Influence of the Azulene Ring on the Enantioseparation of 1,5-Diols
Abstract
:1. Introduction
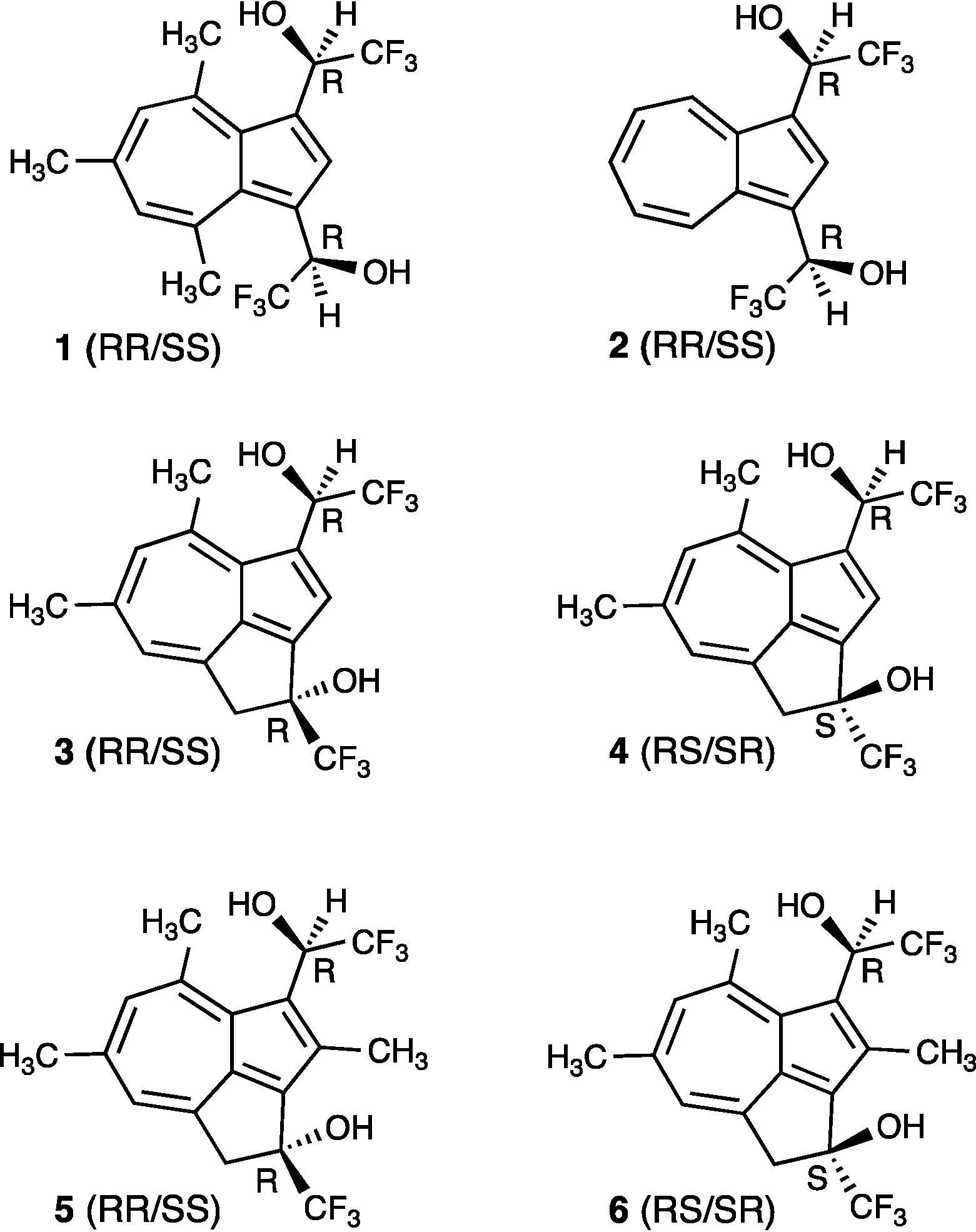
2. Experimental Section
2.1. Instrumentation
2.2. Example Procedure for Separation and Analysis of Enantiomers, 1RS/SR
3. Results and Discussion
| Compound | Rf Value * | Rt 1 (min) | Rt 2 (min) | w1/2 1 (min) | w1/2 2 (min) | Dead Volume | Rs | α |
|---|---|---|---|---|---|---|---|---|
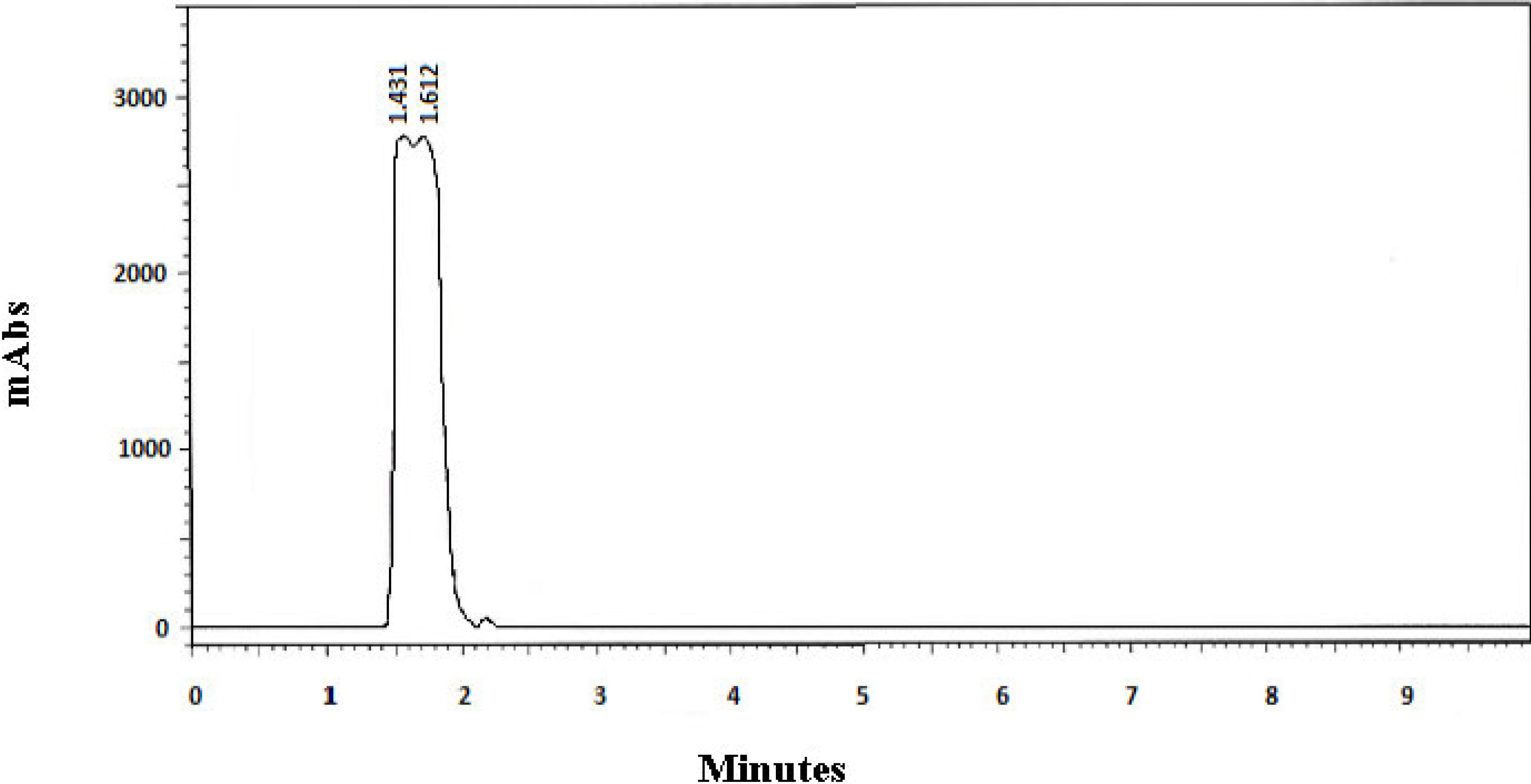
| 1 | 0.59 | 1.43 f | 1.61 f | 0.30 | 0.30 | 0.41 mL | 0.35 | 1.17 |
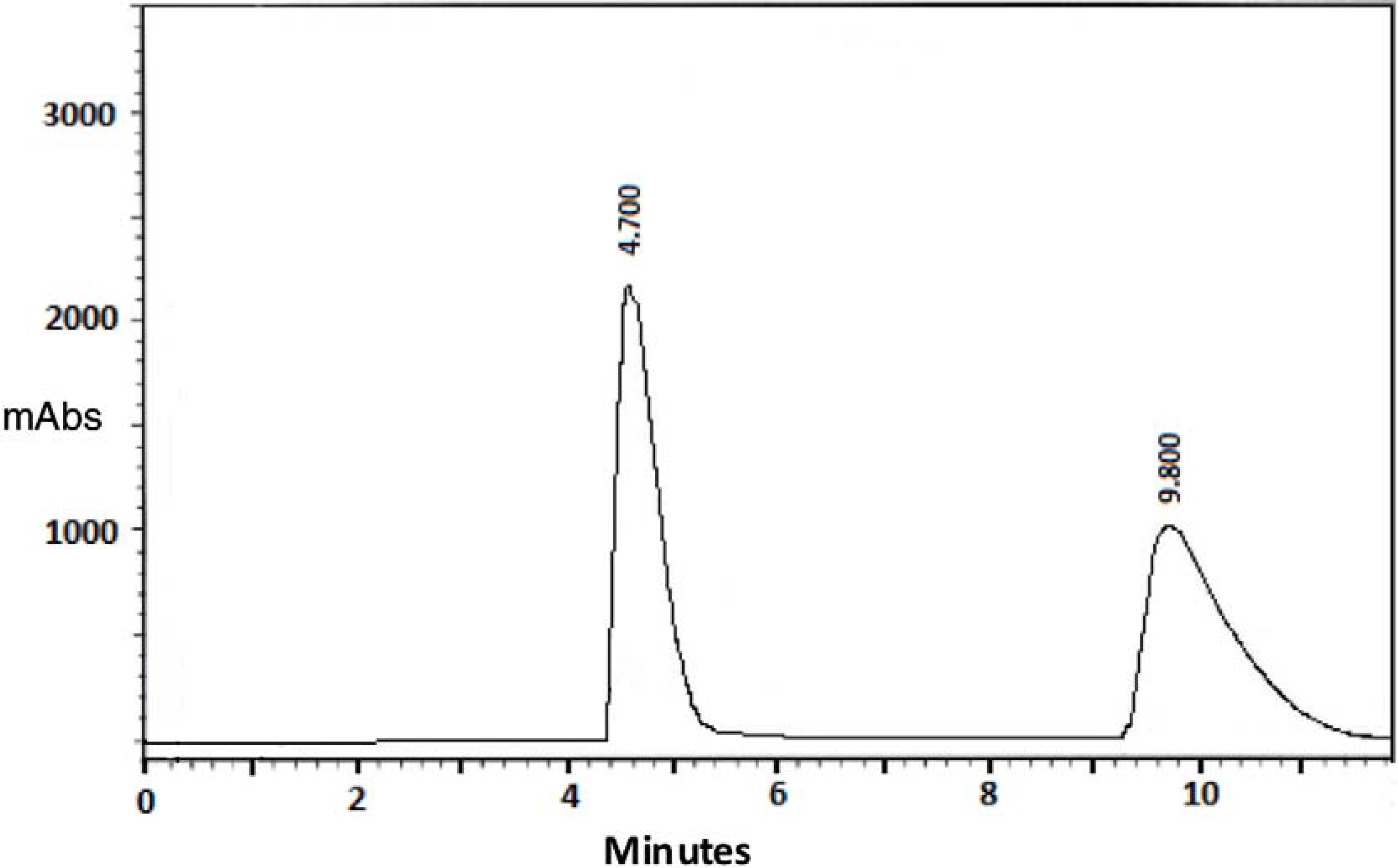
| 2 | 0.39 | 4.70 e | 9.80 e | 0.41 | 0.90 | 0.38 | 4.61 | 2.18 |
| 3 | 0.63 | 1.75 f | 2.97 f | 0.16 | 0.31 | 0.41 | 3.10 | 1.91 |
| 4 | 0.40 | 1.30 g | 8.13 g | 0.21 | 0.76 | 0.38 | 8.28 | 8.43 |
| 5 | 0.58 | 1.88 b | 4.73 b | 0.27 | 0.63 | 0.40 | 3.74 | 2.93 |
| 6 | 0.38 | 1.87 c | 11.15 c | 0.39 | 0.97 | 0.38 | 8.05 | 7.25 |
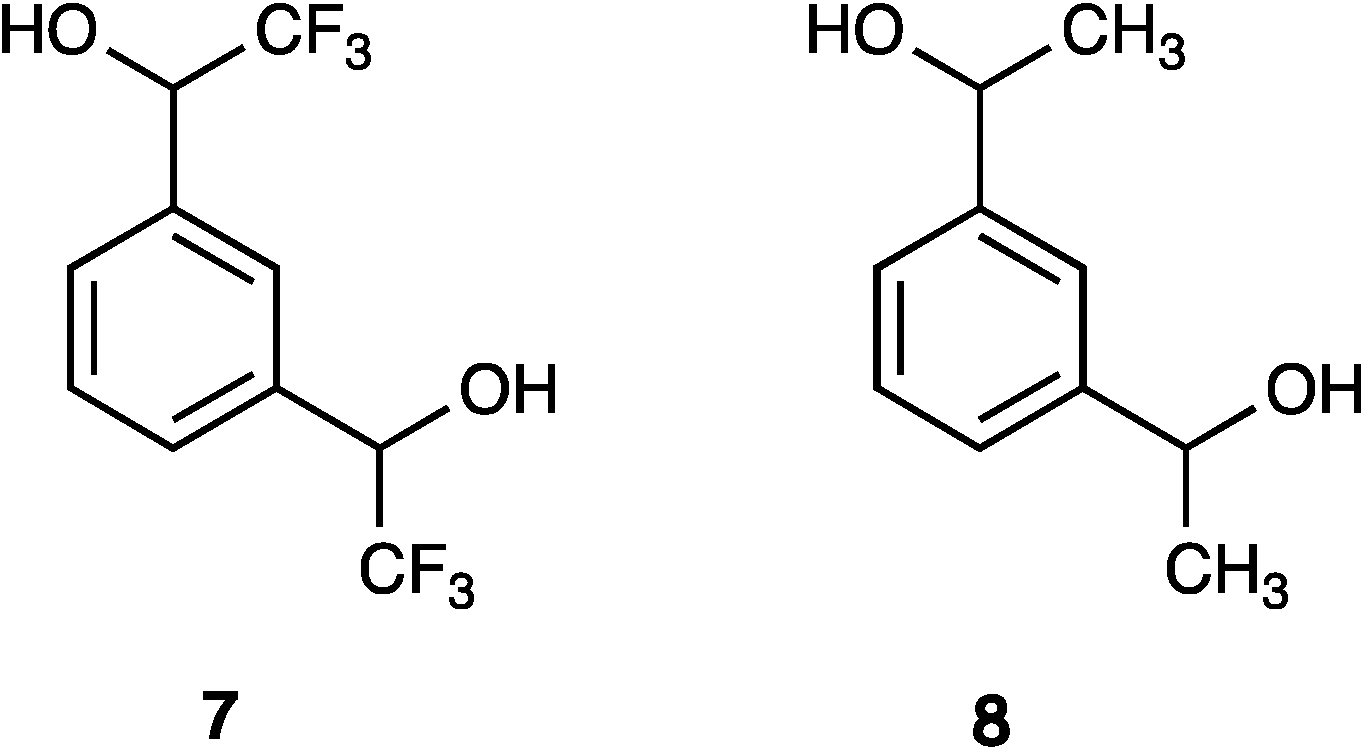
| 7 | 0.31 | 6.92 b | 7.68 b | 0.70 | 1.14 | 0.46 | 0.49 | 1.12 |
| 8 | 0.10 | 2.78 a | 3.82 a | 0.16 | 0.30 | 0.80 | 2.65 | 1.53 |
| Compound | Rf value * | Rt 1 (min) | Rt 2 (min) | w1/2 1 (min) | w1/2 2 (min) | Dead Volume | Rs | α | ||
|---|---|---|---|---|---|---|---|---|---|---|
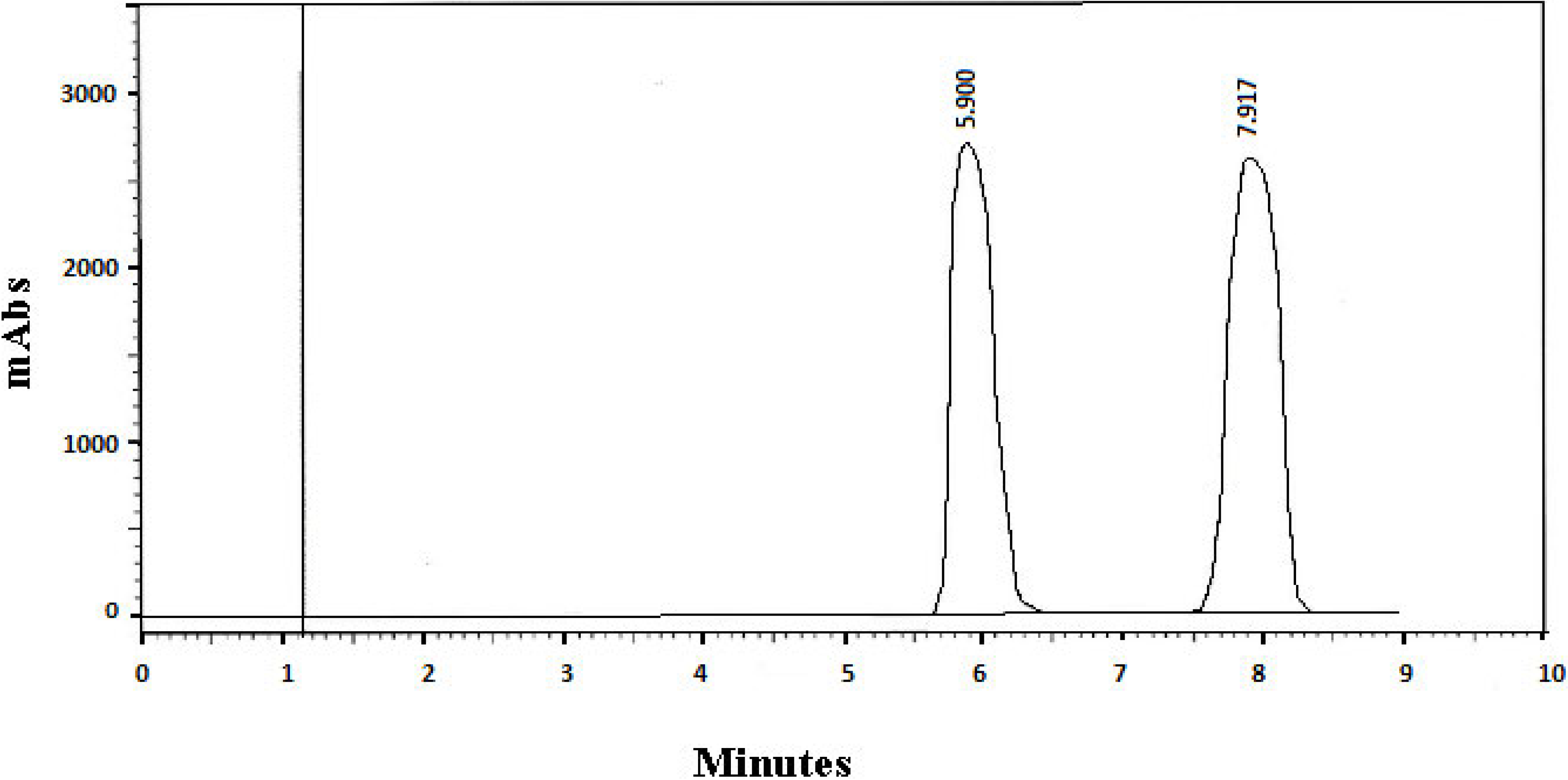
| 1 | 0.59 | 5.90 d | 7.92 d | 0.16 | 0.19 | 1.14 mL | 8.71 | 1.42 | ||
| 2 | 0.39 | 3.23 c | 3.53 c | 0.46 | 0.46 | 1.13 | 0.38 | 1.14 | ||
| 3 | 0.63 | 5.32 b | 6.10 b | 0.21 | 0.24 | 1.25 | 2.06 | 1.19 | ||
| 4 | 0.40 | 2.28 c | 2.85 c | 0.55 | 0.55 | 1.15 | 0.61 | 1.50 | ||
| 5 | 0.58 | 3.82 b | 4.93 b | 0.19 | 0.25 | 1.45 | 2.98 | 1.47 | ||
| 6 | 0.38 | 6.65 b | 7.77 b | 0.36 | 0.38 | 1.40 | 1.78 | 1.21 | ||
| 7 | 0.31 | 3.75 b | 3.98 b | 0.12 | 0.24 | 1.27 | 0.76 | 1.09 | ||
| 8 | 0.10 | 9.70 b | 10.4 b | 0.51 | 0.70 | 1.20 | 0.63 | 1.08 | ||
| Compound | Column Name | Stationary Phase | Solvent System | Rs | α |
|---|---|---|---|---|---|
| 1 | Phenomenex Lux 5u Cellulose 4 | Cellulose tris-(4-chloro-3-methylphenylcarbamate) | 60:40:0.1 AmmBi:ACN:DEA c | 4.40 | 1.23 |
| 1 | Phenomenex Lux 5u Cellulose 3 | Cellulose tris-(4-methylbenzoate | 60:40:0.1 AmmBi:ACN:DEA c | 2.90 | 1.21 |
| 1 | Phenomenex Lux 5u Cellulose 2 | Cellulose tris-(3-chloro-4-methylphenylcarbamate) | 60:40:0.1 AmmBi:ACN:DEA c | 1.03 | 1.14 |
| 1 | Phenomenex Lux 5u Amylose 2 | Amylose tris-(5-chloro-2-methylphenylcarbamate) | 60:40:0.1 AmmBi:ACN:DEA c | 0.82 | 1.07 |
| 1 | Phenomenex Lux 5u Cellulose 2 | Cellulose tris-(3-chloro-4-methylphenylcarbamate) | 90:10:0.1 Hex:IPA:DEA c | 4.45 | 1.52 |
| 1 | Phenomenex Lux 5u Amylose 2 | Amylose tris-(5-chloro-2-methylphenylcarbamate) | 90:10:0.1 Hex:IPA:DEA c | 3.20 | 1.80 |
| 2 | (S,S)-Whelk-O 1 (Regis Tech.) | 4-(3,5-dinitrobenzamido) tetrahydrophenathrene on silica | 90:10 Hex:IPA | 4.22 | 1.23 |
| 2 | RegisPack a | Tris-(3,5-dimethylphenyl) carbamoyl amylose | 90:10 Hex:MeOH | 4.64 | 1.22 |
| 2 | RegisCell b | Tris-(3,5-dimethylphenyl) carbamoyl cellulose | 85:15 Hex:EtOH c | 9.51 | 1.78 |
| 4 | CHIRALPAK IB-3 (Chiral Tech.) | Cellulose tris-(3,5-dimethylphenylcarbamate) | 80:20 Hex:EtOH c | 10.64 | 1.31 |
| Compound | λ (nm) | Δε | |
|---|---|---|---|
| 1 | Faster—1.43 min | 349.8 | 11.64 |
| Slower—1.61 min | 349.8 | −6.47 | |
| 2 | Faster—4.70 min | 356.3 | 11.23 |
| Slower—9.80 min | 356.3 | −10.66 | |
| 3 | Faster—1.75 min | 349.7 | −7.45 |
| Slower—2.97 min | 349.7 | 8.60 | |
| 4 | Faster—1.30 min | 345.0 | −2.83 |
| Slower—8.13 min | 345.0 | 4.17 | |
| 5 | Faster—1.88 min | 314 | −3.56 |
| Slower—4.73 min | 314 | 7.12 | |
| 6 | Faster—1.87 min | 339.7 | −9.67 |
| Slower—11.15 min | 339.7 | 10.70 | |

4. Conclusions
Acknowledgments
Authors Contributions
Conflicts of Interest
References
- Chiral Separations: Methods and Protocols (Methods in Molecular Biology Volume 243); Gübitz, G.; Schmid, M.G. (Eds.) Humana Press: Totowa, NJ, USA, 2004; pp. 1–424.
- Principles of Asymmetric Synthesis, 2nd ed.; Gawley, R.A.; Aubé, J. (Eds.) Elsevier: Oxford, UK, 2012; pp. 76–79.
- Dou, L.; Zeng, J.; Gerochi, D.; Duda, M.; Stuting, H. Chiral high-performance liquid chromatography methodology for quality control monitoring of dexfenfluramine. J. Chrom. A 1994, 679, 367–374. [Google Scholar] [CrossRef]
- Ribeiro, A.R.; Castro, P.M.L.; Tiritan, M.E. Chiral pharmaceuticals in the environment. Environ. Chem. Lett. 2012, 10, 239–253. [Google Scholar] [CrossRef]
- Anderson, A.G.; Strecker, B.M. Azulene VIII. A study of the absorption spectra and dipole moments of some 1- and 1,3-substituted azulenes. J. Am. Chem. Soc. 1959, 81, 4941–4946. [Google Scholar] [CrossRef]
- Liu, R.S.H. Colorful azulene and its equally colorful derivatives. J. Chem. Educ. 2002, 79, 183–185. [Google Scholar] [CrossRef]
- Zhang, L-Y.; Yang, F.; Shi, W-Q.; Zhang, P.; Li, Y.; Yin, S-F. Synthesis and antigastric ulcer activity of novel 5-isopropyl-3,8-dimethylazulene derivatives. Bioorgan. Med. Chem. Lett. 2011, 21, 5722–5725. [Google Scholar] [CrossRef]
- Okano, K. Synthesis and application of chiral hydrobenzoin. Tetrahedron 2011, 67, 2483–2572. [Google Scholar] [CrossRef]
- Naoshima, Y.; Kimura, T.; Mori, Y.; Kamezawa, M.; Tachibana, H.; Kohara, K.; Ohtani, T. The first synthesis of chiral azulene alcohol possessing a trifluoromethyl group by lipase-mediated biotransformation. Recent Res. Devel. Org. Bioorgan. Chem. 2004, 6, 1–9. [Google Scholar]
- Redl, F.X.; Köthe, O.; Röckl, K.; Bauerr, W.; Daub, J. Azulene appended cellulose: Synthesis, optical and chiroptical properties, film formation by electrochemical oxidation. Macromol. Chem. Phys. 2000, 201, 2091–2000. [Google Scholar] [CrossRef]
- Oki, M.; Nakamura, N. Restricted Rotation Involving the Tetrahedral Carbon. II. 2-Substituted 4,6,8-Trimethylazulenes. Bull. Chem. Soc. Jpn. 1971, 44, 1880–1885. [Google Scholar]
- Anderson, A.G., Jr.; Anderson, R.G.; Hollander, G.T. Reactions of some 1-trihaloacetyl-8-acetylazulenes with base. J. Org. Chem. 1965, 30, 131–138. [Google Scholar]
- Chen, A.H.; Yen, H.H.; Kuo, Y.C.; Chen, W.Z. Asymmetric synthesis and characterization of chiral 2,2′diamino-3,3′-diethoxycarbonyl-8–8′-diphenyl-1,1′-biazulene. Synth. Commun. 2007, 37, 2975–2987. [Google Scholar] [CrossRef]
- Horgen, D.A.; Klausmeyer, K.K.; Carson, C.; Shen, H.; Garner, C.M. Remarkably large remote-stereocenter induced chromatographic differences: Azulene 1,5-diols. Tetrahedron Lett. 2014, 55, 128–132. [Google Scholar] [CrossRef]
- Prakash, G.K.S.; Krishnamurti, R.; Olah, G.A. Synthetic methods and reactions. 141. Fluoride-induced trifluoromethylation of carbonyl compounds with trifluoromethyltrimethylsilane (TMS-CF3). A trifluoromethide equivalent. J. Am. Chem. Soc. 1989, 111, 393–395. [Google Scholar] [CrossRef]
- Johnston, H.W.; Williams, J.L.R. Preparation of m-divinylbenzene. J. Am. Chem. Soc. 1947, 69, 2065–2065. [Google Scholar] [CrossRef]
- Yang, S.; Li, C.; Wang, S.; Zhao, L.; Hou, Z.; Lou, H.; Ren, D. Chiral separation of two diastereomeric pairs of enantiomers of novel alkaloid-ligand hybrids from Lobelia chinensis and determination of the tentative absolute configuration. J. Chrom. A 2013, 1311, 134–139. [Google Scholar] [CrossRef]
- Tabata, H.; Yoneda, T.; Oshitari, T.; Takahashi, H.; Natsugari, H. Stereochemistry of 1,5-Benzothiazepin-4-one S-Oxide: Insight into the Stereogenic Elements at the Sulfur Atom and Axis. J. Org. Chem. 2013, 78, 6264–6270. [Google Scholar]
- Christov, P.P.; Hawkins, E.K.; Kett, N.R.; Rizzo, C.J. Simplified synthesis of individual stereoisomers of the 4-hydroxynonenal adducts of deoxyguanosine. Tetrahedron Lett. 2013, 54, 4289–4291. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Horgen, D.A.; Garner, C.M. Influence of the Azulene Ring on the Enantioseparation of 1,5-Diols. Chromatography 2014, 1, 65-74. https://doi.org/10.3390/chromatography1020065
Horgen DA, Garner CM. Influence of the Azulene Ring on the Enantioseparation of 1,5-Diols. Chromatography. 2014; 1(2):65-74. https://doi.org/10.3390/chromatography1020065
Chicago/Turabian StyleHorgen, Dana A., and Charles M. Garner. 2014. "Influence of the Azulene Ring on the Enantioseparation of 1,5-Diols" Chromatography 1, no. 2: 65-74. https://doi.org/10.3390/chromatography1020065





