Characterization of Botanical and Geographical Origin of Corsican “Spring” Honeys by Melissopalynological and Volatile Analysis
Abstract
:1. Introduction
2. Experimental Section
2.1. Honey and Flower Sampling
2.2. Melissopalynological Analysis
2.3. Physicochemical Analysis
2.4. HS-SPME Extraction
2.5. GC-FID and GC-MS Analysis
2.6. Statistical Analysis
3. Results and Discussion
3.1. Determination of Geographical and Botanical Origins of Corsican “Spring” Honeys
| No a | Type b | Taxa | PR c | Relative frequency (RF) d | BC g | ||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | Min. | Max. | SD e | CV f | |||||
| T1 | P | Quercus sp. | 100 | 13.2 | 0.8 | 35.7 | 9.7 | 73.8 | 21-35-55-58 |
| T2 | P | Cistus sp. | 100 | 8.5 | 0.3 | 33.3 | 6.3 | 74.4 | 21-29 |
| T3 | P | Castanea sativa h | 90 | 10.3 | 0.3 | 33.8 | 8.7 | 84.2 | 59 |
| T4 | P | Fraxinus ornus | 90 | 3.3 | 0.3 | 22.3 | 5.0 | 153.2 | 58 |
| T5 | N, P | Erica arborea | 85 | 7.8 | 0.2 | 35.5 | 8.7 | 112.3 | 21 |
| T6 | N, P | Genista form i | 83 | 6.0 | 0.3 | 31.5 | 8.0 | 134.8 | 14-21-29-51-62 |
| T7 | N, P | Lotus sp. | 76 | 5.3 | 0.3 | 52.8 | 9.5 | 178.3 | 21-51 |
| T8 | N, P | Salix sp. | 73 | 6.3 | 0.2 | 29.9 | 7.4 | 117.1 | 51-52 |
| T9 | N, P | Trifolium sp. | 71 | 14.2 | 0.4 | 53.5 | 16.8 | 117.9 | 21-31-51 |
| T10 | N, P | Rubus sp. | 71 | 3.6 | 0.4 | 11.7 | 3.4 | 94.6 | 31-35 |
| T11 | N, P | Prunus form j | 66 | 3.0 | 0.2 | 24.1 | 4.7 | 155.6 | 99-54 |
| T12 | P | Eucalyptus sp. | 63 | 2.1 | 0.3 | 15.5 | 3.1 | 148.4 | 99 |
| T13 | N, P | Echium sp. | 59 | 10.5 | 0.6 | 71.1 | 15.6 | 148.2 | 31 |
| T14 | N, P | Apiaceae | 59 | 4.0 | 0.2 | 17.5 | 4.4 | 109.6 | nd |
| T15 | P | Actinidia sinensis | 49 | 4.2 | 0.3 | 16.1 | 4.5 | 107.7 | 99 |
| T16 | N, P | Brassicaceae others | 49 | 2.8 | 0.3 | 14.7 | 3.3 | 118.6 | nd |
| T17 | N, P | Lavandula stoechas | 49 | 1.8 | 0.4 | 10.1 | 2.2 | 124.1 | 21 |
| T18 | N, P | Citrus sp. | 44 | 6.1 | 0.2 | 16.1 | 5.2 | 86.6 | 99 |
| T19 | N, P | Vicia form | 44 | 3.0 | 0.3 | 11.8 | 3.2 | 107.1 | nd |
| T20 | P | Pistacia lentiscus | 44 | 3.0 | 0.5 | 9.3 | 2.6 | 88.0 | 29 |
| T21 | N, P | Asteraceae Galactites form | 44 | 1.9 | 0.2 | 5.2 | 1.7 | 93.6 | 21 |
| T22 | N, P | Asphodelus ramosus subsp. ramosus | 44 | 0.7 | 0.2 | 2.9 | 0.7 | 96.5 | 21 |
| T23 | P | Scrophulariaceae others | 39 | 0.9 | 0.3 | 4.5 | 1.0 | 114.4 | nd |
| T24 | P | Phillyrea sp. | 37 | 3.0 | 0.3 | 13.3 | 3.8 | 125.5 | 25 |
| T25 | P | Olea sp. | 37 | 1.0 | 0.4 | 3.6 | 0.8 | 74.3 | 21 |
| T26 | N, P | Viburnum tinus | 34 | 1.9 | 0.3 | 16.2 | 4.2 | 225.9 | 21 |
| T27 | N, P | Asteraceae (fenestrated type) | 29 | 1.1 | 0.3 | 3.2 | 1.0 | 94.0 | 21-94 |
| T28 | N, P | Rosa sp. | 27 | 1.2 | 0.3 | 4.5 | 1.3 | 108.1 | 31-51 |
| T29 | P | Myrtus communis | 24 | 0.9 | 0.3 | 1.6 | 0.5 | 52.0 | 21 |
| T30 | N, P | Fabaceae others/Dorycnopis form | 24 | 0.6 | 0.3 | 1.4 | 0.3 | 54.5 | nd |
| T31 | P | Plantago sp. | 24 | 0.5 | 0.3 | 0.9 | 0.2 | 33.8 | nd |
| T32 | N, P | Asteraceae Achillea form | 22 | 0.8 | 0.2 | 2.6 | 0.7 | 94.0 | 21-94 |
| T33 | P | Poaceae | 22 | 0.6 | 0.2 | 1.2 | 0.3 | 54.0 | nd |
| T34 | N, P | Crataegus monogyna | 20 | 2.1 | 0.3 | 7.9 | 2.7 | 130.8 | 51 |
| T35 | N, P | Jasione montana | 17 | 2.0 | 0.3 | 10.0 | 3.5 | 176.0 | 54 |
| T36 | N, P | Rosaceae others | 17 | 1.2 | 0.3 | 3.0 | 1.0 | 85.6 | nd |
| T37 | N, P | Asteraceae Dittrichia form | 17 | 1.0 | 0.2 | 1.9 | 0.8 | 74.4 | 21-94 |
| T38 | N, P | Rhamnus sp. | 15 | 1.0 | 0.3 | 3.3 | 1.1 | 117.6 | 21 |
| T39 | N, P | Psoralea bituminosa | 15 | 0.7 | 0.3 | 1.6 | 0.5 | 75.6 | 31 |
| T40 | N, P | Knautia sp. | 15 | 0.5 | 0.3 | 0.9 | 0.3 | 52.8 | 31 |
| T41 | N, P | Lupinus angustifolius | 12 | 4.8 | 0.3 | 18.9 | 8.0 | 166.1 | 21 |
| T42 | P | Cytinus hypocistis | 12 | 0.9 | 0.4 | 1.8 | 0.7 | 77.6 | 29 |
| T43 | N, P | Hedera helix | 12 | 0.8 | 0.3 | 1.3 | 0.4 | 46.1 | 65 |
| T44 | N, P | Liliaceae others | 12 | 0.4 | 0.3 | 0.6 | 0.2 | 45.8 | nd |
| T45 | N, P | Allium sp. | 12 | 0.4 | 0.3 | 0.6 | 0.2 | 42.9 | 21-25 |
| T46 | N, P | Acacia dealbata | 12 | 0.4 | 0.3 | 0.4 | 0 | 13.3 | 99 |
| T47 | P | Alnus sp. | 7 | 2.3 | 0.3 | 6.1 | 3.3 | 146.5 | 51 |
| T48 | N, P | Dorycnium sp. | 10 | 1.5 | 0.3 | 3.2 | 1.2 | 83.3 | 35 |
| T49 | N, P | Rosmarinus officinalis | 10 | 1.7 | 0.4 | 3.0 | 1.3 | 77.9 | 21 |
| T50 | P | Vitis vinifera | 7 | 1.5 | 0.4 | 3.0 | 1.3 | 89.4 | 99 |
| Melissopalynological Data | Group I—“Clementine” Honeys 18 Samples (1–18) | Group II—Other “Spring” Honeys 23 Samples (19–41) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RF c | RF c | |||||||||||||
| No. a | Type b | Taxa | PR | Mean | Min. | Max. | SD | CV | PR | Mean | Min. | Max. | SD | CV |
| Main nectariferous taxa | ||||||||||||||
| T18 | N, P | Citrus sp. | 100 | 6.1 | 0.2 | 16.1 | 5.2 | 86.6 | - | - | - | - | - | - |
| T5 | N, P | Erica arborea | 89 | 7.5 | 0.3 | 35.5 | 10.2 | 135.4 | 83 | 8.0 | 0.2 | 29.7 | 7.6 | 94.9 |
| T8 | N, P | Salix sp. | 78 | 5.8 | 0.6 | 12.9 | 4.5 | 77.7 | 70 | 6.7 | 0.2 | 29.9 | 9.3 | 139.4 |
| T11 | N, P | Prunus form | 78 | 4.8 | 0.2 | 24.1 | 6.1 | 128.4 | 57 | 1.2 | 0.3 | 3.3 | 0.9 | 75.5 |
| T7 | N, P | Lotus sp. | 72 | 2.7 | 0.4 | 8.7 | 2.5 | 91.9 | 78 | 7.2 | 0.3 | 52.8 | 12.1 | 167.5 |
| T10 | N, P | Rubus sp. | 67 | 2.3 | 0.4 | 6.5 | 1.8 | 77.9 | 74 | 4.5 | 0.4 | 11.7 | 4.0 | 88.4 |
| T9 | N, P | Trifolium sp. | 67 | 12.9 | 0.5 | 53.5 | 19.4 | 150.7 | 74 | 15.2 | 0.4 | 44.1 | 15.2 | 100.2 |
| T6 | N, P | Genista form | 67 | 3.1 | 0.3 | 11.8 | 3.4 | 107.1 | 96 | 7.5 | 0.6 | 31.5 | 9.4 | 125.3 |
| T13 | N, P | Echium sp. | 44 | 19.3 | 1.3 | 71.1 | 23.3 | 121.0 | 70 | 6.1 | 0.6 | 30.1 | 7.5 | 123.2 |
| T26 | N, P | Viburnum tinus | 22 | 4.5 | 0.3 | 16.2 | 7.8 | 174.5 | 43 | 0.8 | 0.3 | 3.7 | 1.1 | 127.7 |
| T14 | N, P | Apiaceae | 28 | 2.2 | 0.2 | 4.5 | 2.0 | 88.8 | 83 | 4.5 | 0.3 | 17.5 | 4.8 | 106.0 |
| T22 | N, P | Asphodelus ramosus subsp. ramosus | 11 | 0.9 | 0.5 | 1.3 | 0.5 | 57.3 | 70 | 0.7 | 0.2 | 2.9 | 0.7 | 104.1 |
| T16 | N, P | Brassicaceae others | 33 | 1.2 | 0.3 | 2.7 | 1.0 | 77.9 | 61 | 3.5 | 0.4 | 14.7 | 3.8 | 108.2 |
| T17 | N, P | Lavandula stoechas | 39 | 1.7 | 0.5 | 4.4 | 1.4 | 81.9 | 57 | 1.8 | 0.4 | 10.1 | 2.6 | 142.4 |
| T19 | N, P | Vicia form | 28 | 2.3 | 0.7 | 6.0 | 2.3 | 102.4 | 57 | 3.3 | 0.3 | 11.8 | 3.6 | 107.6 |
| T41 | N, P | Lupinus angustifolius | 17 | 1.5 | 0.3 | 3.5 | 1.8 | 119.1 | 9 | 9.8 | 0.7 | 18.9 | 12.9 | 131.6 |
| Other nectariferous taxa | 100 | 3.3 | 0.3 | 11.0 | 3.0 | 89.9 | 100 | 5.2 | 1.0 | 10.0 | 2.8 | 53.5 | ||
| Main only-polleniferous taxa | ||||||||||||||
| T15 | P | Actinidia sinensis | 100 | 4.6 | 0.3 | 16.1 | 4.6 | 99.7 | 9 | 0.5 | 0.3 | 0.7 | 0.3 | 55.3 |
| T2 | P | Cistus sp. | 100 | 6.9 | 0.3 | 20.4 | 5.2 | 76.0 | 100 | 9.8 | 0.3 | 33.3 | 6.9 | 70.9 |
| T1 | P | Quercus sp. | 100 | 16.5 | 0.8 | 35.7 | 10.7 | 64.9 | 100 | 10.6 | 1.3 | 29.0 | 8.1 | 77.2 |
| T3 | P | Castanea sativa | 89 | 7.9 | 0.3 | 25.0 | 7.3 | 91.7 | 91 | 12.1 | 0.3 | 33.8 | 9.4 | 77.4 |
| T4 | P | Fraxinus ornus | 89 | 6.1 | 0.5 | 22.3 | 6.6 | 108.4 | 91 | 1.1 | 0.3 | 3.3 | 1.0 | 87.6 |
| T12 | P | Eucalyptus sp. | 72 | 3.3 | 0.4 | 15.5 | 4.1 | 125.9 | 57 | 1.0 | 0.3 | 3.4 | 1.0 | 99.3 |
| T25 | P | Olea sp. | 50 | 0.8 | 0.4 | 1.3 | 0.3 | 38.9 | 26 | 1.3 | 0.6 | 3.6 | 1.1 | 84.3 |
| T20 | P | Pistacia lentiscus | 11 | 0.6 | 0.6 | 0.6 | 0.0 | 8.2 | 70 | 3.3 | 0.5 | 9.3 | 2.7 | 80.4 |
| T24 | P | Phillyrea sp. | 17 | 4.7 | 0.3 | 13.3 | 7.4 | 157.4 | 52 | 2.6 | 0.4 | 9.9 | 2.7 | 103.5 |
| Other only-polleniferous taxa | 100 | 1.9 | 0.3 | 8.9 | 2.1 | 110.0 | 78 | 1.7 | 0.3 | 8.5 | 2.0 | 117.8 | ||
| Pollen density (103 PG/10 g) d | 68 | 20 | 202 | 52 | 77 | 107 | 22 | 603 | 126 | 118 | ||||
| Physico-chemical data e | ||||||||||||||
| Color | 26.4 | 11.0 | 55.0 | 13.6 | 51.5 | 33.3 | 18.0 | 71.0 | 16.3 | 48.7 | ||||
| Electrical conductivity | 0.25 | 0.15 | 0.42 | 0.07 | 27.72 | 0.24 | 0.13 | 0.45 | 0.09 | 36.96 | ||||
3.2. Physicochemical Characteristics of Corsican “Spring” Honeys
3.3. Chemical Variability of Corsican “Spring” Honeys
| No a | Components | RI b | Group I “Clementine” Honeys c | Group II “Not-Clementine” Honeys c | Sample 35 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IIa | IIb | |||||||||||
| Mean ± SD d | Min. | Max. | Mean ± SD d | Min. | Max. | Mean ± SD d | Min. | Max. | ||||
| C1 | 3-Methyl-3-buten-1-ol | 704 | 2.9 ± 2.76 | 0.3 | 11.3 | 1.8 ± 1.19 | 0.7 | 3.7 | 1.8 ± 1.44 | 0.4 | 6.0 | 2.8 |
| C2 | Methyl-benzene | 741 | 6.5 ± 4.45 | 1.5 | 15.6 | 4.1 ± 2.08 | 2.4 | 7.1 | 6.2 ± 4.08 | 1.5 | 17.3 | 10.4 |
| C3 | Hexanal | 773 | 1.1 ± 0.46 | 0.5 | 2.0 | 1.6 ± 2.62 | 0.1 | 6.3 | 1.6 ± 1.13 | 0.3 | 4.5 | 1.9 |
| C4 | Octane | 790 | 1.4 ± 1.08 | 0.3 | 4.7 | 0.9 ± 0.77 | 0.3 | 2.2 | 2.6 ± 1.63 | 0.7 | 5.6 | 1.4 |
| C5 | 3-Furaldehyde | 800 | 2.8 ± 1.56 | 0.6 | 5.9 | 3.5 ± 1.37 | 2.5 | 5.9 | 3.5 ± 1.78 | 1.9 | 8.3 | 18.5 |
| C6 | 2-Methyl butanoic acid | 858 | 0.8 ± 0.91 | 0.1 | 3.3 | 4.6 ± 3.21 | 1.1 | 7.6 | 2.9 ± 4.59 | 0.1 | 20.4 | 2.4 |
| C7 | 2-Methyl octane | 873 | 0.4 ± 0.28 | 0.1 | 0.9 | 0.5 ± 0.44 | 0.1 | 1.2 | 0.7 ± 0.38 | 0.1 | 1.7 | 1.6 |
| C8 | Nonane | 893 | 0.7 ± 0.65 | 0.2 | 2.5 | 1.2 ± 0.35 | 0.8 | 1.7 | 1.5 ± 0.92 | 0.2 | 3.5 | 3.8 |
| C9 | Benzaldehyde | 924 | 5.5 ± 3.56 | 2.4 | 17.9 | 10.4 ± 3.85 | 5.4 | 14.8 | 8.8 ± 4.89 | 2.5 | 18.4 | 3.0 |
| C10 | Hexanoic acid | 969 | 0.7 ± 0.25 | 0.4 | 1.4 | 1.7 ± 1.68 | 0.3 | 3.9 | 1.2 ± 0.75 | 0.4 | 3.3 | - |
| C11 | Octanal | 982 | 1.0 ± 0.47 | 0.2 | 2.1 | 0.6 ± 0.12 | 0.5 | 0.7 | 1.6 ± 1.45 | 0.5 | 6.6 | 1.0 |
| C12 | 2,2,4,6,6-Pentamethylheptane | 992 | 1.1 ± 0.60 | 0.4 | 2.4 | 0.6 ± 0.53 | 0.1 | 1.3 | 2.3 ± 1.83 | 0.2 | 5.2 | 15.6 |
| C13 | p-Methylanisol | 995 | 0.9 ± 1.13 | 0.1 | 4.9 | 1.1 ± 1.10 | 0.3 | 3.0 | 0.9 ± 1.48 | 0.1 | 6.1 | - |
| C14 | Phenylacetaldehyde | 1006 | 10.1 ± 10.68 | 0.8 | 39.1 | 16.5 ± 6.93 | 7.2 | 25.7 | 21.2 ± 7.83 | 3.7 | 36.2 | 13.0 |
| C15 | p-Cymene | 1008 | 0.7 ± 0.29 | 0.1 | 1.0 | 0.9 ± 0.42 | 0.6 | 1.2 | - | - | - | - |
| C16 | Acetophenone | 1037 | 0.2 ± 0.08 | 0.1 | 0.4 | 0.4 ± 0.21 | 0.2 | 0.5 | 0.3 ± 0.10 | 0.2 | 0.4 | - |
| C17 | trans-Furanoid-linaloxide | 1049 | 1.5 ± 1.00 | 0.8 | 4.0 | 1.3 ± 1.25 | 0.5 | 3.5 | 2.4 ± 1.20 | 1.0 | 6.3 | - |
| C18 | cis-Furanoid-linaloxide | 1064 | 1.1 ± 0.40 | 0.7 | 2.0 | 1.0 ± 0.29 | 0.7 | 1.4 | 1.1 ± 0.26 | 0.5 | 1.6 | - |
| C19 | β-Phenylethanol | 1077 | 4.2 ± 1.54 | 2.2 | 5.8 | 1.6 ± 0.00 | 1.6 | 1.6 | 3.3 ± 1.67 | 2.1 | 5.8 | - |
| C20 | Nonanal | 1079 | 2.7 ± 1.73 | 0.9 | 7.6 | 1.8 ± 1.28 | 0.4 | 3.5 | 3.0 ± 2.29 | 0.5 | 7.2 | 2.9 |
| C21 | Linalol | 1084 | 2.4 ± 1.82 | 0.2 | 6.6 | 1.3 ± 1.62 | 0.3 | 3.2 | 12.5 ± 10.42 | 2.1 | 32.3 | tr |
| C22 | Hotrienol | 1085 | 4.1 ± 4.39 | 0.7 | 10.5 | - | - | - | 9.7 ± 0.00 | 9.7 | 9.7 | - |
| C23 | Isophorone | 1087 | 2.8 ± 1.54 | 0.2 | 4.9 | 18.2 ± 8.02 | 8.8 | 29.3 | 3.3 ± 3.5 | 0.1 | 9.6 | - |
| C24 | 4-Oxoisophorone | 1102 | 0.9 ± 0.33 | 0.3 | 1.4 | 4.2 ± 1.87 | 2.3 | 6.4 | 1.5 ± 0.99 | 0.3 | 5.0 | - |
| C25 | (2S,2′S,5′S)-Lilac aldehyde | 1112 | 5.4 ± 2.36 | 1.3 | 8.9 | - | - | - | 1.5 ± 0.00 | 1.5 | 1.5 | - |
| C26 | Dihydrolinalool | 1116 | 1.2 ± 0.66 | 0.5 | 3.0 | - | - | - | 1.1 ± 0.00 | 1.1 | 1.1 | - |
| C27 | (2R,2′S,5′S)-Lilac aldehyde | 1121 | 10.5 ± 3.66 | 4.9 | 16.5 | - | - | - | 2.4 ± 0.00 | 2.4 | 2.4 | - |
| C28 | (2R,2′R,5′S)-Lilac aldehyde | 1134 | 4.8 ± 1.85 | 2.2 | 8.1 | - | - | - | 1.1 ± 0.00 | 1.1 | 1.1 | - |
| C29 | Octanoic acid | 1167 | 1.7 ± 1.55 | 0.3 | 6.0 | 0.9 ± 0.38 | 0.3 | 1.3 | 1.6 ± 0.78 | 0.7 | 3.8 | 4.7 |
| C30 | Decanal | 1174 | 1.2 ± 0.67 | 0.2 | 2.8 | 0.6 ± 0.25 | 0.2 | 0.8 | 1.5 ± 0.54 | 0.6 | 2.3 | - |
| C31 | p-Menth-1-en-9-al (isomer 1) | 1184 | 1.9 ± 0.41 | 1.2 | 2.7 | - | - | - | - | - | - | - |
| C32 | p-Menth-1-en-9-al (isomer 2) | 1186 | 1.7 ± 0.46 | 0.5 | 2.5 | - | - | - | - | - | - | - |
| C33 | p-Anisaldehyde | 1208 | 0.7 ± 1.19 | 0.1 | 4.6 | 0.9 ± 0.37 | 0.3 | 1.1 | 0.4 ± 0.21 | 0.2 | 0.8 | - |
| C34 | 2,3,5-Trimethylphenol | 1248 | 0.4 ± 0.30 | 0.1 | 1.1 | 1.0 ± 0.69 | 0.4 | 2.0 | 0.8 ± 0.68 | 0.1 | 2.0 | - |
| C35 | 4-n-Propylanisol | 1264 | 1.6 ± 1.78 | 0.2 | 5.7 | 2.4 ± 1.29 | 0.8 | 4.3 | 3.8 ± 2.45 | 1.4 | 6.3 | - |
| C36 | Nonanoic acid | 1271 | 2.7 ± 1.39 | 0.5 | 4.9 | 2.6 ± 0.94 | 1.4 | 3.7 | 3.1 ± 1.24 | 1.3 | 6.4 | - |
| C37 | 3,4,5-Trimethylphenol | 1290 | 0.5 ± 0.32 | 0.2 | 1.4 | 5.4 ± 2.71 | 2.9 | 9.4 | 0.5 ± 0.67 | 0.1 | 2.0 | - |
| C38 | Methyl anthranilate | 1300 | 1.4 ± 0.96 | 0.2 | 3.5 | - | - | - | - | - | - | - |
| C39 | cis-p-Mentha-1(7),8-dien-1-hydroperoxide | 1348 | 0.4 ± 0.14 | 0.2 | 0.7 | - | - | - | - | - | - | - |
| C40 | Decanoic acid | 1362 | 1.2 ± 0.45 | 0.6 | 2.1 | 1.3 ± 0.75 | 0.1 | 1.9 | 1.7 ± 1.50 | 0.6 | 6.8 | 3.8 |
| C41 | Methyl 3,5-dimethoxybenzoate | 1494 | - | - | - | 0.4 ± 0.26 | 0.2 | 0.7 | 0.5 ± 0.19 | 0.3 | 0.8 | - |
| C42 | Methyl syringate | 1722 | - | - | - | 0.5 ± 0.50 | 0.1 | 1.4 | 0.9 ± 1.17 | 0.1 | 4.1 | - |
| C43 | Tricosane | 2305 | 0.3 ± 0.17 | 0.1 | 0.5 | 0.5 ± 0.00 | 0.5 | 0.5 | 0.5 ± 0.22 | 0.2 | 0.7 | - |
| Total identification (%) | 84.2 ± 6.95 | 71.5 | 94.5 | 91.2 ± 5.43 | 84.2 | 96.8 | 86.3 ± 5.49 | 78.8 | 96.7 | 86.8 | ||
| Total peak area(106) e | 3.8 ± 1.96 | 1.3 | 7.4 | 2.9 ± 1.27 | 1.6 | 4.5 | 2.4 ± 1.08 | 0.8 | 4.4 | 0.3 | ||
| Hydrocarbons | 10.6 ± 5.23 | 4.7 | 20.8 | 7.7 ± 3.59 | 4.8 | 13.5 | 13.3 ± 5.88 | 5.0 | 23.9 | 32.8 | ||
| Oxygenated compounds | 73.6 ± 7.23 | 58.2 | 82.8 | 83.6 ± 4.11 | 79.2 | 90.3 | 73.7 ± 7.56 | 58.1 | 81.7 | 54.0 | ||
| Phenolic compounds | 29.4 ± 12.3 | 12.6 | 59.3 | 43.0 ± 7.39 | 34.8 | 53.0 | 39.9 ± 11.22 | 23.2 | 60.4 | 26.4 | ||
| Furan compounds | 26.2 ± 7.85 | 12.2 | 38.6 | 5.8 ± 2.83 | 3.9 | 10.8 | 7.5 ± 1.99 | 4.3 | 11.0 | 18.5 | ||
| Linear compounds | 21.0 ± 7.08 | 11.3 | 36.6 | 19.2 ± 3.88 | 14.8 | 23.4 | 26.2 ± 9.63 | 11.3 | 53.4 | 41.9 | ||
| Terpenic compounds | 31.0 ± 9.88 | 15.4 | 52.2 | 3.5 ± 2.64 | 1.7 | 8.1 | 13.6 ± 13.05 | 1.5 | 45.8 | 0 | ||
| Ketones | 2.5 ± 2.14 | 0 | 6.0 | 22.5 ± 9.67 | 11.6 | 35.7 | 4.3 ± 3.77 | 0.8 | 11.6 | 0 | ||
| Aldehydes | 49.1 ± 8.03 | 34.4 | 63.1 | 35.6 ± 9.73 | 26.1 | 47.7 | 40.7 ± 9.78 | 22.1 | 52.3 | 40.3 | ||
| Esters | 1.4 ± 0.96 | 0.2 | 3.5 | 0.2 ± 0.29 | 0 | 0.7 | 0.3 ± 0.27 | 0 | 0.8 | 0 | ||
| Alcohols | 9.7 ± 6.17 | 3.3 | 27.8 | 9.2 ± 3.3 | 4.9 | 12.7 | 12.8 ± 10.40 | 3.0 | 40.2 | 2.8 | ||
| Acids | 6.8 ± 3.31 | 0.4 | 15.4 | 9.7 ± 5.23 | 3.0 | 15.1 | 10.2 ± 5.28 | 5.6 | 27.0 | 10.9 | ||
| Oxides | 5.2 ± 2.76 | 2.5 | 12.6 | 6.3 ± 2.45 | 3.0 | 9.3 | 5.5 ± 2.64 | 3.3 | 14.3 | 0 | ||
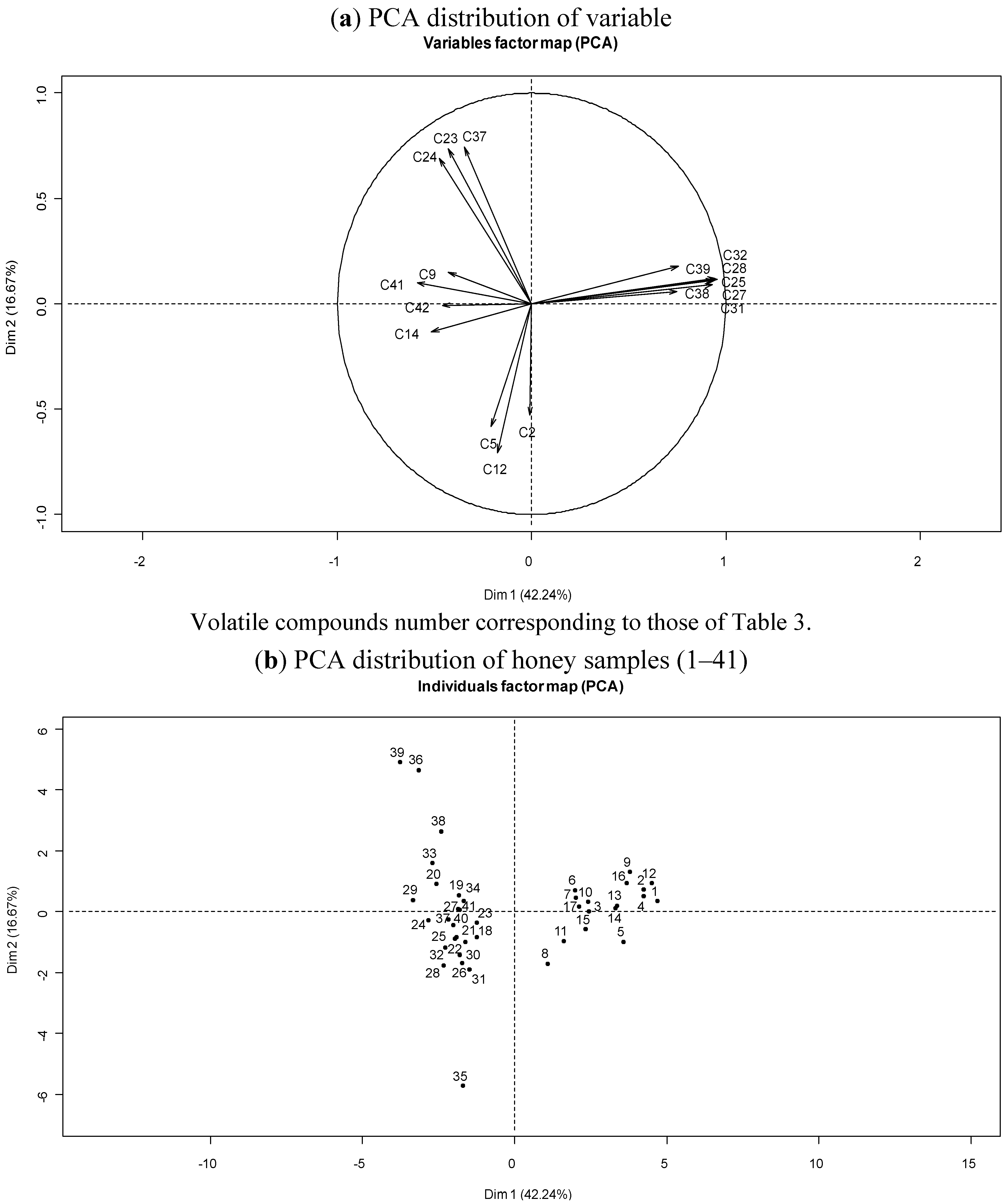
3.4. Botanical Origin and Volatile Composition of Corsican “Spring” Honeys
| Components a | RI(Lit) b | RI c | Clementine Flower d | Asphodel Flower e | Identification g | ||||
|---|---|---|---|---|---|---|---|---|---|
| Mean ± SD f | Min. | Max. | Mean ± SD f | Min. | Max. | ||||
| 3-Furaldehyde | 799 | 800 | - | - | - | 1.0 ± 0.87 | 0.5 | 2.7 | RI, MS |
| Furfural | 831 | 836 | - | - | - | 3.5 ± 1.26 | 1.7 | 5.2 | RI, MS |
| 2-Furanmethanol | 839 | 842 | - | - | - | 2.1 ± 1.48 | 0.8 | 4.7 | RI, MS, Ref |
| Heptanal | 882 | 876 | - | - | - | 5.4 ± 2.46 | 3.1 | 9.5 | RI, MS |
| α-Thujene | 924 | 922 | 1.2 ± 0.21 | 1.0 | 1.4 | - | - | - | RI, MS |
| α-Pinene | 932 | 931 | 3.6 ± 2.46 | 2.0 | 6.4 | - | - | - | RI, MS |
| Benzaldehyde | 929 | 933 | - | - | - | 2.7 ± 0.78 | 1.4 | 3.7 | RI, MS |
| Tetrahydro-citronellene | 937 | 935 | 6.8 ± 4.90 | 3.3 | 12.4 | - | - | - | RI, MS, Ref |
| β-Citronellene | 943 | 940 | 2.2 ± 0.15 | 2.0 | 2.3 | - | - | - | RI, MS |
| Octen-3-ol | 962 | 955 | - | - | - | 0.2 ± 0.05 | 0.1 | 0.2 | RI, MS |
| Furfuryl acetate | 964 | 959 | - | - | - | 0.7 ± 0.31 | 0.5 | 1.3 | RI, MS, Ref |
| Sabinene | 973 | 958 | 16.8 ± 3.14 | 13.4 | 19.6 | - | - | - | RI, MS |
| 2-Pentylfuran | 973 | 966 | - | - | - | 0.8 ± 1.00 | 0.2 | 2.8 | RI, MS |
| β-Pinene | 978 | 972 | 1.5 ± 1.36 | 0.4 | 3.0 | - | - | - | RI, MS |
| Myrcene | 987 | 979 | 6.1 ± 0.45 | 5.6 | 6.5 | - | - | - | RI, MS |
| Octanal | 981 | 980 | - | - | - | 7.0 ± 3.12 | 3.5 | 12.6 | RI, MS |
| (Z)-3-Hexenyl acetate | 989 | 984 | - | - | - | 21.6 ± 14.27 | 5.2 | 41.8 | RI, MS |
| (E)-3-Hexenyl acetate | 1002 | 994 | - | - | - | 0.8 ± 0.54 | 0.1 | 1.5 | RI, MS |
| α-Phellandrene | 1002 | 995 | 1.5 ± 0.23 | 1.4 | 1.8 | 0.3 ± 0.12 | 0.1 | 0.4 | RI, MS |
| α-Terpinene | 1013 | 1008 | 0.6 ± 0.44 | 0.3 | 1.1 | - | - | - | RI, MS |
| Phenylacetaldehyde | 1012 | 1009 | - | - | - | 0.9 ± 0.67 | 0.2 | 2.1 | RI, MS |
| p-Cymene | 1015 | 1011 | 0.6 ± 0.10 | 0.5 | 0.7 | - | - | - | RI, MS |
| p-Menth-1-ene | 1017 | 1018 | 0.5 ± 0.15 | 0.4 | 0.7 | - | - | - | RI, MS |
| Limonene | 1025 | 1020 | 1.5 ± 0.70 | 0.8 | 2.2 | - | - | - | RI, MS |
| (Z)-β-Ocimene | 1029 | 1024 | 0.1 ± 0.06 | 0.1 | 0.2 | - | - | - | RI, MS |
| (E)-2-Octenal | 1034 | 1034 | - | - | - | 0.4 ± 0.29 | 0.1 | 0.8 | RI, MS |
| (E)-β-Ocimene | 1041 | 1036 | 2.6 ± 2.21 | 0.9 | 5.1 | - | - | - | RI, MS |
| γ-Terpinene | 1051 | 1047 | 1.1 ± 0.51 | 0.5 | 1.5 | - | - | - | RI, MS |
| trans-Sabinene hydrate | 1053 | 1050 | 1.0 ± 0.36 | 0.7 | 1.4 | - | - | - | RI, MS |
| 1-Octanol | 1063 | 1057 | - | - | - | 6.0 ± 1.93 | 2.8 | 8.8 | RI, MS |
| Terpinolene | 1082 | 1078 | 0.1 ± 0.06 | 0.1 | 0.2 | - | - | - | RI, MS |
| Nonanal | 1076 | 1081 | - | - | - | 25.8 ± 10.1 | 16.5 | 38.2 | RI, MS |
| Linalool | 1086 | 1086 | 17.8 ± 7.14 | 9.6 | 22.6 | 1.7 ± 0.21 | 1.5 | 1.8 | RI, MS |
| Tetrahydrolinalool | 1099 | 1095 | 4.1 ± 3.07 | 0.7 | 6.7 | - | - | - | RI, MS, Ref |
| Dihydrolinalool | 1118 | 1114 | 10.8 ± 3.50 | 8.5 | 14.8 | - | - | - | RI, MS, Ref |
| (E)-2-Nonen-1-ol | 1149 | 1153 | - | - | - | 2.2 ± 1.65 | 0.6 | 4.6 | RI, MS |
| 1-Phenylethyl acetate | 1166 | 1163 | - | - | - | 0.1 ± 0.05 | 0.1 | 0.2 | RI, MS |
| Terpinen-4-ol | 1164 | 1164 | 0.3 ± 0.20 | 0.1 | 0.5 | - | - | - | RI, MS |
| α-Terpineol | 1176 | 1173 | tr | tr | tr | - | - | - | RI, MS |
| Decanal | 1180 | 1182 | - | - | - | 1.6 ± 0.74 | 0.7 | 2.5 | RI, MS |
| Undecanal | 1285 | 1285 | - | - | - | 1.0 ± 1.03 | 0.2 | 2.8 | RI, MS |
| Methyl anthranilate | 1308 | 1302 | 0.2 ± 0.10 | 0.1 | 0.3 | - | - | - | RI, MS |
| (E)-Jasmone | 1356 | 1360 | tr | tr | tr | - | - | - | RI, MS |
| Isocaryophyllene | 1409 | 1405 | tr | tr | tr | - | - | - | RI, MS |
| (E)-β-Farnesene | 1446 | 1442 | tr | tr | tr | - | - | - | RI, MS |
| (E,E)-α-Farnesene | 1498 | 1492 | 0.1 ± 0.00 | 0.1 | 0.1 | - | - | - | RI, MS |
| (E)-Nerolidol | 1553 | 1548 | tr | tr | tr | - | - | - | RI, MS |
| Heptadecane | 1700 | 1698 | 0.2 ± 0.10 | 0.1 | 0.3 | - | - | - | RI, MS |
| Total identification (%) | 81.2 ± 5.75 | 75.5 | 87.0 | 85.9 ± 2.66 | 82.4 | 90.1 | |||
| Hydrocarbons | 48.0 ± 8.16 | 39.7 | 56.0 | - | - | - | |||
| Oxygenated compounds | 33.1 ± 11.88 | 19.5 | 41.3 | 85.9 ± 2.66 | 82.4 | 90.1 | |||
| Phenolic compounds | 0.2 ± 0.1 | 0.1 | 0.3 | 3.7 ± 0.99 | 1.8 | 4.5 | |||
| Furan compounds | - | - | - | 8.2 ± 4.42 | 4.3 | 16.7 | |||
| Linear compounds | 0.2 ± 0.1 | 0.1 | 0.3 | 73.9 ± 5.41 | 65.0 | 79.3 | |||
| Terpenic compounds | 80.8 ± 5.75 | 75.1 | 86.6 | 0.8 ± 0.85 | 0.1 | 1.9 | |||
| Ketones | tr | - | tr | - | - | - | |||
| Aldehydes | - | - | - | 43.8 ± 13.47 | 31.2 | 62.8 | |||
| Esters | 0.2 ± 0.1 | 0.1 | 0.3 | 23.2 ± 14.58 | 5.8 | 43.1 | |||
| Alcohols | 32.9 ± 11.8 | 19.4 | 41.1 | 11 ± 4.13 | 4.3 | 17 | |||
3.5. Correlation of Melissopalynological and Chemical Data
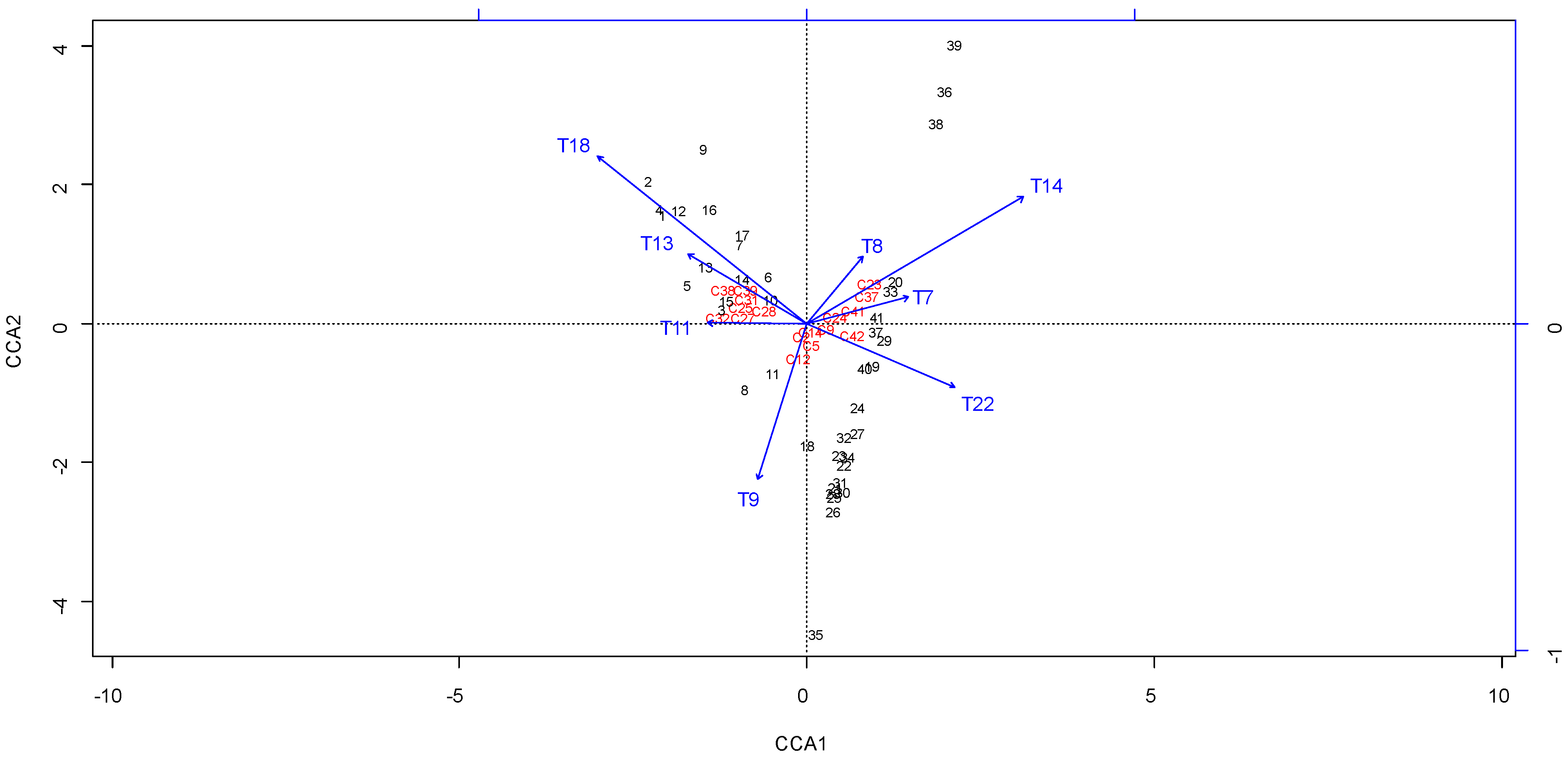
4. Conclusions
Supplementary Materials
Supplementary File 1Acknowledgments
Conflicts of Interest
References
- Battesti, M.J.; Gamisans, J.; Piana, L. Définition du Périmètre de Production—Rapport des Experts en Vue de la Mise à l’Enquête. Demande de Reconnaissance en A.O.C. <Miel de Corse-Mele di Corsica>; Institut National des Appelations d’Origine (INAO): Corte, France, 1997. [Google Scholar]
- Décret n° 2010–1045 du 31 Août 2010 Relatif à l’Appellation d’Origine Contrôlée <Miel de Corse-Mele di Corsica>. Available online: http://www.legifrance.gouv.fr/affichTexte.do?cidTexte=JORFTEXT000022783277 (accessed on 1 september 2013).
- Council Regulation (EC) No 510/2006 on the Protection of Geographical Indications and Designations of Origin for Agricultural Products and Foodstuffs. “Miel de Corse/Mele di Corsica”. EC No: FR-PDO-0105-0066-20.04.2011. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:C:2013:134:0039:0048:EN:PDF (accessed on 1 September 2013).
- Piana, L.; Persano Oddo, L.; Bentabol, A.; Bruneau, E.; Bogdanov, S.; Guyot Declerck, C. Sensory analysis applied to honey: State of the art. Apidologie 2004, 35, 26–27. [Google Scholar] [CrossRef]
- Battesti, M.J. Contribution à la Melissopalynologie Méditerranéenne: Les Miels Corses. Ph.D. Thesis, University of Marseille St. Jérôme, Marseille, France, 1990. [Google Scholar]
- Persano Oddo, L.; Piana, L.; Bogdanov, S.; Bentabol, A.; Gotsiou, P.; Kerkvliet, J. Botanical species giving unifloral honey in Europe. Apidologie 2004, 26, 82–93. [Google Scholar]
- Persano Oddo, L.; Piro, R. Main European unifloral honeys: Descriptive sheets. Apidologie 2004, 35, 38–81. [Google Scholar] [CrossRef]
- Persano Oddo, L.; Sabatini, A.G.; Accorti, M.; Colombo, R.; Marcazzan, G.L.; Piana, L.; Piazza, M.G.; Pulcini, P. I Mieli Uniflorali Italiani—Nuove Schede di Caratterizzazione; Ministero delle Politiche Agricole e Forestali: Gradoli, Italy, 2000. [Google Scholar]
- Floris, I.; Palmieri, N.; Satta, A. Caratteristiche Melissopalinologiche dei Mieli di Sardegna. In I Mieli Regionali Italiani—Caratterizzazion Melissopalinologica; Ministero delle Politiche Agricole Alimentari e Forestali & C.R.A. Istituto Sperimentale per la Zoologia Agraria, Sezione di Apicoltura: Roma, Italy, 2007. [Google Scholar]
- Jeanmonod, D.; Gamisans, J. Flora Corsica; Edisud: Aix-en-Provence, France, 2007. [Google Scholar]
- Battesti, M.J.; Goeury, C. Efficacité de l’analyse mélitopalynologique quantitative pour la certification des origines géographique et botanique des miels: Le modèle des miels corses. Rev. Palaeobot. Palynol. 1992, 75, 77–102. [Google Scholar] [CrossRef]
- Von Der Ohe, W.; Persano Oddo, L.; Piana, M.L.; Morlot, M.; Martin, P. Harmonized methods of melissopalynology. Apidologie 2004, 35, 18–23. [Google Scholar] [CrossRef]
- Cuevas-Glory, L.F.; Pino, J.A.; Santiago, L.S.; Sauri-Duch, E. A review of volatile analytical methods for determining the botanical origin of honey. Food Chem. 2007, 103, 1032–1043. [Google Scholar] [CrossRef]
- Sesta, G.; Piana, M.L.; Persano Oddo, L.; Lusco, L.; Belligoli, P. Methyl anthranilate in Citrus honey. Analytical methode and suitability as a chemical marker. Apidologie 2008, 39, 334–342. [Google Scholar] [CrossRef]
- Alissandrakis, E.; Tarantilis, P.A.; Harizanis, P.C.; Polissiou, M. Aroma investigation of unifloral Greek citrus honey using solid-phase microextraction coupled go gas chromatographic-mass spectrometric analysis. Food Chem. 2007, 100, 396–404. [Google Scholar] [CrossRef]
- Alissandrakis, E.; Daferera, D.; Tarantilis, P.A.; Polissiou, M.; Harizanis, P.C. Ultrasound-assisted extraction of volatile compounds from citrus flowers and citrus honey. Food Chem. 2003, 82, 575–582. [Google Scholar] [CrossRef]
- Ferreres, F.; Giner, J.M.; Tomas-Barberan, F.A. A comparative study of hesperetin and methyl anthranilate as markers of the floral origin of citrus honey. J. Sci. Food Agric. 1994, 65, 371–372. [Google Scholar] [CrossRef]
- Jerkovic, I.; Tuberoso, C.I.G.; Kasum, A.; Marijanovic, Z. Volatile compounds of Asphodelus microcarpus Salzm. et Viv. Honey obtained by HS-SPME and USE analyzed by GC/MS. Chem. Biodivers. 2011, 8, 587–598. [Google Scholar] [CrossRef]
- Tuberoso, C.I.G.; Bifulco, E.; Jerkovic, I.; Caboni, P.; Cabras, P.; Floris, I. Methyl syringate: A chemical marker of asphodel (Asphodelus microcarpus Salzm. et Viv.) monofloral honey. J. Agric. Food Chem. 2009, 57, 3895–3900. [Google Scholar] [CrossRef]
- Donarski, J.A.; Jones, S.A.; Charlton, A.J. Application of cryoprobe 1H Nuclear Magnetic Resonance spectroscopy and multivariate analysis for the verification of Corsican honey. J. Agric. Food Chem. 2008, 56, 5451–5456. [Google Scholar] [CrossRef]
- Woodcock, T.; Downey, G.; O’Donnell, C. Near infrared spectral fingerprinting for confirmation of claimed PDO provenance of honey. Food Chem. 2009, 114, 742–746. [Google Scholar] [CrossRef]
- Stanimirova, I.; Üstün, B.; Cajka, T.; Riddelova, K.; Hajslova, J.; Buydens, L.M.C. Tracing the geographical origin of honeys based on volatile compounds profiles assessment using pattern recognition techniques. Food Chem. 2010, 118, 171–176. [Google Scholar] [CrossRef]
- Gonnet, M.; Vache, G. Le Goût du Miel; U.N.A.F.: Paris, France, 1985. [Google Scholar]
- Yang, Y.; Battesti, M.J.; Djabou, N.; Muselli, A.; Paolini, J.; Tomi, P.; Costa, J. Melissopalynological origin determination and volatile composition analysis of Corsican “chestnut grove” honeys. Food Chem. 2012, 132, 2144–2154. [Google Scholar] [CrossRef]
- Aubert, S.; Gonnet, M. Mesure de la couleur des miels. Apidologie 1983, 14, 105–118. [Google Scholar] [CrossRef]
- Bogdanov, S. Charakterisierung von Schweizer Sortenhonigen. Agrarforschung 1997, 4, 427–430. [Google Scholar]
- Konig, W.A.; Hochmuth, D.H.; Joulain, D. Terpenoids and Related Constituents of Essential oils; Library of Mass Finder 2.1, Institute of Organic Chemistry: Hamburg, Germany, 2001. [Google Scholar]
- National Institute of Standards and Technology (NIST). Spectral Database for Organic Compounds. NIST Chemistry WebBook. Available online: http://webbook.nist.gov/chemistry (accessed on 1 September 2013).
- Yang, Y.; Battesti, M.J.; Paolini, J.; Muselli, A.; Tomi, P.; Costa, J. Melissopalynological origin determination and volatile composition analysis of Corsican “Erica arborea spring maquis” honeys. Food Chem. 2012, 134, 37–47. [Google Scholar] [CrossRef]
- Perez, R.A.; Sanchez-Brunete, C.; Calvo, R.M.; Tadeo, J.L. Analysis of volatiles from Spanish honeys by solid-phase microextraction and gas chromatography-mass spectrometry. J. Agric. Food Chem. 2002, 50, 2633–2637. [Google Scholar] [CrossRef]
- Castro-Vazquez, L.; Diaz-Maroto, M.C.; Perez-Coello, M.S. Aroma composition and new chemical markers of Spanish citrus honeys. Food Chem. 2007, 103, 601–606. [Google Scholar] [CrossRef]
- Castro-Vazquez, L.; Diaz-Maroto, M.C.; Gonzalez-Vinas, M.A.; Perez-Coello, M.S. Differentiation of monofloral citrus, rosemary, eucalyptus, lavender, thyme, and heather honeys based on volatile composition and sensory descriptive analysis. Food Chem. 2009, 112, 1022–1030. [Google Scholar] [CrossRef]
- Aliferis, K.A.; Tarantilis, P.A.; Harizanis, P.C.; Alissandrakis, E. Botanical discrimination and classification of honey samples applying gas chromatography/mass spectrometry fingerprinting of headspace volatile compounds. Food Chem. 2010, 121, 856–862. [Google Scholar] [CrossRef]
- Maurizio, A.; Louveaux, J. Pollens de Plantes Mellifères d’Europe; Union des Groupements Apicoles Français: Paris, France, 1965. [Google Scholar]
- De la Fuente, E.; Sanz, M.L.; Martinez-Castro, I.; Sanz, J.; Ruiz-Matute, A.I. Volatile and carbohydrate composition of rare unifloral honeys from Spain. Food Chem. 2007, 105, 84–93. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, Y.; Battesti, M.-J.; Costa, J.; Paolini, J. Characterization of Botanical and Geographical Origin of Corsican “Spring” Honeys by Melissopalynological and Volatile Analysis. Foods 2014, 3, 128-148. https://doi.org/10.3390/foods3010128
Yang Y, Battesti M-J, Costa J, Paolini J. Characterization of Botanical and Geographical Origin of Corsican “Spring” Honeys by Melissopalynological and Volatile Analysis. Foods. 2014; 3(1):128-148. https://doi.org/10.3390/foods3010128
Chicago/Turabian StyleYang, Yin, Marie-José Battesti, Jean Costa, and Julien Paolini. 2014. "Characterization of Botanical and Geographical Origin of Corsican “Spring” Honeys by Melissopalynological and Volatile Analysis" Foods 3, no. 1: 128-148. https://doi.org/10.3390/foods3010128
APA StyleYang, Y., Battesti, M.-J., Costa, J., & Paolini, J. (2014). Characterization of Botanical and Geographical Origin of Corsican “Spring” Honeys by Melissopalynological and Volatile Analysis. Foods, 3(1), 128-148. https://doi.org/10.3390/foods3010128




