The Content of Tocols in South African Wheat; Impact on Nutritional Benefits
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Extraction of Tocols
2.3. Analytical High Performance Liquid Chromatography (HPLC)
2.4. Data Analysis
3. Results
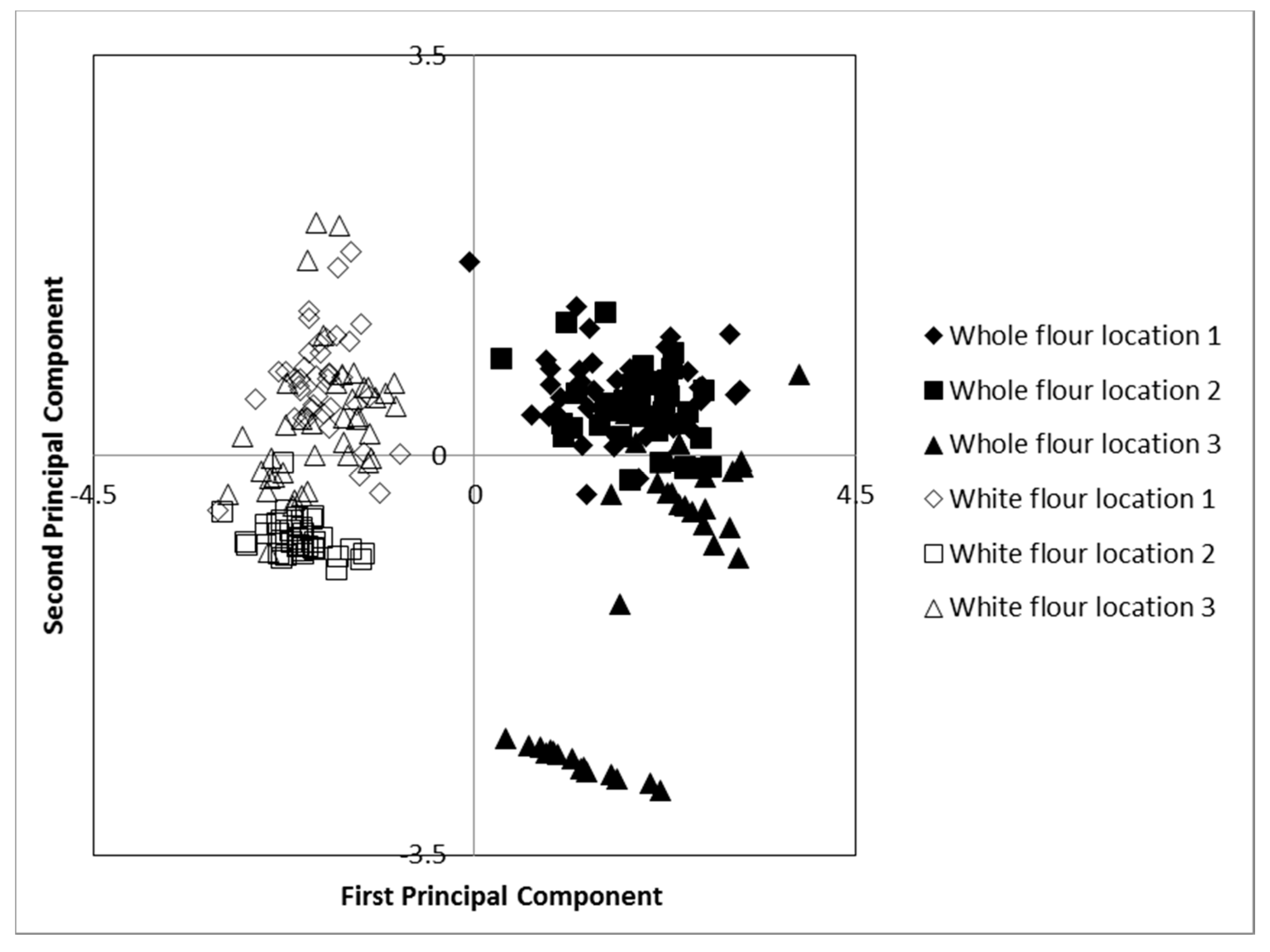
3.1. Importance of Genotype, Environment, and Fractionation on Tocols Content and Composition
3.2. Effect of Flour Type on Content and Composition of Tocols
3.3. Effect of Cultivar and Growing Location on Content and Composition of Tocols
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tilahun, D.; Shiferaw, E.; Johansson, E.; Hailu, F. Genetic variability of Ethiopian bread wheat genotypes (Triticum aestivum L.) using agro-morphological traits and their gliadin content. Afr. J. Agric. Res. 2016, 11, 330–339. [Google Scholar]
- Husenov, B.; Makhkamov, M.; Garkava-Gustavsson, L.; Muminjanov, H.; Johansson, E. Breeding for wheat quality to assure food security of a staple crop: The case study of Tajikistan. Agric. Food Secur. 2015, 4, 9. [Google Scholar] [CrossRef]
- Zielinski, H.; Kozlowska, H. Antioxidant activity and total phenolics in selected cereal grains and their different morphological fractions. J. Agric. Food Chem. 2000, 48, 2008–2016. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.; Larsson, H.; Kuktaite, R.; Johansson, E. Mineral composition of organically grown wheat genotypes: contribution to daily merals intake. Int. J. Environ. Res. Public Health 2010, 7, 3442–3456. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.; Larsson, H.; Kuktaite, R.; Olsson, M.E.; Johansson, E. Carotenoid content in organically produced wheat: Relevance for human nutritional health on consumption. Int. J. Environ. Res. Public Health 2015, 12, 14068–14083. [Google Scholar] [CrossRef]
- Johansson, E.; Hussain, A.; Kuktaite, R.; Andersson, S.C.; Olsson, M. Contribution of organically grown crops to human health. Int. J. Environ. Res. Public Health 2014, 11, 3870–3898. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Scientific opinion on dietary reference values for vitamin E as α-tocopherol. EFSA Panel on dietetic products, nutrition and allergies (NDA). EFSA J. 2015, 13, 4149. [Google Scholar]
- Wolf, G. The discovery of the antioxidant function of vitamin E: The contribution of Henry A. Mattill. J. Nutr. 2005, 135, 363–366. [Google Scholar] [PubMed]
- Dörmann, P. Functional diversity of tocochromanols in plants. Planta 2007, 225, 269–276. [Google Scholar]
- United States Department of Agriculture. USDA National Nutrient Database for Standard Reference. Available online: https://ndb.nal.usda.gov/ndb/ (accessed on 1 August 2017).
- Hussain, A.; Larsson, H.; Olsson, M.E.; Kuktaite, R.; Grausgruber, H.; Johansson, E. Is organically produced wheat a source of tocopherols and tocotrienols for health food? Food Chem. 2012, 132, 1789–1795. [Google Scholar] [CrossRef]
- Dror, D.K.; Allen, L.H. Vitamin E deficiency in developing countries. Food Nutr. Bull. 2011, 2, 124–143. [Google Scholar] [CrossRef] [PubMed]
- Piironen, V.; Lampi, A.; Ekholm, P.; Salmenkallio-Marttila, M.; Liukkonen, K. Micronutrients and phytochemicals in wheat grain. In Wheat Chemistry and Technology, 4th ed.; Khalil, K.K., Shewry, P.R., Eds.; American Association of Cereal Chemists, Inc.: Eagan, MN, USA, 2009; pp. 179–210. [Google Scholar]
- Lampi, A.; Nurmi, T.; Piironen, V. Efects of the environment and genotype on tocopherols and tocotrienols in wheat in the HEALTHGRAIN diversity screen. J. Agric. Food Chem. 2010, 58, 9306–9313. [Google Scholar] [CrossRef] [PubMed]
- Wrigley, C.W. Wheat: A unique grain for the world. In Wheat Chemistry and Technology, 4th ed.; Khalil, K.K., Shewry, P.R., Eds.; American Association of Cereal Chemists, Inc.: Eagan, MN, USA, 2009; pp. 1–15. [Google Scholar]
- Bramley, P.M.; Elmadfa, I.; Kafatos, A.; Kelly, F.J.; Manios, Y.; Roxborough, H.E.; Schuch, W.; Sheehy, P.J.A.; Wagner, K.H. Vitamin E. J. Sci. Food Agric. 2000, 80, 913–938. [Google Scholar] [CrossRef]
- Fratianni, A.; Caboni, M.F.; Irano, M.; Panfili, G. A critical comparison between traditional methods and supercritical carbon dioxide extraction for the determination of tocochromanols in cereals. Eur. Food Res. Technol. 2002, 215, 353–358. [Google Scholar] [CrossRef]
- Labuschagne, M.T.; Mkhatywa, N.; Wentzel, B.; Johansson, E.; Van Biljon, A. Tocochromanol concentration, protein composition and baking quality of white flour of South African wheat cultivars. J. Food Compos. Anal. 2014, 33, 127–131. [Google Scholar] [CrossRef]
- Panfili, G.; Fratianni, A.; Irano, M. Normal phase-high performance liquid chromatography method for the determination of tocopherols and tocotrienols in cereals. J. Agric. Food Chem. 2003, 51, 3940–3944. [Google Scholar] [CrossRef] [PubMed]
- Malik, A.H.; Kuktaite, R.; Johansson, E. Combined effect of genetic and environmental factors on the accumulation of proteins in the wheat grain and their relationships to bread-making quality. J. Cereal Sci. 2013, 57, 170–174. [Google Scholar] [CrossRef]
- Moreira-Ascarrunz, S.D.; Larsson, H.; Prieto-Linde, M.L.; Johansson, E. Mineral nutritional yield and nutrient density of locally adapted wheat genotypes under organic production. Foods 2016, 5, 89. [Google Scholar] [CrossRef] [PubMed]
- European Parliament and Council. Regulation (EU) No 1169/2011 of the European Parliament and of the Council of 25 October 2011 on the Provision of Food Information to Consumers. Off. J. Eur. Commun. 2011, L304, 18. [Google Scholar]
- Hidalgo, A.; Brandolino, A. Tocol stability during bread, biscuit and pasta processing from wheat. J. Cereal Sci. 2010, 52, 254–259. [Google Scholar] [CrossRef]
- Zielinski, H.; Ciska, E.; Kozlowska, H. The cereal grains: Focus on vitamin E. Czech J. Food Sci. 2001, 19, 182–188. [Google Scholar]
- Nurit, E.; Lyan, B.; Pujos-Guillot, E.; Branlard, G.; Piquet, A. Change in B and E vitamin and lutein, β-sitosterol contents in industrial milling fractiona and during toasted bread production. J. Cereal Sci. 2016, 69, 290–296. [Google Scholar] [CrossRef]
- Andersson, S.C.; Olsson, M.E.; Gustavsson, K.-E.; Johansson, E.; Rumpunen, K. Tocopherols in rose hips (Rosa spp.) during ripening. J. Sci. Food Agric. 2012, 92, 2116–2121. [Google Scholar] [CrossRef] [PubMed]
- Andersson, S.C.; Rumpunen, K.; Johansson, E.; Olsson, M.E. Tocopherols and tocotrienols in Sea Buchthorn (Hoppophae rhamnoides) berries during ripening. J. Agric. Food Chem. 2008, 56, 6701–6706. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Zamarreno, M.M.; Bustamante-Rangel, M.; Sierra-Manzano, S.; Verdugo-Jara, M.; Carabias-Martinez, R. Simultaneous extraction of tocotrienols and tocopherols from cereals using pressurized liquid extraction prior to LC determination. J. Sep. Sci. 2009, 32, 1430–1436. [Google Scholar] [CrossRef] [PubMed]
- Sramkova, Z.; Gregova, A.; Sturdik, E. Chemical composition and nutritional quality of wheat grain. Acta Chim. Slovaca 2009, 2, 115–138. [Google Scholar]
- Tiwari, U.; Cummins, E. Nutritional importance and effect of processing on tocols in cereals. Trends Food Sci. Technol. 2009, 20, 511–520. [Google Scholar] [CrossRef]
- Serbinova, E.; Kagan, V.; Han, D.; Packer, L. Free-radical recycling and intramembrane mobility in the antioxidant properties of alpha-tocopherol and alpha-tocotrienol. Free Radic. Biol. Med. 1991, 10, 263–275. [Google Scholar] [CrossRef]
- Schaffer, S.; Müller, W.E.; Eckert, G.P. Tocotrienols: Constitutional effects in aging and disease. J. Nutr. 2005, 135, 151–154. [Google Scholar] [PubMed]
- Shibata, A.; Kobayashi, T.; Asai, A.; Eitsuka, T.; Oikawa, S.; Miyazawa, T.; Nakagawa, K. High purity tocotrienols attenuate atherosclerotic lesion formation in apoE-KO mice. J. Nutr. Biochem. 2017, 48, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Alawin, O.A.; Ahmed, R.A.; Dronamraju, V.; Briski, K.; Sylvester, P.W. γ-Tocotrienol-indusced disruption of lipid rafts in human breast cancer cells in associated with a reduction in exosome heregulin content. J. Nutr. Biochem. 2017, 48, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Allen, L.; Ramlingam, L.; Menikdiwela, K.; Scoggin, S.; Shen, C.-L.; Tomison, M.D.; Kaur, G.; Dufour, J.M.; Chung, E.; Kalupahana, N.S.; et al. Effects of delta-tocotrienol on obesity-related adipocyte hypertrophy, inflammation and hepatic steatosis in high-fat-fed mice. J. Nutr. Biochem. 2017, 48, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.-Y.; Che, H.-L.; Tan, D.M.-Y.; Teng, K.-T. Bioavailability to tocotrienols: Evidence in human studies. Nutr. Metab. 2014, 11, 5. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, T.; Shibata, A.; Sookwong, P.; Kawakami, Y.; Eitsuka, T.; Asai, A.; Oikawa, S.; Nakagawa, K. Antiangiogenic and anticancer potential of unsaturated vitamin E (tocotrienol). J. Nutr. Biochem. 2009, 20, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Sen, C.K.; Khanna, S.; Roy, S.; Packer, L. Molecular basis of vitamin E action—Tocotrienol potently inhibits glutamate-induced pp60(c-Src) kinase activation and death of HT4 neuronal cells. J. Biol. Chem. 2000, 275, 13049–13055. [Google Scholar] [CrossRef] [PubMed]
- Tonini, T.; Rossi, F.; Claudio, P.P. Molecular basis of angiogenesis and cancer. Oncogene 2003, 22, 6549–6556. [Google Scholar] [CrossRef] [PubMed]
- Miscellaneous Nutrients. Available online: https://www.dcnutrition.com/miscellaneous-nutrients/tocotrienols/ (accessed on 1 August 2017).
- Lampi, A.; Nurmi, T.; Ollilainen, V.; Piironen, V. Tocopherols and tocotrienols in wheat genotypes in the HEALTHGRAIN diversity screen. J. Agric. Food Chem. 2008, 56, 9716–9721. [Google Scholar] [CrossRef] [PubMed]
- Okarter, N.; Liu, C.; Sorrells, M.; Liu, R.H. Phytaochemical content and antioxidant activity of six diverse varieties of whole wheat. Food Chem. 2010, 119, 249–257. [Google Scholar] [CrossRef]
- Wong, R.S.; Radhakrishnan, A.K. Tocotrienol research: Past into present. Nutr. Res. 2012, 70, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Fardet, A.; Rock, E.; Remesy, C. Is the in vitro antioxidant potential of whole-grain cereals and cereal products well reflected in vivo? J. Cereal Sci. 2008, 48, 258–276. [Google Scholar] [CrossRef]
- Shewry, P.R.; Piironen, V.; Lampi, A.-M.; Edelmann, M.; Kariluoto, S.; Nurmi, T.; Fernandez-Orozco, R.; Ravel, C.; Charmet, G.; Andersson, A.A.M.; et al. The HEALTHGRAIN wheat diversity screen: Effects of genotype and environment on phytochemicals and dietary fiber components. J. Agric. Food Chem. 2010, 58, 9291–9298. [Google Scholar] [CrossRef] [PubMed]
- Miller, H.E.; Rigelhof, F.; Marquart, L.; Prakash, A.; Kanter, M. Antioxidant content of whole grain beakfast cereals, fruits and vegetables. J. Am. Coll. Nutr. 2000, 19, 312–319. [Google Scholar] [CrossRef]
- Baublis, A.J.; Lu, C.; Clydesdale, F.M.; Decker, E.A. Potential of wheat-based breakfast cereals as a source of dietary antioxidants. J. Am. Coll. Nutr. 2000, 19, 308–311. [Google Scholar] [CrossRef]
- Perez-Jimenez, J.; Saura-Calixto, F. Literature data may underestimate the actual antioxidant capacity of cerreals. J. Agric. Food Chem. 2005, 53, 5036–5040. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, A.; Brandolino, A. Protein, ash, lutein and tocols distribution in einkorn (Triticum monococcum L. subsp. monococcum) seed fractions. Food Chem. 2008, 107, 444–448. [Google Scholar] [CrossRef]
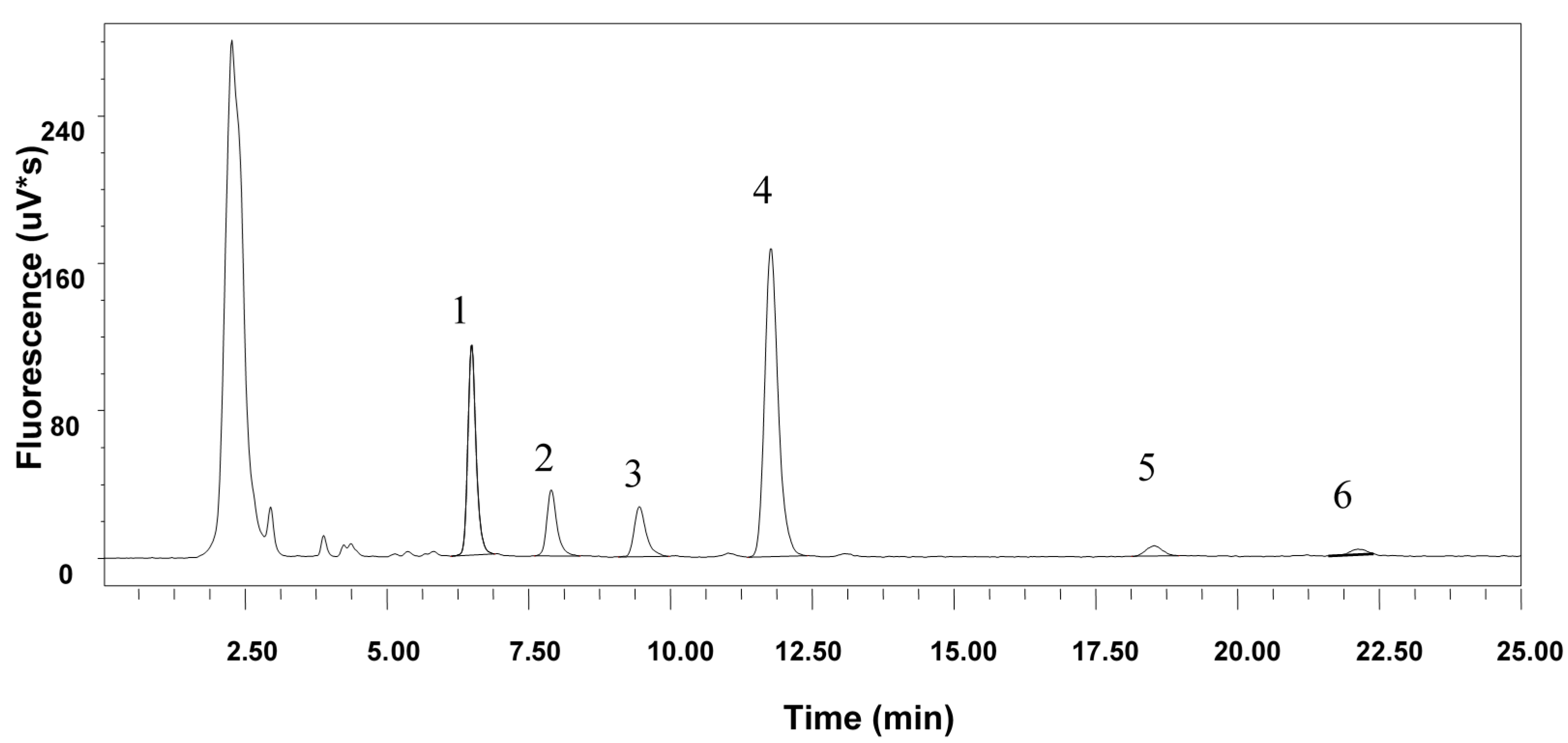
| α-TP | β-TP | α-TT | β-TT | δ-TT | TP | TT | Tot | |
|---|---|---|---|---|---|---|---|---|
| Flour | 89.0 | 89.6 | 87.8 | 83.5 | 11.2 | 89.9 | 85.8 | 90.3 |
| Variety | 1.06 | 1.45 | 3.53 | 1.46 | 4.29 | 1.18 | 2.06 | 1.64 |
| Location | 0.90 | 0.01 | 0.46 | 1.38 | 15.1 | 0.49 | 1.16 | 0.70 |
| F, V, L | 90.9 | 91.1 | 91.8 | 86.4 | 30.7 | 91.6 | 89.0 | 92.6 |
| Flour | α-TP | β-TP | α-TT | β-TT | δ-TT | TT/TP | Tot |
|---|---|---|---|---|---|---|---|
| White | 3.34 b | 2.00 b | 1.24 b | 12.7 b | 0.45 b | 2.83 a | 19.7 b |
| Whole | 13.3 a | 6.43 a | 4.93 a | 24.0 a | 0.60 a | 1.49 b | 49.4 a |
| Flour Type | Cultivar | Bultfontein | Clarens | Ladybrand | Average |
|---|---|---|---|---|---|
| White | Betta-DN | 21.2 | 17.5 | 19.0 | 19.3 cd |
| Caledon | 26.4 | 24.1 | 25.9 | 25.5 a | |
| Elands | 16.4 | 20.6 | 23.8 | 20.3 bc | |
| Gariep | 20.1 | 20.0 | 24.7 | 21.6 b | |
| Komati | 20.6 | 21.4 | 23.6 | 21.9 b | |
| Limpopo | 17.9 | 20.7 | 17.4 | 18.6 cde | |
| Matlabas | 18.8 | 18.7 | 18.3 | 18.6 cde | |
| C1PAN3118 | 17.1 | 17.0 | 15.3 | 16.5 f | |
| C2PAN3349 | 18.0 | 16.6 | 15.9 | 16.8 f | |
| C3PAN3377 | 18.2 | 17.8 | 17.8 | 18.1 def | |
| Average | 19.5 a | 19.4 a | 20.2 a | ||
| Whole | Betta-DN | 48.0 | 51.2 | 52.8 | 50.6 bc |
| Caledon | 52.0 | 55.0 | 57.6 | 55.0 a | |
| Elands | 49.9 | 51.4 | 59.8 | 53.7 a | |
| Gariep | 40.9 | 48.6 | 49.6 | 46.3 d | |
| Komati | 43.8 | 51.8 | 52.3 | 49.3 bc | |
| Limpopo | 55.7 | 49.5 | 55.7 | 53.7 a | |
| Matlabas | 39.4 | 52.8 | 52.0 | 48.2 cd | |
| C1PAN3118 | 43.5 | 42.9 | 45.4 | 43.9 e | |
| C2PAN3349 | 40.4 | 41.7 | 43.6 | 41.9 e | |
| C3PAN3377 | 50.2 | 47.7 | 54.5 | 51.0 b | |
| Average | 46.5 c | 49.2 b | 52.3 a |
| Flour Type | Characteristic | Bultfontein | Clarens | Ladybrand |
|---|---|---|---|---|
| White | α-Tocopherol | 3.02 b | 3.59 a | 3.36 a |
| β-Tocopherol | 1.88 b | 2.08 a | 2.02 a | |
| α-Tocotrienol | 1.32 a | 1.12 b | 1.28 a | |
| β-Tocotrienol | 12.7 a | 12.4 a | 13.0 a | |
| TT/TP | 3.07 a | 2.51 b | 2.92 a | |
| Whole | α-Tocopherol | 11.8 b | 14.1 a | 14.0 a |
| β-Tocopherol | 6.30 b | 6.62 a | 6.33 b | |
| α-Tocotrienol | 4.80 b | 4.45 c | 5.52 a | |
| β-Tocotrienol | 22.7 c | 23.4 b | 26.0 a | |
| TT/TP | 1.53 a | 1.36 b | 1.57 a |
| Flour Origin | Vitamin E Activity (mg kg−1) | Recommended Daily Intake (mg) | Percentage of Recommended Vitamin E from 200 g of Wheat Flour (%) |
|---|---|---|---|
| White | 3.3 | 12 | 5.5 |
| Whole | 13.3 | 12 | 22.2 |
| Whole flour at various locations | |||
| Bultfontein | 11.9 | 12 | 19.8 |
| Clarence | 14.1 | 12 | 23.5 |
| Whole flour from Clarence in various cultivars | |||
| Caledon | 16.1 | 12 | 26.8 |
| C1PAN3118 | 12.3 | 12 | 20.5 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Labuschagne, M.; Mkhatywa, N.; Johansson, E.; Wentzel, B.; Van Biljon, A. The Content of Tocols in South African Wheat; Impact on Nutritional Benefits. Foods 2017, 6, 95. https://doi.org/10.3390/foods6110095
Labuschagne M, Mkhatywa N, Johansson E, Wentzel B, Van Biljon A. The Content of Tocols in South African Wheat; Impact on Nutritional Benefits. Foods. 2017; 6(11):95. https://doi.org/10.3390/foods6110095
Chicago/Turabian StyleLabuschagne, Maryke, Nomcebo Mkhatywa, Eva Johansson, Barend Wentzel, and Angeline Van Biljon. 2017. "The Content of Tocols in South African Wheat; Impact on Nutritional Benefits" Foods 6, no. 11: 95. https://doi.org/10.3390/foods6110095






