Regulation by Light of Chemotaxis to Nitrite during the Sexual Life Cycle in Chlamydomonas reinhardtii
Abstract
:1. Introduction
2. Results
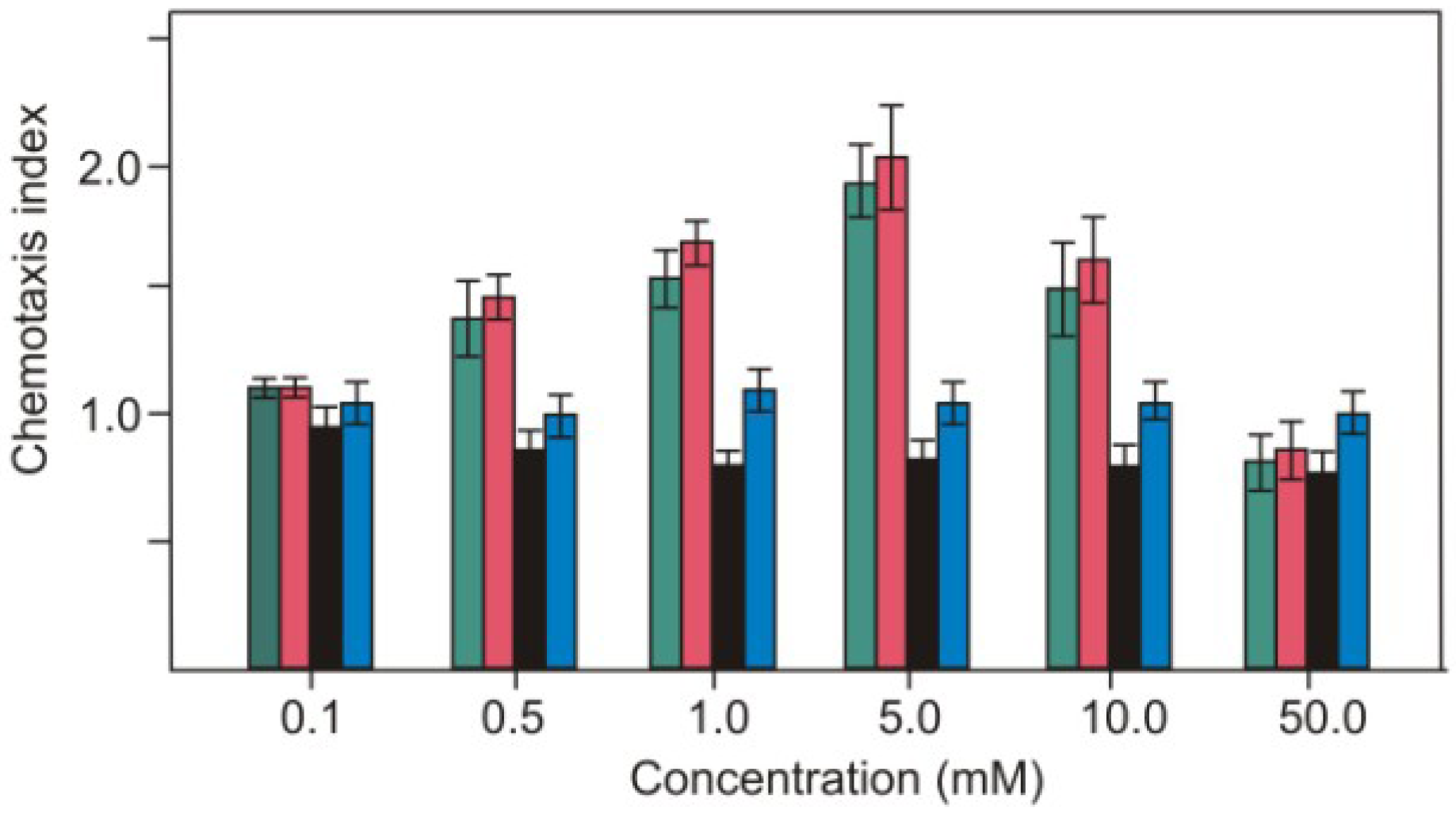
2.1. Nitrite as Chemoattractant
2.2. Role of Light in Change of Chemotaxis to Nitrite during Gametogenesis


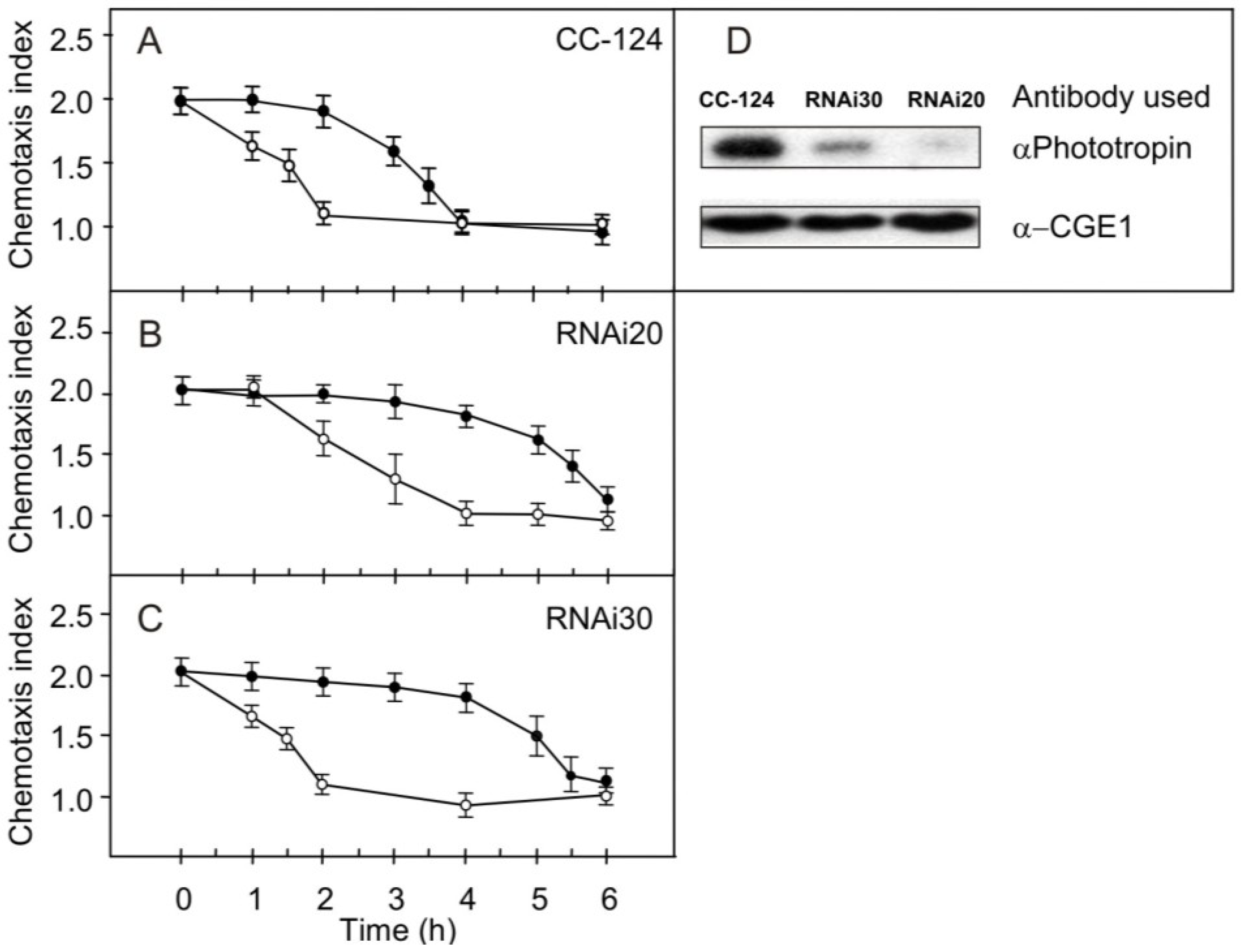
2.3. Effect of Reduced Phototropin Levels on the Kinetics of Changes in Chemotaxis to Nitrite
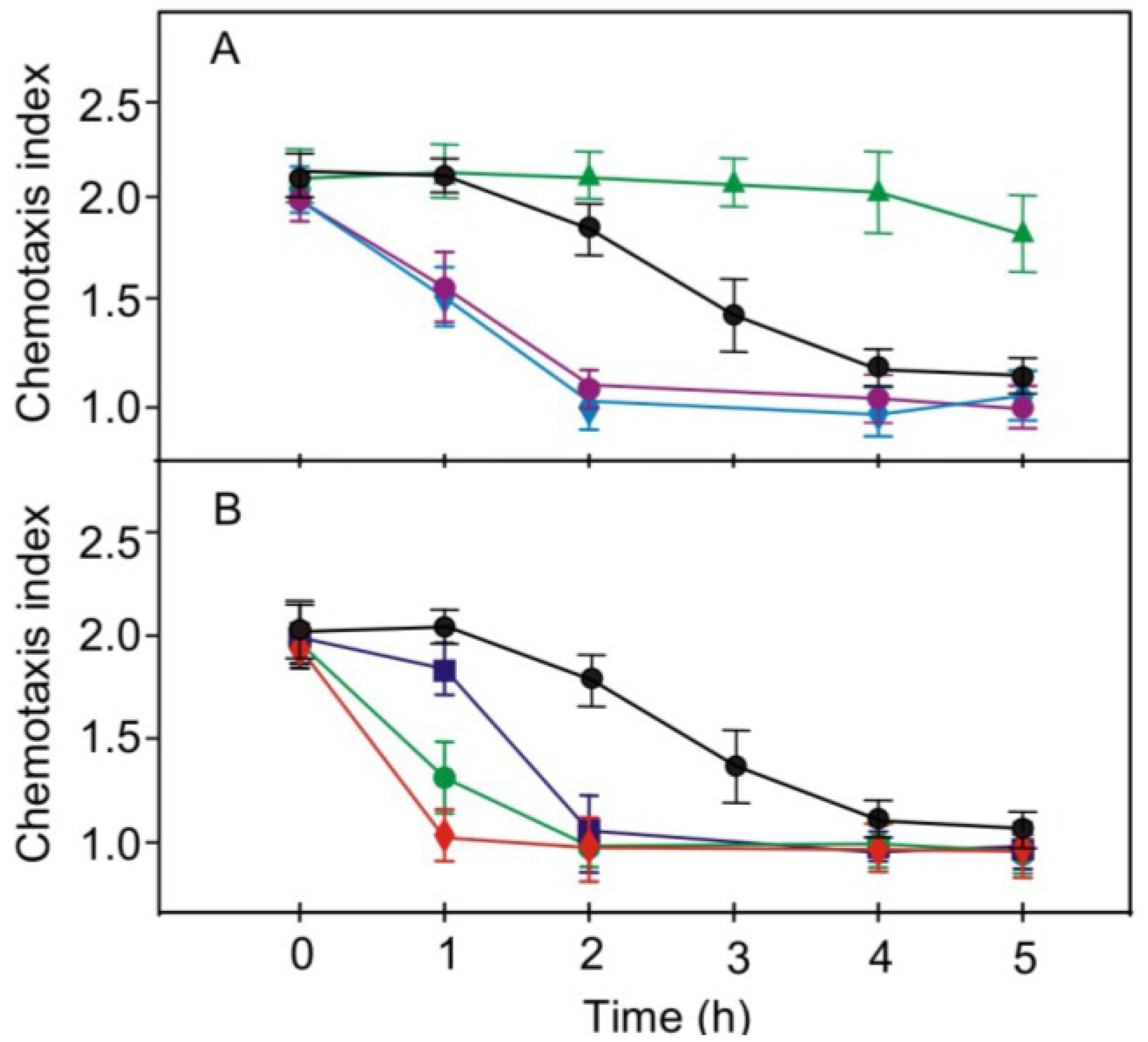
2.4. Effects of Pharmacological Compounds on the Kinetics of Changes in Chemotaxis to Nitrite in the Strain RNAi20

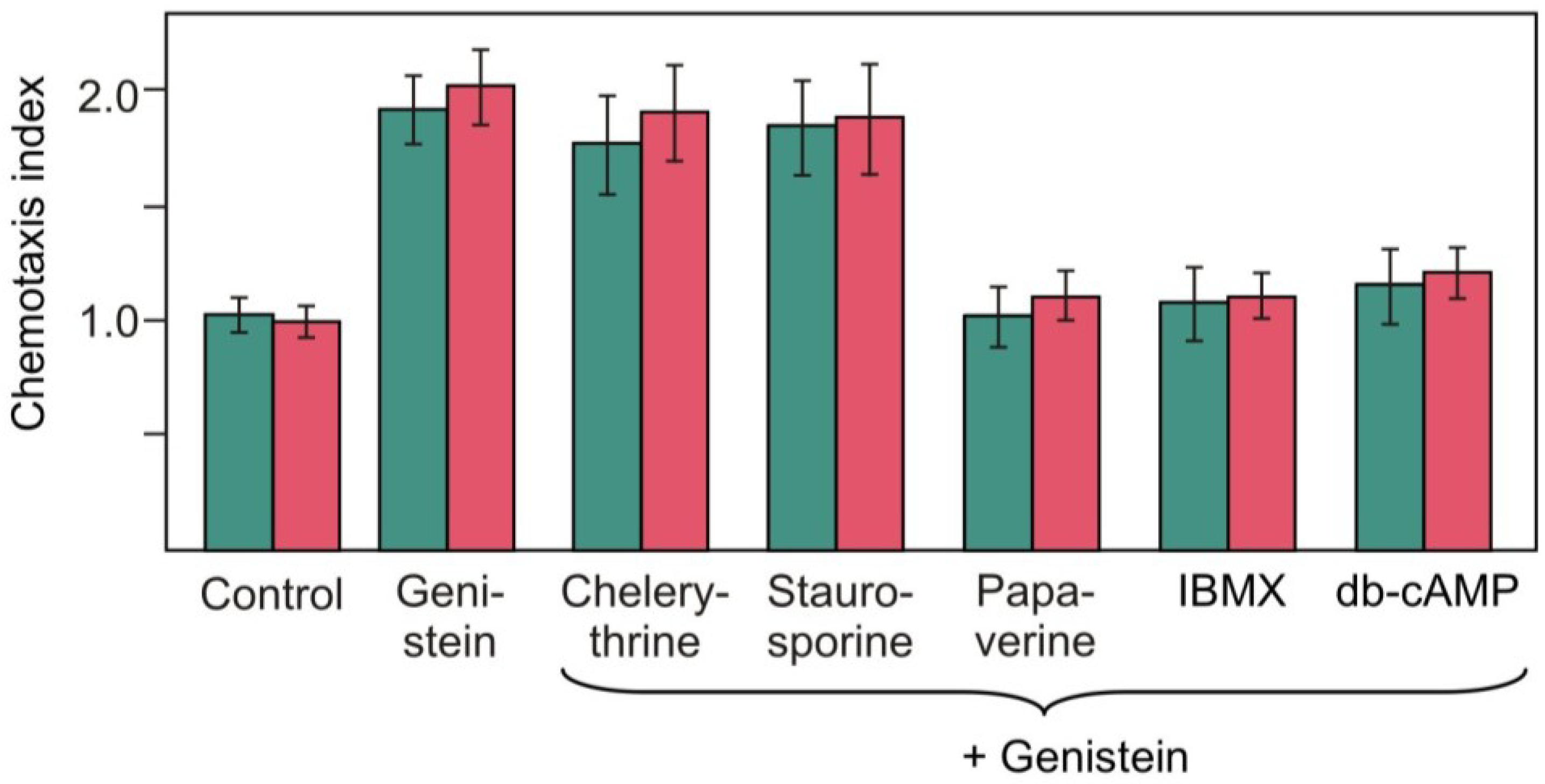
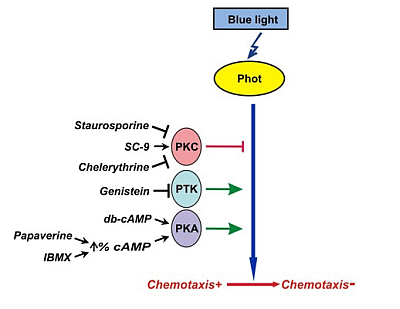
3. Discussion
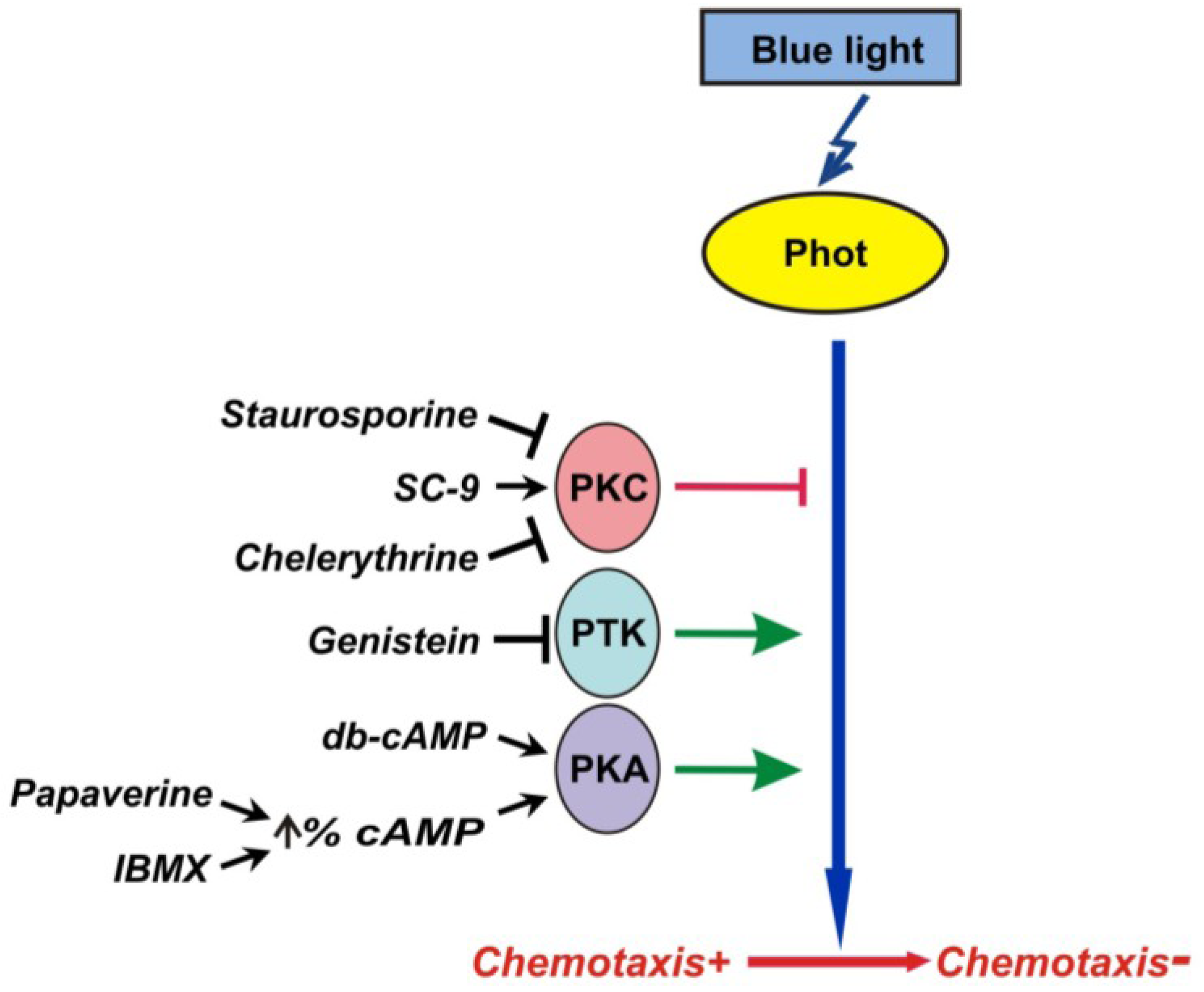
4. Experimental
4.1. Strains and Culture Conditions
4.2. Reagents
| Compound | Mode of action | Effective concentration | Reference |
|---|---|---|---|
| Papaverine | Phosphodiesterase inhibitor | 50 µM | [24,33] |
| 3-isobutyl-1-methylxanthine | Phosphodiesterase inhibitor | 50 µM | [15,24] |
| db-cAMP | PKA activator | 15 mM | [34] |
| Genistein | PTK inhibitor | 200 µM | [15,24,35] |
| Staurosporine | PKA inhibitor | 20 nM | [15,36,37] |
| Chelerythrine | PKC inhibitor | 1 µM | [24,38] |
| SC-9 | PKC activator | 200 µM | [15,39] |
4.3. Generation of Pregametes and Gametes
4.4. Determination of Mating Competence
4.5. Chemotaxis Assay

4.6. Protein Isolation, SDS-PAGE and Immunoblot Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Byrne, T.E.; Wells, M.R.; Johnson, C.H. Circadian rhythms of chemotaxis and of methylammonium uptake in by Chlamydomonas reinhardtii. Plant Physiol. 1992, 98, 879–886. [Google Scholar] [CrossRef]
- Ermilova, E.V.; Zalutskaya, Z.M.; Lapina, T.V.; Nikitin, M.M. Chemotactic behavior of Chlamydomonas reinhardtii is altered during gametogenesis. Curr. Microbiol. 2003, 46, 261–264. [Google Scholar] [CrossRef]
- Ermilova, E.V.; Zalutskaya, Z.M.; Nikitin, M.M.; Lapina, T.V.; Fernández, E. Regulation by light of ammonium transport systems in Chlamydomonas reinhardtii. Plant Cell Environ. 2010, 33, 1049–1056. [Google Scholar] [CrossRef]
- Furukawa, T.; Watanabe, M.; Shihira-Ishikawa, I. Blue and green light chloroplast migration in the centric diatom Pleurosira laevis. Protoplasma 1998, 203, 214–220. [Google Scholar] [CrossRef]
- Herman, C.A.; Im, C.-S. Light-regulated expression of the gsa gene encoding the chlorophyll biosynthetic enzyme glutamate 1-semialdehyde aminotransferase in carotenoid-deficient Chlamydomonas reinhardtii cells. Plant Mol. Biol. 1999, 39, 289–297. [Google Scholar] [CrossRef]
- Huang, K.; Merkle, T.; Beck, C.F. Isolation and characterization of a Chlamydomonas gene that encodes a putative blue-light photoreceptor of the phototropin family. Physiol Plant. 2002, 115, 613–622. [Google Scholar]
- Johnson, C.H.; Suzuki, L. Algae know the time of day: Circadian and photoperiodic programs. J. Phycol. 2001, 37, 933–942. [Google Scholar] [CrossRef]
- Oldenhof, H.; Bisová, K.; van den Ende, H.; Zachleder, V. Effect of red and blue light on the timing of cyclin-dependent kinase activity and the timing of cell division in Chlamydomonas reinhardtii. Plant Physiol. Biochem. 2004, 42, 341–348. [Google Scholar] [CrossRef]
- Hegemann, P. Algal sensory photoreceptors. Annu. Rev. Plant Biol. 2008, 59, 167–189. [Google Scholar] [CrossRef]
- Beck, C.; Haring, M. Gametic differentiation of Chlamydomonas. Int. Rev. Cytol. 1996, 168, 259–302. [Google Scholar] [CrossRef]
- Beck, C.F.; Acker, A. Gametic differentiation of Chlamydomonas reinhardtii. Plant Physiol. 1992, 98, 822–826. [Google Scholar] [CrossRef]
- Treier, U.; Fuchs, S.; Weber, M.; Wakarchuk, W.W.; Beck, C. Gametic differentiation in Chlamydomonas reinhardtii: Light dependence and gene expression patterns. Arch. Microbiol. 1989, 152, 572–577. [Google Scholar] [CrossRef]
- Im, C.-S.; Eberhard, S.; Huang, K.; Beck, C.F.; Grossman, A.R. Phototropin involvement in the expression of genes encoding chlorophyll and carotenoid biosynthesis enzymes and LHC apoproteins in Chlamydomonas reinhardtii. Plant J. 2006, 48, 1–16. [Google Scholar] [CrossRef]
- Trippens, J.; Greiner, A.; Schellwat, J.; Neukam, M.; Rottmann, T.; Lu, Y.; Kateriya, S.; Hegemann, P.; Kreimer, G. Phototropin influence on eyespot development and regulation of phototactic behavior in Chlamydomonas reinhardtii. Plant Cell 2012, 24, 4687–4702. [Google Scholar] [CrossRef]
- Pan, J.M.; Haring, M.A.; Beck, C.F. Dissection of the blue-light dependent signal transduction pathway involved in gametic differentiation of Chlamydomonas reinhardtii. Plant Physiol. 1996, 115, 1241–1249. [Google Scholar]
- Ermilova, E.; Nikitin, M.; Fernandez, E. Chemotaxis to ammonium/methylammonium in Chlamydomonas reinhardtii: The role of transport systems for ammonium/methylammonium. Planta 2007, 226, 1323–1332. [Google Scholar] [CrossRef]
- Ermilova, E.V.; Zalutskaya, Z.M.; Lapina, T.V. Chemotaxis of Chlamydomonas reinhardtii to nitrate is changed during gametogenesis. Protistology 2009, 6, 79–84. [Google Scholar]
- Ermilova, E.V.; Zalutskaya, Z.M.; Huang, K.; Beck, C.F. Ptototropin plays a crucial role in controlling changes in chemotaxis during the initial phase of the sexual life cycle in Chlamydomonas. Planta 2004, 219, 420–427. [Google Scholar]
- Matsuda, Y.; Shimada, T.; Sakamoto, Y. Ammonium ions control gametic differentiation and dedifferentiation in Chlamydomonas reinhardtii. Plant Cell Physiol. 1992, 33, 909–914. [Google Scholar]
- Pozuelo, M.; Merchán, F.; Macías, M.I.; Beck, C.F.; Galván, A.; Fernández, E. The negative effect of nitrate on gametogenesis is independent of nitrate assimilation in Chlamydomonas reinhardtii. Planta 2000, 211, 287–292. [Google Scholar] [CrossRef]
- Galvan, A.; Fernandez, E. Eukaryotic nitrate and nitrite transporters. Cell Mol. Life Sci. 2001, 58, 225–233. [Google Scholar] [CrossRef]
- Lengeler, K.B.; Davidson, R.C.; D’souza, C.; Toshiaki, H.; Shen, W.-C.; Wang, P.; Pan, X.; Waugh, M.; Heitman, J. Signal transduction cascades regulating fungal development and virulence. Microbiol. Mol. Biol. Rev. 2000, 64, 746–785. [Google Scholar] [CrossRef]
- Treier, U.; Beck, C.F. Changes in gene expression patterns during the sexual life cycle of Chlamydomonas reinhardtii. Physiol. Plant. 1991, 83, 633–639. [Google Scholar] [CrossRef]
- Ermilova, E.V.; Zalutskaya, Z.M.; Baibus, D.M.; Beck, C.F. Characterization of phototropin-controlled signaling components that regulate chemotaxis towards ammonium in Chlamydomonas. Protistology 2006, 4, 301–310. [Google Scholar]
- Camargo, A.; Llamas, A.; Schnell, R.A.; Higuera, J.J.; Gonzáles-Ballester, D.; Lefevbre, P.A.; Fernández, E.; Galván, A. Nitrate signaling by the regulatory gene NIT2 in Chlamydomonas. Plant Cell 2007, 19, 3491–3503. [Google Scholar] [CrossRef]
- Ho, C.H.; Lin, C.H.; Hu, H.C.; Tsay, Y.F. CHL1 functions as a nitrate sensor in plants. Cell 2009, 138, 1184–1194. [Google Scholar] [CrossRef]
- Fernandez, E.; Llamas, A.; Galvan, A. Nitrogen Assimilation and Its Regulation. In The Chlamydomonas Sourcebook, 2nd ed.; Harris, S., Ed.; Elsevier Press: Oxford, UK, 2009; Volume II, pp. 69–113. [Google Scholar]
- Llamas, A.; Igeño, M.I.; Galván, A.; Fernández, E. Nitrate signaling on the nitrate reductase gene promoter depends directly on the activity of the nitrate transport systems in Chlamydomonas. Plant J. 2002, 30, 261–271. [Google Scholar] [CrossRef]
- Pan, J.M.; Haring, M.A.; Beck, C.F. Characterization of blue light signal transduction chains that control development and maintenance of sexual competence in Chlamydomonas reinhardtii. Plant Physiol. 1997, 115, 1241–1249. [Google Scholar]
- Huang, K.; Beck, C.F. Phototropin is the blue-light receptor that controls multiple steps in the sexual life cycle of the green alga Chlamydomonas reinhardtii. Proc. Natl. Acad. Sci. USA 2003, 100, 6269–6274. [Google Scholar] [CrossRef]
- Harris, E.H. The Chlamydomonas Sourcebook; Academic Press: San Diego, CA, USA, 1989; pp. 1–24. [Google Scholar]
- Gorman, D.S.; Levine, R.P. Cytochrome and plastocyanin: Their sequences in the photosynthetic electron transport chain of Chlamydomonas reinhardtii. Proc. Natl. Acad. Sci. USA 1965, 54, 1665–1669. [Google Scholar] [CrossRef]
- Pasquale, S.M.; Goodenough, U.W. Cyclic AMP functions as a primary sexual signal in gametes of Chlamydomonas reinhardtii. J. Cell Biol. 1987, 105, 2279–2292. [Google Scholar] [CrossRef]
- Lando, M.; Abemayor, E.; Verity, M.A.; Sidell, N. Modulation of intracellular cyclic adenosine monophosphate levels and the differentiation response of human neuroblastoma cells. Cancer Res. 1990, 50, 722–777. [Google Scholar]
- Bowler, C.; Yamagata, H.; Neuhaus, G.; Chua, N.H. Phytochrome mediated signal transduction pathways are regulated by reciprocal control mechanisms. Genes Dev. 1994, 8, 2188–2202. [Google Scholar] [CrossRef]
- Zhang, Y.; Snell, W.J. Flagellar adhesion-dependent regulation of Chlamydomonas adenylyl cyclase in vitro: A possible role for protein kinases in sexual signaling. J. Cell Biol. 1994, 125, 617–624. [Google Scholar] [CrossRef]
- Meggio, F.; Donella Deana, A.; Ruzzene, M.; Brunati, A.M.; Cesaro, L.; Guerra, B.; Meyer, T.; Mett, H.; Fabro, D.; Furet, P.; et al. Different susceptibility of protein kinases to staurosporine inhibition. Eur. J. Biochem. 1995, 234, 317–322. [Google Scholar]
- Herbert, J.M.; Augereau, J.M.; Gleye, J.; Maffrand, J.P. Chelerythrine is a potent and specific inhibitor of protein kinase C. Biochem. Biophys. Res. Commun. 1990, 172, 993–999. [Google Scholar] [CrossRef]
- Ito, M.; Tanaka, T.; Inagaki, M.; Nakanishi, K.; Hidaka, H.N. (6-Phenylhexyl)-5-chloro-1-naphthalenesulfonamide, a novel activator of protein kinase C. Biochemistry 1986, 25, 4179–4184. [Google Scholar] [CrossRef]
- Ermilova, E.V.; Zalutskaya, Z.M.; Munnik, T.; van den Ende, H.; Gromov, B.V. Calcium in the control of chemotaxis in Chlamydomonas. Biol. Bratislava 1998, 53, 577–581. [Google Scholar]
- Popov, N.; Schmitt, S.; Matthices, H. Eine störungsfreie Mikromethode zur Bestimmung des Proteingahalts in Gewebshomogenaten. Acta Biol. Germ. 1975, 34, 1441–1446. [Google Scholar]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Schroda, M.; Vallon, O.; Whitelegge, J.P.; Beck, C.F.; Wollman, F.-A. The chloroplastic GrpE homolog of Chlamydomonas. Two isoforms generated by differential splicing. Plant Cell 2001, 13, 2823–2839. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ermilova, E.; Zalutskaya, Z. Regulation by Light of Chemotaxis to Nitrite during the Sexual Life Cycle in Chlamydomonas reinhardtii. Plants 2014, 3, 113-127. https://doi.org/10.3390/plants3010113
Ermilova E, Zalutskaya Z. Regulation by Light of Chemotaxis to Nitrite during the Sexual Life Cycle in Chlamydomonas reinhardtii. Plants. 2014; 3(1):113-127. https://doi.org/10.3390/plants3010113
Chicago/Turabian StyleErmilova, Elena, and Zhanneta Zalutskaya. 2014. "Regulation by Light of Chemotaxis to Nitrite during the Sexual Life Cycle in Chlamydomonas reinhardtii" Plants 3, no. 1: 113-127. https://doi.org/10.3390/plants3010113