Expression and Function of CD44 in Epithelial Ovarian Carcinoma
Abstract
:1. Introduction
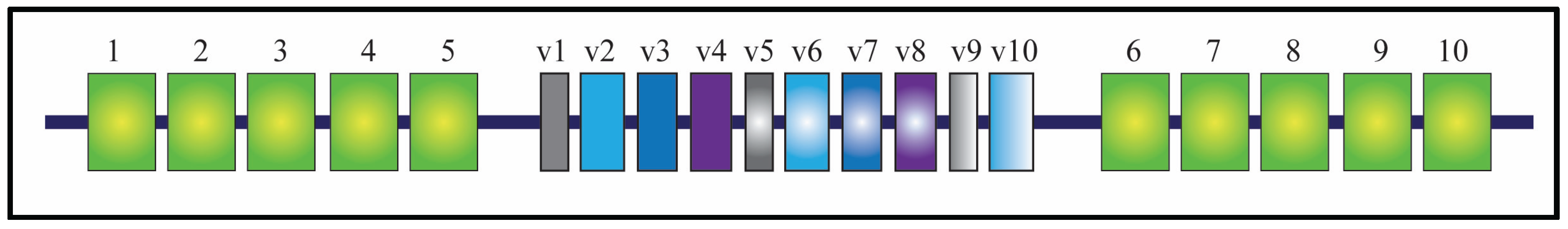
2. CD44 Expression
| Sample Type | Method(s) & Antibodies | Results | Reference |
|---|---|---|---|
| 31 epithelial ovarian tumors (24 serous, 5 endometrioid, 2 clear cell) | RT-PCR, IHC pan-CD44 (clone 25.32) CD44v3 (clone BBA11) CD44v4 (clone 11.10) CD44v6 (clone 11.9) CD44v9 (clone 11.24) | High expression of CD44s and CD44v9 in EOC; lack of association between CD44v and prognosis | [17] |
| 21 epithelial ovarian tumors (13 serous, 2 endometrioid, 6 mucinous) | RT-PCR | CD44s amplified in all specimens; CD44R1/s ratio higher in metastases than in primary tumors | [44] |
| 22 epithelial ovarian tumors (9 serous, 8 mucinous, 5 undifferentiated) | IHC CD44v5 (clone VFF-8) CD44v6 (clone VFF-7) CD44v7-8 (clone VFF-17) | Low CD44v5, v6, and v7-8 expression | [45] |
| 44 epithelial ovarian tumors (26 serous, 12 mucinous, 6 other) | IHC CD44 v3-10 (clone 1.1ASML) | Association between increased CD44v expression and reduced survival | [24] |
| 11 ovarian tumors (6 serous, 1 endometrioid, 2 clear cell, 1 yolk sac, 1 dysgerminoma) | IHC CD44v1-10 CD44v6 (clone 2F10) CD44v9 | Expression of CD44v6 involved in nodal metastasis | [42] |
| 76 epithelial ovarian tumors (20 benign, 36 borderline; 20 carcinoma: 10 serous, 10 mucinous) | IHC pan-CD44 (clone 2C5) CD44v3 (clone 3G5) CD44v6 (clone 2F10) | CD44 isoforms in ovarian borderline tumors of littlediagnostic and prognostic value | [46] |
| 43 epithelial ovarian tumors (29 serous, 6 mucinous, 5 clear cell, 3 endometrioid) | IHC CD44v6 (clone BBA13) | CD44v6 expression greater in recurrent tumors compared to primary | [47] |
| 56 epithelial ovarian tumors (38 serous, 6 poorly differentiated, 5 mucinous, 3 clear cell, 2 mixed Müllerian, 2 endometrioid | IHC pan-CD44 (clone SFF-304) | CD44 expression was significantly associated with poorer overall survival | [15] |
| 115 epithelial ovarian tumors carcinomas (50 serous, 19 mucinous, 16 endometrioid, 30 clear cell), 32 low malignant potential (LMP) tumors (3 serous, 29 mucinous), 53 cystadenomas (14 serous, 39 mucinous) | RT-PCR, IHC pan-CD44 (clone DF1485) CD44v3 (clone 3G5) CD44v6 (clone 2F10) | CD44 expression upregulated during ovarian carcinoma development, but down-regulated during progression; loss of CD44v3 expression predicts poor prognosis | [13] |
| 56 epithelial ovarian tumors (40 serous, 5 mucinous, 3 endometrioid, 5 clear cell, 3 undifferentiated) | IHC CD44v6 (clone 2F10) | No significant change in CD44v6 expression in primary and matched metastatic sites | [39] |
| 50 epithelial ovarian tumors (25 serous, 10 mucinous, 9 endometrioid, 6 other) | IHC pan-CD44 (clone SFF-2) CD44v5 (clone VFF-8) CD44v6 (clone VFF-7) CD44v7-8 (clone VFF-9) CD44v10 (clone VFF-14) | High expression of CD44v5; CD44v5 and v6 associated with intraperitoneal implantation | [48] |
| 28 epithelial ovarian tumors (all serous; 1 well-differentiated, 12 moderately differentiated, 15 poorly differentiated) | IHC pan-CD44 CD44v5 (clone VFF-8) CD44v6 (clone VFF-7) CD44v7-8 (clone VFF-17) | Increased expression of CD44s and CD44v5 in stage III compared with stage I tumors. | [25] |
| 101 epithelial ovarian tumors (45 serous, 9 endometroid, 5 clear cell 5 mucinous; 18 LMP: 13 serous, 4 mucinous-GI type, 1 endometrioid) | IHC pan-CD44 (clone A3D8) | Decreased expression of CD44s associated with shortened survival | [26] |
| 142 epithelial ovarian tumors (93 serous, 15 mucinous, 14 endometrioid, 21 LMP) | IHC pan-CD44 (clone 2C5) CD44v3 (clone 3G5) CD44v4 (ATCC 257-HB) CD44v5 (clone VFF-8) CD44v6 (ATCC 256-HB) CD44v9 (ATCC 258-HB) CD44v10 (clone VFF-14) | Expression of CD44s, CD44v4, CD44v6, and CD44v10 in primary EOC associated with increased survival | [27] |
| 307 epithelial ovarian tumors (109 serous, 31 mucinous, 82 endometrioid, 32 clear cell, 53 other) | IHC pan-CD44(clones 2C5, DF1485) | High CD44 expression associated with favorable prognosis | [16] |
| 83 epithelial ovarian tumors (31 serous, 4 mucinous, 3 mixed, 3 clear cell, 7 poorly differentiated; 14 borderline, 21 benign) | IHC pan-CD44 (clone DF1485) | No correlation between CD44 and survival | [49] |
| 158 epithelial ovarian tumors (95 malignant serous, 23 benign, 40 borderline) | IHC pan-CD44 (clone DF1485) CD44v6 (clone VFF-7) | Overexpression of CD44 associated with high grade, advanced stage, and low overall survival rate | [14] |
| 57 epithelial ovarian tumors (all serous) | IHC CD44v6 (VFF-18) | CD44 expression higher in borderline and carcinomas compared to benign, but no significant correlation with clinicopathological factors | [35] |
| 62 epithelial ovarian tumors (45 serous, 5 mucinous, 8 endometrioid, 4 undifferentiated) | IHC CD44v6 (clone VFF-7) | CD44v6 an important molecular marker for poor prognosis | [36] |
| 65 epithelial ovarian tumors (all serous) | IHC pan-CD44 (clone IM7) CD44v6 (clone VFF-18) | CD44v6 expression associated with progression, metastasis, and relapse | [37] |
| 72 epithelial ovarian tumors (all serous: 14 well-differentiated, 36 moderately differentiated, 22 poorly differentiated) | IHC pan-CD44 (clone DF1485) | No change in expression between primary tumors and peritoneal metastases; Increased CD44 expression in patients with metastases | [30] |
| 483 primary epithelial ovarian tumors (393 serous, 35 endometrioid, 16 clear cell, 11 transitional cell, 28 other) 127 paired primary and recurrent tumors | IHC pan-CD44(clone 156-3C11) | CD44 expression associated with high grade and advanced stage carcinoma, but not with overall survival; no statistical difference in CD44 expression between primary and recurrent disease | [33] |
| 26 paired primary, metastatic, and recurrent epithelial ovarian tumors from well-characterized late-stage ovarian cancer patients | IHC pan-CD44(clone 156-3C11) | Significant association between CD44 expression and both disease-free and overall survival | [32] |
3. Post-Translational Modifications of CD44
4. CD44 and Stemness
5. CD44 in Peritoneal Adhesion
6. Therapeutic Targeting of CD44
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ponta, H.; Sherman, L.; Herrlich, P.A. CD44: From adhesion molecules to signalling regulators. Nat. Rev. Mol. Cell Biol. 2003, 4, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Naor, D.; Nedvetzki, S.; Golan, I.; Melnik, L.; Faitelson, Y. CD44 in cancer. Crit. Rev. Clin. Lab. Sci. 2002, 39, 527–579. [Google Scholar] [CrossRef] [PubMed]
- Stickeler, E.; Runnebaum, I.B.; Mobus, V.J.; Kieback, D.G.; Kreienberg, R. Expression of CD44 standard and variant isoforms v5, v6 and v7 in human ovarian cancer cell lines. Anticancer Res. 1997, 17, 1871–1876. [Google Scholar] [PubMed]
- Bennett, K.L.; Modrell, B.; Greenfield, B.; Bartolazzi, A.; Stamenkovic, I.; Peach, R.; Jackson, D.G.; Spring, F.; Aruffo, A. Regulation of CD44 binding to hyaluronan by glycosylation of variably spliced exons. J. Cell Biol. 1995, 131, 1623–1633. [Google Scholar] [CrossRef] [PubMed]
- Di Sante, G.; Migliara, G.; Valentini, M.; Delogu, G.; Ria, F. Regulation of and regulation by CD44: A paradigm complex regulatory network. Int. Trends Immun. 2013, 1, 33–42. [Google Scholar]
- Naor, D.; Sionov, R.V.; Ish-Shalom, D. CD44: Structure, function and association with the malignant process. In Advances in Cancer Research; George, F.V.W., George, K., Eds.; Academic Press: New York, NY, USA, 1997; pp. 241–319. [Google Scholar]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2015. CA Cancer J. Clin. 2015, 65, 5–29. [Google Scholar] [CrossRef] [PubMed]
- Panici, P.B.; Angioli, R. Role of lymphadenectomy in ovarian cancer. Best Pract. Res. Clin. Obstet. Gynaecol. 2002, 16, 529–551. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, S.; Kim, S.W.; Wu, S.Y.; Nishimura, M.; Chaluvally-Raghavan, P.; Miyake, T.; Pecot, C.V.; Kim, S.J.; Choi, H.J.; Bischoff, F.Z.; et al. Hematogenous metastasis of ovarian cancer: Rethinking mode of spread. Cancer Cell 2014, 26, 77–91. [Google Scholar] [CrossRef] [PubMed]
- Lengyel, E. Ovarian cancer development and metastasis. Am. J. Pathol. 2010, 177, 1053–1064. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.R.; Shih Ie, M. Ovarian cancer. Annu. Rev. Pathol. 2009, 4, 287–313. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.S.P.; Agarwal, R.; Kaye, S.B. Mechanisms of transcoelomic metastasis in ovarian cancer. Lancet Oncol. 2006, 7, 925–934. [Google Scholar] [CrossRef]
- Saegusa, M.; Machida, D.; Hashimura, M.; Okayasu, I. CD44 expression in benign, premalignant, and malignant ovarian neoplasms: Relation to tumour development and progression. J. Pathol. 1999, 189, 326–337. [Google Scholar] [CrossRef]
- Cho, E.Y.; Choi, Y.; Chae, S.W.; Sohn, J.H.; Ahn, G.H. Immunohistochemical study of the expression of adhesion molecules in ovarian serous neoplasms. Pathol. Int. 2006, 56, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Kayastha, S.; Freedman, A.N.; Piver, M.S.; Mukkamalla, J.; Romero-Guittierez, M.; Werness, B.A. Expression of the hyaluronan receptor, CD44s, in epithelial ovarian cancer is an independent predictor of survival. Clin. Cancer Res. 1999, 5, 1073–1076. [Google Scholar] [PubMed]
- Sillanpaa, S.; Anttila, M.A.; Voutilainen, K.; Tammi, R.H.; Tammi, M.I.; Saarikoski, S.V.; Kosma, V.M. CD44 expression indicates favorable prognosis in epithelial ovarian cancer. Clin. Cancer Res. 2003, 9, 5318–5324. [Google Scholar] [PubMed]
- Cannistra, S.A.; Abu-Jawdeh, G.; Niloff, J.; Strobel, T.; Swanson, L.; Andersen, J.; Ottensmeier, C. CD44 variant expression is a common feature of epithelial ovarian cancer: Lack of association with standard prognostic factors. J. Clin. Oncol. 1995, 13, 1912–1921. [Google Scholar] [PubMed]
- Paik, D.Y.; Janzen, D.M.; Schafenacker, A.M.; Velasco, V.S.; Shung, M.S.; Cheng, D.; Huang, J.; Witte, O.N.; Memarzadeh, S. Stem-like epithelial cells are concentrated in the distal end of the fallopian tube: A site for injury and serous cancer initiation. Stem Cells 2012, 30, 2487–2497. [Google Scholar] [CrossRef] [PubMed]
- Cannistra, S.A.; Kansas, G.S.; Niloff, J.; DeFranzo, B.; Kim, Y.; Ottensmeier, C. Binding of ovarian cancer cells to peritoneal mesothelium in vitro is partly mediated by CD44H. Cancer Res. 1993, 53, 3830–3838. [Google Scholar] [PubMed]
- Bourguignon, L.Y.; Zhu, H.; Zhou, B.; Diedrich, F.; Singleton, P.A.; Hung, M.C. Hyaluronan promotes CD44v3-Vav2 interaction with Grb2-p185(HER2) and induces Rac1 and Ras signaling during ovarian tumor cell migration and growth. J. Biol. Chem. 2001, 276, 48679–48692. [Google Scholar] [CrossRef] [PubMed]
- Volz, Y.; Koschut, D.; Matzke-Ogi, A.; Dietz, M.S.; Karathanasis, C.; Richert, L.; Wagner, M.G.; Mely, Y.; Heilemann, M.; Niemann, H.H.; et al. Direct binding of hepatocyte growth factor and vascular endothelial growth factor to CD44v6. Biosci. Rep. 2015. [Google Scholar] [CrossRef] [PubMed]
- Preca, B.T.; Bajdak, K.; Mock, K.; Sundararajan, V.; Pfannstiel, J.; Maurer, J.; Wellner, U.; Hopt, U.T.; Brummer, T.; Brabletz, S.; et al. A self-enforcing CD44s/ZEB1 feedback loop maintains emt and stemness properties in cancer cells. Int. J. Cancer 2015, 137, 2566–2577. [Google Scholar] [CrossRef] [PubMed]
- Ween, M.P.; Oehler, M.K.; Ricciardelli, C. Role of versican, hyaluronan and CD44 in ovarian cancer metastasis. Int. J. Mol. Sci. 2011, 12, 1009–1029. [Google Scholar] [CrossRef] [PubMed]
- Uhl-Steidl, M.; Muller-Holzner, E.; Zeimet, A.G.; Adolf, G.R.; Daxenbichler, G.; Marth, C.; Dapunt, O. Prognostic value of CD44 splice variant expression in ovarian cancer. Oncology 1995, 52, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Afify, A.M.; Ferguson, A.W.; Davila, R.M.; Werness, B.A. Expression of CD44s and CD44v5 is more common in stage III than in stage I serous ovarian carcinomas. Appl. Immunohistochem. Mol. Morphol. 2001, 9, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.S.; Sheehan, C.E.; Williams, S.S.; Malfetano, J.H.; Szyfelbein, W.M.; Kallakury, B.V. Decreased CD44 standard form expression correlates with prognostic variables in ovarian carcinomas. Am. J. Clin. Pathol. 2001, 116, 122–128. [Google Scholar] [PubMed]
- Rodriguez-Rodriguez, L.; Sancho-Torres, I.; Mesonero, C.; Gibbon, D.G.; Shih, W.J.; Zotalis, G. The CD44 receptor is a molecular predictor of survival in ovarian cancer. Med. Oncol. 2003, 20, 255–263. [Google Scholar] [CrossRef]
- Speiser, P.; Wanner, C.; Breitenecker, G.; Kohlberger, P.; Kainz, C. CD44 is not involved in the metastatic spread of ovarian cancer in vivo. Anticancer Res. 1995, 15, 2767–2769. [Google Scholar] [PubMed]
- Sanchez Lockhart, M.; Hajos, S.E.; Basilio, F.M.; Mongini, C.; Alvarez, E. Splice variant expression of CD44 in patients with breast and ovarian cancer. Oncol. Rep. 2001, 8, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Ryabtseva, O.D.; Lukianova, N.Y.; Shmurakov, Y.A.; Polishchuk, L.Z.; Antipova, S.V. Significance of adhesion molecules expression for estimation of serous ovarian cancer prognosis. Exp. Oncol. 2013, 35, 211–218. [Google Scholar] [PubMed]
- Chen, H.; Hao, J.; Wang, L.; Li, Y. Coexpression of invasive markers (uPA, CD44) and multiple drug-resistance proteins (MDR1, MRP2) is correlated with epithelial ovarian cancer progression. Br. J. Cancer 2009, 101, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Foster, R.; Yang, X.; Feng, Y.; Shen, J.K.; Mankin, H.J.; Hornicek, F.J.; Amiji, M.M.; Duan, Z. Up-regulation of CD44 in the development of metastasis, recurrence and drug resistance of ovarian cancer. Oncotarget 2015, 6, 9313–9326. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chang, B.; Liu, J. CD44 standard form expression is correlated with high-grade and advanced-stage ovarian carcinoma but not prognosis. Hum. Pathol. 2013, 44, 1882–1889. [Google Scholar] [CrossRef] [PubMed]
- Bar, J.K.; Grelewski, P.; Popiela, A.; Noga, L.; Rabczynski, J. Type IV collagen and CD44v6 expression in benign, malignant primary and metastatic ovarian tumors: Correlation with Ki-67 and p53 immunoreactivity. Gynecol. Oncol. 2004, 95, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.C.; Song, J.Y.; Lee, J.K.; Lee, N.W.; Kim, S.H.; Yeom, B.W.; Lee, K.W. Significance of CD44v6 expression in gynecologic malignancies. J. Obstet. Gynaecol. Res. 2006, 32, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.X.; Liu, Y.X.; Xue, Y.H. Expression of CD44v6 and its association with prognosis in epithelial ovarian carcinomas. Pathol. Res. Int. 2012. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Zhou, Z.; Di, W.; Li, N. Correlation of CD44v6 expression with ovarian cancer progression and recurrence. BMC Cancer 2013, 13, 182. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Lu, L.; Wang, Y.; Sun, Y.; Zhang, Y.; Guo, C.; Gu, Y.; Liu, A. Expression and clinicopathologic significance of CD44v6/CD24 in ovarian serous carcinomas. Chin. J. Pathol. 2014, 43, 20–24. (In Chinese) [Google Scholar]
- Sakai, K.; Kaku, T.; Kamura, T.; Kinukawa, N.; Amada, S.; Shigematsu, T.; Hirakawa, T.; Kobayashi, H.; Ariyoshi, K.; Nakano, H. Comparison of p53, Ki-67, and CD44v6 expression between primary and matched metastatic lesions in ovarian cancer. Gynecol. Oncol. 1999, 72, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Berner, H.S.; Davidson, B.; Berner, A.; Risberg, B.; Kristensen, G.B.; Trope, C.G.; van de Putte, G.; Nesland, J.M. Expression of CD44 in effusions of patients diagnosed with serous ovarian carcinoma—Diagnostic and prognostic implications. Clin. Exp. Metastasis 2000, 18, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Kainz, C.; Tempfer, C. CD44 as a differentiation marker and metastasis promoter in gynecological malignancies. Cancer J. 1997, 10, 306–309. [Google Scholar]
- Yorishima, T.; Nagai, N.; Ohama, K. Expression of CD44 alternative splicing variants in primary and lymph node metastatic lesions of gynecological cancer. Hiroshima J. Med. Sci. 1997, 46, 21–29. [Google Scholar] [PubMed]
- Tjhay, F.; Motohara, T.; Tayama, S.; Narantuya, D.; Fujimoto, K.; Guo, J.; Sakaguchi, I.; Honda, R.; Tashiro, H.; Katabuchi, H. CD44 variant 6 is correlated with peritoneal dissemination and poor prognosis in patients with advanced epithelial ovarian cancer. Cancer Sci. 2015, 106, 1421–1428. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kang, Y.S.; Kim, B.G.; Park, S.Y.; Lee, E.D.; Lee, K.H.; Park, K.B. Expression of the CD44 adhesion molecule in primary and metastatic gynecologic malignancies and their cell lines. Int. J. Gynecol. Cancer 1995, 5, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Sliutz, G.; Tempfer, C.; Winkler, S.; Kohlberger, P.; Reinthaller, A.; Kainz, C. Immunohistochemical and serological evaluation of CD44 splice variants in human ovarian cancer. Br. J. Cancer 1995, 72, 1494–1497. [Google Scholar] [CrossRef] [PubMed]
- Darai, E.; Walker-Combrouze, F.; Fauconnier, A.; Madelenat, P.; Potet, F.; Scoazec, J.Y. Analysis of CD44 expression in serous and mucinous borderline tumours of the ovary: Comparison with cystadenomas and overt carcinomas. Histopathology 1998, 32, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Kamura, T.; Sakai, K.; Kaku, T.; Kobayashi, H.; Mitsumoto, M.; Tsuneyoshi, M.; Nakano, H. Comparison of p53 and CD44 variant 6 expression between paired primary and recurrent ovarian cancer: An immunohistochemical analysis. Oncol. Rep. 1999, 6, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Schroder, W.; Rudlowski, C.; Biesterfeld, S.; Knobloch, C.; Hauptmann, S.; Rath, W. Expression of CD44(v5-10) splicing variants in primary ovarian cancer and lymph node metastases. Anticancer Res. 1999, 19, 3901–3906. [Google Scholar] [PubMed]
- Zagorianakou, N.; Stefanou, D.; Makrydimas, G.; Zagorianakou, P.; Briasoulis, E.; Karavasilis, B.; Agnantis, N.J. CD44s expression, in benign, borderline and malignant tumors of ovarian surface epithelium. Correlation with p53, steroid receptor status, proliferative indices (PCNA, MIB1) and survival. Anticancer Res. 2004, 24, 1665–1670. [Google Scholar] [PubMed]
- Sneath, R.J.; Mangham, D.C. The normal structure and function of CD44 and its role in neoplasia. Mol. Pathol. 1998, 51, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, M.; Ma, B.Y.; Murai, R.; Nakamura, N.; Baba, M.; Kawasaki, N.; Hodohara, K.; Asano, S.; Kawasaki, T. Glycosylation-dependent interactions of C-type lectin DC-SIGN with colorectal tumor-associated lewis glycans impair the function and differentiation of monocyte-derived dendritic cells. J. Immunol. 2008, 180, 3347–3356. [Google Scholar] [CrossRef] [PubMed]
- Walsh, C. Posttranslational Modification of Proteins: Expanding Nature’s Inventory; Roberts and Co. Publishers: Englewood, CO, USA, 2006. [Google Scholar]
- Casey, R.C.; Oegema, T.R., Jr.; Skubitz, K.M.; Pambuccian, S.E.; Grindle, S.M.; Skubitz, A.P. Cell membrane glycosylation mediates the adhesion, migration, and invasion of ovarian carcinoma cells. Clin. Exp. Metastasis 2003, 20, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Catterall, J.B.; Jones, L.M.; Turner, G.A. Membrane protein glycosylation and CD44 content in the adhesion of human ovarian cancer cells to hyaluronan. Clin. Exp. Metastasis 1999, 17, 583–591. [Google Scholar] [CrossRef] [PubMed]
- Goupille, C.; Marionneau, S.; Bureau, V.; Hallouin, F.; Meichenin, M.; Rocher, J.; le Pendu, J. Alpha1,2Fucosyltransferase increases resistance to apoptosis of rat colon carcinoma cells. Glycobiology 2000, 10, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Yan, L.; Lin, B.; Gao, J.; Liang, X.; Wang, Y.; Liu, J.; Zhang, S.; Iwamori, M. Enhancive effects of Lewis y antigen on CD44-mediated adhesion and spreading of human ovarian cancer cell line RMG-I. J. Exp. Clin. Cancer Res. 2011. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Gao, J.; Zhang, D.; Liu, Q.; Yan, L.; Gao, L.; Liu, J.; Liu, D.; Zhang, S.; Lin, B. High expression of Lewis y antigen and CD44 is correlated with resistance to chemotherapy in epithelial ovarian cancers. PLoS ONE 2013. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.C.; Gao, J.; Hu, Z.H.; Schwab, C.L.; Zhuang, H.Y.; Tan, M.Z.; Yan, L.M.; Liu, J.J.; Zhang, D.Y.; Lin, B. Membranous expressions of Lewis y and CAM-DR-related markers are independent factors of chemotherapy resistance and poor prognosis in epithelial ovarian cancer. Am. J. Cancer Res. 2015, 5, 830–843. [Google Scholar] [PubMed]
- Rahman, M.; Deleyrolle, L.; Vedam-Mai, V.; Azari, H.; Abd-El-Barr, M.; Reynolds, B.A. The cancer stem cell hypothesis: Failures and pitfalls. Neurosurgery 2011, 68, 531–545. [Google Scholar] [CrossRef] [PubMed]
- Zoller, M. CD44: Can a cancer-initiating cell profit from an abundantly expressed molecule? Nat. Rev. Cancer 2011, 11, 254–267. [Google Scholar] [CrossRef] [PubMed]
- Marquez, R.T.; Baggerly, K.A.; Patterson, A.P.; Liu, J.; Broaddus, R.; Frumovitz, M.; Atkinson, E.N.; Smith, D.I.; Hartmann, L.; Fishman, D.; et al. Patterns of gene expression in different histotypes of epithelial ovarian cancer correlate with those in normal fallopian tube, endometrium, and colon. Clin. Cancer Res. 2005, 11, 6116–6126. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C.; Yo, Y.T.; Lee, H.Y.; Liao, Y.P.; Chao, T.K.; Su, P.H.; Lai, H.C. ALDH1-bright epithelial ovarian cancer cells are associated with CD44 expression, drug resistance, and poor clinical outcome. Am. J. Pathol. 2012, 180, 1159–1169. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Balch, C.; Chan, M.W.; Lai, H.C.; Matei, D.; Schilder, J.M.; Yan, P.S.; Huang, T.H.; Nephew, K.P. Identification and characterization of ovarian cancer-initiating cells from primary human tumors. Cancer Res. 2008, 68, 4311–4320. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wang, J.; Chen, D.; Yang, J.; Yang, C.; Zhang, Y.; Zhang, H.; Dou, J. Evaluation of characteristics of CD44+CD117+ ovarian cancer stem cells in three dimensional basement membrane extract scaffold versus two dimensional monocultures. BMC Cell Biol. 2013, 14, 7. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wang, J.; Zhang, Y.; Chen, D.; Yang, C.; Kai, C.; Wang, X.; Shi, F.; Dou, J. Observation of ovarian cancer stem cell behavior and investigation of potential mechanisms of drug resistance in three-dimensional cell culture. J. Biosci. Bioeng. 2014, 118, 214–222. [Google Scholar] [CrossRef] [PubMed]
- He, Q.Z.; Luo, X.Z.; Wang, K.; Zhou, Q.; Ao, H.; Yang, Y.; Li, S.X.; Li, Y.; Zhu, H.T.; Duan, T. Isolation and characterization of cancer stem cells from high-grade serous ovarian carcinomas. Cell. Physiol. Biochem. 2014, 33, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.R.; Chen, Y.; Gao, Y.; Niu, X.L.; Li, Y.J.; Deng, W.M. Effects and mechanisms of anti-CD44 monoclonal antibody A3D8 on proliferation and apoptosis of sphere-forming cells with stemness from human ovarian cancer. Int. J. Gynecol. Cancer 2013, 23, 1367–1375. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Liu, T.; Wan, X.; Gao, Y.; Wang, H. MicroRNA-199a targets CD44 to suppress the tumorigenicity and multidrug resistance of ovarian cancer-initiating cells. FEBS J. 2012, 279, 2047–2059. [Google Scholar] [CrossRef] [PubMed]
- Gardner, M.J.; Catterall, J.B.; Jones, L.M.; Turner, G.A. Human ovarian tumour cells can bind hyaluronic acid via membrane CD44: A possible step in peritoneal metastasis. Clin. Exp. Metastasis 1996, 14, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Lessan, K.; Aguiar, D.J.; Oegema, T.; Siebenson, L.; Skubitz, A.P. CD44 and beta1 integrin mediate ovarian carcinoma cell adhesion to peritoneal mesothelial cells. Am. J. Pathol. 1999, 154, 1525–1537. [Google Scholar] [CrossRef]
- Ween, M.P.; Hummitzsch, K.; Rodgers, R.J.; Oehler, M.K.; Ricciardelli, C. Versican induces a pro-metastatic ovarian cancer cell behavior which can be inhibited by small hyaluronan oligosaccharides. Clin. Exp. Metastasis 2011, 28, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Desjardins, M.; Xie, J.; Gurler, H.; Muralidhar, G.G.; Sacks, J.D.; Burdette, J.E.; Barbolina, M.V. Versican regulates metastasis of epithelial ovarian carcinoma cells and spheroids. J. Ovarian Res. 2014. [Google Scholar] [CrossRef] [PubMed]
- Haria, D.; Trinh, B.Q.; Ko, S.Y.; Barengo, N.; Liu, J.; Naora, H. The homeoprotein DLX4 stimulates NF-κB activation and CD44-mediated tumor-mesothelial cell interactions in ovarian cancer. Am. J. Pathol. 2015, 185, 2298–2308. [Google Scholar] [CrossRef] [PubMed]
- Henry, J.C.; Park, J.K.; Jiang, J.; Kim, J.H.; Nagorney, D.M.; Roberts, L.R.; Banerjee, S.; Schmittgen, T.D. MiR-199a-3p targets CD44 and reduces proliferation of CD44 positive hepatocellular carcinoma cell lines. Biochem. Biophys. Res. Commun. 2010, 403, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Feng, Y.; Shen, J.K.; Lin, M.; Choy, E.; Cote, G.M.; Harmon, D.C.; Mankin, H.J.; Hornicek, F.J.; Duan, Z. CD44 is a direct target of miR-199a-3p and contributes to aggressive progression in osteosarcoma. Sci. Rep. 2015. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Kelnar, K.; Liu, B.; Chen, X.; Calhoun-Davis, T.; Li, H.; Patrawala, L.; Yan, H.; Jeter, C.; Honorio, S.; et al. The microRNA miR-34a inhibits prostate cancer stem cells and metastasis by directly repressing CD44. Nat. Med. 2011, 17, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Saini, S.; Majid, S.; Shahryari, V.; Arora, S.; Yamamura, S.; Chang, I.; Zaman, M.S.; Deng, G.; Tanaka, Y.; Dahiya, R. MiRNA-708 control of CD44+ prostate cancer-initiating cells. Cancer Res. 2012, 72, 3618–3630. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Wang, J.; Qian, J.; Kong, X.; Tang, J.; Wang, Y.; Chen, H.; Hong, J.; Zou, W.; Chen, Y.; et al. Long noncoding RNA gaplinc regulates CD44-dependent cell invasiveness and associates with poor prognosis of gastric cancer. Cancer Res. 2014, 74, 6890–6902. [Google Scholar] [CrossRef] [PubMed]
- Ishimoto, T.; Sugihara, H.; Watanabe, M.; Sawayama, H.; Iwatsuki, M.; Baba, Y.; Okabe, H.; Hidaka, K.; Yokoyama, N.; Miyake, K.; et al. Macrophage-derived reactive oxygen species suppress miR-328 targeting CD44 in cancer cells and promote redox adaptation. Carcinogenesis 2014, 35, 1003–1011. [Google Scholar] [CrossRef] [PubMed]
- Orian-Rousseau, V. CD44 acts as a signaling platform controlling tumor progression and metastasis. Front. Immunol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Misra, S.; Heldin, P.; Hascall, V.C.; Karamanos, N.K.; Skandalis, S.S.; Markwald, R.R.; Ghatak, S. Hyaluronan-CD44 interactions as potential targets for cancer therapy. FEBS J. 2011, 278, 1429–1443. [Google Scholar] [CrossRef] [PubMed]
- Orian-Rousseau, V.; Sleeman, J. CD44 is a multidomain signaling platform that integrates extracellular matrix cues with growth factor and cytokine signals. Adv. Cancer Res. 2014, 123, 231–254. [Google Scholar] [PubMed]
- Yu, Q.; Toole, B.P.; Stamenkovic, I. Induction of apoptosis of metastatic mammary carcinoma cells in vivo by disruption of tumor cell surface CD44 function. J. Exp. Med. 1997, 186, 1985–1996. [Google Scholar] [CrossRef] [PubMed]
- Ghatak, S.; Misra, S.; Toole, B.P. Hyaluronan oligosaccharides inhibit anchorage-independent growth of tumor cells by suppressing the phosphoinositide 3-kinase/Akt cell survival pathway. J. Biol. Chem. 2002, 277, 38013–38020. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Hao, X.; Qin, J.; Tang, W.; He, F.; Smith, A.; Zhang, M.; Simeone, D.M.; Qiao, X.T.; Chen, Z.N.; et al. Antibody against CD44s inhibits pancreatic tumor initiation and postradiation recurrence in mice. Gastroenterology 2014, 146, 1108–1118. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Hope, K.J.; Zhai, Q.; Smadja-Joffe, F.; Dick, J.E. Targeting of CD44 eradicates human acute myeloid leukemic stem cells. Nat. Med. 2006, 12, 1167–1174. [Google Scholar] [CrossRef] [PubMed]
- Heider, K.H.; Sproll, M.; Susani, S.; Patzelt, E.; Beaumier, P.; Ostermann, E.; Ahorn, H.; Adolf, G.R. Characterization of a high-affinity monoclonal antibody specific for CD44v6 as candidate for immunotherapy of squamous cell carcinomas. Cancer Immunol. Immunother. 1996, 43, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Iida, J.; Clancy, R.; Dorchak, J.; Somiari, R.I.; Somiari, S.; Cutler, M.L.; Mural, R.J.; Shriver, C.D. DNA aptamers against exon v10 of CD44 inhibit breast cancer cell migration. PLoS ONE 2014, 9, e88712. [Google Scholar] [CrossRef] [PubMed]
- Tremmel, M.; Matzke, A.; Albrecht, I.; Laib, A.M.; Olaku, V.; Ballmer-Hofer, K.; Christofori, G.; Heroult, M.; Augustin, H.G.; Ponta, H.; et al. A CD44v6 peptide reveals a role of CD44 in VEGFR-2 signaling and angiogenesis. Blood 2009, 114, 5236–5244. [Google Scholar] [CrossRef] [PubMed]
- Shah, V.; Taratula, O.; Garbuzenko, O.B.; Taratula, O.R.; Rodriguez-Rodriguez, L.; Minko, T. Targeted nanomedicine for suppression of CD44 and simultaneous cell death induction in ovarian cancer: An optimal delivery of siRNA and anticancer drug. Clin. Cancer Res. 2013, 19, 6193–6204. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Song, X.; Yi, T.; Li, S.; Deng, H.; Chen, X.; Li, Z.; Bai, Y.; Zhong, Q.; Wei, Y.; et al. Administration of PLGA nanoparticles carrying shRNA against focal adhesion kinase and CD44 results in enhanced antitumor effects against ovarian cancer. Cancer Gene Ther. 2013, 20, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Nakshatri, H.; Srour, E.F.; Badve, S. Breast cancer stem cells and intrinsic subtypes: Controversies rage on. Curr. Stem Cell Res. Ther. 2009, 4, 50–60. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sacks, J.D.; Barbolina, M.V. Expression and Function of CD44 in Epithelial Ovarian Carcinoma. Biomolecules 2015, 5, 3051-3066. https://doi.org/10.3390/biom5043051
Sacks JD, Barbolina MV. Expression and Function of CD44 in Epithelial Ovarian Carcinoma. Biomolecules. 2015; 5(4):3051-3066. https://doi.org/10.3390/biom5043051
Chicago/Turabian StyleSacks, Joelle D., and Maria V. Barbolina. 2015. "Expression and Function of CD44 in Epithelial Ovarian Carcinoma" Biomolecules 5, no. 4: 3051-3066. https://doi.org/10.3390/biom5043051
APA StyleSacks, J. D., & Barbolina, M. V. (2015). Expression and Function of CD44 in Epithelial Ovarian Carcinoma. Biomolecules, 5(4), 3051-3066. https://doi.org/10.3390/biom5043051





