Food Quality Improvement of Soy Milk Made from Short-Time Germinated Soybeans
Abstract
:1. Introduction
2. Materials and Methods
2.1. Soybean Materials
2.2. Chemicals and Reagents
2.3. Germination of Soybean
2.4. Preparation of Soy Milk
2.5. Determination of Quality Attributes
2.5.1. pH Value
2.5.2. Total Solids, Moisture and Ash
2.5.3. Crude Protein, Fat and Carbohydrate
2.5.4. Viscosity
2.6. Assay for Total Phenolic Content (TPC)
2.7. Assay for Phytic Acid
2.8. Assay for Trypsin Inhibitor Activity (TIA)
2.9. Statistical Analysis
3. Results and Discussion
3.1. Effect of Germination on TPC in Soy Milk
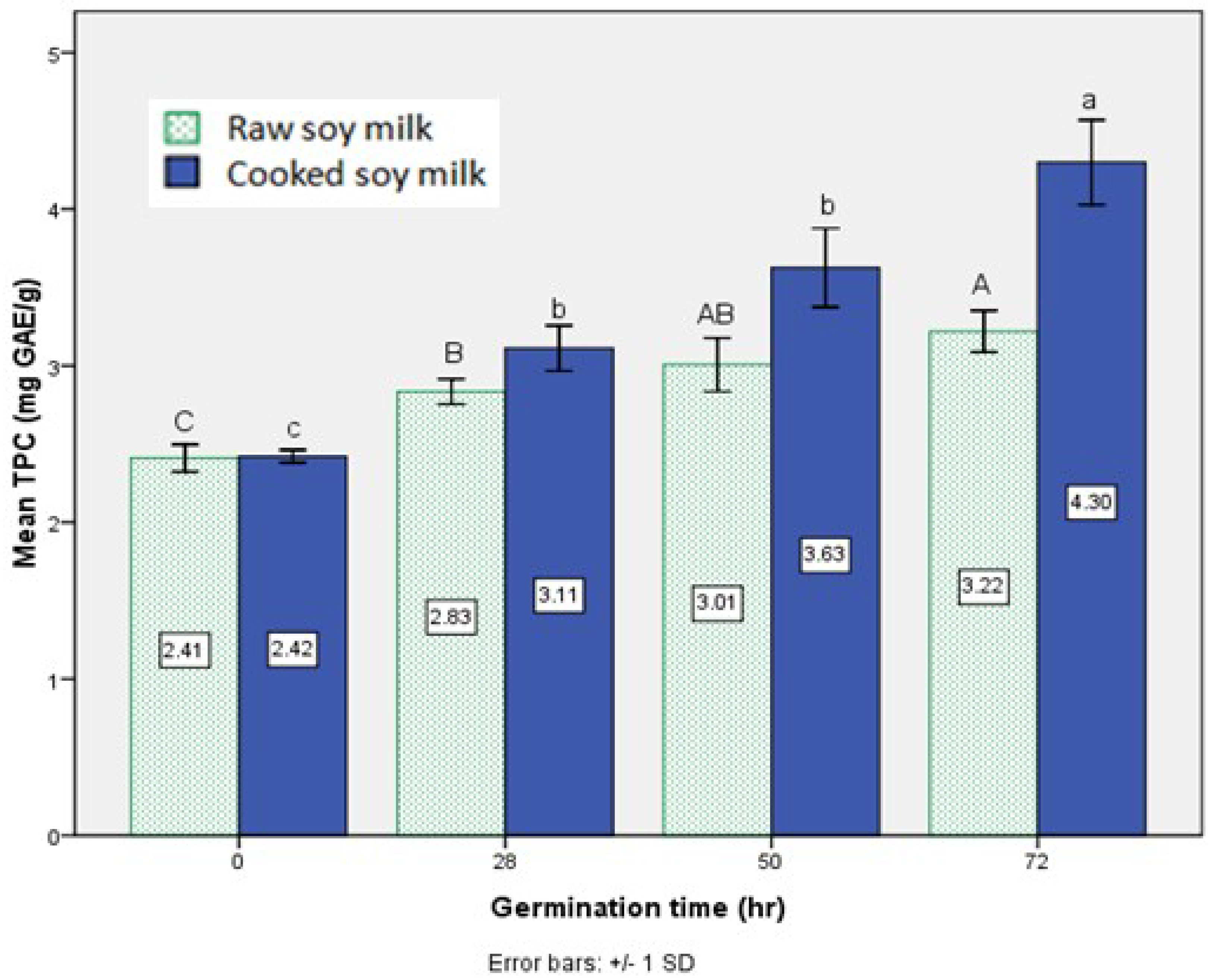
3.2. Effect of Germination on Phytic Acid Content
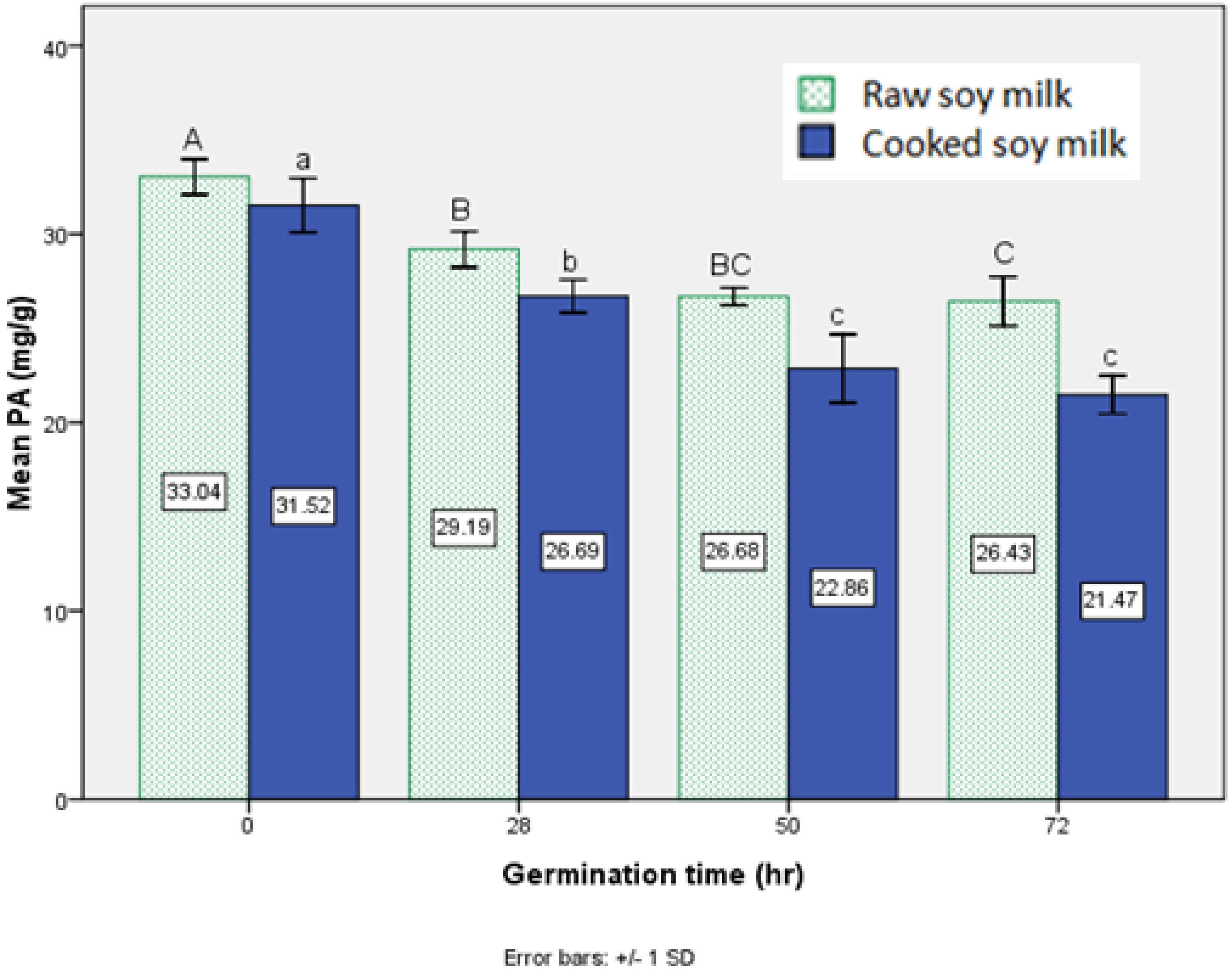
3.3. Effect of Germination on Trypsin Inhibitor Activity in Soy Milk
| Germination time (h) | TIU/g | mg TI/g | Residue% | |||
|---|---|---|---|---|---|---|
| Raw | Cooked | Raw | Cooked | Raw | Cooked | |
| 0 | 56,245 ± 2741 | 8280 ± 485 | 29.60 ± 1.44 A | 4.36 ± 0.26 a | 100 ± 4.9 | 14.7 ± 0.9 |
| 28 | 40,552 ± 325 | 4685 ± 885 | 21.34 ± 0.17 B | 2.47 ± 0.45 b | 72.1 ± 0.6 | 8.3 ± 1.5 |
| 50 | 43,001 ± 2121 | 5397 ± 497 | 22.63 ± 1.12 B | 2.84 ± 0.26 b | 76.5 ± 3.8 | 9.6 ± 0.9 |
| 72 | 45,243 ± 1244 | 5894 ± 18 | 23.81 ± 0.65 B | 3.10 ± 0.01 b | 80.4 ± 2.2 | 10.5 ± 0.0 |
3.4. Selection of Optimum Germination Condition
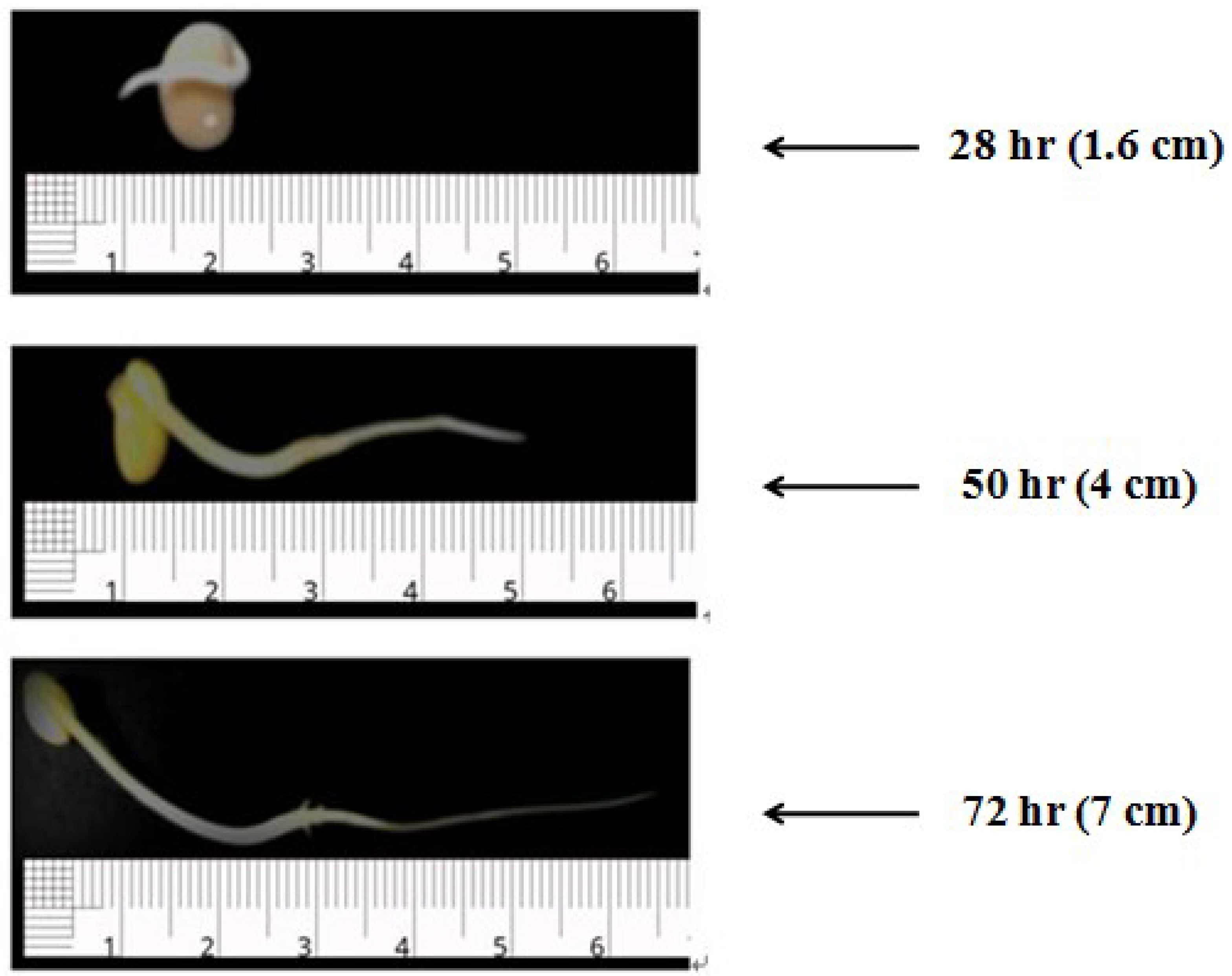
3.5. Profile of Soy Milk Made from Germinated Soybean
| Non-germinated soybean milk | Germinated soybean milk | |
|---|---|---|
| Yield of milk (g of milk/100 g dry beans) | 651.11 (0.96) A | 619.44 (4.19) B |
| pH | 6.72 | 6.51 |
| Specific gravity | 1.008 (0.006) A | 1.002 (0.003) A |
| Viscosity (centipoises/cP) | 3.46 (0.01) A | 2.51 (0.06) B |
| Total solids (g/100 g liquid soy milk) | 5.86 (0.39) A | 4.76 (0.34) B |
| Moisture (g/100 g liquid soy milk) | 94.14 | 95.24 |
| Ash (g/100 g liquid soy milk) | 0.33 (0.01) A | 0.23 (0.01) B |
| Protein (g/100 g liquid soy milk) | 2.19 (0.05) B | 2.66 (0.02) A |
| Fat (g/100 g liquid soy milk) | 1.37 (0.02) A | 1.33 (0.04) A |
| Carbohydrate (g/100 g liquid soy milk) | 1.96 | 0.54 |
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Onuorah, C.E.; Adejare, A.O.; Uhiara, N.S. Comparative physico-chemical evaluation of soy milk and soya cake produced by three different methods. Niger. Food J. 2007, 25, 28–38. [Google Scholar]
- Zieliński, H. Contribution of low molecular weight antioxidants to the antioxidant screen of germinated soybean seeds. Plant Foods Hum. Nutr. 2003, 58, 1–20. [Google Scholar] [CrossRef]
- Kaushik, G.; Naik, S.N.; Satya, S. Effect of domestic processing techniques on the mineral content in soybean. Asian J. Chem. 2010, 22, 5159–5164. [Google Scholar]
- Martinez, A.P.C.; Martinesz, P.C.C.; Souza, M.C.; Canniatti-Brazaca, S.G. Chemical change in soybean grains with germination. Cienc. Tecnol. Aliment. 2011, 31, 23–30. [Google Scholar] [CrossRef]
- Sattar, A.; Akhtar, M.A. Irradiation and germination effects on phytate, protein and amino acids of soybean. Plant Foods Hum. Nutr. 1990, 40, 185–194. [Google Scholar] [CrossRef]
- Paucar-Menacho, L.M.; Berhow, M.A.; Mandarino, J.M.G.; Chang, Y.K.; Mejia, E. Effect of time and temperature on bioactive compounds in germinated Brazilian soybean cultivar BRS 258. Food Res. Int. 2010, 43, 1856–1865. [Google Scholar] [CrossRef]
- Liu, Z.S.; Chang, S.K.C. Nutritional profile and physiochemical properties of commercial soy milk. J. Food Proc. Preserv. 2012. [Google Scholar] [CrossRef]
- China National Standardization Committee of Light Industry. In Plant Protein Beverage—Soymilk and Soy Drink, QB/T 2132–2008; China Light Industry Press: Beijing, China, 2008.
- Xu, B.J.; Chang, S.K.C. A comparative study on phenolic profiles and antioxidant activities of legumes as affected by extraction solvents. J. Food Sci. 2007, 72, S159–S166. [Google Scholar] [CrossRef]
- Gao, Y.; Shang, M.A.; Maroof, S.; Biyashev, R.M.; Grabau, E.A.; Kwanyuen, P.; Burton, J.W.; Buss, G.R. A modified colorimetric method for phytic acid analysis in soybean. Crop Sci. 2007, 47, 1797–1803. [Google Scholar] [CrossRef]
- Hamerstrand, G.E.; Black, L.T.; Glover, J.D. Trypsin inhibitors in soy products: Modification of the standard analytical procedure. Cereal Chem. 1981, 58, 42–45. [Google Scholar]
- Randhir, R.; Lin, Y.T.; Shetty, K. Stimulation of phenolics, antioxidant and antimicrobial activities in dark germinated mung bean sprouts in response to peptide and phytochemical elicitors. Proc. Biochem. 2004, 39, 637–647. [Google Scholar] [CrossRef]
- Lin, P.Y.; Lai, H.M. Bioactive compounds in legumes and their germinated products. J. Agric. Food Chem. 2006, 54, 3807–3814. [Google Scholar] [CrossRef]
- Xu, B.J.; Chang, S.K.C. Isoflavones, Flavan-3-ols, phenolic acids, total phenolic profiles, and antioxidant capacities of soy milk as affected by ultrahigh-temperature and traditional processing methods. J. Agric. Food Chem. 2009, 57, 4706–4717. [Google Scholar] [CrossRef]
- Randhir, R.; Kwon, Y.I.; Lin, Y.T.; Shetty, K. Effect of thermal processing on the phenolic associated health-relevant functionality of selected legume sprouts and seedlings. J. Food Biochem. 2009, 33, 89–112. [Google Scholar] [CrossRef]
- Shamsuddin, A.M. Anti-cancer function of phytic acid. Int. J. Food Sci. Technol. 2002, 37, 769–782. [Google Scholar] [CrossRef]
- Kumar, V.; Sinha, A.K.; Makkar, H.P.S.; Becker, K. Dietary roles of phytate and phytase in human nutrition: A review. Food Chem. 2010, 120, 945–959. [Google Scholar] [CrossRef]
- Rasha, M.K.; Abou-Arab, E.A.; Gibriel, A.Y.; Rasmy, N.M.H.; Abu-Salem, F.M. Effect of legume processing treatments individually or in combination on their phytic acid content. Afr. J. Food Sci. Technol. 2011, 2, 36–46. [Google Scholar]
- Rouhana, A.; Adler-Nissen, J.; Cogan, U.; Frøkiær, H. Heat inactivation kinetics of trypsin inhibitors during high temperature-short time processing of soy milk. J. Food Sci. 1996, 61, 256–269. [Google Scholar]
- Yuan, S.; Chang, S.K.C.; Liu, Z.; Xu, B.J. Elimination of trypsin inhibitor activity and beany flavor in soy milk by consecutive blanching and ultrahigh-temperature (UHT) processing. J. Agric. Food Chem. 2008, 56, 7957–7963. [Google Scholar] [CrossRef]
- Hackler, L.R.; van Buren, J.P.; Steinkraus, K.H.; EI Rawi, I.; Hand, D.B. Effect of heat treatment on nutritive value of soy milk protein fed to weanling rats. J. Food Sci. 1965, 30, 723–728. [Google Scholar] [CrossRef]
- Kwok, K.C.; Shiu, W.W.; Yeung, C.H.; Niranjan, K. Effect of thermal processing on available lysine, thiamine and riboflavin content in soy milk. J. Sci. Food Agric. 1998, 77, 473–478. [Google Scholar] [CrossRef]
- Sugawara, M.; Ito, D.; Yamamoto, K.; Akita, M.; Oguri, S.; Momonoki, Y.S. Kunitz soybean trypsin inhibitor is modified at its C-terminus by novel soybean thiol protease (protease T1). Plant Prod. Sci. 2007, 10, 314–321. [Google Scholar] [CrossRef]
- McGrain, A.K.; Chen, J.C.; Wilson, K.A.; Tan-Wilson, A.L. Proteolytic activities degrading the Bowman-Birk trypsin inhibitors during soybean germination. Phytochemistry 1989, 28, 1013–1017. [Google Scholar] [CrossRef]
- Kumar, V.; Rani, A.; Pandey, V.; Chauhan, G.S. Changes in lipoxygenase isozymes and trypsin inhibitor activity in soybean during germination at different temperatures. Food Chem. 2006, 99, 563–568. [Google Scholar] [CrossRef]
- Wilson, K.A. The release of proteinase inhibitors from legume seeds during germination. Phytochemistry 1980, 19, 2517–2519. [Google Scholar] [CrossRef]
- Momonoki, Y.S.; Sugawara, M.; Watanabe, T. Change in activity of soybean trypsin inhibitor by removed of C-terminal amino acid residues during seed germination. Plant Prod. Sci. 2002, 5, 51–57. [Google Scholar] [CrossRef]
- Chauhan, O.P.; Chauhan, G.S. Development of anti-nutrients free soy beverage using germinated soybean. J. Food Sci. Technol. 2007, 44, 62–65. [Google Scholar]
- Liu, Z.S.; Chang, S.K.C. Effect of soy milk characteristics and cooking conditions on coagulant requirements for making filled tofu. J. Agric. Food Chem. 2004, 52, 3405–3411. [Google Scholar] [CrossRef]
- Harjai, N.; Singh, G. Evaluation of different soybean varieties for manufacture of soy milk. Int. J. Food Sci.Technol. Nutr. 2007, 2, 71–77. [Google Scholar]
- Mostafa, M.M.; Rahma, E.H.; Rady, A.H. Chemical and nutritional changes in soybean during germination. Food Chem. 1987, 23, 257–265. [Google Scholar] [CrossRef]
- Liu, Z.S.; Chang, S.K.C. Soy milk viscosity as influenced by heating methods and soybean varieties. J. Food Proc. Preserv. 2007, 31, 320–333. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jiang, S.; Cai, W.; Xu, B. Food Quality Improvement of Soy Milk Made from Short-Time Germinated Soybeans. Foods 2013, 2, 198-212. https://doi.org/10.3390/foods2020198
Jiang S, Cai W, Xu B. Food Quality Improvement of Soy Milk Made from Short-Time Germinated Soybeans. Foods. 2013; 2(2):198-212. https://doi.org/10.3390/foods2020198
Chicago/Turabian StyleJiang, Susu, Weixi Cai, and Baojun Xu. 2013. "Food Quality Improvement of Soy Milk Made from Short-Time Germinated Soybeans" Foods 2, no. 2: 198-212. https://doi.org/10.3390/foods2020198
APA StyleJiang, S., Cai, W., & Xu, B. (2013). Food Quality Improvement of Soy Milk Made from Short-Time Germinated Soybeans. Foods, 2(2), 198-212. https://doi.org/10.3390/foods2020198






