Differentiating Hepatocellular Carcinoma from Hepatitis C Using Metabolite Profiling
Abstract
:1. Introduction
2. Experimental Methods
2.1. Chemicals
2.2. Serum Sample Collection and Storage
| HCC (Hepatocellular carcinoma) | HCV (Hepatitis C Virus) | |
|---|---|---|
| Samples | 40 | 22 |
| Average Age | 54.6 ± 9.8 | 52.2 ± 8.1 |
| Gender (F/M) | 0.21 | 0.46 |
| Ethnicity | ||
| Caucasian | 32 | 20 |
| African American | 1 | 2 |
| Hispanic | 3 | 0 |
| Unknown | 4 | 0 |
2.3. Sample Preparation and Acquisition of NMR Spectra
2.4. Statistical Analysis
3. Results
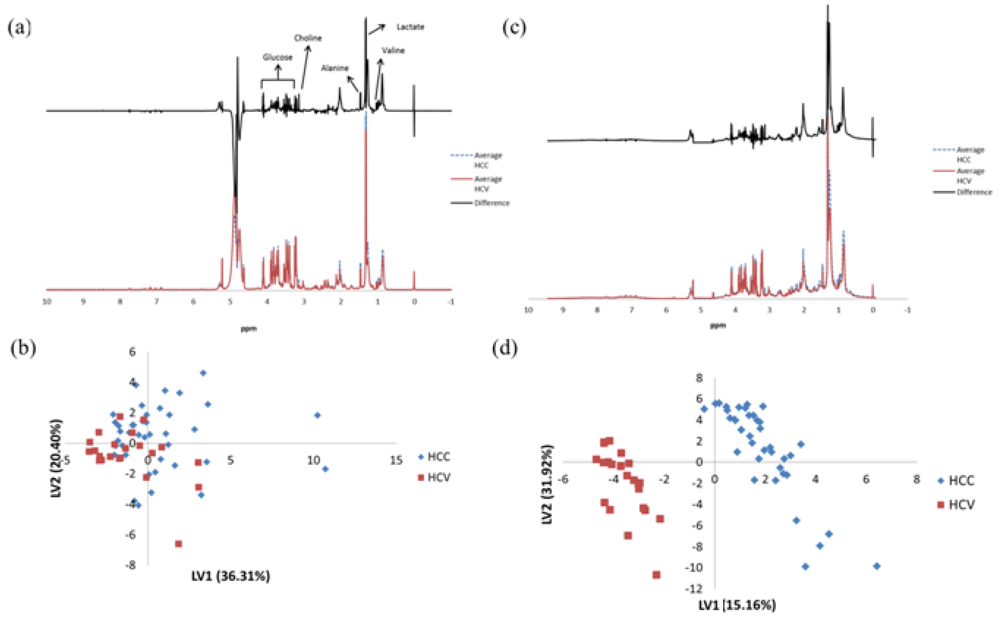
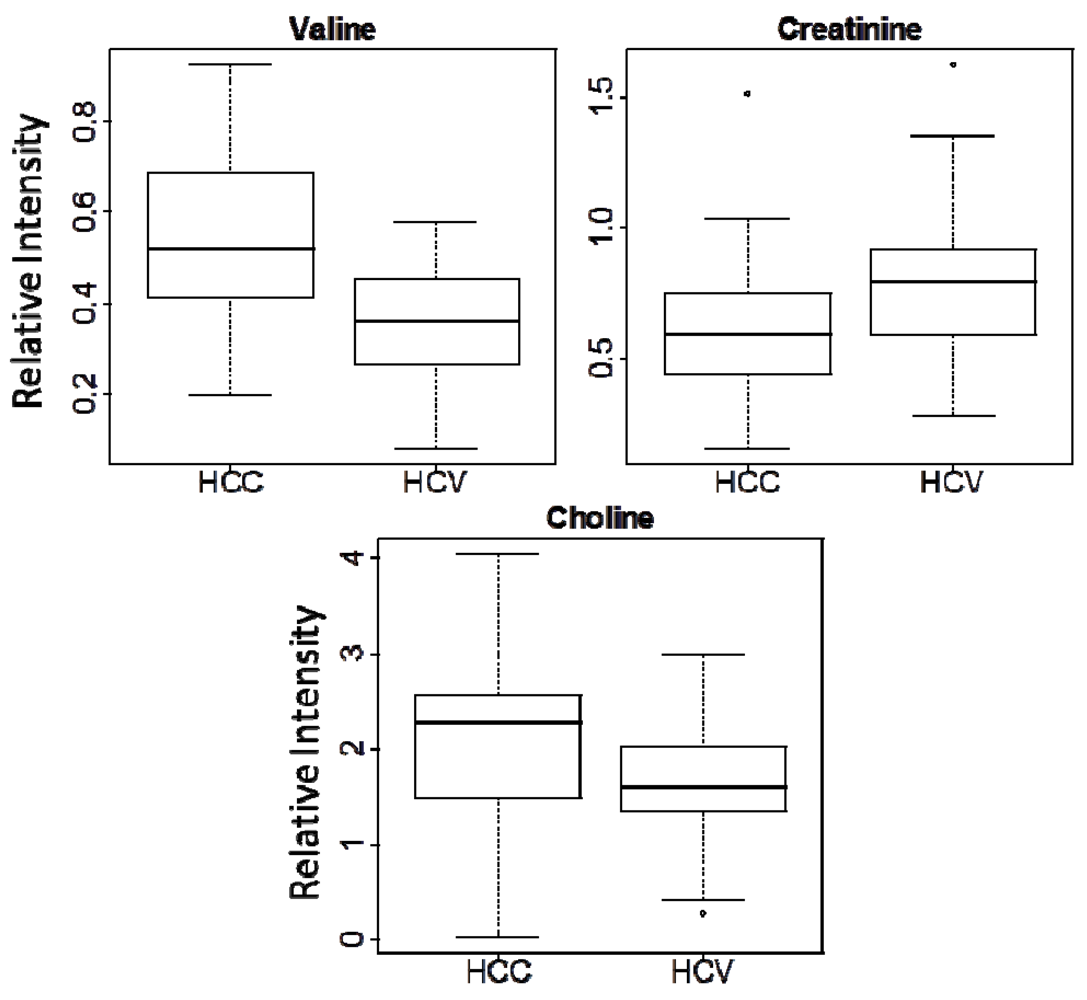
| Metabolite | Chemical Shift (ppm) | Multiplicity | p-valuea | Fold changeb |
|---|---|---|---|---|
| (HCC vs. HCV) | ||||
| Choline | 3.20 | s | 0.0200 | 1.32 |
| Valine | 1.03 | d | 5.67 × 10−6 | 1.53 |
| Creatinine | 3.03 | s | 0.0279 | -1.28 |
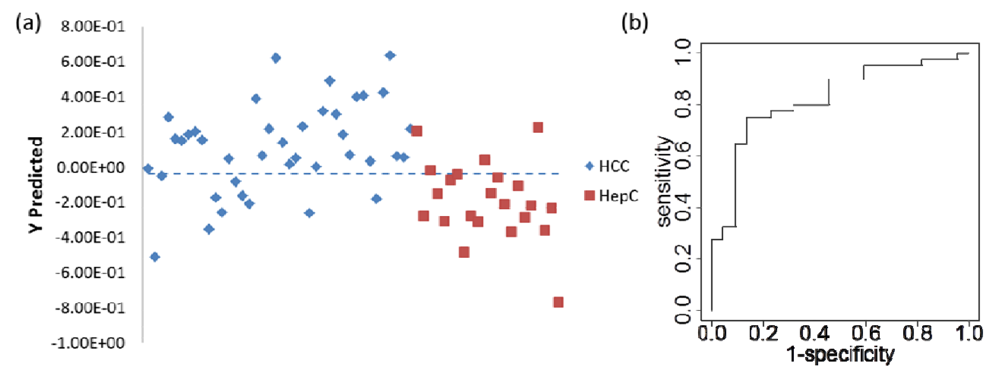
| Predicted class | |||
|---|---|---|---|
| True class | Total number of samples | HCC | HCV |
| HCC | 8000 (8000) | 5674 (4349) | 2326 (3651) |
| HCV | 4400 (4400) | 1195 (2735) | 3205 (1665) |
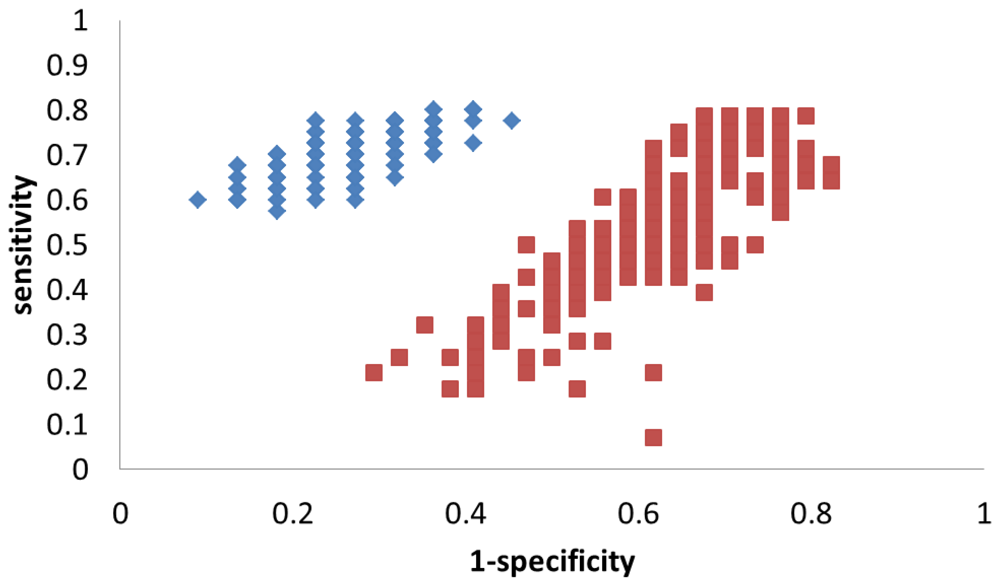
4. Discussion
5. Conclusions
Conflict of Interest
Acknowledgments
References
- Shariff, M.I.; Cox, I.J.; Gomaa, A.I.; Khan, S.A.; Gedroyc, W.; Taylor-Robinson, S.D. Hepatocellular carcinoma: Current trends in worldwide epidemiology, risk factors, diagnosis and therapeutics. Expert Rev. Gastroenterol. Hepatol. 2009, 3, 353–367. [Google Scholar] [CrossRef]
- El-Serag, H.B. Current concepts hepatocellular carcinoma. N. Engl. J. Med. 2011, 365, 1118–1127. [Google Scholar] [CrossRef]
- El-Serag, H.B.; Marrero, J.A.; Rudolph, L.; Reddy, K.R. Diagnosis and treatment of hepatocellular carcinoma. Gastroenterology 2008, 134, 1752–1763. [Google Scholar]
- Tang, Z.Y.; Yu, Y.Q.; Zhou, X.D. An important approach to prolonging survival further after radical resection of AFP-positive hepatocellular-carcinoma. J. Exp. Clin. Cancer Res. 1984, 3, 359–366. [Google Scholar]
- Donato, F.; Tagger, A.; Gelatti, U.; Parrinello, G.; Boffetta, P.; Albertini, A.; Decarli, A.; Trevisi, P.; Ribero, M.L.; Martelli, C.; et al. Alcohol and hepatocellular carcinoma: The effect of lifetime intake and hepatitis virus infections in men and women. Am. J. Epidemiol. 2002, 155, 323–331. [Google Scholar] [CrossRef]
- Kew, M.C.; Purves, L.R.; Bersohn, I. Serum alpha-fetoprotein levels in acute viral-hepatitis. Gut 1973, 14, 939–942. [Google Scholar] [CrossRef]
- Tong, M.J.; Elfarra, N.S.; Reikes, A.R.; Co, R.L. Clinical outcomes after transfusion-associated hepatitis-C. N. Engl. J. Med. 1995, 332, 1463–1466. [Google Scholar] [CrossRef]
- Bayati, N.; Silverman, A.L.; Gordon, S.C. Serum alpha-fetoprotein levels and liver histology in patients with chronic hepatitis C. Am. J. Gastroenterol. 1998, 93, 2452–2456. [Google Scholar] [CrossRef]
- Hu, K.Q.; Kyulo, N.L.; Lim, N.; Elhazin, B.; Hillebrand, D.J.; Bock, T. Clinical significance of elevated alpha-fetoprotein (AFP) in patients with chronic hepatitis c, but not hepatocellular carcinoma. Am. J. Gastroenterol. 2004, 99, 860–865. [Google Scholar] [CrossRef]
- Gomaa, A.I.; Khan, S.A.; Leen, E.L.S.; Waked, I.; Taylor-Robinson, S.D. Diagnosis of hepatocellular carcinoma. World J. Gastroenterol. 2009, 15, 1301–1314. [Google Scholar] [CrossRef]
- Marrero, J.A.; Feng, Z.D.; Wang, Y.H.; Nguyen, M.H.; Befeler, A.S.; Roberts, L.R.; Reddy, K.R.; Harnois, D.; Llovet, J.M.; Normolle, D.; et al. Alpha-fetoprotein, des-gamma carboxyprothrombin, and lectin-bound alpha-fetoprotein in early hepatocellular carcinoma. Gastroenterology 2009, 137, 110–118. [Google Scholar]
- Mita, Y.; Aoyagi, Y.; Yanagi, M.; Suda, T.; Suzuki, Y.; Asakura, H. The usefulness of determining des-gamma-carboxy prothrombin by sensitive enzyme immunoassay in the early diagnosis of patients with hepatocellular carcinoma. Cancer 1998, 82, 1643–1648. [Google Scholar] [CrossRef]
- Hsieh, S.Y.; He, J.R.; Yu, M.C.; Lee, W.C.; Chen, T.C.; Lo, S.J.; Bera, R.; Sung, C.M.; Chiu, C.T. Secreted RRBB3 isoforms are serum markers for early hepatoma in patients with chronic hepatitis and cirrhosis. J. Proteome Res. 2011, 10, 4715–4724. [Google Scholar] [CrossRef]
- Kumada, T.; Toyoda, H.; Kiriyama, S.; Tanikawa, M.; Hisanaga, Y.; Kanamori, A.; Tada, T.; Tanaka, J.; Yoshizawa, H. Predictive value of tumor markers for hepatocarcinogenesis in patients with hepatitis C virus. J. Gastroenterol. 2011, 46, 536–544. [Google Scholar] [CrossRef]
- Kaddurah-Daouk, R.; Kristal, B.S.; Weinshilboum, R.M. Metabolomics: A global biochemical approach to drug response and disease. Annu. Rev. Pharmacol. 2008, 48, 653–683. [Google Scholar] [CrossRef]
- Gowda, G.A.N.; Zhang, S.C.; Gu, H.W.; Asiago, V.; Shanaiah, N.; Raftery, D. Metabolomics-based methods for early disease diagnostics. Expert Rev. Mol. Diagn. 2008, 8, 617–633. [Google Scholar] [CrossRef]
- Gibney, M.J.; Walsh, M.; Brennan, L.; Roche, H.M.; German, B.; van Ommen, B. Metabolomics in human nutrition: Opportunities and challenges. Am. J. Clin. Nutr. 2005, 82, 497–503. [Google Scholar]
- Griffin, J.L. Metabonomics: NMR spectroscopy and pattern recognition analysis of body fluids and tissues for characterisation of xenobiotic toxicity and disease diagnosis. Curr. Opin. Chem. Biol. 2003, 7, 648–654. [Google Scholar] [CrossRef]
- Rocha, C.M.; Carrola, J.; Barros, A.S.; Gil, A.M.; Goodfellow, B.J.; Carreira, I.M.; Bernardo, J.; Gomes, A.; Sousa, V.; Carvalho, L.; et al. Metabolic signatures of lung cancer in biofluids: Nmr-based metabonomics of blood plasma. J. Proteome Res. 2011, 10, 4314–4324. [Google Scholar]
- Carrola, J.; Rocha, C.M.; Barros, A.S.; Gil, A.M.; Goodfellow, B.J.; Carreira, I.M.; Bernardo, J.; Gomes, A.; Sousa, V.; Carvalho, L.; et al. Metabolic signatures of lung cancer in biofluids: Nmr-based metabonomics of urine. J. Proteome Res. 2011, 10, 221–230. [Google Scholar] [CrossRef]
- Sreekumar, A.; Poisson, L.M.; Rajendiran, T.M.; Khan, A.P.; Cao, Q.; Yu, J.; Laxman, B.; Mehra, R.; Lonigro, R.J.; Li, Y.; et al. Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature 2009, 457, 910–914. [Google Scholar]
- Chan, E.C.Y.; Koh, P.K.; Mal, M.; Cheah, P.Y.; Eu, K.W.; Backshall, A.; Cavill, R.; Nicholson, J.K.; Keun, H.C. Metabolic profiling of human colorectal cancer using high-resolution magic angle spinning nuclear magnetic resonance (HR-MAS NMR) spectroscopy and gas chromatography mass spectrometry (GS/MS). J. Proteome Res. 2009, 8, 352–361. [Google Scholar] [CrossRef]
- Slupsky, C.M.; Steed, H.; Wells, T.H.; Dabbs, K.; Schepansky, A.; Capstick, V.; Faught, W.; Sawyer, M.B. Urine metabolite analysis offers potential early diagnosis of ovarian and breast cancers. Clin. Cancer Res. 2010, 16, 5835–5841. [Google Scholar] [CrossRef]
- Asiago, V.M.; Alvarado, L.Z.; Shanaiah, N.; Gowda, G.A.N.; Owusu-Sarfo, K.; Ballas, R.A.; Raftery, D. Early detection of recurrent breast cancer using metabolite profiling. Cancer Res. 2010, 70, 8309–8318. [Google Scholar]
- Zhang, J.; Liu, L..; Wei, S.; Gowda, G.A.N.; Hammoud, Z.; Kesler, K.A.; Raftery, D. Metabolomics study of esophageal adenocarcinoma. J. Thorac. Cardiovasc. Surg. 2011, 141, 469–475. [Google Scholar] [CrossRef]
- Zhang, J.; Bowers, J.; Liu, L.; Wei, S.; Gowda, G.A.N.; Hammoud, Z.; Raftery, D. Esophageal cancer metabolite biomarkers detected by LC-MS and NMR methods. PloS One 2012, 7, e30181. [Google Scholar]
- Yang, Y.X.; Li, C.L.; Nie, X.; Feng, X.S.; Chen, W.X.; Yue, Y.; Tang, H.R.; Deng, F. Metabonomic studies of human hepatocellular carcinoma using high-resolution magic-angle spinning 1H NMR spectroscopy in conjunction with multivariate data analysis. J. Proteome Res. 2007, 6, 2605–2614. [Google Scholar] [CrossRef]
- Shariff, M.I.F.; Ladep, N.G.; Cox, I.J.; Williams, H.R.T.; Okeke, E.; Malu, A.; Thillainayagam, A.V.; Crossey, M.M.E.; Khan, S.A.; Thomas, H.C.; et al. Characterization of urinary biomarkers of hepatocellular carcinoma using magnetic resonance spectroscopy in a Nigerian population. J. Proteome Res. 2010, 9, 1096–1103. [Google Scholar] [CrossRef]
- Yang, J.; Xu, G.W.; Zheng, Y.F.; Kong, H.W.; Pang, T.; Lv, S.; Yang, Q. Diagnosis of liver cancer using hplc-based metabonomics avoiding false-positive result from hepatitis and hepatocirrhosis diseases. J. Chromatogr. B 2004, 813, 59–65. [Google Scholar] [CrossRef]
- Wu, H.; Xue, R.Y.; Dong, L.; Liu, T.T.; Deng, C.H.; Zeng, H.Z.; Shen, X.Z. Metabolomic profiling of human urine in hepatocellular carcinoma patients using gas chromatography/mass spectrometry. Anal. Chim. Acta 2009, 648, 98–104. [Google Scholar] [CrossRef]
- Chen, F.; Xue, J.H.; Zhou, L.F.; Wu, S.S.; Chen, Z. Identification of serum biomarkers of hepatocarcinoma through liquid chromatography/mass spectrometry-based metabonomic method. Anal. Bioanal. Chem. 2011, 401, 1899–1904. [Google Scholar] [CrossRef]
- Patterson, A.D.; Maurhofer, O.; Beyoglu, D.; Lanz, C.; Krausz, K.W.; Pabst, T.; Gonzalez, F.J.; Dufour, J.F.; Idle, J.R. Aberrant lipid metabolism in hepatocellular carcinoma revealed by plasma metabolomics and lipid profiling. Cancer Res. 2011, 71, 6590–6600. [Google Scholar]
- Chen, T.L.; Xie, G.X.; Wang, X.Y.; Fan, J.; Qui, Y.P.; Zheng, X.J.; Qi, X.; Cao, Y.; Su, M.M.; Wang, X.Y.; et al. Serum and urine metabolite profiling reveals potential biomarkers of human hepatocellular carcinoma. Mol. Cell. Proteomics 2011, 10. 110.004945. [Google Scholar]
- Human Metabolome Database. Available online: www.hmdb.ca 30 September 2012.
- Gupta, S.; Bent, S.; Kohlwes, J. Test characteristics of alpha-fetoprotein for detecting hepatocellular carcinoma in patients with hepatitis C—A systematic review and critical analysis. Ann. Intern. Med. 2003, 139, 46–50. [Google Scholar]
- El-Serag, H.B.; Kramer, J.R.; Chen, G.J.; Duan, Z.G.; Richardson, P.A.; Davila, J.A. Effectiveness of afp and ultrasound tests on hepatocellular carcinoma mortality in hcv-infected patients in the USA. Gut 2011, 60, 992–997. [Google Scholar] [CrossRef]
- Chen, J.; Wang, W.Z.; Lv, S.; Yin, P.Y.; Zhao, X.J.; Lu, X.; Zhang, F.X.; Xu, G.W. Metabonomics study of liver cancer based on ultra performance liquid chromatography coupled to mass spectrometry with hilic and rplc separations. Anal. Chim. Acta 2009, 650, 3–9. [Google Scholar] [CrossRef]
- OuYang, D.; Xu, J.J.; Huang, H.G.; Chen, Z. Metabolomic profiling of serum from human pancreatic cancer patients using 1H NMR spectroscopy and principal component analysis. Appl. Biochem. Biotechnol. 2011, 165, 148–154. [Google Scholar] [CrossRef]
- Cheng, Y.; Xie, G.; Chen, T.; Qiu, Y.; Zou, X.; Zheng, M.; Tan, B.; Feng, B.; Dong, T.; He, P.; et al. Distinct urinary metabolic profile of human colorectal cancer. J. Proteome Res. 2012, 11, 1354–1363. [Google Scholar] [CrossRef]
- Hultman, E.; Soderlund, K.; Timmons, J.A.; Cederblad, G.; Greenhaff, P.L. Muscle creatine loading in men. J. Appl. Physiol. 1996, 81, 232–237. [Google Scholar]
- Xue, R.Y.; Lin, Z.X.; Deng, C.H.; Dong, L.; Liu, T.T.; Wang, J.Y.; Shen, X.Z. A serum metabolomic investigation on hepatocellular carcinoma patients by chemical derivatization followed by gas chromatography/mass spectrometry. Rapid Commun. Mass Spectrom. 2008, 22, 3061–3068. [Google Scholar]
- Xue, R.Y.; Dong, L.; Wu, H.; Liu, T.T.; Wang, J.Y.; Shen, X.Z. Gas chromatography/mass spectrometry screening of serum metabolomic biomarkers in hepatitis B virus infected cirrhosis patients. Clin. Chem. Lab. Med. 2009, 47, 305–310. [Google Scholar]
- Taniguchi, K.; Nonami, T.; Nakao, A.; Harada, A.; Kurokawa, T.; Sugiyama, S.; Fujitsuka, N.; Shimomura, Y.; Hutson, S.M.; Harris, R.A.; et al. The valine catabolic pathway in human liver: Effect of cirrhosis on enzyme activities. Hepatology 1996, 24, 1395–1398. [Google Scholar] [CrossRef]
- Taniguchi, K.; Nonami, T.; Takagi, H.; Shimomura, Y.; Fujitsuka, N.; Harris, R.A. Enzyme-activity in the valine catabolic pathway of human liver. Faseb J. 1995, 9, A1319–A1319. [Google Scholar]
- Ishigure, K.; Shimomura, Y.; Murakami, T.; Kaneko, T.; Takeda, S.; Inoue, S.; Nomoto, S.; Koshikawa, K.; Nonami, T.; Nakao, A. Human liver disease decreases methacrylyl-coa hydratase and beta-hydroxyisobutyryl-coa hydrolase activities in valine catabolism. Clin. Chim. Acta 2001, 312, 115–121. [Google Scholar] [CrossRef]
- Tiziani, S.; Lopes, V.; Gunther, U.L. Early stage diagnosis of oral cancer using 1H NMR-based metabolomics. Neoplasia 2009, 11, 269–276. [Google Scholar]
- Wu, H.; Xue, R.Y.; Tang, Z.Q.; Deng, C.H.; Liu, T.T.; Zeng, H.Z.; Sun, Y.H.; Shen, X.Z. Metabolomic investigation of gastric cancer tissue using gas chromatography/mass spectrometry. Anal. Bioanal. Chem. 2010, 396, 1385–1395. [Google Scholar] [CrossRef]
- Gao, H.C.; Lu, Q.; Liu, X.; Cong, H.; Zhao, L.C.; Wang, H.M.; Lin, D.H. Application of 1H NMR-based metabonomics in the study of metabolic profiling of human hepatocellular carcinoma and liver cirrhosis. Cancer Sci. 2009, 100, 782–785. [Google Scholar] [CrossRef]
- Kuo, Y.T.; Li, C.W.; Chen, C.Y.; Jao, J.C.; Wu, D.K.; Liu, G.C. In vivo proton magnetic resonance spectroscopy of large focal hepatic lesions and metabolite change of hepatocellular carcinoma before and after transcatheter arterial chemoembolization using 3.0-T MR scanner. J. Magn. Reson. Imaging 2004, 19, 598–604. [Google Scholar]
- Michel, V.; Yuan, Z.F.; Ramsubir, S.; Bakovic, M. Choline transport for phospholipid synthesis. Exp. Biol. Med. 2006, 231, 490–504. [Google Scholar]
- Daly, P.F.; Lyon, R.C.; Faustino, P.J.; Cohen, J.S. Phospholipid metabolism in cancer cells monitored by 31p nmr spectroscopy. J. Biol. Chem. 1987, 262, 14875–14878. [Google Scholar]
- Miller, B.L. A review of chemical issues in 1H NMR spectroscopy: N-acetyl-l-aspartate, creatine and choline. NMR Biomed. 1991, 4, 47–52. [Google Scholar] [CrossRef]
- Kuesel, A.C.; Graschew, G.; Hull, W.E.; Lorenz, W.; Thielmann, H.W. 31P NMR studies of cultured human tumor cells. Influence of pH on phospholipid metabolite levels and the detection of cytidine 5'-diphosphate choline. NMR Biomed. 1990, 3, 78–89. [Google Scholar]
- Chen, W.X.; Zhou, X.Y.; Huang, D.; Chen, F.; Du, X. Metabolic profiling of human colorectal cancer using high resolution 1H nuclear magnetic resonance spectroscopy. Chin. J. Chem. 2011, 29, 2511–2519. [Google Scholar]
- Bianchi, L.; De Micheli, E.; Bricolo, A.; Ballini, C.; Fattori, M.; Venturi, C.; Pedata, F.; Tipton, K.F.; Della Corte, L. Extracellular levels of amino acids and choline in human high grade gliomas: An intraoperative microdialysis study. Neurochem. Res. 2004, 29, 325–334. [Google Scholar] [CrossRef]
- Katz-Brull, R.; Seger, D.; Rivenson-Segal, D.; Rushkin, E.; Degani, H. Metabolic markers of breast cancer: Enhanced choline metabolism and reduced choline-ether-phospholipid synthesis. Cancer Res. 2002, 62, 1966–1970. [Google Scholar]
Supplementary Files
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wei, S.; Suryani, Y.; Gowda, G.A.N.; Skill, N.; Maluccio, M.; Raftery, D. Differentiating Hepatocellular Carcinoma from Hepatitis C Using Metabolite Profiling. Metabolites 2012, 2, 701-716. https://doi.org/10.3390/metabo2040701
Wei S, Suryani Y, Gowda GAN, Skill N, Maluccio M, Raftery D. Differentiating Hepatocellular Carcinoma from Hepatitis C Using Metabolite Profiling. Metabolites. 2012; 2(4):701-716. https://doi.org/10.3390/metabo2040701
Chicago/Turabian StyleWei, Siwei, Yuliana Suryani, G. A. Nagana Gowda, Nicholas Skill, Mary Maluccio, and Daniel Raftery. 2012. "Differentiating Hepatocellular Carcinoma from Hepatitis C Using Metabolite Profiling" Metabolites 2, no. 4: 701-716. https://doi.org/10.3390/metabo2040701





