Macular Carotenoid Supplementation Improves Visual Performance, Sleep Quality, and Adverse Physical Symptoms in Those with High Screen Time Exposure
Abstract
:1. Introduction
2. Materials and Methods
2.1. Macular Carotenoid Supplementation
2.2. Measurement of Macular Pigment Optical Density (MPOD)
2.3. Measurement of Temporal Vision (CFF)
2.4. Physical Indicators of Excessive Screen Time
2.5. Sleep Quality
2.6. Contrast Sensitivity Testing
2.7. Disability Glare/Photostress Recovery
2.8. Statistical Analysis
3. Results
3.1. Baseline Results
3.2. Effects of Supplementation
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gentile, D.A.; Reimer, R.A.; Nathanson, A.I.; Walsh, D.A.; Eisenmann, J.C. Protective effects of parental monitoring of children’s media use: A prospective study. JAMA Pediatr. 2014, 168, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Tosini, G.; Ferguson, I.; Tsubota, K. Effects of blue light on the circadian system and eye physiology. Mol Vis. 2016, 22, 61–72. [Google Scholar] [PubMed]
- Chang, A.M.; Aeschbach, D.; Duffy, J.F.; Czeisler, C.A. Evening use of light-emitting eReaders negatively affects sleep, circadian timing, and next-morning alertness. Proc. Natl. Acad. Sci. USA 2015, 112, 1232–1237. [Google Scholar] [CrossRef] [PubMed]
- The Vision Council Digital Eye Strain Report. 2015. Available online: http://www.thevisioncouncil.org/sites/default/files/2416_VC_2016EyeStrain_Report_WEB.pdf (accessed on 14 April 2017).
- The Nielsen Total Audience Report, Q1 2016. Available online: www.nielsen.com/us/en/insights/reports/2016/the-total-audience-report-q1-2016.html (accessed on 10 April 2017).
- Hansraj, K.K. Assessment of stresses in the cervical spine caused by posture and position of the head. Surg. Technol. Int. 2014, 25, 277–279. [Google Scholar] [PubMed]
- Toomingas, A.; Hagberg, M.; Heiden, M.; Richter, H.; Westergren, K.E.; Tornqvist, E.W. Risk factors, incidence and persistence of symptoms from the eyes among professional computer users. Work 2014, 47, 291–301. [Google Scholar] [PubMed]
- Porcar, E.; Pons, A.M.; Lorente, A. Visual and ocular effects from the use of flat-panel displays. Int. J. Ophthalmol. 2016, 9, 881–885. [Google Scholar] [PubMed]
- Chen, J.; Liang, Y.; Mai, C.; Zhong, X.; Qu, C. General Deficit in Inhibitory Control of Excessive Smartphone Users: Evidence from an Event-Related Potential Study. Front Psychol. 2016, 7, 511. [Google Scholar] [CrossRef] [PubMed]
- Gowrisankaran, S.; Sheedy, J.E. Computer vision syndrome: A review. Work 2015, 52, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Hiromoto, K.; Kuse, Y.; Tsuruma, K.; Tadokoro, N.; Kaneko, N.; Shimazawa, M.; Hara, H. Colored lenses suppress blue light-emitting diode light-induced damage in photoreceptor-derived cells. J. Biomed. Opt. 2016, 21, 35004. [Google Scholar] [CrossRef] [PubMed]
- Chamorro, E.; Bonnin-Arias, C.; Pérez-Carrasco, M.J.; de Luna, J.M.; Vázquez, D.; Sánchez-Ramos, C. Effects of light-emitting diode radiations on human retinal pigment epithelial cells in vitro. Photochem. Photobiol. 2013, 89, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Humphries, J.M.; Khachik, F. Distribution of lutein, zeaxanthin, and related geometrical isomers in fruit, vegetables, wheat, and pasta products. J. Agric. Food Chem. 2003, 51, 1322–1327. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Landrum, J.T.; Friedes, L.M.; Gomez, C.M.; Kilburn, M.D.; Menendez, E.; Vidal, I.; Wang, W. Distribution of lutein and zeaxanthin stereoisomers in the human retina. Exp. Eye Res. 1997, 64, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Seddon, J.M.; Ajani, U.A.; Sperduto, R.D.; Hiller, R.; Blair, N.; Burton, T.C.; Farber, M.D.; Gragoudas, E.S.; Haller, J.; Miller, D.T.; et al. Dietary carotenoids, vitamins A, C, and E, and advanced age-related macular degeneration. Eye-disease Case-control Study Group. JAMA 1994, 272, 1413–1420. [Google Scholar] [CrossRef] [PubMed]
- Richer, S.; Stiles, W.; Statkute, L.; Pulido, J.; Frankowski, J.; Rudy, D.; Pei, K.; Tsipursky, M.; Nyland, J. Double-masked, placebo-controlled, randomized trial of lutein and antioxidant supplementation in the intervention of atrophic age-related macular degeneration: The Veterans LAST study (Lutein Antioxidant Supplementation Trial). Optometry 2004, 75, 216–230. [Google Scholar] [CrossRef]
- Futterman, S. Metabolism and Photochemistry in the Retina. In Adler’s Physiology of the Eye, 6th ed.; Moses, R.A., Ed.; C.V. Mosby Company: St. Louis, MO, USA, 1975; pp. 406–419. [Google Scholar]
- Stringham, J.M.; O’Brien, K.J.; Stringham, N.T. Contrast Sensitivity and Lateral Inhibition Are Enhanced With Macular Carotenoid Supplementation. Invest. Ophthalmol. Vis. Sci. 2017, 58, 2291–2295. [Google Scholar] [CrossRef] [PubMed]
- Nolan, J.M.; Power, R.; Stringham, J.; Dennison, J.; Stack, J.; Kelly, D.; Moran, R.; Akuffo, K.O.; Corcoran, L.; Beatty, S. Enrichment of Macular Pigment Enhances Contrast Sensitivity in Subjects Free of Retinal Disease: Central Retinal Enrichment Supplementation Trials-Report 1. Invest. Ophthalmol. Vis. Sci. 2016, 57, 3429–3439. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Qiu, Q.H.; Wu, X.W.; Cai, Z.Y.; Xu, S.; Liang, X.Q. Lutein supplementation improves visual performance in Chinese drivers: 1-year randomized, double-blind, placebo-controlled study. Nutrition 2013, 29, 958–964. [Google Scholar] [CrossRef] [PubMed]
- Loughman, J.; Nolan, J.M.; Howard, A.N.; Connolly, E.; Meagher, K.; Beatty, S. The impact of macular pigment augmentation on visual performance using different carotenoid formulations. Invest. Ophthalmol. Vis. Sci. 2012, 53, 7871–7880. [Google Scholar] [CrossRef] [PubMed]
- Hammond, B.R., Jr.; Wooten, B.R. CFF thresholds: Relation to macular pigment optical density. Ophthalmic Physiol. Opt. 2005, 25, 315–319. [Google Scholar] [CrossRef] [PubMed]
- Renzi, L.M.; Hammond, B.R., Jr. The relation between the macular carotenoids, lutein and zeaxanthin, and temporal vision. Ophthalmic Physiol. Opt. 2010, 30, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Stringham, N.T.; Stringham, J.M. Temporal Visual Mechanisms May Mediate Compensation for Macular Pigment. Perception 2015, 44, 1400–1415. [Google Scholar] [CrossRef] [PubMed]
- Stringham, J.M.; Fuld, K.; Wenzel, A.J. Action spectrum for photophobia. J. Opt. Soc. Am. A Opt. Image Sci. Vis. 2003, 10, 1852–1858. [Google Scholar] [CrossRef]
- Wenzel, A.J.; Fuld, K.; Stringham, J.M.; Curran-Celentano, J. Macular pigment optical density and photophobia light threshold. Vis. Res. 2006, 28, 4615–4622. [Google Scholar] [CrossRef] [PubMed]
- Stringham, J.M.; Garcia, P.V.; Smith, P.A.; McLin, L.N.; Foutch, B.K. Macular pigment and visual performance in glare: benefits for photostress recovery, disability glare, and visual discomfort. Invest Ophthalmol. Vis. Sci. 2011, 52, 7406–7415. [Google Scholar] [CrossRef] [PubMed]
- Hammond, B.R., Jr.; Fletcher, L.M.; Elliott, J.G. Glare disability, photostress recovery, and chromatic contrast: Relation to macular pigment and serum lutein and zeaxanthin. Invest. Ophthalmol. Vis. Sci. 2013, 54, 476–481. [Google Scholar] [CrossRef] [PubMed]
- Stringham, J.M.; Hammond, B.R. Macular pigment and visual performance under glare conditions. Optom. Vis Sci. 2008, 85, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Curran-Celentano, J.; Hammond, B.R., Jr.; Ciulla, T.A.; Cooper, D.A.; Pratt, L.M.; Danis, R.B. Relation between dietary intake, serum concentrations, and retinal concentrations of lutein and zeaxanthin in adults in a Midwest population. Am. J. Clin. Nutr. 2001, 74, 796–802. [Google Scholar] [PubMed]
- Stringham, J.M.; Stringham, N.T. Serum and retinal responses to three different doses of macular carotenoids over 12 weeks of supplementation. Exp. Eye Res. 2016, 151, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wooten, B.R.; Hammond, B.R.; Land, R.; Snodderly, D.M. A practical method of measuring macular pigment optical density. Invest. Ophthalmol. Vis. Sci. 1999, 40, 2481–2489. [Google Scholar] [PubMed]
- Wooten, B.R.; Hammond, B.R.; Smollon, W. Assessment of the validity of heterochromatic flicker photometry for measuring macular pigment optical density in normal subjects. Optom. Vis. Sci. 2005, 82, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Stringham, J.M.; Hammond, B.R.; Nolan, J.M.; Wooten, B.R.; Mammen, A.; Smollon, W.; Snodderly, D.M. The utility of using customized heterochromatic flicker photometry (cHFP) to measure macular pigment in patients with age-related macular degeneration. Exp. Eye Res. 2008, 87, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Smollon, W.E., Jr.; Wooten, B.R.; Hammond, B.R. Stimulus edge effects in the measurement of macular pigment using heterochromatic flicker photometry. J. Biomed. Opt. 2015, 20, 115004. [Google Scholar] [CrossRef] [PubMed]
- Buysse, D.J.; Reynolds, C.F., III; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Hurvich, L.M.; Jameson, D. An opponent-process theory of color vision. Psychol. Rev. 1957, 64, 384–404. [Google Scholar] [CrossRef]
- Vielma, A.; Delgado, L.; Elgueta, C.; Osorio, R.; Palacios, A.G.; Schmachtenberg, O. Nitric oxide amplifies the rat electroretinogram. Exp. Eye Res. 2010, 91, 700–709. [Google Scholar] [CrossRef] [PubMed]
- Reddy, S.C.; Low, C.K.; Lim, Y.P.; Low, L.L.; Mardina, F.; Nursaleha, M.P. Computer vision syndrome: A study of knowledge and practices in university students. Nepal. J. Ophthalmol. 2013, 5, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Tsai, J.W.; Hannibal, J.; Hagiwara, G.; Colas, D.; Ruppert, E.; Ruby, N.F.; Heller, H.C.; Franken, P.; Bourgin, P. Melanopsin as a sleep modulator: Circadian gating of the direct effects of light on sleep and altered sleep homeostasis in Opn4(-/-) mice. PLoS Biol. 2009, 7, e1000125. [Google Scholar] [CrossRef] [PubMed]
- Hannibal, J.; Christiansen, A.T.; Heegaard, S.; Fahrenkrug, J.; Kiilgaard, J.F. Melanopsin-expressing human retinal ganglion cells: Subtypes, distribution, and intraretinal connectivity. J. Comp. Neurol. 2017, 525, 1934–1961. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Robredo, P.; Sádaba, L.M.; Salinas-Alamán, A.; Recalde, S.; Rodríguez, J.A.; García-Layana, A. Effect of lutein and antioxidant supplementation on VEGF expression, MMP-2 activity, and ultrastructural alterations in apolipoprotein E-deficient mouse. Oxid. Med. Cell. Longev. 2013, 2013, 213505. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Kijlstra, A.; van der Veen, R.L.; Makridaki, M.; Murray, I.J.; Berendschot, T.T. The effect of lutein supplementation on blood plasma levels of complement factor D, C5a, and C3d. PLoS ONE. 2013, 8, e73387. [Google Scholar] [CrossRef] [PubMed]
- Stringham, N.T.; Holmes, P.V.; Stringham, J.M. Supplementation with macular carotenoids reduces psychological stress, serum cortisol, and suboptimal symptoms of physical and emotional health in young adults. Nutr. Neurosci. 2017, 15, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Akerstedt, T. Psychosocial stress and impaired sleep. Scand. J. Work Environ. Health 2006, 32, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Duncan, G.; Wormstone, I.M.; Davies, P.D. The aging human lens: Structure, growth, and physiological behaviour. Br. J. Ophthalmol. 1997, 81, 818–823. [Google Scholar] [CrossRef] [PubMed]
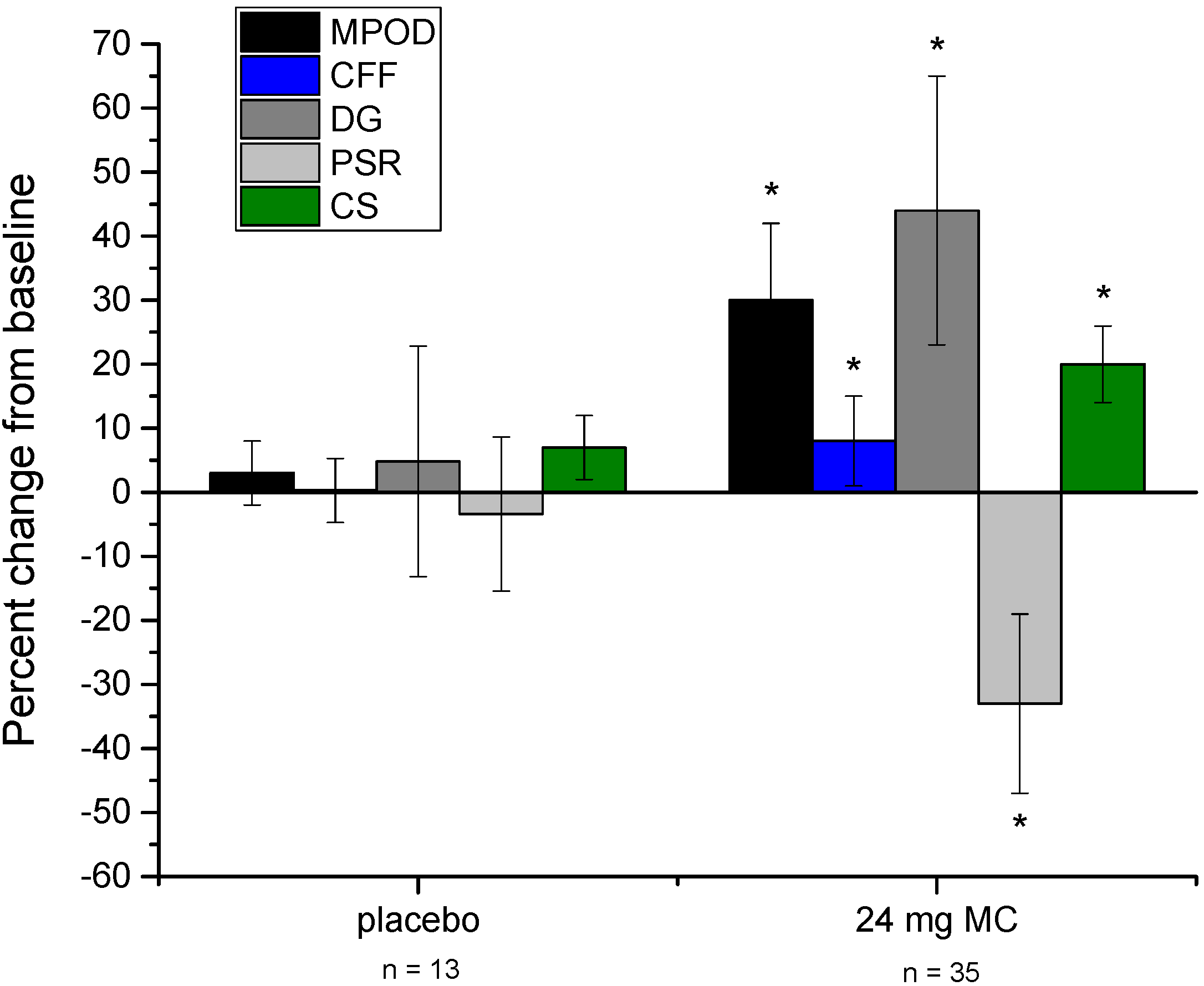
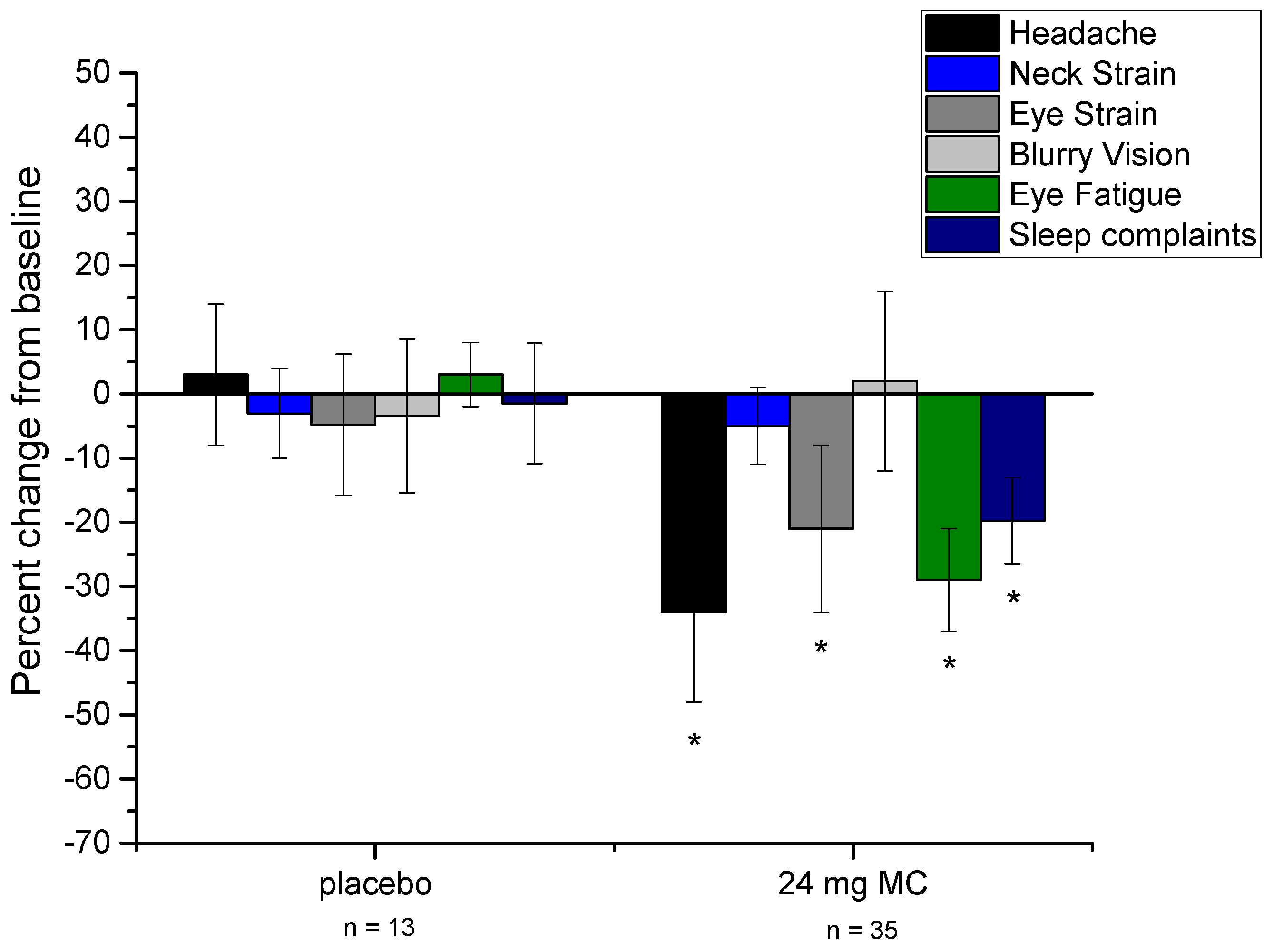
| Variable | Placebo Group (n = 13) | Macular Carotenoid Group (n = 35) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline | SD | 3 months | SD | 6 months | SD | Baseline | SD | 3 months | SD | 6 months | SD | |
| MPOD | 0.372 | 0.119 | 0.368 | 0.135 | 0.383 | 0.132 | 0.376 | 0.133 | 0.421 | 0.151 | 0.472* | 0.144 |
| Total Number of Undesirable Sleep Symptoms | 5.46 | 2.06 | 5.69 | 2.69 | 5.38 | 2.02 | 4.74 | 2.11 | 3.89* | 1.55 | 3.8* | 1.51 |
| CFF (Hz) | 25.23 | 1.8 | 25.33 | 1.74 | 25.38 | 1.84 | 25.05 | 2.13 | 26.31* | 1.69 | 27.12* | 1.96 |
| CS threshold (percent contrast) | 4.25 | 0.38 | 4.21 | 0.33 | 4.01 | 0.31 | 4.63 | 0.46 | 4.13 | 0.39 | 3.73* | 0.34 |
| Headache Frequency (weekly) | 2.65 | 1.21 | 2.57 | 1.16 | 2.73 | 1.19 | 2.78 | 1.12 | 2.02 | 0.94 | 1.83* | 0.91 |
| Eye strain (weekly) | 2.11 | 2.61 | 1.96 | 2.54 | 2.01 | 2.72 | 2.4 | 2.42 | 2.04 | 2.11 | 1.71* | 1.82 |
| Eye Fatigue (weekly) | 2.88 | 2.43 | 2.91 | 2.75 | 2.96 | 2.62 | 3.03 | 2.77 | 2.61 | 2.64 | 2.18* | 2.23 |
| Blurry vision (weekly) | 1.33 | 2.46 | 1.37 | 2.55 | 1.29 | 2.29 | 1.43 | 2.54 | 1.47 | 2.48 | 1.46 | 2.59 |
| Neck strain (weekly) | 3.36 | 3.81 | 3.41 | 3.77 | 3.27 | 3.69 | 3.55 | 3.9 | 3.53 | 3.74 | 3.38 | 3.49 |
| DG (nominal light output) | 101.78 | 29.15 | 105.44 | 31.25 | 103.87 | 33.35 | 81.67 | 23.85 | 106.31 | 29.83 | 117.66* | 35.42 |
| PSR (seconds) | 10.54 | 3.41 | 10.79 | 4.02 | 10.11 | 3.61 | 11.03 | 3.99 | 8.89 | 3.41 | 7.41* | 2.91 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stringham, J.M.; Stringham, N.T.; O’Brien, K.J. Macular Carotenoid Supplementation Improves Visual Performance, Sleep Quality, and Adverse Physical Symptoms in Those with High Screen Time Exposure. Foods 2017, 6, 47. https://doi.org/10.3390/foods6070047
Stringham JM, Stringham NT, O’Brien KJ. Macular Carotenoid Supplementation Improves Visual Performance, Sleep Quality, and Adverse Physical Symptoms in Those with High Screen Time Exposure. Foods. 2017; 6(7):47. https://doi.org/10.3390/foods6070047
Chicago/Turabian StyleStringham, James M., Nicole T. Stringham, and Kevin J. O’Brien. 2017. "Macular Carotenoid Supplementation Improves Visual Performance, Sleep Quality, and Adverse Physical Symptoms in Those with High Screen Time Exposure" Foods 6, no. 7: 47. https://doi.org/10.3390/foods6070047
APA StyleStringham, J. M., Stringham, N. T., & O’Brien, K. J. (2017). Macular Carotenoid Supplementation Improves Visual Performance, Sleep Quality, and Adverse Physical Symptoms in Those with High Screen Time Exposure. Foods, 6(7), 47. https://doi.org/10.3390/foods6070047





