Green Process Engineering as the Key to Future Processes
Abstract
:1. Introduction
1.1. Issues and Challenges of Green Process Engineering
2. Applications of Green Process Engineering (GPE)
2.1. Production of Biodiesel and Biofuels
2.2. Green Synthesis of Organic Carbonates from Carbon Dioxide
2.3. Production of Commercially Important Epoxide Building Blocks

2.4. Green Chemical Processing Using Supercritical CO2 (SC-CO2)


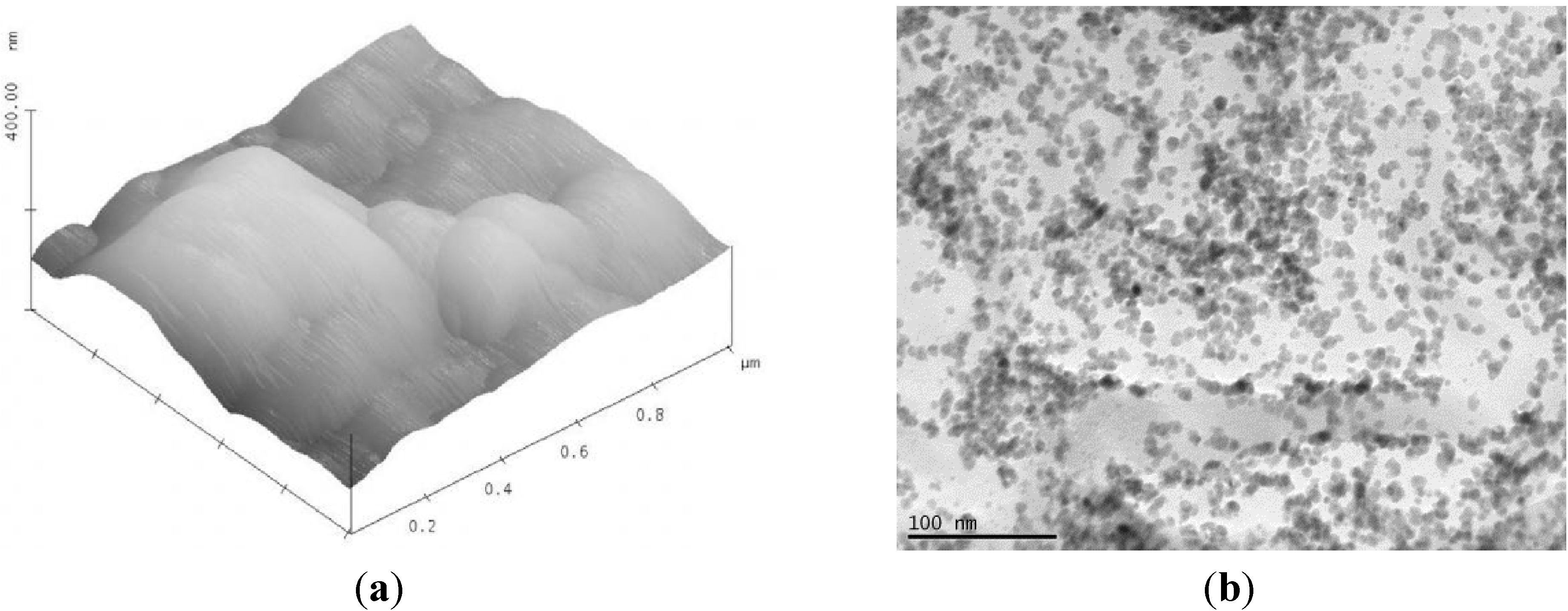
2.5. Continuous Hydrothermal Flow Synthesis (CHFS)
2.6. Supercritical Water Oxidation (SCWO) Process
2.7. Green Process Using Biphasic Catalysis
3. Conclusions
Acknowledgments
Conflicts of Interest
References
- Website of U.S. Environmental Protection Agency, Washington, D.C. Available online: http://www.epa.gov/oppt/greenengineering/pubs/whats_ge.html (accessed on 3 February 2014).
- Jenck, J.F.; Agterberg, F.; Droesher, M.J. Products and processes for a sustainable chemical industry: A review of achievements and prospects. Green Chem. 2004, 6, 544–556. [Google Scholar] [CrossRef]
- Anastas, P.T.; Zimmerman, J.B. Design through the 12 principles of green engineering. Environ. Sci. Technol. 2003, 37, 94A–101A. [Google Scholar] [CrossRef]
- Abraham, M.A.; Nguyen, N. “Green engineering: Defining the principles”—Resdts from the sandestin conference. Environ. Prog. 2003, 22, 233–236. [Google Scholar] [CrossRef]
- Patel, D.; Kellici, S.; Saha, B. Some novel aspects of green process engineering. Chim. Oggi 2013, 31, 57–61. [Google Scholar]
- Website of Engineering and Physical Sciences Research Council (EPSRC). Available online: http://www.epsrc.ac.uk/newsevents/news/2012/Pages/greenengineering.aspx (accessed on 3 February 2014).
- Liu, C.; Li, F.; Ma, L.P.; Cheng, H.M. Advanced materials for energy storage. Adv. Mater. 2010, 22, E28–E62. [Google Scholar] [CrossRef]
- Diwekar, U.M. Greener by design. Environ. Sci. Technol. 2003, 37, 5432–5444. [Google Scholar] [CrossRef]
- Grimm, N.B.; Faeth, S.H.; Golubiewski, N.E.; Redman, C.L.; Wu, J.G.; Bai, X.M.; Briggs, J.M. Global change and the ecology of cities. Science 2008, 319, 756–760. [Google Scholar] [CrossRef]
- Lotero, E.; Liu, Y.J.; Lopez, D.E.; Suwannakarn, K.; Bruce, D.A.; Goodwin, J.G. Synthesis of biodiesel via acid catalysis. Ind. Eng. Chem. Res. 2005, 44, 5353–5363. [Google Scholar] [CrossRef]
- Abbaszaadeh, A.; Ghobadian, B.; Omidkhah, M.R.; Najafi, G. Current biodiesel production technologies: A comparative review. Energy Convers. Manag. 2012, 63, 138–148. [Google Scholar] [CrossRef]
- Rawat, I.; Kumar, R.R.; Mutanda, T.; Bux, F. Biodiesel from microalgae: A critical evaluation from laboratory to large scale production. Appl. Energy 2013, 103, 444–467. [Google Scholar] [CrossRef]
- Abidin, S.Z.; Haigh, K.F.; Saha, B. Esterification of free fatty acids in used cooking oil using ion-exchange resins as catalysts: An efficient pretreatment method for biodiesel feedstock. Ind. Eng. Chem. Res. 2012, 51, 14653–14664. [Google Scholar] [CrossRef]
- Haigh, K.F.; Abidin, S.Z.; Saha, B.; Vladisavljević, G.T. Pretreatment of used cooking oil for the preparation of biodiesel using heterogeneous catalysis. Prog. Colloid Polym. Sci. 2012, 139, 19–22. [Google Scholar]
- Demirbaş, A. Biodiesel fuels from vegetable oils via catalytic and non-catalytic supercritical alcohol transesterifications and other methods: A survey. Energy Convers. Manag. 2003, 44, 2093–2109. [Google Scholar] [CrossRef]
- Abidin, S.Z.; Vladisavljević, G.; Saha, B. Two-stage esterification-transesterification process for synthesis of biodiesel from used cooking oil (UCO) using novel heterogeneous catalysts. In Proceedings of the 9th World Congress of Chemical Engineering (WCCE9), Seoul, Korea, 18–23 August 2013; Paper number ThO-T409-6. Korean Institute of Chemical Engineers (KIChE): Seoul, Korea, 2013. [Google Scholar]
- Dorado, M.P.; Ballesteros, E.; Mittelbach, M.; Lopez, F.J. Kinetic parameters affecting the alkali-catalyzed transesterification process of used olive oil. Energy Fuels 2004, 18, 1457–1462. [Google Scholar] [CrossRef]
- Haigh, K.F.; Abidin, S.Z.; Vladisavljević, G.T.; Saha, B. Comparison of Novozyme 435 and Purolite D5081 as heterogeneous catalysts for the pretreatment of used cooking oil for biodiesel production. Fuel 2013, 111, 186–193. [Google Scholar] [CrossRef]
- Kim, H.J.; Kang, B.S.; Kim, M.J.; Park, Y.M.; Kim, D.K.; Lee, J.S.; Lee, K.Y. Transesterification of vegetable oil to biodiesel using heterogeneous base catalyst. Catal. Today 2004, 93–95, 315–320. [Google Scholar]
- Du, W.; Xu, Y.Y.; Liu, D.H. Lipase-catalysed transesterification of soya bean oil for biodiesel production during continuous batch operation. Biotechnol. Appl. Biochem. 2003, 38, 103–106. [Google Scholar] [CrossRef]
- Haigh, K.F.; Vladisavljević, G.T.; Reynolds, J.C.; Nagy, Z.; Saha, B. Kinetics of the pre-treatment of used cooking oil using Novozyme 435 for biodiesel production. Chem. Eng. Res. Des. 2014. [Google Scholar] [CrossRef]
- Hoekman, S.K.; Broch, A.; Robbins, C.; Ceniceros, E.; Natarajan, M. Review of biodiesel composition, properties, and specifications. Renew. Sustain. Energy Rev. 2012, 16, 143–169. [Google Scholar] [CrossRef]
- Atadashi, I.M.; Aroua, M.K.; Aziz, A.R.A.; Sulaiman, N.M.N. The effects of catalysts in biodiesel production: A review. J. Ind. Eng. Chem. 2013, 19, 14–26. [Google Scholar] [CrossRef]
- Gog, A.; Roman, M.; Toşa, M.; Paizs, C.; Irimie, F.D. Biodiesel production using enzymatic transesterification—Current state and perspectives. Renew. Energy 2012, 39, 10–16. [Google Scholar] [CrossRef]
- Talebian-Kiakalaieh, A.; Amin, N.A.S.; Mazaheri, H. A review on novel processes of biodiesel production from waste cooking oil. Appl. Energy 2013, 104, 683–710. [Google Scholar] [CrossRef]
- Abidin, S.Z.; Patel, D.; Saha, B. Quantitative analysis of fatty acids composition in the used cooking oil (UCO) by gas chromatography-mass spectrometry (GC-MS). Can. J. Chem. Eng. 2013, 91, 1896–1903. [Google Scholar] [CrossRef]
- Jachuck, R.; Pherwani, G.; Gorton, S.M. Green engineering: Continuous production of biodiesel using an alkaline catalyst in an intensified narrow channel reactor. J. Environ. Monit. 2009, 11, 642–647. [Google Scholar]
- Demirbaş, A.; Demirbaş, M.F. Importance of algae oil as a source of biodiesel. Energy Convers. Manag. 2011, 52, 163–170. [Google Scholar] [CrossRef]
- Blatti, J.L.; Burkart, M.D. Releasing stored solar energy within pond scum: Biodiesel from algal lipids. J. Chem. Educ. 2012, 89, 239–242. [Google Scholar] [CrossRef]
- Pfromm, P.H.; Amanor-Boadu, V.; Nelson, R. Sustainability of algae derived biodiesel: A mass balance approach. Bioresour. Technol. 2011, 102, 1185–1193. [Google Scholar] [CrossRef]
- Gallagher, B.J. The economics of producing biodiesel from algae. Renew. Energy 2011, 36, 158–162. [Google Scholar] [CrossRef]
- Lee, D.H. Algal biodiesel economy and competition among bio-fuels. Bioresour. Technol. 2011, 102, 43–49. [Google Scholar] [CrossRef]
- Wiley, P.E.; Campbell, J.E.; Mckuin, B. Production of biodiesel and biogas from algae: A review of process train options. Water Environ. Res. 2011, 83, 326–338. [Google Scholar] [CrossRef]
- Halim, R.; Danquah, M.K.; Webley, P.A. Extraction of oil from microalgae for biodiesel production: A review. Biotechnol. Adv. 2012, 30, 709–732. [Google Scholar] [CrossRef]
- Chen, L.; Liu, T.Z.; Zhang, W.; Chen, X.L.; Wang, J.F. Biodiesel production from algae oil high in free fatty acids by two-step catalytic conversion. Bioresour. Technol. 2012, 111, 208–214. [Google Scholar] [CrossRef]
- Suganya, T.; Gandhi, N.N.; Renganathan, S. Production of algal biodiesel from marine macroalgae Enteromorpha compressa by two step process: Optimization and kinetic study. Bioresour. Technol. 2013, 128, 392–400. [Google Scholar] [CrossRef]
- Krohn, B.J.; McNeff, C.V.; Yan, B.W.; Nowlan, D. Production of algae-based biodiesel using the continuous catalytic Mcgyan® process. Bioresour. Technol. 2011, 102, 94–100. [Google Scholar] [CrossRef]
- Patil, P.D.; Gude, V.G.; Mannarswamy, A.; Deng, S.G.; Cooke, P.; Munson-McGee, S.; Rhodes, I.; Lammers, P.; Nirmalakhandan, N. Optimization of direct conversion of wet algae to biodiesel under supercritical methanol conditions. Bioresour. Technol. 2011, 102, 118–122. [Google Scholar] [CrossRef]
- Sathish, A.; Sims, R.C. Biodiesel from mixed culture algae via a wet lipid extraction procedure. Bioresour. Technol. 2012, 118, 643–647. [Google Scholar] [CrossRef]
- Martín, M.; Grossmann, I.E. Simultaneous optimization and heat integration for biodiesel production from cooking oil and algae. Ind. Eng. Chem. Res. 2012, 51, 7998–8014. [Google Scholar] [CrossRef]
- Shiels, R.A.; Jones, C.W. Homogeneous and heterogeneous 4-(N,N-dialkylamino)pyridines as effective single component catalysts in the synthesis of propylene carbonate. J. Mol. Catal. A Chem. 2007, 261, 160–166. [Google Scholar] [CrossRef]
- Wang, J.Q.; Yue, X.D.; Cai, F.; He, L.N. Solventless synthesis of cyclic carbonates from carbon dioxide and epoxides catalyzed by silica-supported ionic liquids under supercritical conditions. Catal. Commun. 2007, 8, 167–172. [Google Scholar] [CrossRef]
- Verevkin, S.P.; Emel’yanenko, V.N.; Toktonov, A.V.; Chernyak, Y.; Schäffner, B.; Börner, A. Cyclic alkylene carbonates. Experiment and first principle calculations for prediction of thermochemical properties. J. Chem. Thermodyn. 2008, 40, 1428–1432. [Google Scholar]
- Bian, J.; Xiao, M.; Wang, S.J.; Wang, X.J.; Lu, Y.X.; Meng, Y.Z. Highly effective synthesis of dimethyl carbonate from methanol and carbon dioxide using a novel copper-nickel/graphite bimetallic nanocomposite catalyst. Chem. Eng. J. 2009, 147, 287–296. [Google Scholar] [CrossRef]
- Ulusoy, M.; Kilic, A.; Durgun, M.; Tasci, Z.; Cetinkaya, B. Silicon containing new salicylaldimine Pd(II) and Co(II) metal complexes as efficient catalysts in transformation of carbon dioxide (CO2) to cyclic carbonates. J. Organomet. Chem. 2011, 696, 1372–1379. [Google Scholar] [CrossRef]
- Bhanage, B.M.; Fujita, S.; Ikushima, Y.; Arai, M. Synthesis of dimethyl carbonate and glycols from carbon dioxide, epoxides, and methanol using heterogeneous basic metal oxide catalysts with high activity and selectivity. Appl. Catal. A 2001, 219, 259–266. [Google Scholar] [CrossRef]
- Sun, J.M.; Fujita, S.; Arai, M. Development in the green synthesis of cyclic carbonate from carbon dioxide using ionic liquids. J. Organomet. Chem. 2005, 690, 3490–3497. [Google Scholar] [CrossRef]
- Sakakura, T.; Choi, J.C.; Yasuda, H. Transformation of carbon dioxide. Chem. Rev. 2007, 107, 2365–2387. [Google Scholar] [CrossRef]
- Dai, W.L.; Chen, L.; Yin, S.F.; Luo, S.L.; Au, C.T. 3-(2-Hydroxyl-ethyl)-1-propylimidazolium bromide immobilized on SBA-15 as efficient catalyst for the synthesis of cyclic carbonates via the coupling of carbon dioxide with epoxides. Catal. Lett. 2010, 135, 295–304. [Google Scholar] [CrossRef]
- Song, J.; Zhang, B.; Jiang, T.; Yang, G.; Han, B. Synthesis of cyclic carbonates and dimethyl carbonate using CO2 as a building block catalyzed by MOF-5/KI and MOF-5/KI/K2CO3. Front. Chem. China 2011, 6, 21–30. [Google Scholar] [CrossRef]
- Kim, H.S.; Kim, J.J.; Kwon, H.N.; Chung, M.J.; Lee, B.G.; Jang, H.G. Well-defined highly active heterogeneous catalyst system for the coupling reactions of carbon dioxide and epoxides. J. Catal. 2002, 205, 226–229. [Google Scholar] [CrossRef]
- Alvaro, M.; Baleizao, C.; Carbonell, E.; El Ghoul, M.; García, H.; Gigante, B. Polymer-bound aluminium salen complex as reusable catalysts for CO2 Insertion into epoxides. Tetrahedron 2005, 61, 12131–12139. [Google Scholar] [CrossRef]
- Yasuda, H.; He, L.N.; Takahashi, T.; Sakakura, T. Non-halogen catalysts for propylene carbonate synthesis from CO2 under supercritical conditions. Appl. Catal. A 2006, 298, 177–180. [Google Scholar] [CrossRef]
- Udayakumar, S.; Park, S.W.; Park, D.W.; Choi, B.S. Immobilization of ionic liquid on hybrid MCM-41 system for the chemical fixation of carbon dioxide on cyclic carbonate. Catal. Commun. 2008, 9, 1563–1570. [Google Scholar] [CrossRef]
- Yamaguchi, K.; Ebitani, K.; Yoshida, T.; Yoshida, H.; Kaneda, K. Mg-Al mixed oxides as highly active acid-base catalysts for cycloaddition of carbon dioxide to epoxides. J. Am. Chem. Soc. 1999, 121, 4526–4527. [Google Scholar] [CrossRef]
- Barbarini, A.; Maggi, R.; Mazzacani, A.; Mori, G.; Sartori, G.; Sartorio, R. Cycloaddition of CO2 to epoxides over both homogeneous and silica-supported guanidine catalysts. Tetrahedron Lett. 2003, 44, 2931–2934. [Google Scholar] [CrossRef]
- Wang, J.Q.; Kong, D.L.; Chen, J.Y.; Cai, F.; He, L.N. Synthesis of cyclic carbonates from epoxides and carbon dioxide over silica-supported quaternary ammonium salts under supercritical conditions. J. Mol. Catal. A Chem. 2006, 249, 143–148. [Google Scholar] [CrossRef]
- Zhang, X.H.; Zhao, N.; Wei, W.; Sun, Y.H. Chemical fixation of carbon dioxide to propylene carbonate over amine-functionalized silica catalysts. Catal. Today 2006, 115, 102–106. [Google Scholar] [CrossRef]
- Adeleye, A.I.; Patel, D.; Niyogi, D.; Saha, B. An efficient and greener synthesis of propylene carbonate from carbon dioxide and propylene oxide. Unpublished work (in preparation). 2014. [Google Scholar]
- Saada, R.; Kellici, S.; Saha, B. Synthesis of dimethyl carbonate from carbon dioxide and methanol using ceria-zirconia doped graphene catalyst. Unpublished work (in preparation). 2014. [Google Scholar]
- Sienel, G.; Rieth, R.; Rowbottom, K.T. Epoxides. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2000. [Google Scholar]
- Bauer, K.; Garbe, D.; Surburg, H. Common Fragrance and Flavour Materials, 4th ed.; Wiley-VCH: Weinheim, Germany, 2001; pp. 143–145. [Google Scholar]
- Andrei, K.Y. Aziridines and Epoxides in Organic Synthesis; Wiley-VCH: Weinheim, Germany, 2006; p. 516. [Google Scholar]
- Ambroziak, K.; Mbeleck, R.; He, Y.; Saha, B.; Sherrington, D.C. Investigation of batch alkene epoxidations catalyzed by polymer-supported Mo(VI) complexes. Ind. Eng. Chem. Res. 2009, 48, 3293–3302. [Google Scholar] [CrossRef]
- Serafimidou, A.; Stamatis, A.; Louloudi, M. Manganese(II) complexes of imidazole based-acetamide as homogeneous and heterogenised catalysts for alkene epoxidation with H2O2. Catal. Commun. 2008, 9, 35–39. [Google Scholar] [CrossRef]
- Sözen-Aktaş, P.; Manoury, E.; Demirhan, F.; Poli, R. Molybdenum versus tungsten for the epoxidation of cyclooctene catalyzed by [Cp*2M2O5]. Eur. J. Inorg. Chem. 2013, 2013, 2728–2735. [Google Scholar]
- Kollar, J. Epoxidation Process. US Patent 3351635, 7 November 1967. [Google Scholar]
- Salavati-Niasari, M.; Esmaeili, E.; Seyghalkar, H.; Bazarganipour, M. Cobalt(II) Schiff base complex on multi-wall carbon nanotubes (MWNTs) by covalently grafted method: Synthesis, characterization and liquid phase epoxidation of cyclohexene by air. Inorg. Chim. Acta 2011, 375, 11–19. [Google Scholar] [CrossRef]
- Dell’Anna, M.M.; Mastrorilli, P.; Nobile, C.F.; Suranna, G.P. Epoxidation of olefins catalyzed by polymer-supported metal beta-ketoesterato complexes of iron(III), nickel(II) and cobalt(II). J. Mol. Catal. A Chem. 1995, 103, 17–22. [Google Scholar] [CrossRef]
- Antony, R.; Tembe, G.L.; Ravindranathan, M.; Ram, R.N. Polymer supported Ru(III) complexes, synthesis and catalytic activity. Polymer 1998, 39, 4327–4333. [Google Scholar] [CrossRef]
- Deleuze, H.; Schultze, X.; Sherrington, D.C. Reactivity of some polymer-supported titanium catalysts in transesterification and epoxidation reactions. J. Mol. Catal. A Chem. 2000, 159, 257–267. [Google Scholar] [CrossRef]
- Brulé, E.; de Miguel, Y.R.; Hii, K.K. Chemoselective epoxidation of dienes using polymer-supported manganese porphyrin catalysts. Tetrahedron 2004, 60, 5913–5918. [Google Scholar] [CrossRef]
- Grivani, G.; Tangestaninejad, S.; Halili, A. A readily prepared, highly reusable and active polymer-supported molybdenum carbonyl Schiff base complex as epoxidation catalyst. Inorg. Chem. Commun. 2007, 10, 914–917. [Google Scholar] [CrossRef]
- Islam, S.M.; Roy, A.S.; Mondal, P.; Mobarok, M.; Roy, B.; Salam, N.; Paul, S.; Mondal, S. Olefin epoxidation with tert-butyl hydroperoxide catalyzed by functionalized polymer-supported copper(II) Schiff base complex. Monat. Chem. 2012, 143, 815–823. [Google Scholar] [CrossRef]
- Sherrington, D.C. Polymer-supported metal complex alkene epoxidation catalysts. Catal. Today 2000, 57, 87–104. [Google Scholar] [CrossRef]
- Mbeleck, R.; Ambroziak, K.; Saha, B.; Sherrington, D.C. Stability and recycling of polymer-supported Mo(VI) alkene epoxidation catalysts. React. Funct. Polym. 2007, 67, 1448–1457. [Google Scholar] [CrossRef]
- Tangestaninejad, S.; Mirkhani, V.; Moghadam, M.; Grivani, G. Readily prepared heterogeneous molybdenum-based catalysts as highly recoverable, reusable and active catalysts for alkene epoxidation. Catal. Commun. 2007, 8, 839–844. [Google Scholar] [CrossRef]
- Barrio, L.; Campos-Martin, J.M.; de Frutos, M.P.; Fierro, J.L.G. Alkene epoxidation with ethylbenzene hydroperoxides using molybdenum heterogeneous catalysts. Ind. Eng. Chem. Res. 2008, 47, 8016–8024. [Google Scholar] [CrossRef]
- Ambroziak, K.; Mbeleck, R.; Saha, B.; Sherrington, D.C. Greener and sustainable method for alkene epoxidations by polymer-supported Mo(VI) catalysts. Int. J. Chem. React. Eng. 2010, 8, A125:1–A125:13. [Google Scholar]
- Mbeleck, R.; Mohammed, M.L.; Ambroziak, K.; Sherrington, D.C.; Saha, B. Cleaner and efficient alkenes/terpenes epoxidation process catalysed by novel polymer-supported Mo(VI) complexes. In Proceedings of the IEX 2012, Cambridge, UK, 19–21 September 2012; Society of Chemical Industry (SCI): London, UK, 2012. [Google Scholar]
- Grivani, G.; Akherati, A. Polymer-supported bis (2-hydroxyanyl) acetylacetonato molybdenyl Schiff base catalyst as effective, selective and highly reusable catalyst in epoxidation of alkenes. Inorg. Chem. Commun. 2013, 28, 90–93. [Google Scholar] [CrossRef]
- Mohammed, M.L.; Mbeleck, R.; Sherrington, D.C.; Saha, B. Greener and efficient alkene epoxidation process. In Proceedings of the 9th European Congress of Chemical Engineering (ECCE9), Hague, The Netherlands, 21–24 April 2013; Paper number 794, ECCE9-ECAB2 App. MCI-Eurocongress v.o.f.: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Mbeleck, R.; Mohammed, M.L.; Sherrington, D.C.; Saha, B. Environmentally benign alkene epoxidation process. In Proceedings of the 9th World Congress of Chemical Engineering (WCCE9), Seoul, Korea, 18–23 August 2013; Paper number FrO-T104-3. Paper number FrO-T104-3, Korean Institute of Chemical Engineers (KIChE): Seoul, Korea, 2013. [Google Scholar]
- Mohammed, M.L.; Patel, D.; Mbeleck, R.; Niyogi, D.; Sherrington, D.C.; Saha, B. Optimisation of alkene epoxidation catalysed by polymer supported Mo(VI) complexes and application of artificial neural network for prediction of catalytic performances. Appl. Catal. A 2013, 466, 142–152. [Google Scholar] [CrossRef]
- Mbeleck, R.; Mohammed, M.L.; Ambroziak, K.; Sherrington, D.C.; Saha, B. Efficient epoxidation of cyclododecene and dodecene catalysed by polybezimidazole-supported Mo(VI) complex. Unpublished work (in preparation). 2014. [Google Scholar]
- Oku, N.; Seo, T. Process for Producing Propylene Oxide. U.S. Patent 6646138, 11 November 2003. [Google Scholar]
- Saha, B.; Ambroziak, K.; Sherrington, D.C.; Mbeleck, R. Process. Publication Number WO/2011/012869, International Patent Application Number PCT/GB2010/001458. 3 February 2011. [Google Scholar]
- Mbeleck, R.; Ambroziak, K.; Sherrington, D.C.; Saha, B. Production of commercially important epoxide building blocks by catalytic distillation. Unpublished work (in preparation). 2015. [Google Scholar]
- Saha, B. Continuous clean alkene epoxidation process technology for the production of commercially important epoxide building blocks. In Proceedings of the 3rd International Conference of the Flow Chemistry Society, Munich, Germany, 19–20 March 2013; Select Biosciences Ltd.: Sudbury, UK, 2013. [Google Scholar]
- Mohammed, M.L.; Mbeleck, R.; Niyogi, D.; Sherrington, D.C.; Saha, B. Continuous alkene epoxidation process catalysed by polymer supported molybdenum (VI) complex. Unpublished work (in preparation). 2014. [Google Scholar]
- Browne, D.L.; Baumann, M.; Harji, B.H.; Baxendale, I.R.; Ley, S.V. A new enabling technology for convenient laboratory scale continuous flow processing at low temperatures. Org. Lett. 2011, 13, 3312–3315. [Google Scholar] [CrossRef]
- Kelly, C.B.; Lee, C.; Mercadante, M.A.; Leadbeater, N.E. A continuous-flow approach to palladium-catalyzed alkoxycarbonylation reactions. Org. Process Res. Dev. 2011, 15, 717–720. [Google Scholar] [CrossRef]
- Nagy, K.D.; Jensen, K.F. Catalytic processes in small scale flow reactors Status and opportunities. Chim. Oggi 2011, 29, 18–21. [Google Scholar]
- Palde, P.B.; Jamison, T.F. Safe and efficient tetrazole synthesis in a continuous-flow microreactor. Angew. Chem. Int. Ed. 2011, 50, 3525–3528. [Google Scholar] [CrossRef] [Green Version]
- Savage, P.E.; Gopalan, S.; Mizan, T.I.; Martino, C.J.; Brock, E.E. Reactions at supercritical conditions: Applications and fundamentals. AIChE J. 1995, 41, 1723–1778. [Google Scholar] [CrossRef]
- Darr, J.A.; Poliakoff, M. New directions in inorganic and metal-organic coordination chemistry in supercritical fluids. Chem. Rev. 1999, 99, 495–541. [Google Scholar] [CrossRef]
- Darr, J.A.; Kellici, S.; Rehman, I.U. Titania nanospheres from supercritical fluids. IEE Proc. Nanobiotechnol. 2005, 152, 109–111. [Google Scholar] [CrossRef]
- Jung, J.; Perrut, M. Particle design using supercritical fluids: Literature and patent survey. J. Supercrit. Fluids 2001, 20, 179–219. [Google Scholar] [CrossRef]
- Sheldon, R.A. Green solvents for sustainable organic synthesis: State of the art. Green Chem. 2005, 7, 267–278. [Google Scholar] [CrossRef]
- Eckert, C.A.; Knutson, B.L.; Debenedetti, P.G. Supercritical fluids as solvents for chemical and materials processing. Nature 1996, 383, 313–318. [Google Scholar] [CrossRef]
- Kendall, J.L.; Canelas, D.A.; Young, J.L.; DeSimone, J.M. Polymerizations in supercritical carbon dioxide. Chem. Rev. 1999, 99, 543–563. [Google Scholar] [CrossRef]
- Cooper, A.I. Polymer synthesis and processing using supercritical carbon dioxide. J. Mater. Chem. 2000, 10, 207–234. [Google Scholar] [CrossRef]
- Kazarian, S.G. Polymer processing with supercritical fluids. Polym. Sci, Ser. C 2000, 42, 78–101. [Google Scholar]
- Gregorowicz, J.; Wawrzyńska, E.P.; Parzuchowski, P.G.; Fraś, Z.; Rokicki, G.; Wojciechowski, K.; Wieczorek, S.A.; Wiśniewska, A.; Plichta, A.; Dąbrowski, K.; et al. Synthesis, characterization, and solubility in supercritical carbon dioxide of hyperbranched copolyesters. Macromolecules 2013, 46, 7180–7195. [Google Scholar] [CrossRef]
- Tryznowski, M.; Tomczyk, K.; Fraś, Z.; Gregorowicz, J.; Rokicki, G.; Wawrzyńska, E.; Parzuchowski, P.G. Aliphatic hyperbranched polycarbonates: Synthesis, characterization, and solubility in supercritical carbon dioxide. Macromolecules 2012, 45, 6819–6829. [Google Scholar] [CrossRef]
- Smith, C.A.; Cloutet, E.; Tassaing, T.; Cramail, H. Synthesis of polyurethane particles in supercritical carbon dioxide using organocatalysts or organocatalytic surfactants. Green Chem. 2013, 15, 2769–2776. [Google Scholar] [CrossRef]
- Mohamed, A.L.; Er-Rafik, M.; Moller, M. Supercritical carbon dioxide assisted silicon based finishing of cellulosic fabric: A novel approach. Carbohydr. Polym. 2013, 98, 1095–1107. [Google Scholar] [CrossRef]
- Liu, W.W.; Zhang, B.; Li, Y.Z.; He, Y.M.; Zhang, H.C. An environmentally friendly approach for contaminants removal using supercritical CO2 for remanufacturing industry. Appl. Surf. Sci. 2014, 292, 142–148. [Google Scholar] [CrossRef]
- Matson, D.W.; Fulton, J.L.; Petersen, R.C.; Smith, R.D. Rapid expansion of supercritical fluid solutions: Solute formation of powders, thin films, and fibers. Ind. Eng. Chem. Res. 1987, 26, 2298–2306. [Google Scholar] [CrossRef]
- Beckman, E.J. Supercritical and near-critical CO2 in green chemical synthesis and processing. J. Supercrit. Fluids 2004, 28, 121–191. [Google Scholar] [CrossRef]
- Johnston, K.P.; Harrison, K.L.; Clarke, M.J.; Howdle, S.M.; Heitz, M.P.; Bright, F.V.; Carlier, C.; Randolph, T.W. Water-in-carbon dioxide microemulsions: An environment for hydrophiles including proteins. Science 1996, 271, 624–626. [Google Scholar]
- Kellici, S.; Rehman, I.; Darr, J.A. Controlled growth of titania nanospheres in supercritical carbon dioxide using a novel surfactant stabilised precursor. J. Mater. Chem. 2006, 16, 159–161. [Google Scholar] [CrossRef]
- Reverchon, E.; Adami, R. Nanomaterials and supercritical fluids. J. Supercrit. Fluids 2006, 37, 1–22. [Google Scholar] [CrossRef]
- Poe, D.P.; Veit, D.; Ranger, M.; Kaczmarski, K.; Tarafder, A.; Guiochon, G. Pressure, temperature and density drops along supercritical fluid chromatography columns in different thermal environments. III. Mixtures of carbon dioxide and methanol as the mobile phase. J. Chromatogr. A 2014, 1323, 143–156. [Google Scholar] [CrossRef]
- Yang, Q.Y.; de Vries, M.H.; Picchioni, F.; Loos, K. A novel method of preparing metallic Janus silica particles using supercritical carbon dioxide. Nanoscale 2013, 5, 10420–10427. [Google Scholar] [CrossRef]
- Yan, K.; Jarvis, C.; Lafleur, T.; Qiao, Y.X.; Xie, X.M. Novel synthesis of Pd nanoparticles for hydrogenation of biomass-derived platform chemicals showing enhanced catalytic performance. RSC Adv. 2013, 3, 25865–25871. [Google Scholar] [CrossRef]
- Xie, M.; Zhang, X.X.; Deng, S.X.; Wang, Y.Z.; Wang, H.; Liu, J.B.; Yan, H.; Laakso, J.; Levänen, E. The effects of supercritical carbon dioxide treatment on the morphology and electrochemical performance of LiFePO4 cathode materials. RSC Adv. 2013, 3, 12786–12793. [Google Scholar]
- Romang, A.H.; Watkins, J.J. Supercritical fluids for the fabrication of semiconductor devices: Emerging or missed opportunities? Chem. Rev. 2010, 110, 459–478. [Google Scholar] [CrossRef]
- Wang, W.; Wang, Y.; Gao, Y.; Zhao, Y. Control of number of graphene layers using ultrasound in supercritical CO2 and their application in lithium-ion batteries. J. Supercrit. Fluids 2014, 85, 95–101. [Google Scholar] [CrossRef]
- Li, C.Q.; Yang, G.H.; Deng, H.; Wang, K.; Zhang, Q.; Chen, F.; Fu, Q. The preparation and properties of polystyrene/functionalized graphene nanocomposite foams using supercritical carbon dioxide. Polym. Int. 2013, 62, 1077–1084. [Google Scholar]
- Zhao, J.; Zhang, L.Q.; Chen, T.; Yu, H.; Zhang, L.; Xue, H.; Hu, H.Q. Supercritical carbon-dioxide-assisted deposition of Pt nanoparticles on graphene sheets and their application as an electrocatalyst for direct methanol fuel cells. J. Phys. Chem. C 2012, 116, 21374–21381. [Google Scholar] [CrossRef]
- Zhao, J.; Zhang, L.; Xue, H.; Wang, Z.B.; Hu, H.Q. Methanol electrocatalytic oxidation on highly dispersed platinum-ruthenium/graphene catalysts prepared in supercritical carbon dioxide-methanol solution. RSC Adv. 2012, 2, 9651–9659. [Google Scholar] [CrossRef]
- Nguyen, V.H.; Kim, B.K.; Jo, Y.L.; Shim, J.J. Preparation and antibacterial activity of silver nanoparticles-decorated graphene composites. J. Supercrit. Fluids 2012, 72, 28–35. [Google Scholar] [CrossRef]
- Saha, B. The Centre for Green Process Engineering (CGPE) opens with launch event at London South Bank University (LSBU). Green Process. Synth. 2013, 2, 169–174. [Google Scholar]
- Adeleye, A.I.; Kellici, S.; Saha, B. Synthesis of propylene carbonate by graphene-inorganic nanoparticles. Unpublished work (in preparation). 2014. [Google Scholar]
- Weingärtner, H.; Franck, E.U. Supercritical water as a solvent. Angew. Chem. Int. Ed. 2005, 44, 2672–2692. [Google Scholar] [CrossRef]
- Shaw, R.W.; Brill, T.B.; Clifford, A.A.; Eckert, C.A.; Franck, E.U. Supercritical water—A medium for chemistry. Chem. Eng. News 1991, 69, 26–39. [Google Scholar]
- Byrappa, K.; Adschiri, T. Hydrothermal technology for nanotechnology. Prog. Cryst. Growth Charact. Mater. 2007, 53, 117–166. [Google Scholar] [CrossRef]
- Cabañas, A.; Poliakoff, M. The continuous hydrothermal synthesis of nano-particulate ferrites in near critical and supercritical water. J. Mater. Chem. 2001, 11, 1408–1416. [Google Scholar] [CrossRef]
- Blood, P.J.; Denyer, J.P.; Azzopardi, B.J.; Poliakoff, M.; Lester, E. A versatile flow visualisation technique for quantifying mixing in a binary system: Application to continuous supercritical water hydrothermal synthesis (SWHS). Chem. Eng. Sci. 2004, 59, 2853–2861. [Google Scholar] [CrossRef]
- Chaudhry, A.A.; Haque, S.; Kellici, S.; Boldrin, P.; Rehman, I.; Fazal, A.K.; Darr, J.A. Instant nano-hydroxyapatite: A continuous and rapid hydrothermal synthesis. Chem. Commun. 2006, 21, 2286–2288. [Google Scholar]
- Hakuta, Y.; Adschiri, T.; Suzuki, T.; Chida, T.; Seino, K.; Arai, K. Flow method for rapidly producing barium hexaferrite particles in supercritical water. J. Am. Ceram. Soc. 1998, 81, 2461–2464. [Google Scholar]
- Lester, E.; Blood, P.; Denyer, J.; Giddings, D.; Azzopardi, B.; Poliakoff, M. Reaction engineering: The supercritical water hydrothermal synthesis of nano-particles. J. Supercrit. Fluids 2006, 37, 209–214. [Google Scholar] [CrossRef]
- Boldrin, P.; Hebb, A.K.; Chaudhry, A.A.; Otley, L.; Thiebaut, B.; Bishop, P.; Darr, J.A. Direct synthesis of nanosized NiCo2O4 spinel and related compounds via continuous hydrothermal synthesis methods. Ind. Eng. Chem. Res. 2007, 46, 4830–4838. [Google Scholar] [CrossRef]
- Weng, X.; Boldrin, P.; Abrahams, I.; Skinner, S.J.; Darr, J.A. Direct syntheses of mixed ion and electronic conductors La4Ni3O10 and La3Ni2O7 from nanosized coprecipitates. Chem. Mater. 2007, 19, 4382–4384. [Google Scholar] [CrossRef]
- Middelkoop, V.; Boldrin, P.; Peel, M.; Buslaps, T.; Barnes, P.; Darr, J.A.; Jacques, S.D.M. Imaging the inside of a continuous nanoceramic synthesizer under supercritical water conditions using high-energy synchrotron X-Radiation. Chem. Mater. 2009, 21, 2430–2435. [Google Scholar] [CrossRef]
- Weng, X.L.; Boldrin, P.; Abrahams, I.; Skinner, S.J.; Kellici, S.; Darr, J.A. Direct syntheses of Lan+1NinO3n+1 phases (n = 1, 2, 3 and ∞) from nanosized co-crystallites. J. Solid State Chem. 2008, 181, 1123–1132. [Google Scholar] [CrossRef]
- Lin, T.; Kellici, S.; Gong, K.; Thompson, K.; Evans, J.R.G.; Wang, X.; Darr, J.A. Rapid automated materials synthesis instrument: Exploring the composition and heat-treatment of nanoprecursors toward low temperature red phosphors. J. Comb. Chem. 2010, 12, 383–392. [Google Scholar] [CrossRef]
- Zhang, Z.C.; Brown, S.; Goodall, J.B.M.; Weng, X.L.; Thompson, K.; Gong, K.N.; Kellici, S.; Clark, R.J.H.; Evans, J.R.G.; Darr, J.A. Direct continuous hydrothermal synthesis of high surface area nanosized titania. J. Alloys Compd. 2009, 476, 451–456. [Google Scholar]
- Ma, C.Y.; Wang, X.Z.; Tighe, C.J.; Gruar, R.I.; Darr, J.A. Simulation of hydrodynamics and heat transfer in confined jet reactors of different size scales for nanomaterial production. Comput.-Aided Chem. Eng. 2012, 31, 1236–1240. [Google Scholar] [CrossRef]
- Weng, X.L.; Cockcroft, J.K.; Hyett, G.; Vickers, M.; Boldrin, P.; Tang, C.C.; Thompson, S.P.; Parker, J.E.; Knowles, J.C.; Rehman, I.; et al. High-throughput continuous hydrothermal synthesis of an entire nanoceramic phase diagram. J. Comb. Chem. 2009, 11, 829–834. [Google Scholar] [CrossRef]
- Kellici, S.; Gong, K.A.; Lin, T.A.; Brown, S.; Clark, R.J.H.; Vickers, M.; Cockcroft, J.K.; Middelkoop, V.; Barnes, P.; Perkins, J.M.; et al. High-throughput continuous hydrothermal flow synthesis of Zn-Ce oxides: Unprecedented solubility of Zn in the nanoparticle fluorite lattice. Philos. T. R. Soc. A 2010, 368, 4331–4349. [Google Scholar] [CrossRef]
- Gruar, R.I.; Tighe, C.J.; Muir, J.; Kittler, J.T.; Wodjak, M.; Kenyon, A.J.; Darr, J.A. Continuous hydrothermal synthesis of surface-functionalised nanophosphors for biological imaging. RSC Adv. 2012, 2, 10037–10047. [Google Scholar] [CrossRef]
- Gruar, R.I.; Tighe, C.J.; Darr, J.A. Scaling-up a confined jet reactor for the continuous hydrothermal manufacture of nanomaterials. Ind. Eng. Chem. Res. 2013, 52, 5270–5281. [Google Scholar] [CrossRef]
- Hawthorne, S.B.; Yang, Y.; Miller, D.J. Extraction of organic pollutants from environmental solids with sub- and supercritical water. Anal. Chem. 1994, 66, 2912–2920. [Google Scholar]
- Gloyna, E.F.; Li, L.X. Supercritical water oxidation research and development update. Environ. Prog. 1995, 14, 182–192. [Google Scholar] [CrossRef]
- Cabeza, P.; Bermejo, M.D.; Jiménez, C.; Cocero, M.J. Experimental study of the supercritical water oxidation of recalcitrant compounds under hydrothermal flames using tubular reactors. Water Res. 2011, 45, 2485–2495. [Google Scholar] [CrossRef]
- Jimenez-Espadafor, F.; Portela, J.R.; Vadillo, V.; Sánchez-Oneto, J.; Villanueva, J.A.B.; García, M.T.; de la Ossa, E.J.M. Supercritical water oxidation of oily wastes at pilot plant: Simulation for energy recovery. Ind. Eng. Chem. Res. 2011, 50, 775–784. [Google Scholar] [CrossRef]
- Zhao, Q.C.; Dong, X.; Zhou, M.H.; Wang, J.C. Dioxin destruction by supercritical water oxidation. Adv. Mater. Res. 2013, 864–867, 1709–1712. [Google Scholar]
- Kritzer, P.; Dinjus, E. An assessment of supercritical water oxidation (SCWO): Existing problems, possible solutions and new reactor concepts. Chem. Eng. J. 2001, 83, 207–214. [Google Scholar] [CrossRef]
- Vadillo, V.; Sánchez-Oneto, J.; Portela, J.R.; de la Ossa, E.J.M. Problems in supercritical water oxidation process and proposed solutions. Ind. Eng. Chem. Res. 2013, 52, 7617–7629. [Google Scholar]
- Baker, R.T.; Tumas, W. Homogeneous catalysis—Toward greener chemistry. Science 1999, 284, 1477–1479. [Google Scholar] [CrossRef]
- Jutz, F.; Andanson, J.M.; Baiker, A. Ionic liquids and dense carbon dioxide: A beneficial biphasic system for catalysis. Chem. Rev. 2011, 111, 322–353. [Google Scholar] [CrossRef]
- Kohlpaintner, C.W.; Fischer, R.W.; Cornils, B. Aqueous biphasic catalysis: Ruhrchemie/Rhone-Poulenc oxo process. Appl. Catal., A 2001, 221, 219–225. [Google Scholar] [CrossRef]
- Yu, Y.Y.; Lu, B.; Wang, X.G.; Zhao, J.X.; Wang, X.Z.; Cai, Q.H. Highly selective oxidation of benzyl alcohol to benzaldehyde with hydrogen peroxide by biphasic catalysis. Chem. Eng. J. 2010, 162, 738–742. [Google Scholar] [CrossRef]
- Mao, H.; Yu, H.; Chen, J.; Liao, X.P. Biphasic catalysis using amphiphilic polyphenols-chelated noble metals as highly active and selective catalysts. Sci. Rep. 2013, 3, A2226:1–A2226:5. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Patel, D.; Kellici, S.; Saha, B. Green Process Engineering as the Key to Future Processes. Processes 2014, 2, 311-332. https://doi.org/10.3390/pr2010311
Patel D, Kellici S, Saha B. Green Process Engineering as the Key to Future Processes. Processes. 2014; 2(1):311-332. https://doi.org/10.3390/pr2010311
Chicago/Turabian StylePatel, Dipesh, Suela Kellici, and Basudeb Saha. 2014. "Green Process Engineering as the Key to Future Processes" Processes 2, no. 1: 311-332. https://doi.org/10.3390/pr2010311




