Fluorescence Chemosensory Determination of Cu2+ Using a New Rhodamine–Morpholine Conjugate
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Solutions
2.2. Instrumentation and Apparatus
2.3. Synthesis of the Chemosensor RECM
2.3.1. Synthesis of Rhodamine-B-lactam-ethylenediamine (RE)
2.3.2. Synthesis of RE–Carbonyl Morpholine (RECM)
2.4. Measurement Procedures
2.4.1. Selectivity Measurements
2.4.2. Spectral Measurements
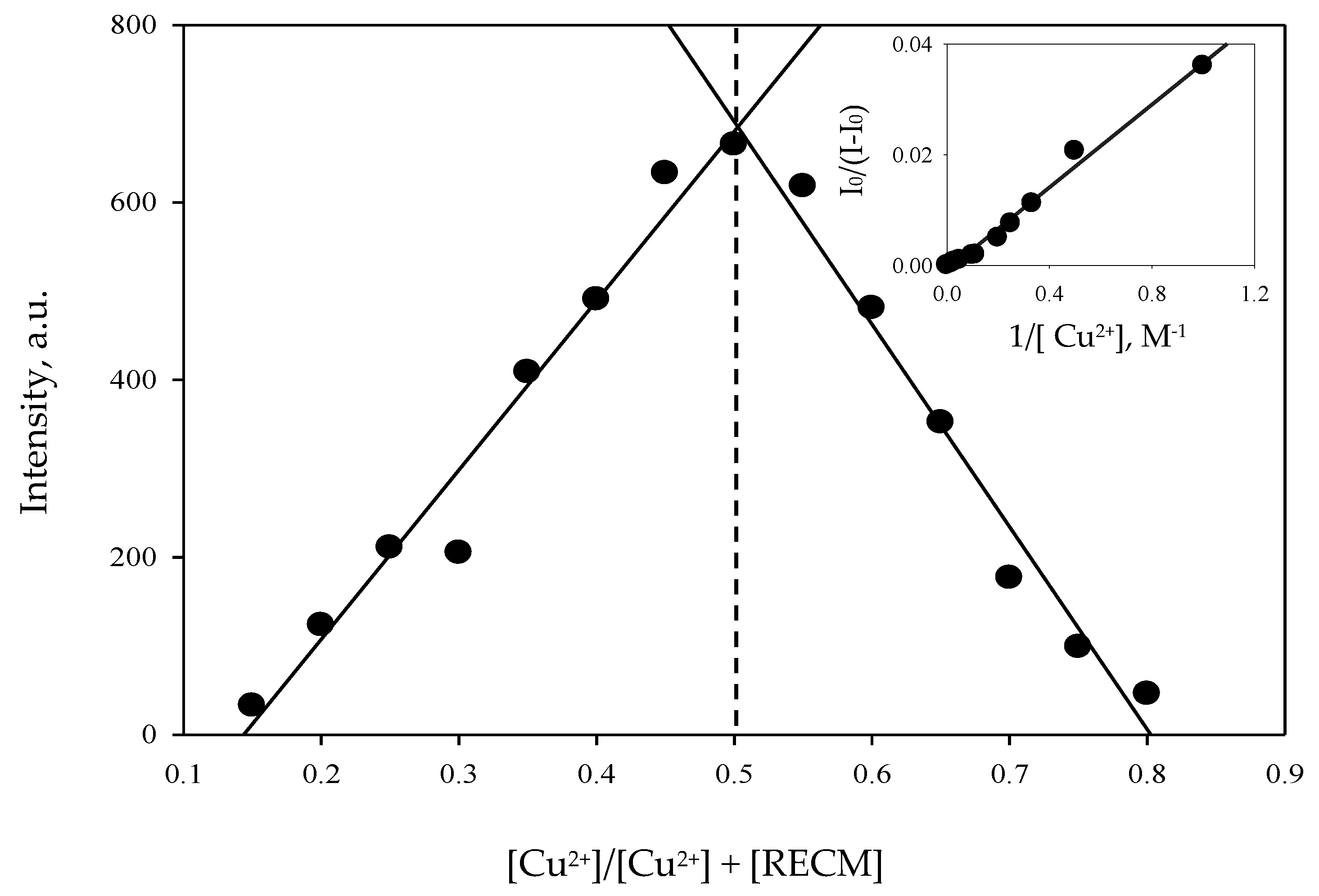
2.4.3. Job Plot Measurements
3. Results and Discussion
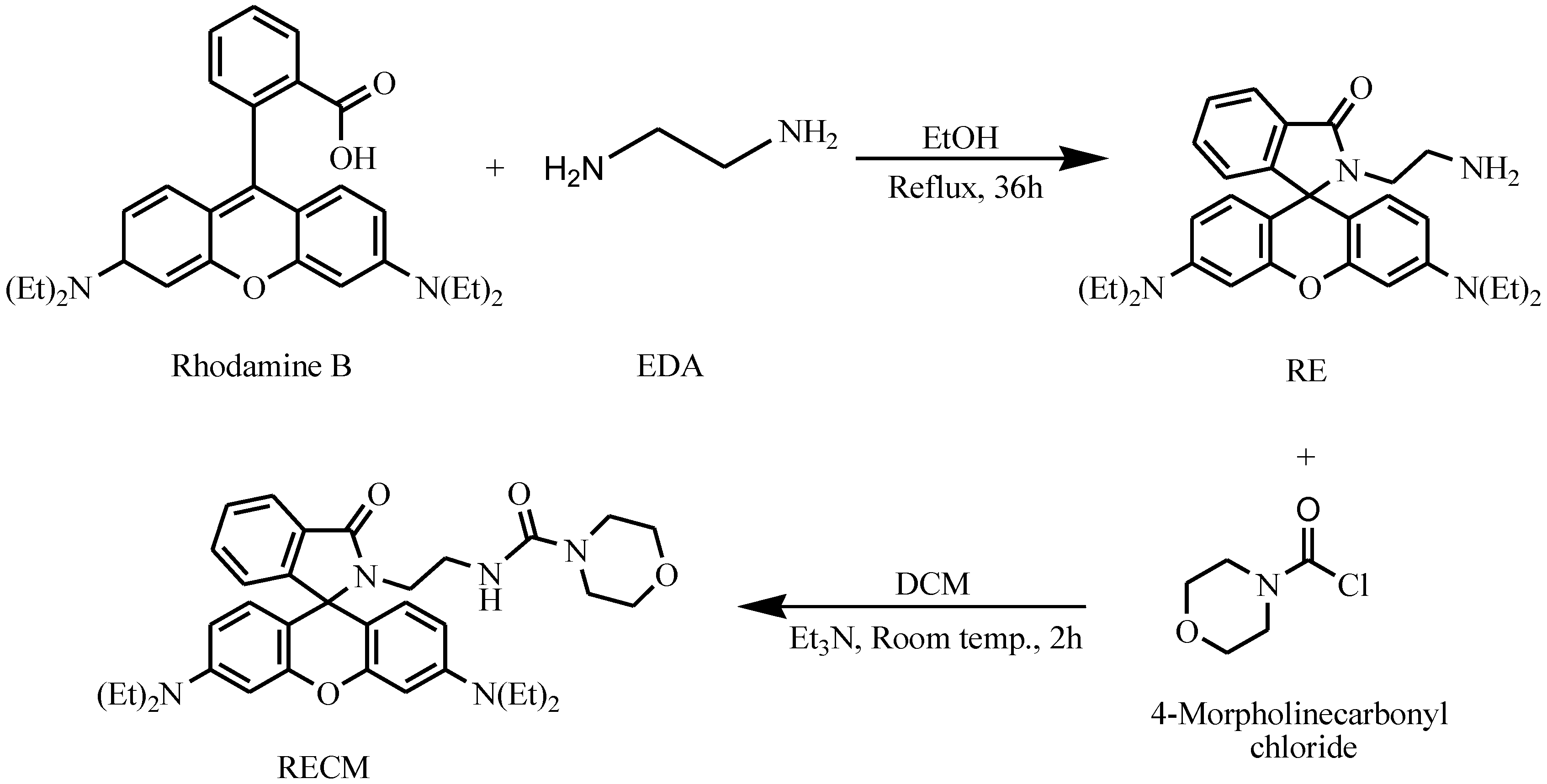
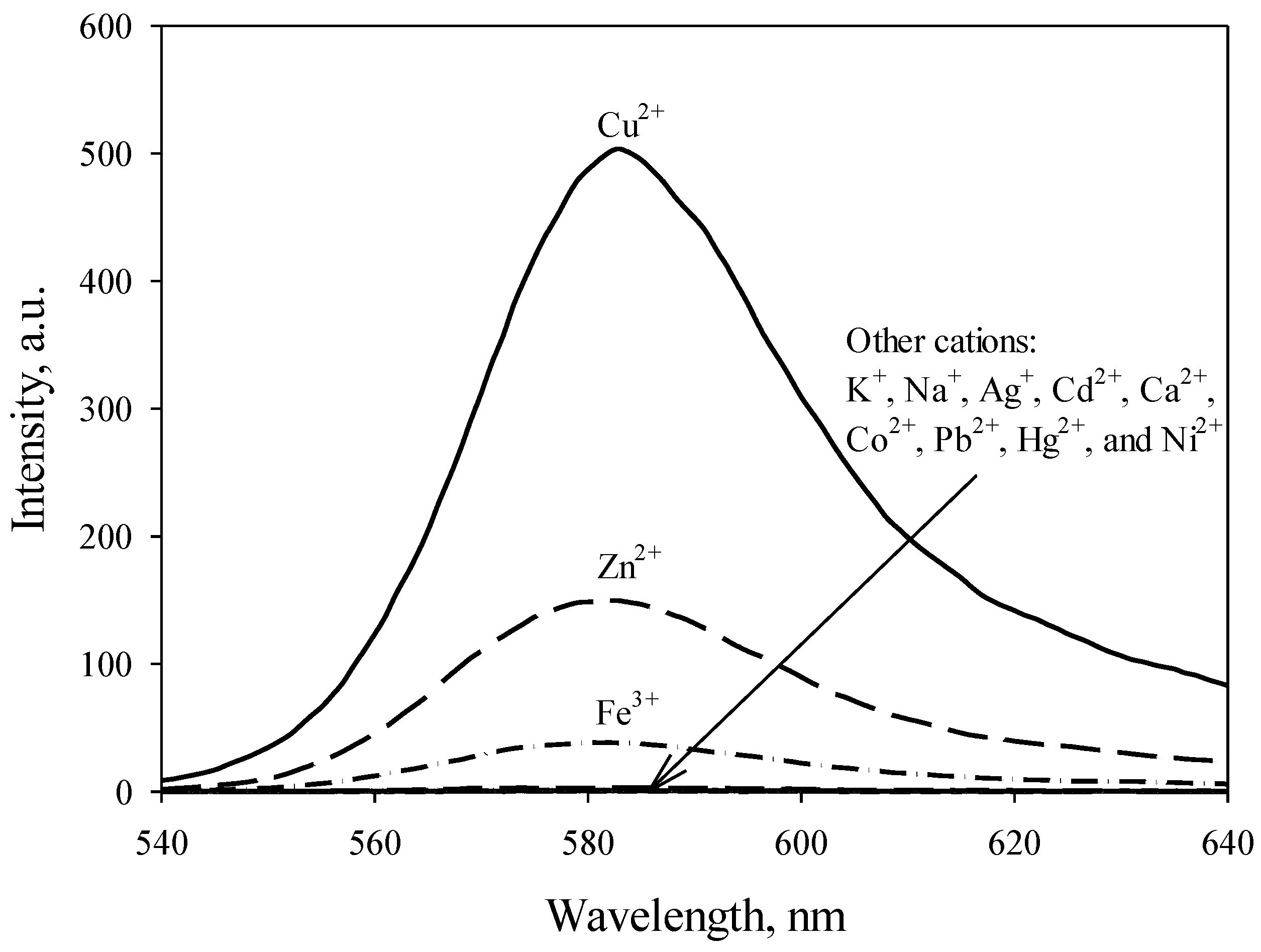
3.1. Selectivity
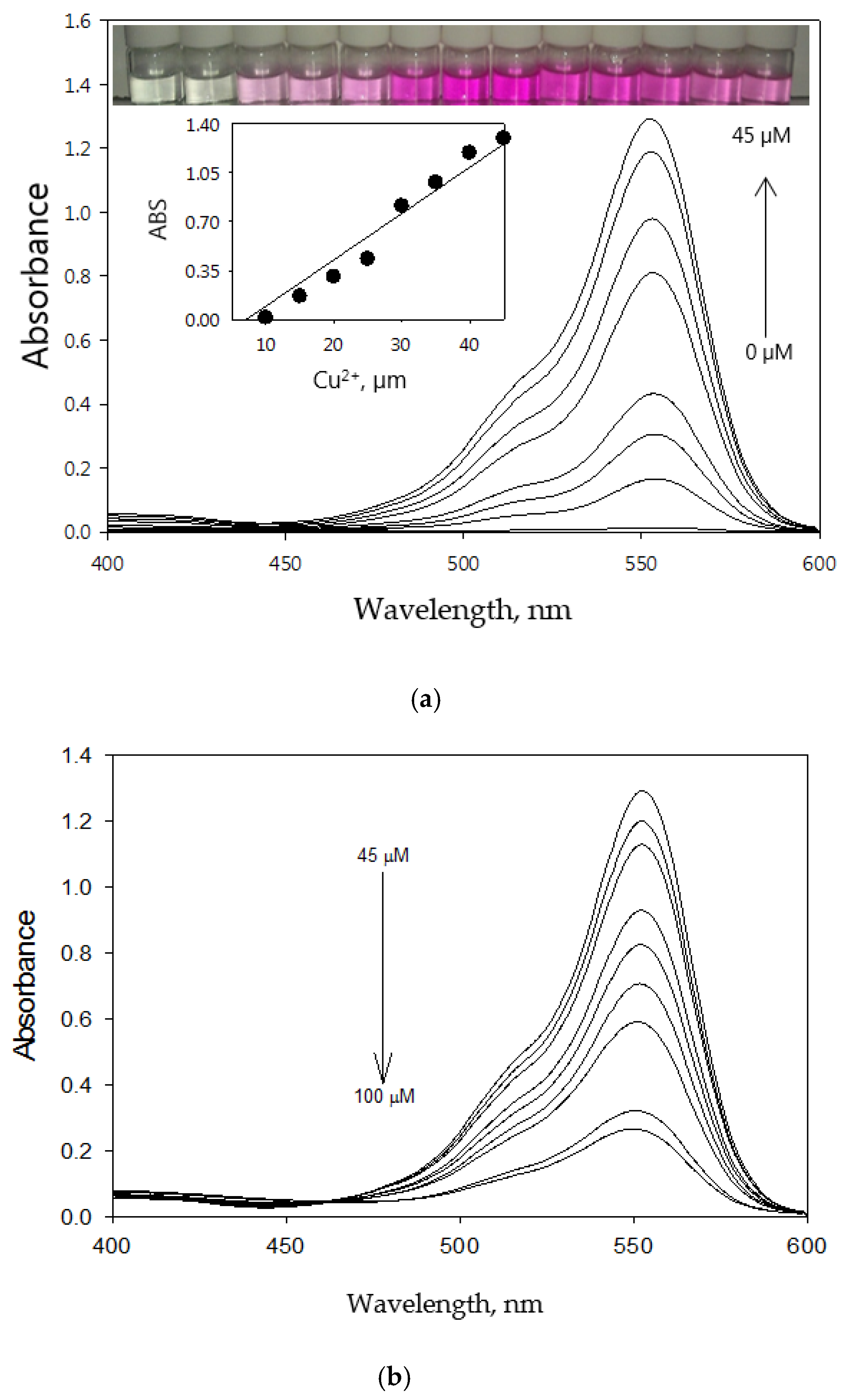
3.2. Optical Sensing Properties
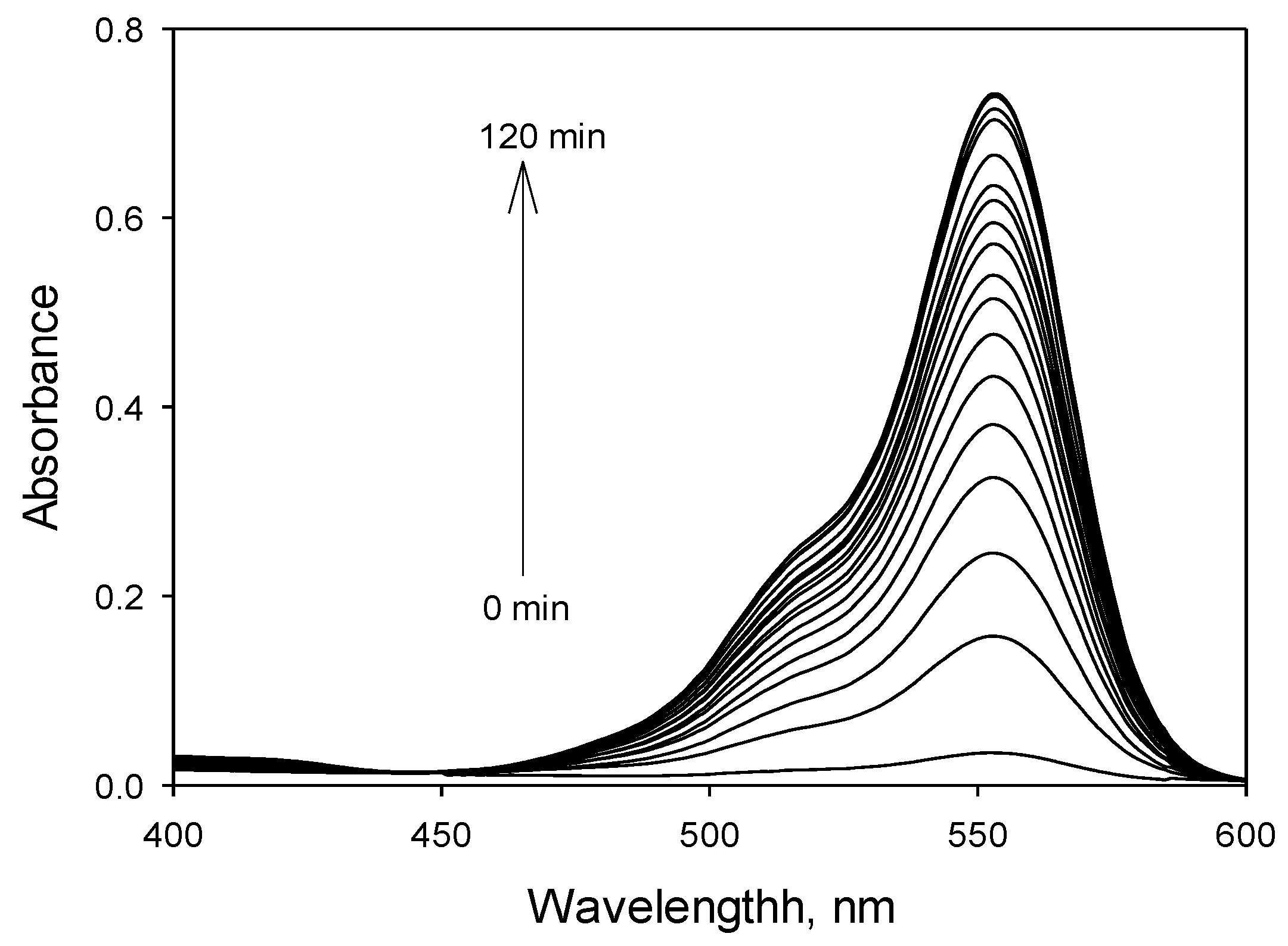
3.3. Response Time
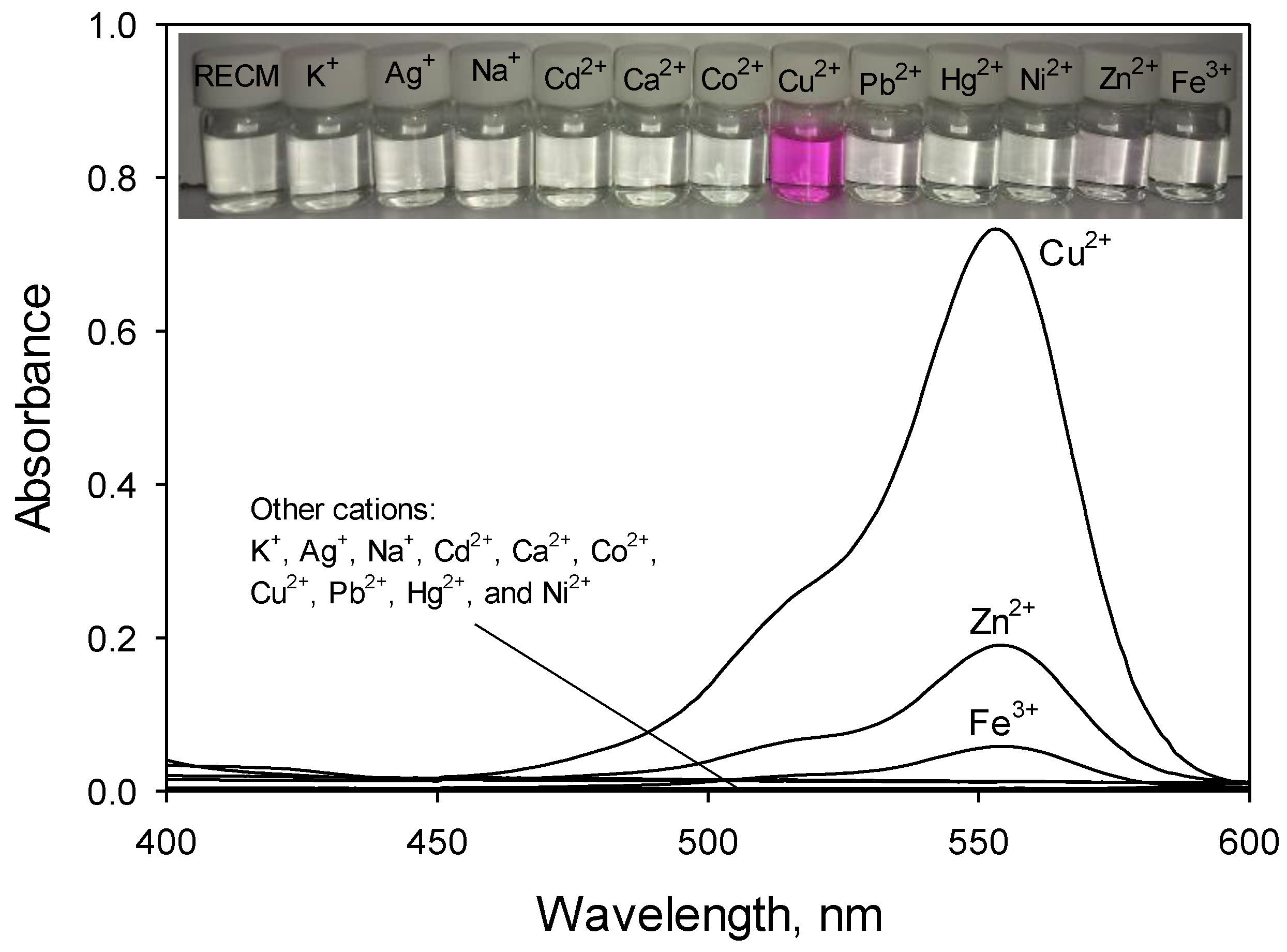
3.4. Binding Capability
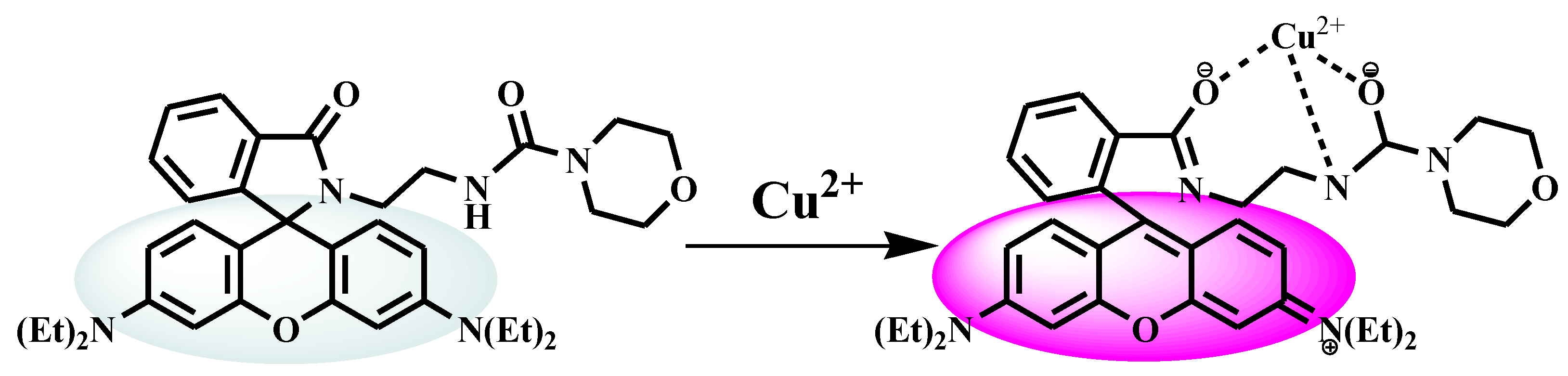
3.5. Structure and Complexation Mechanism
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Duarte, K.; Justino, C.I.; Freitas, A.C.; Gomes, A.M.; Duarte, A.C.; Rocha-Santos, T.A. Disposable sensors for environmental monitoring of lead, cadmium and mercury. TrAC Trends Anal. Chem. 2015, 64, 183–190. [Google Scholar]
- Gaggelli, E.; Kozlowski, H.; Valensin, D.; Valensin, G. Copper homeostasis and neurodegenerative disorders (Alzheimer’s, Prion, and Parkinson’s diseases and amyotrophic lateral sclerosis). Chem. Rev. 2006, 106, 1995–2044. [Google Scholar] [PubMed]
- Mathie, A.; Sutton, G.L.; Clarke, C.E.; Veale, E.L. Zinc and copper: Pharmacological probes and endogenous modulators of neuronal excitability. Pharmacol. Ther. 2006, 111, 567–583. [Google Scholar] [PubMed]
- Crichton, R.R.; Dexter, D.T.; Ward, R.J. Metal based neurodegenerative diseases—From molecular mechanisms to therapeutic strategies. Coord. Chem. Rev. 2008, 252, 1189–1199. [Google Scholar]
- Puangploy, P.; Smanmoo, S.; Surareungchai, W. A new rhodamine derivative-based chemosensor for highly selective and sensitive determination of Cu2+. Sens. Actuators B Chem. 2014, 193, 679–686. [Google Scholar]
- Bachan Upadhyay, L.S.; Verma, N. Enzyme inhibition based biosensors: A review. Anal. Lett. 2012, 46, 225–241. [Google Scholar]
- Ke, J.; Li, X.; Shi, Y.; Zhao, Q.; Jiang, X. A facile and highly sensitive probe for Hg(II) based on metal-induced aggregation of ZnSe/ZnS quantum dots. Nanoscale 2012, 4, 4996–5001. [Google Scholar] [PubMed]
- Yan, G.; Viraraghavan, T. Heavy metal removal in a biosorption column by immobilized M. rouxii biomass. Bioresour. Technol. 2001, 78, 243–249. [Google Scholar] [PubMed]
- El-Safty, S.A.; Shenashen, M.A. Mercury-ion optical sensors. TrAC Trends Anal. Chem. 2012, 38, 98–115. [Google Scholar]
- Zhang, X.; Shiraishi, Y.; Hirai, T. Cu(II)-selective green fluorescence of a rhodamine−diacetic acid conjugate. Org. Lett. 2007, 9, 5039–5042. [Google Scholar] [PubMed]
- Xie, P.; Guo, F.; Li, D.; Liu, X.; Liu, L. A Cu2+ chemodosimeter based on amplified fluorescence in the red region. J. Lumin. 2011, 131, 104–108. [Google Scholar]
- Wang, X.; Tao, J.; Chen, X.; Yang, H. An ultrasensitive and selective “off-on” rhodamine-based colorimetric and fluorescent chemodosimeter for the detection of Cu2+. Sens. Actuators B Chem. 2017, 244, 709–716. [Google Scholar]
- Xie, P.; Xiao, Y.; Yao, D.; Jin, Q.; Guo, F. A novel colorimetric and fluorescent “off-on” chemosensor for Cu2+ based on a rhodamine derivative bearing naphthyridine group. J. Fluoresc. 2013, 23, 265–271. [Google Scholar] [PubMed]
- Yang, Y.; Gao, C.Y.; Zhang, N.; Dong, D. Tetraphenylethene functionalized rhodamine chemosensor for Fe3+ and Cu2+ ions in aqueous media. Sens. Actuators B Chem. 2016, 222, 741–746. [Google Scholar]
- Fang, Y.; Zhou, Y.; Rui, Q.; Yao, C. Rhodamine–ferrocene conjugate chemosensor for selectively sensing copper(II) with multisignals: Chromaticity, fluorescence, and electrochemistry and its application in living cell imaging. Organometallics 2015, 34, 2962–2970. [Google Scholar]
- Zhang, J.F.; Zhou, Y.; Yoon, J.; Kim, Y.; Kim, S.J.; Kim, J.S. Naphthalimide modified rhodamine derivative: Ratiometric and selective fluorescent sensor for Cu2+ based on two different approaches. Org. Lett. 2010, 12, 3852–3855. [Google Scholar] [PubMed]
- Ganguly, A.; Paul, B.K.; Ghosh, S.; Kar, S.; Guchhait, N. Selective fluorescence sensing of Cu(II) and Zn(II) using a new Schiff base-derived model compound: Naked eye detection and spectral deciphering of the mechanism of sensory action. Analyst 2013, 138, 6532–6541. [Google Scholar] [PubMed]
- Soh, J.H.; Swamy, K.M.K.; Kim, S.K.; Kim, S.; Lee, S.H.; Yoon, J. Rhodamine urea derivatives as fluorescent chemosensors for Hg2+. Tetrahedron Lett. 2007, 48, 5966–5969. [Google Scholar]
- Kaewtong, C.; Noiseephum, J.; Uppa, Y.; Morakot, N.; Morakot, N.; Wanno, B.; Tuntulani, T.; Pulpoka, B. A reversible Em-FRET rhodamine-based chemosensor for carboxylate anions using a ditopic receptor strategy. New J. Chem. 2010, 34, 1104–1108. [Google Scholar]
- Liu, B.; Zeng, F.; Wu, G.; Wu, S. A FRET-based ratiometric sensor for mercury ions in water with multi-layered silica nanoparticles as the scaffold. Chem. Commun. 2011, 47, 8913–8915. [Google Scholar]
- Ma, C.; Zeng, F.; Huang, L.; Wu, S. FRET-based ratiometric detection system for mercury ions in water with polymeric particles as scaffolds. J. Phys. Chem. B 2011, 115, 874–882. [Google Scholar] [PubMed]
- Montalbetti, C.A.; Falque, V. Amide bond formation and peptide coupling. Tetrahedron 2005, 61, 10827–10852. [Google Scholar]
- Yano, K.; Nishi, T. A novel route to highly monodispersed mesoporous silica spheres consisting of nano-sized particles. Microporous Mesoporous Mater. 2012, 158, 257–263. [Google Scholar]
- Xiang, Y.; Tong, A.; Jin, P.; Ju, Y. New fluorescent rhodamine hydrazone chemosensor for Cu(II) with high selectivity and sensitivity. Org. Lett. 2006, 8, 2863–2866. [Google Scholar] [PubMed]
- Yang, Y.; Gao, C.; Li, B.; Xu, L.; Duan, L. A rhodamine-based colorimetric and reversible fluorescent chemosensor for selectively detection of Cu2+ and Hg2+ ions. Sens. Actuators B Chem. 2014, 199, 121–126. [Google Scholar]
- Chen, C.; Men, G.; Bu, W.; Liang, C.; Sun, H.; Jiang, S. A colorimetric and fluorescent probe for multiple transition metal ions (Cu2+, Zn2+ and Ni2+): Fast response and high selectivity. Sens. Actuators B Chem. 2015, 220, 463–471. [Google Scholar]
- Kacmaz, S.; Ertekin, K.; Mercan, D.; Oter, O.; Cetinkaya, E.; Celik, E. An ultra sensitive fluorescent nanosensor for detection of ionic copper. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 135, 551–559. [Google Scholar]
- Basa, P.N.; Sykes, A.G. Differential sensing of Zn(II) and Cu(II) via two independent mechanisms. J. Org. Chem. 2012, 77, 8428–8434. [Google Scholar] [PubMed]
- Chatterjee, N.; Mahaling, B.; Sivakumar, S.; Bharadwaj, P.K. A highly selective and sensitive “turn-on” fluorescence chemosensor for the Cu2+ ion in aqueous ethanolic medium and its application in live cell imaging. J. Photochem. Photobiol. A Chem. 2016, 330, 110–116. [Google Scholar]
- Sun, Y.; Liu, Y.; Guo, W. Fluorescent and chromogenic probes bearing salicylaldehyde hydrazone functionality for cyanide detection in aqueous solution. Sens. Actuators B Chem. 2009, 143, 171–176. [Google Scholar]
- United States Environmental Protection Agency (USEPA). Basic Information about Copper in Drinking Water. Available online: http://water.epa.gov/drink/contaminants/basicinformation/copper.cfm (accessed on 5 January 2017).
- Kim, H.S.; Angupillai, S.; Son, Y.A. A dual chemosensor for both Cu2+ and Al3+: A potential Cu2+ and Al3+ switched YES logic function with an INHIBIT logic gate and a novel solid sensor for detection and extraction of Al3+ ions from aqueous solution. Sens. Actuators B Chem. 2016, 222, 447–458. [Google Scholar]
- Kaur, M.; Cho, M.J.; Choi, D.H. A phenothiazine-based “naked-eye” fluorescent probe for the dual detection of Hg2+ and Cu2+: Application as a solid state sensor. Dyes Pigments 2016, 125, 1–7. [Google Scholar]
- Erdemir, S.; Malkondu, S. Novel “turn on” fluorescent sensors based on anthracene and carbazone units for Cu(II) ion in CH3CN–H2O. J. Lumin. 2015, 158, 86–90. [Google Scholar]
- Peralta-Domínguez, D.; Rodriguez, M.; Ramos-Ortiz, G.; Maldonado, J.L.; Luna-Moreno, D.; Ortiz-Gutierrez, M.; Barba, V. A Schiff base derivative used as sensor of copper through colorimetric and surface plasmon resonance techniques. Sens. Actuators B Chem. 2016, 225, 221–227. [Google Scholar]
- Lu, D.; Lei, J.; Tian, Z.; Wang, L.; Zhang, J. Cu2+ fluorescent sensor based on mesoporous silica nanosphere. Dyes Pigments 2012, 94, 239–246. [Google Scholar]
- Zhou, Z.; Zheng, Y.; Wang, Q. Thiazole derivative based terbium(III) covalent silica nanosphere and its sensing property. Inorg. Chim. Acta 2013, 394, 127–131. [Google Scholar]
- Nugent, J.W.; Reibenspies, J.H.; Hancock, R.D. Controlling the fluorescence response of pet sensors via the metal-ion π-contacting ability of the fluorophore: Coumarin, a weaker π contacter. Inorg. Chem. 2015, 54, 9976–9988. [Google Scholar] [PubMed]
- Tobita, S.; Arakawa, M.; Tanaka, I. The paramagnetic metal effect on the ligand localized S1 .apprx. .fwdarw. T1 intersystem crossing in the rare-earth-metal complexes with methyl salicylate. J. Phys. Chem. 1985, 89, 5649–5654. [Google Scholar]
- Formica, M.; Fusi, V.; Giorgi, L.; Micheloni, M. New fluorescent chemosensors for metal ions in solution. Coord. Chem. Rev. 2012, 256, 170–192. [Google Scholar]
- Shao, N.; Zhang, Y.; Cheung, S.; Yang, R.; Chan, W.; Mo, T.; Li, K.A.; Liu, F. Copper ion-selective fluorescent sensor based on the inner filter effect using a spiropyran derivative. Anal. Chem. 2005, 77, 7294–7303. [Google Scholar] [PubMed]
- Su, Y.; Shi, B.; Liao, S.; Qin, Y.; Zhang, L.; Huang, M.; Zhao, S. Facile preparation of fluorescent polydihydroxyphenylalanine nanoparticles for label-free detection of copper ions. Sens. Actuators B Chem. 2016, 225, 334–339. [Google Scholar]
- Ghaedi, M.; Shahamiri, A.; Mirtamizdoust, B.; Hajati, S.; Taghizadeh, F. A novel polyvinyl chloride-membrane optical sensor for the determination of Cu2+ ion based on synthesized (N′1E,N′2E)-N′1,N′2-bis(pyridine-2-ylmethylene)oxalohydrazide: Experimental design and optimization. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 138, 878–884. [Google Scholar]
- Kim, S.; Kwak, J.; Lee, S.Y. Optical sensing of copper (II) ions using a biofunctional bolaamphiphile self-assembly: Selective binding of copper (II) ions to tyrosine moieties. Sens. Actuators B Chem. 2014, 203, 483–489. [Google Scholar]
- Huang, L.; Wang, X.; Xie, G.; Xi, P.; Li, Z.; Xu, M.; Wu, Y.; Bai, D.; Zeng, Z. A new rhodamine-based chemosensor for Cu2+ and the study of its behaviour in living cells. R. Soc. Chem. Dalton Trans. 2010, 39, 7894–7896. [Google Scholar]
- Swamy, K.M.K.; Ko, S.; Kwon, S.K.; Lee, H.N.; Mao, C.; Kim, J.; Lee, K.; Kim, J.; Shin, I.; Yoon, J. Boronic acid-linked fluorescent and colorimetric probes for copper ions. Chem. Commun. 2008, 5915–5917. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shekari, Z.; Younesi, H.; Heydari, A.; Tajbakhsh, M.; Chaichi, M.J.; Shahbazi, A.; Saberi, D. Fluorescence Chemosensory Determination of Cu2+ Using a New Rhodamine–Morpholine Conjugate. Chemosensors 2017, 5, 26. https://doi.org/10.3390/chemosensors5030026
Shekari Z, Younesi H, Heydari A, Tajbakhsh M, Chaichi MJ, Shahbazi A, Saberi D. Fluorescence Chemosensory Determination of Cu2+ Using a New Rhodamine–Morpholine Conjugate. Chemosensors. 2017; 5(3):26. https://doi.org/10.3390/chemosensors5030026
Chicago/Turabian StyleShekari, Zeinab, Habibollah Younesi, Akbar Heydari, Mahmood Tajbakhsh, Mohammad Javad Chaichi, Afsaneh Shahbazi, and Dariush Saberi. 2017. "Fluorescence Chemosensory Determination of Cu2+ Using a New Rhodamine–Morpholine Conjugate" Chemosensors 5, no. 3: 26. https://doi.org/10.3390/chemosensors5030026




