A Novel Cell Seeding Chamber for Tissue Engineering and Regenerative Medicine
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Seeding Function

2.2. Blood Substitute
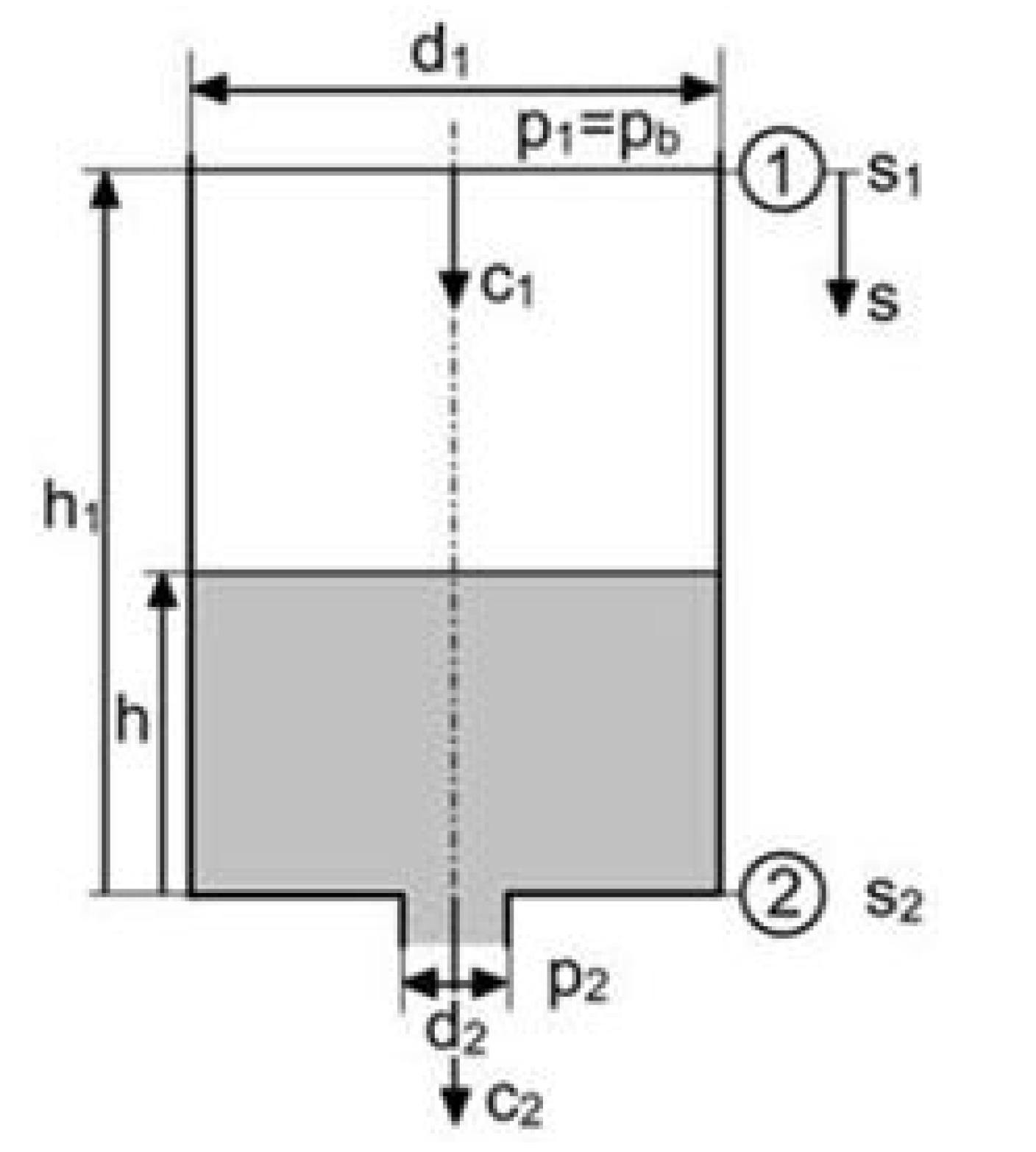
2.3. Analytical Approach of the Cell Suspension Flow
- At first a cell suspension flows out of a reservoir with a variable cell suspension level (h(t));
- The second section is a cell suspension flow through the outflow channel;
- At last there is a drop formation at the end of the outflow channel;
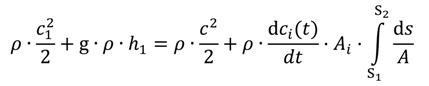
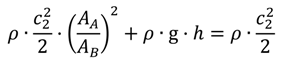
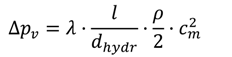
2.4. Cell Seeding Chamber Design

2.5. Media Flow Rate of the Reservoir

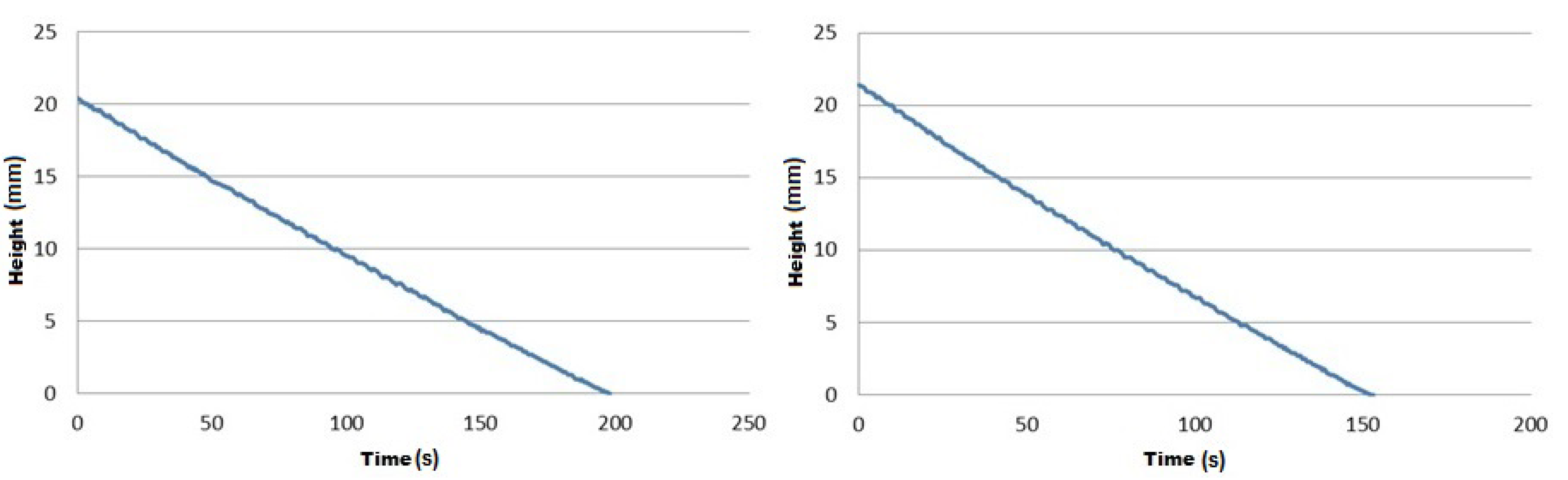
3. Results
Cell Suspension Flow Rate
4. Conclusions
Conflicts of Interest
References
- Schneider, R.K.; Neuss, S.; Knüchel, R.; Perez-Bouza, A. Mesenchymale Stammzellen für das “tissue engineering” des Knochens. Der Phathol. 2010, 31, 138–146. [Google Scholar]
- Poander, S. In-vitro- und In-vivo-Untersuchungen zur Biofunktionalisierung von Knochenersatzmaterialien. Dissertation, Friedrich-Alexander-Universität Erlangen-Nürnberg, Erlangen, Germany, 2009. [Google Scholar]
- Linhart, W.; Briem, D. Knochenersatz 2000 bis 2010. Der Orthop. 2001, 30, 189–192. [Google Scholar] [CrossRef]
- Drosse, I.; Volkmer, E.; Capanna, R.; de Biase, P.; Mutschler, W.; Schieker, M. Tissue engineering for bone defect healing: An update on a multi-component approach. Injury 2008, 39, 9–20. [Google Scholar] [CrossRef]
- Altmann, B.; Löchner, A.; Swain, M.; Kohal, R.J.; Giselbrecht, S.; Gottwald, E.; Steinberg, T.; Tomakidi, P. Differences in morphogenesis of 3D cultured primary human osteoblasts under static and microfluidic growth conditions. Biomaterials 2014, 35, 3208–3219. [Google Scholar] [CrossRef]
- Ordovás, L.; Park, Y.; Verfaillie, C.M. Stem cells and liver engineering. Biotechnol. Adv. 2013, 31, 1094–1107. [Google Scholar] [CrossRef]
- Minuth, W.W.; Sittinger, M.; Kloth, S. Tissue engineering: Generation of differentiated artificial tissues for biomedical applications. Cell Tissue Res. 1998, 291, 1–11. [Google Scholar] [CrossRef]
- Sodian, R.; Lemke, T.; Fritsche, C.; Hoerstrup, S.P.; Fu, P.; Potapov, E.V.; Hausmann, H.; Hetzer, R. Tissue-Engineering Bioreactors: A New Combined Cell-Seeding and Perfusion System for Vascular Tissue Engineering. Tissue Eng. 2002, 8, 863–870. [Google Scholar] [CrossRef]
- Martin, I.; Wendt, D.; Heberer, M. The role of bioreactors in tissue engineering. Trends Biotechnol. 2004, 22, 80–86. [Google Scholar] [CrossRef]
- Martin, Y.; Vermette, P. Bioreactors for tissue mass culture: Design, characterization, and recent advances. Biomaterials 2005, 26, 7481–7503. [Google Scholar]
- Partap, S.; Plunkett, N.A.; O’Brien, F.J. Bioreactors in tissue engineering. In Tissue Engineering; Eberli, D., Ed.; InTech: Dublin, Ireland, 2010; pp. 323–336. [Google Scholar]
- Gardel, L.S.; Serra, L.A.; Reis, R.L.; Gomes, M.E. Use of Perfusion Bioreactors and Large Animal Models for Long Bone Tissue Engineering. Tissue Eng. Part B Rev. 2014. [Google Scholar] [CrossRef]
- Gray, J.D.; Owen, I.; Escudier, M.P. Dynamic scaling of unsteady shear-thinning non-Newtonian cell suspension. Exp. Fluids 2007, 43, 535–546. [Google Scholar] [CrossRef]
- Shibeshi, S.S.; Collins, W.E. The rheology of blood flow in a branched arterial system. Appl. Rheol. 2005, 15, 398–405. [Google Scholar]
- Sousa, P.C.; Pinho, F.T.; Oliveira, M.S.; Alves, M.A. Extensional flow of blood analog solutions in microcell suspensionic devices. Biomicrofluidics 2011. [Google Scholar] [CrossRef] [Green Version]
- Surek, D.; Stempin, S. Angewandte Strömungsmechanik; Teubner Verlag: Wiesbaden, Germany, 2007. [Google Scholar]
- Wagner, W. Strömung und Druckverlust; Vogel: Würzburg, Germany, 2001. [Google Scholar]
- Sart, S.; Agathos, S.N.; Li, Y. Process engineering of stem cell metabolism for large scale expansion and differentiation in bioreactors. Biochem. Eng. J. 2014, 84, 74–82. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hennig, J.; Drescher, P.; Riedl, C.; Schieker, M.; Seitz, H. A Novel Cell Seeding Chamber for Tissue Engineering and Regenerative Medicine. Processes 2014, 2, 361-370. https://doi.org/10.3390/pr2020361
Hennig J, Drescher P, Riedl C, Schieker M, Seitz H. A Novel Cell Seeding Chamber for Tissue Engineering and Regenerative Medicine. Processes. 2014; 2(2):361-370. https://doi.org/10.3390/pr2020361
Chicago/Turabian StyleHennig, Jörn, Philipp Drescher, Christina Riedl, Matthias Schieker, and Hermann Seitz. 2014. "A Novel Cell Seeding Chamber for Tissue Engineering and Regenerative Medicine" Processes 2, no. 2: 361-370. https://doi.org/10.3390/pr2020361
APA StyleHennig, J., Drescher, P., Riedl, C., Schieker, M., & Seitz, H. (2014). A Novel Cell Seeding Chamber for Tissue Engineering and Regenerative Medicine. Processes, 2(2), 361-370. https://doi.org/10.3390/pr2020361





