2.2. Silver Nanoparticle Generation and Particle Characterization
2.3. Histopathogical Evaluation
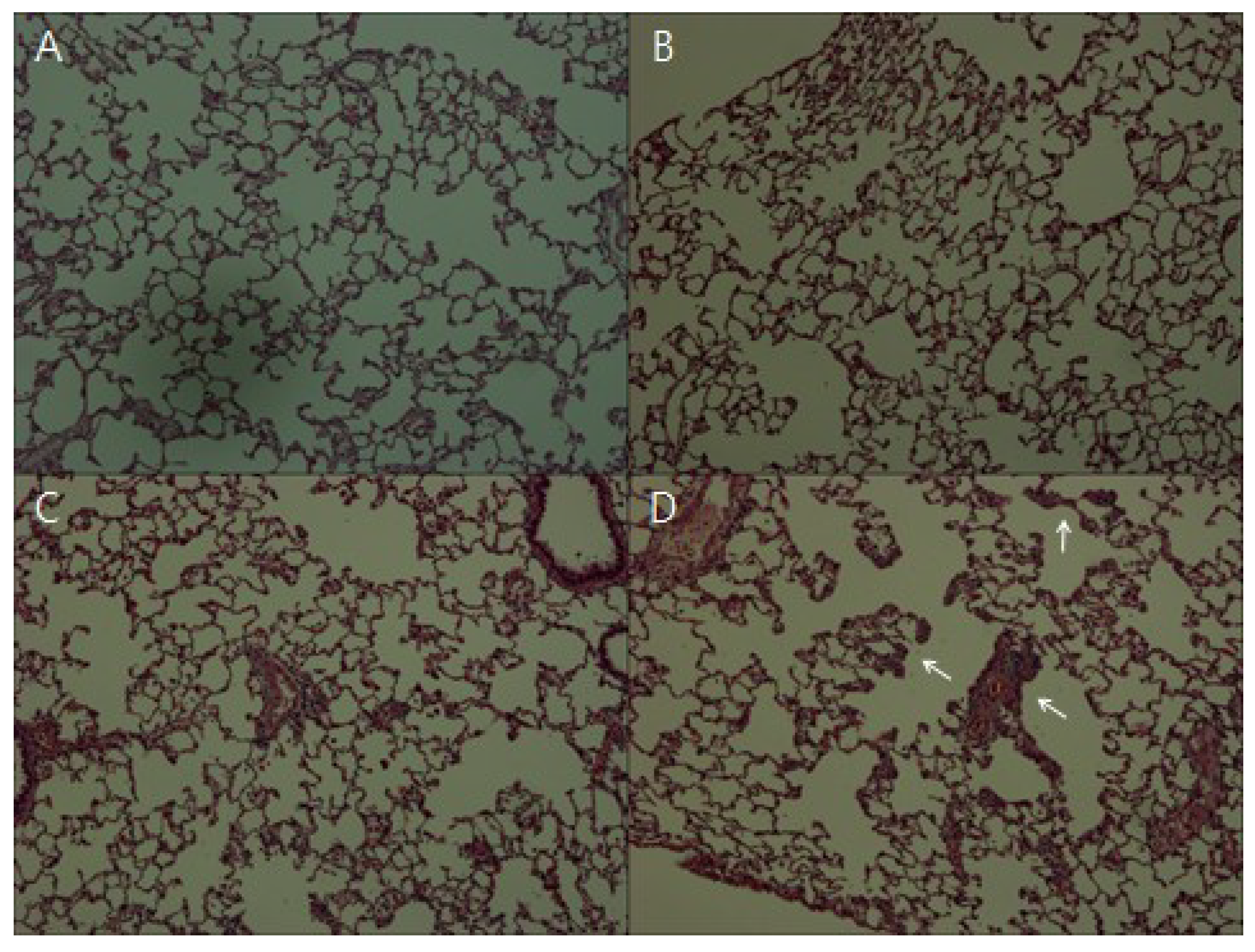
2.4. Silver Concentration in Lungs
2.5. Comet Assay
| Concentration | N | OTM † (Mean ± S.E) |
|---|---|---|
| Control | 5 | 2.93 ± 0.19 |
| Low | 5 | 3.81 ± 0.23 |
| Middle | 4 | 3.40 ± 0.22 |
| High | 4 | 5.16 ± 0.32 ** |
2.6. Other Assays
3. Materials and Methods
3.1. Generation of Silver Nanoparticles
3.2. Monitoring of Inhalation Chamber and Analysis of Silver Nanoparticles
3.3. Animals and Conditions
3.4. Histopathological Evaluation
3.5. Silver Concentration in Lungs
3.6. Cell Isolation
3.7. Single Cell Gel Electrophoresis (Comet Assay)
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Woodrow Wilson International Center for Scholars. A Nanotechnology Consumer Products Inventory. Available online: http://www.nanotechproject.org/consumerproducts (accessed on 25 January 2013).
- Sung, J.H.; Ji, J.H.; Song, K.S.; Lee, J.H.; Choi, K.H.; Lee, S.H.; Yu, I.J. Acute inhalation toxicity of silver nanoparticles. Toxicol. Ind. Health 2010, 27, 149–154. [Google Scholar]
- Ji, J.H.; Jung, J.H.; Kim, S.S.; Yoon, J.U.; Park, J.D.; Choi, B.S.; Chung, Y.H.; Kwon, I.H.; Jeong, J.; Han, B.S.; et al. Twenty-eight-day inhalation toxicity study of silver nanoparticles in Sprague Dawley Rats. Inhal. Toxicol. 2007, 19, 857–871. [Google Scholar] [CrossRef]
- Sung, J.H.; Ji, J.H.; Yun, J.U.; Kim, D.S.; Song, M.Y.; Jeong, J.; Han, B.S.; Han, J.H.; Chung, Y.H.; Kim, J.; et al. Lung function changes in Sprague-Dawley rats after prolonged inhalation exposure to silver nanoparticles. Inhal. Toxicol. 2008, 20, 567–574. [Google Scholar] [CrossRef]
- Sung, J.H.; Ji, J.H.; Park, J.D.; Yoon, J.U.; Kim, D.S.; Jeon, K.S.; Song, M.Y.; Jeong, J.; Han, B.S.; Han, J.H.; et al. Subchronic inhalation toxicity of silver nanoparticles. Toxicol. Sci. 2009, 108, 452–461. [Google Scholar] [CrossRef]
- Song, K.S.; Sung, J.H.; Ji, J.H.; Lee, J.H.; Lee, J.S.; Ryu, H.R.; Lee, J.K.; Chung, Y.H.; Park, H.M.; Shin, B.S.; et al. Recovery from silver-nanoparticle-exposure-induced lung inflammation and lung function changes in Sprague Dawley Rats. Nanotoxicology 2013, 7, 169–180. [Google Scholar] [CrossRef]
- Kim, Y.S.; Kim, J.S.; Cho, H.S.; Rha, D.S.; Kim, J.M.; Park, J.D.; Choi, B.S.; Lim, R.; Chang, H.K.; Chung, Y.H.; et al. Twenty-eight-day oral toxicity, genotoxicity, and gender-related tissue distribution of silver nanoparticles in Sprague-Dawley rats. Inhal. Toxicol. 2008, 20, 575–583. [Google Scholar] [CrossRef]
- Kim, J.S.; Sung, J.H.; Ji, J.H.; Song, K.S.; Lee, J.H.; Kang, C.S.; Yu, I.J. In vivo genotoxicity of silver nanoparticles after 90 day silver nanoparticle inhalation exposure. Saf. Health Work 2011, 2, 65–69. [Google Scholar] [CrossRef]
- Wise, J.P., Sr.; Goodale, B.C.; Wise, S.S.; Craig, G.A.; Pongan, A.F.; Walter, R.B.; Thompson, W.D.; Ng, A.K.; Aboueissa, A.M.; Mitani, H.; et al. Silver nanospheres are cytotoxic and genotoxic to fish cells. Aquat. Toxicol. 2010, 97, 34–41. [Google Scholar] [CrossRef]
- Asharani, P.V.; Hande, M.P.; Valiyaveettil, S. Anti-proliferative activity of silver nanoparticles. BMC Cell Biol. 2009, 10, 65. [Google Scholar] [CrossRef]
- Hussain, S.M.; Hess, K.L.; Gearhart, J.M.; Geiss, K.T.; Schlager, J.J. In vitro toxicity of nanoparticles in BRL 3A rat liver cells. Toxicol. In Vitro 2005, 19, 975–983. [Google Scholar] [CrossRef]
- Kim, S.; Choi, J.E.; Choi, J.; Chung, K.H.; Park, K.; Yi, J.; Ryu, D.Y. Oxidative stress-dependent toxicity of silver nanoparticles in human hepatoma cells. Toxicol. In Vitro 2009, 23, 1076–1084. [Google Scholar] [CrossRef]
- Piao, M.J.; Kang, K.A.; Lee, I.K.; Kim, H.S.; Kim, S.; Choi, J.Y.; Choi, J.; Hyun, J.W. Silver nanoparticles induce oxidative cell damage in human liver cells through inhibition of reduced glutathione and induction of mitochondria-involved apoptosis. Toxicol. Lett. 2011, 201, 92–100. [Google Scholar] [CrossRef]
- Yoon, K.Y.; Hoon, B.J.; Park, J.H.; Hwang, J. Susceptibility constants of Escherichia coli and Bacillus subtilis to silver and copper nanoparticles. Sci. Total Environ. 2007, 373, 572–575. [Google Scholar] [CrossRef]
- Lu, W.; Senapati, D.; Wang, S.; Tovmachenko, O.; Singh, A.K.; Yu, H.; Ray, P.C. Effect of surface coating on the toxicity of silver nanomaterials on human skin keratinocytes. Chem. Phys. Lett. 2010, 487, 92–96. [Google Scholar] [CrossRef]
- Kim, J.S.; Song, K.S.; Sung, J.H.; Ryu, H.R.; Choi, B.G.; Cho, H.S.; Lee, J.K.; Yu, I.J. Genotoxicity, acute oral and dermal toxicity, eye and dermal irritation and corrosion and skin sensitisation evaluation of silver nanoparticles. Nanotoxicology 2013, 7, 953–960. [Google Scholar] [CrossRef]
- Arora, S.; Jain, J.; Rajwade, J.M.; Paknikar, K.M. Interactions of silver nanoparticles with primary mouse fibroblasts and liver cells. Toxicol. Appl. Pharmacol. 2009, 236, 310–318. [Google Scholar] [CrossRef]
- Xia, T.; Kovochich, M.; Brant, J.; Hotze, M.; Sempf, J.; Oberley, T.; Sioutas, C.; Yeh, J.I.; Wiesner, M.R.; Nel, A.E. Comparison of the abilities of ambient and manufactured nanoparticles to induce cellular toxicity according to an oxidative stress paradigm. Nano Lett. 2006, 6, 1794–1807. [Google Scholar] [CrossRef]
- Chi, Z.; Liu, R.; Zhao, L.; Qin, P.; Pan, X.; Sun, F.; Hao, X. A new strategy to probe the genotoxicity of silver nanoparticles combined with cetylpyridine bromide. Spectrochim. Acta A 2009, 72, 577–581. [Google Scholar] [CrossRef]
- Jung, J.H.; Oh, H.C.; Noh, H.S.; Ji, J.H.; Kim, S.S. Metal nanoparticle generation using a small ceramic heater with a local heating area. J. Aerosol Sci. 2006, 37, 1662–1670. [Google Scholar] [CrossRef]
- National Institute for Occupational Safety and Health (NIOSH). NIOSH Manual of Analytical Methods, Method No 7300; Centers for Disease Control and Prevention: Cincinnati, OH, USA, 2003.
- Kumaravel, T.S.; Jha, A.N. Reliable Comet assay measurements for detecting DNA damage induced by ionising radiation and chemicals. Mutat. Res. 2006, 605, 7–16. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cho, H.S.; Sung, J.H.; Song, K.S.; Kim, J.S.; Ji, J.H.; Lee, J.H.; Ryu, H.R.; Ahn, K.; Yu, I.J. Genotoxicity of Silver Nanoparticles in Lung Cells of Sprague Dawley Rats after 12 Weeks of Inhalation Exposure. Toxics 2013, 1, 36-45. https://doi.org/10.3390/toxics1010036
Cho HS, Sung JH, Song KS, Kim JS, Ji JH, Lee JH, Ryu HR, Ahn K, Yu IJ. Genotoxicity of Silver Nanoparticles in Lung Cells of Sprague Dawley Rats after 12 Weeks of Inhalation Exposure. Toxics. 2013; 1(1):36-45. https://doi.org/10.3390/toxics1010036
Chicago/Turabian StyleCho, Hyun Sun, Jae Hyuck Sung, Kyung Seuk Song, Jin Sik Kim, Jun Ho Ji, Ji Hyun Lee, Hyeon Ryol Ryu, Kangho Ahn, and Il Je Yu. 2013. "Genotoxicity of Silver Nanoparticles in Lung Cells of Sprague Dawley Rats after 12 Weeks of Inhalation Exposure" Toxics 1, no. 1: 36-45. https://doi.org/10.3390/toxics1010036
APA StyleCho, H. S., Sung, J. H., Song, K. S., Kim, J. S., Ji, J. H., Lee, J. H., Ryu, H. R., Ahn, K., & Yu, I. J. (2013). Genotoxicity of Silver Nanoparticles in Lung Cells of Sprague Dawley Rats after 12 Weeks of Inhalation Exposure. Toxics, 1(1), 36-45. https://doi.org/10.3390/toxics1010036





