Food Antioxidants and Their Anti-Inflammatory Properties: A Potential Role in Cardiovascular Diseases and Cancer Prevention
Abstract
:1. Introduction
2. Food Preservation, Oxidative Stress and Antioxidant Balance
3. Free Radicals, Oxidants, and Reductants
3.1. Free Radicals
3.2. Oxidants
3.3. Reductants
4. Examples of Free Radical Damage
4.1. Hydroxyl Radical (HO*)
4.2. Nitric Oxide Radical (NO*)
4.3. Superoxide Anion Radical (O2−*)
5. Naturally Occurring Flavonoids, Carotenoids and Pro-Oxidants
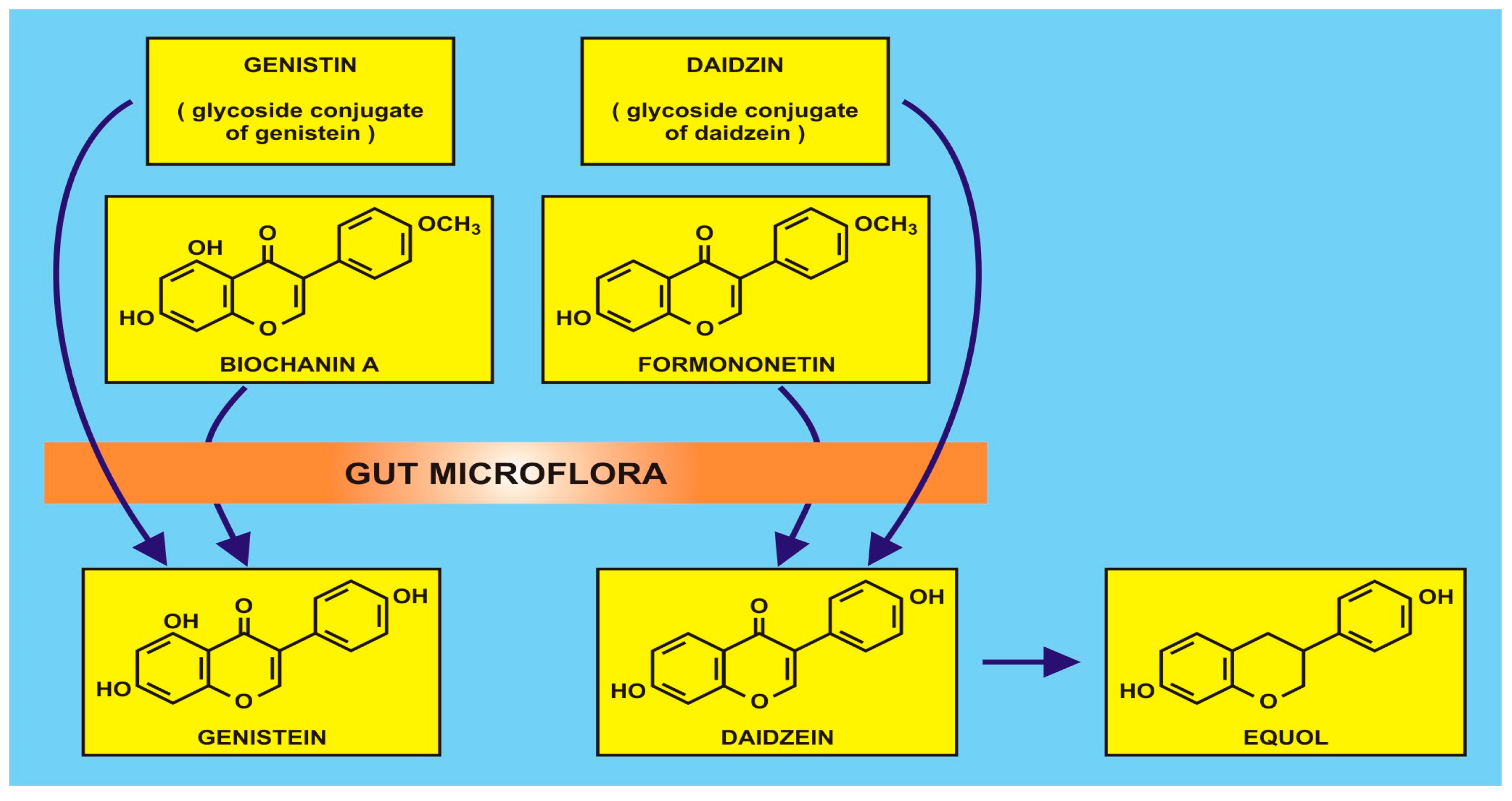
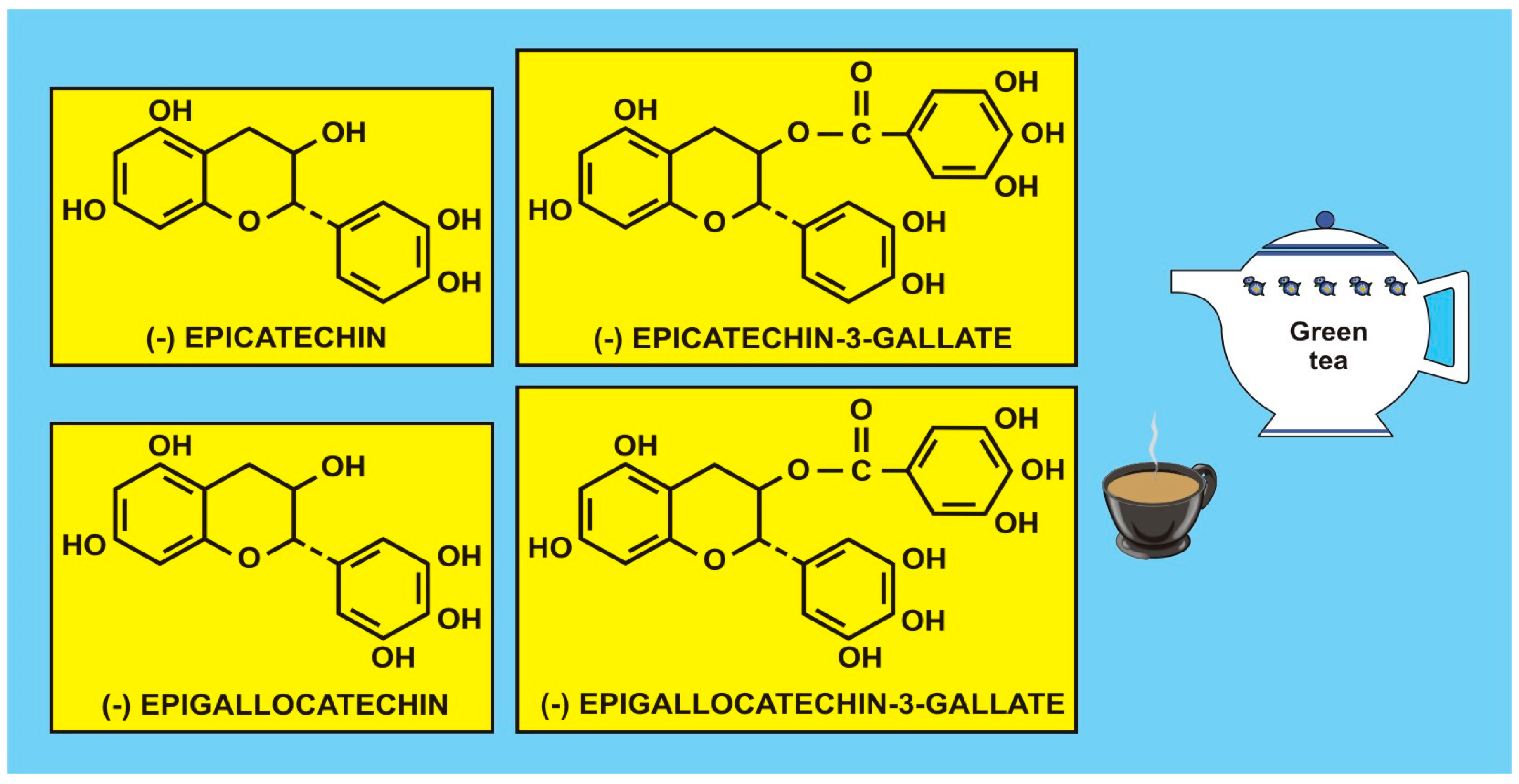
5.1. Flavonoids
5.2. Carotenoids
5.3. Pro-Oxidants
6. Spices, Aromatic Herbs, Antioxidants and Health
7. Conclusions
Acknowledgments
Conflicts of Interest
References
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary prevention of cardiovascular disease with a Mediterranean diet. N. Engl. J. Med. 2013, 368, 1279–1290. [Google Scholar] [CrossRef] [PubMed]
- Toledo, E.; Salas-Salvadó, J.; Donat-Vargas, C.; Buil-Cosiales, P.; Estruch, R.; Ros, E.; Corella, D.; Fitó, M.; Hu, F.B.; Arós, F.; et al. Mediterranean diet and invasive breast cancer risk among women at high cardiovascular risk in the PREDIMED trial: A randomized clinical trial. JAMA Intern. Med. 2015, 5, 1752–1760. [Google Scholar] [CrossRef]
- Salas-Salvadó, J.; Bulló, M.; Babio, N.; Martínez-González, M.Á.; Ibarrola-Jurado, N.; Basora, J.; Estruch, R.; Covas, M.I.; Corella, D.; Arós, F.; et al. Reduction in the incidence of type 2 diabetes with the Mediterranean diet. Diabetes Care 2011, 34, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.B.; Hristova, K.; Fedacko, J.; Singhal, S.; Khan, S.; Wilson, D.W.; Takahashi, T.; Sharma, Z. Antioxidant vitamins and oxidative stress in chronic heart failure. World Heart J. 2015, 7, 257–264. [Google Scholar]
- Rajoria, A.; Kumar, J.; Chauhan, A.K. Anti-oxidative and anti-carcinoginic role of lycopene in human health-A review. J. Dairy. Foods Home Sci. 2010, 29, 3–4. [Google Scholar]
- Ferre, G.M.; Guasch-Ferré, M.; Bulló, M.; Martínez-González, M.Á.; Ros, E.; Corella, D.; Estruch, R.; Fitó, M.; Arós, F.; Wärnberg, J.; et al. Frequency of nut consumption and mortality risk in the PREDIMED nutrition intervention trial. BMC Med. 2013, 11, 164. [Google Scholar] [CrossRef]
- Du, H.; Li, L.; Bennett, D.; Guo, Y.; Key, T.J.; Bian, Z.; Sherliker, P.; Gao, H.; Chen, Y.; Yang, L.; et al. Fresh fruit consumption and major cardiovascular disease in China. N. Engl. J. Med. 2016, 374, 1332–1343. [Google Scholar] [CrossRef] [PubMed]
- De Lorgeril, M.; Renaud, S.; Mamelle, N.; Salen, P.; Martin, J.L.; Monjaud, I.; Guidollet, J.; Touboul, P.; Delaye, J. Mediterranean alpha-linolenic acid-rich diet in secondary prevention of coronary heart disease. Lancet 1994, 343, 1454–1459, Erratum in: Lancet 1995, 345, 738. [Google Scholar] [CrossRef]
- Singh, R.B.; Dubnov, G.; Niaz, M.A.; Ghosh, S.; Singh, R.; Rastogi, S.S.; Manor, O.; Pella, D.; Berry, E.M. Effect of an Indo-Mediterranean diet on progression of coronary disease in high risk patients: a randomized single blind trial. Lancet 2002, 360, 1455–1461. [Google Scholar] [CrossRef]
- Singh, R.B.; Rastogi, S.S.; Verma, R.; Bolaki, L.; Singh, R.; Ghosh, S. An Indian experiment with nutritional modulation in acute myocardial infarction. Am. J. Cardiol. 1992, 69, 879–885. [Google Scholar] [CrossRef]
- Singh, R.B.; Fedacko, J.; Vargova, V.; Niaz, M.A.; Rastogi, S.S.; Ghosh, S. Effect of low W-6/W-3 ratio fatty acid Paleolithic style diet in patients with acute coronary syndromes. A randomized, single blind, controlled trial. World Heart J. 2012, 3, 71–84. [Google Scholar]
- Fitó, M.; Guxens, M.; Corella, D.; Sáez, G.; Estruch, R.; de la Torre, R.; Francés, F.; Cabezas, C.; López-Sabater Mdel, C.; Marrugat, J.; et al. Effect of a traditional Mediterranean diet on lipoprotein oxidation. JAMA Intern. Med. 2007, 167, 1195–1203. [Google Scholar] [CrossRef] [PubMed]
- Siti, H.N.; Kamisah, Y.; Kamsiah, J. The role of oxidative stress, antioxidants and vascular inflammation in cardiovascular disease (a review). Vasc. Pharmacol. 2015, 71, 40–56. [Google Scholar] [CrossRef] [PubMed]
- Guralp, O. Effects of vitamin E on bone remodeling in perimenopausal women: mini review. Maturitas 2014, 79, 476–480. [Google Scholar] [CrossRef] [PubMed]
- Francuz, T.; Czajka-Francuz, P.; Cison-Jurek, S.; Wojnar, J. The role of inflammation in colon cancer pathogenesis. Postep. Hig. Med. Swiadczalnej (Online) 2016, 70, 360–366. [Google Scholar] [CrossRef]
- Griffiths, K.; Denis, L.J.; Turkes, A. Oestrogens, Phyto-Oestrogens, and the Pathogenesis of Prostatic Disease; Martin Dunitz: London, UK, 2002; pp. 43–48. [Google Scholar]
- Ahuja, I.; Kissen, R.; Bones, A.M. Phytoalexins in defense against pathogens. Trends Plant Sci. 2012, 17, 73–90. [Google Scholar] [CrossRef] [PubMed]
- Kuntz, S.; Wenzel, U.; Daniel, H. Comparative analysis of the effects of flavonoids on proliferation, cytotoxicity, and apoptosis in human colon cancer cell lines. Eur. J. Nutr. 1999, 38, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Murtaza, I.; Adhami, V.M.; Hafeez, B.B.; Saleem, M.; Mukhtar, H. Fisetin, a natural flavonoid, targets chemoresistant human pancreatic cancer AsPC-1 cells through DR3-mediated inhibition of NF-kappa B. Int. J. Cancer 2009, 125, 2465–2473. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Tang, L.; Wang, J.-S. Biomarkers of dietary polyphenols in cancer studies: Current evidence and beyond. Oxidative Med. Cell. Longev. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Hwang, K.A.; Choi, K.C. Anticarcinogenic effects of dietary phytoestrogens and their chemopreventive mechanisms. Nutr. Cancer 2015, 67, 796–803. [Google Scholar] [CrossRef] [PubMed]
- Torres, S.; Martha, E.; Saucedo-Vázquez Juan, P.; Kroneck; Peter, M.H. Chapter 1, Section 2 “The rise of dioxygen in the atmosphere”. In Sustaining Life on Planet Earth: Metalloenzymes Mastering Dioxygen and Other Chewy Gases. Metal Ions in Life Sciences 15; Peter, M.H., Kroneck, Martha, E., Torres, S., Eds.; Springer: Cham, Switzerland, 2015; pp. 1–12. [Google Scholar] [CrossRef]
- Williams, G.E. Geological constraints on the Precambrian history of earth’s rotation and Moon’s orbit. Rev. Geophys. 2000, 38, 37–59. [Google Scholar] [CrossRef]
- Lavoisier, L. Traité Élémentaire de Chimie, Cuchet; Paris, 1789; Volume 1, p. 293, English translation by Kerr, R. Elements of Chemistry, Creech, Edinburgh, 1790, reprinted by Dover Press: New York, NY, USA, 1965; p. 66; Available online: http://hdl.handle.net/10630/4374 (accessed on 20 July 2016).
- Idhe, A.J. The history of free radicals and Moses Gomberg’s contributions. Pure Appl. Chem. 1967, 15, 1–14. [Google Scholar] [CrossRef]
- Gomberg, M. Triphenylmethyl, em Fall von Dreiwerthigen Kohlenstoff. Ber. Dtsch. Chem. Ges. 1900, 33, 315. [Google Scholar] [CrossRef]
- Gomberg, M. An instance of trivalent carbon: Triphenylmethyl. J. Am. Chem. Soc. 1900, 22, 757. [Google Scholar] [CrossRef]
- Zavoisky, Y.K. FIAN (FIAN (ФИАН). Ph.D. Thesis, Physical Institute of the Academy of Sciences, Moscow, Russia, 1944. [Google Scholar]
- Zavoisky, E.K. Spin-magnetic resonance in paramagnetics. J. Phys. USSR 1945, 9, 211–216. [Google Scholar]
- Eaton, G.R.; Eaton, S.S. Foundations of Modern EPR; World Scientific: Singapore, 1998; pp. 45–46. [Google Scholar]
- Herzberg, G. The Spectra and Structures of Simple Free Radicals; Dover Publications: New York, NY, USA, 1971. [Google Scholar]
- Ayala, A.; Muñoz, M.F.; Argüelles, S. Lipid peroxidation: Production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxidative Med. Cell Longev. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- De Groot, H. Reactive oxygen species in tissue injury. Hepatogastroenterology 1994, 41, 328–332. [Google Scholar] [PubMed]
- Grace, P.A. Ischaemia-reperfusion injury. Br. J. Surg. 1994, 81, 637–647. [Google Scholar] [CrossRef] [PubMed]
- Tyndall, J. The Copley Medallist of 1871; Longmans, Green, and Co.: London, UK, 1879; Volume I. [Google Scholar]
- Davies, M. Free radicals, oxidants and protein damage. Aust. Biochem. 2012, 43, 8–12. [Google Scholar]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine, 2nd ed.; Clarendon Press: Oxford, UK, 1989. [Google Scholar]
- Halliwell, B.; Cross, C.E.; Gutteridge, J.M.C. Free radicals, antioxidants and human disease: Where are we now? J. Lab. Clin. Med. 1992, 119, 598–620. [Google Scholar] [PubMed]
- Gutteridge, J.M.C. Free radicals in disease processes: A compilation of cause and consequence. Free Radic. Res. Commun. 1993, 19, 141–158. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Gutteridge, J.M.C. Lipid peroxidation, oxygen radicals, cell damage, and antioxidant therapy. Lancet 1984, 1, 1396–1397. [Google Scholar] [CrossRef]
- Halliwell, B. Reactive species and antioxidants. Redox Biology is a fundamental theme of aerobic life. Plant Physiol. 2006, 141, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Nimse, S.B.; Pal, D. Free radicals, natural antioxidants, and their reaction mechanisms. RSC Adv. 2015, 5, 27986–28006. [Google Scholar] [CrossRef]
- Von Sonntag, C. The Chemical Basis of Radiation Biology; Taylor & Francis: London, UK, 1987. [Google Scholar]
- Breen, A.P.; Murphy, J.A. Reactions of oxyl radicals with DNA. Free Radic. Biol. Med. 1995, 18, 1033–1077. [Google Scholar] [CrossRef]
- Dizdaroglu, M.; Jaruga, P. Mechanisms of free radical-induced damage to DNA. Free Radic. Res. 2012, 46, 382–419. [Google Scholar] [CrossRef] [PubMed]
- Fenton, H.J.H. Oxidation of tartaric acid in presence of iron. J. Chem. Soc. Trans. 1894, 65, 899–911. [Google Scholar] [CrossRef]
- Haber, F.; Weiss, J. Über die katalyse des hydroperoxydes. Naturwissenschaften 1932, 20, 948–950. [Google Scholar] [CrossRef]
- Knight, J.A. Free Radicals, Antioxidants, Aging and Disease; AACC Press: Washington, DC, USA, 1999. Available online: http://trove.nla.gov.au/version/39821668 (accessed on 20 July 2016).
- Jomova, K.; Valko, M. Advances in metal-induced oxidative stress and human disease. Toxicology 2011, 283, 65–87. [Google Scholar] [CrossRef] [PubMed]
- Beckman, J.S.; Chen, J.; Ischiropoulos, H.; Crow, J.P. Oxidative chemistry of peroxynitrite. Methods Enzymol. 1994, 233, 229–240. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Hess, D.T.; Qian, Z.; Hausladen, A.; Fonseca, F.; Chaube, R.; Reynolds, J.D.; Stamler, J.S. Hemoglobin βCys93 is essential for cardiovascular function and integrated response to hypoxia. Proc. Natl. Acad. Sci. USA 2015, 112, 6425–6430. [Google Scholar] [CrossRef] [PubMed]
- Mechnikov, I. Nobel Lectures, Physiology or Medicine 1901–1921, 1908; Elsevier Publishing Company: Amsterdam, The Netherlands, 1967. [Google Scholar]
- Johnston, R.B., Jr.; Keele, B.B., Jr.; Misra, H.P.; Lehmeyer, K.J.E.; Webb, L.S.; Baehner, R.L.; Rajagopalan, V. The role of superoxide anion generation in phagocytic bactericidal activity. Studies with normal and chronic granulomatous disease leukocytes. J. Clin. Investig. 1975, 55, 1357–1372. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, K.; Adlercretz, H.; Boyle, P.; Denis, L.; Nicholson, R.I.; Morton, M.S. Nutrition and Cancer; ISIS Medical Media Ltd.: Oxford, UK, 1996. [Google Scholar]
- Pietta, P. Flavonoids as antioxidants. J. Nat. Prod. 2000, 63, 1035–1042. [Google Scholar] [CrossRef] [PubMed]
- Nijveldt, R.J.; van Nood, E.; van Hoorn, D.E.C.; Boelens, P.G.; van Norren, K.; van Leeuwen, P.A. Flavonoids: A review of probable mechanisms of action and potential applications. Am. J. Clin. Nutr. 2001, 74, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Hertog, M.G.; Feskens, E.J.; Hollman, P.C.; Katan, M.B.; Kromhout, D. Dietary antioxidant flavonoids and risk of coronary heart disease: The Zutphen Elderly Study. Lancet 1993, 342, 1007–1011. [Google Scholar] [CrossRef]
- Wang, X.; Ouyang, Y.Y.; Liu, J.; Zhao, G. Flavonoid intake and risk of CVD: A systematic review and meta-analysis of prospective cohort studies. Br. J. Nutr. 2014, 111, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hertog, M.G.; Feskens, E.J.; Hollman, P.C.; Katan, M.B.; Kromhout, D. Dietary flavonoids and cancer risk in the Zutphen Elderly Study. Nutr. Cancer 1994, 22, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Petrick, J.L.; Steck, S.E.; Bradshaw, P.T.; Trivers, K.F.; Abrahamson, P.E.; Engel, L.S.; He, K.; Chow, W.-H.; Mayne, S.T.; Risch, H.A.; et al. Dietary intake of flavonoids and oesophageal and gastric cancer: Incidence and survival in the United States of America (USA). Br. J. Cancer 2015, 112, 1291–1300. [Google Scholar] [CrossRef] [PubMed]
- Romagnolo, D.F.; Selmin, O.I. Flavonoids and cancer prevention: A review of the evidence. J. Nutr. Gerontol. Geriatr. 2012, 31, 206–238. [Google Scholar] [CrossRef] [PubMed]
- Turati, F.; Rossi, M.; Pelucchi, C.; Levi, F.; La Vecchia, C. Fruit and vegetables and cancer risk: A review of southern European studies. Br. J. Nutr. 2015, 113 (Suppl. S2), S102–S110. [Google Scholar] [CrossRef] [PubMed]
- Woo, H.D.; Lee, J.; Choi, I.J.; Kim, C.G.; Lee, J.Y.; Kwon, O.; Kim, J. Dietary flavonoids and gastric cancer risk in a Korean population. Nutrients 2014, 6, 4961–4973. [Google Scholar] [CrossRef] [PubMed]
- Carlson, J.R.; Bauer, B.A.; Vincent, A.; Limburg, P.J.; Wilson, T. Reading the tea leaves: Anticarcinogenic properties of (-)-epigallocatechin-3-gallate. Mayo Clin. Proc. 2007, 82, 725–732. [Google Scholar] [CrossRef]
- Curtis, P.J.; Potter, J.; Kroon, P.A.; Wilson, P.; Dhatariya, K.; Sampson, M.; Cassidy, A. vascular function and atherosclerosis progression after 1 y of flavonoid intake in statin-treated postmenopausal women with type 2 diabetes: A double-blind randomized controlled trial. Am. J. Clin. Nutr. 2013, 97, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Curtis, P.J.; Sampson, M.; Potter, J.; Dhatariya, K.; Kroon, P.A.; Cassidy, A. Chronic ingestion of flavan-3-ols and isoflavones improves insulin sensitivity and lipoprotein status and attenuates estimated 10-year CVD risk in medicated postmenopausal women with type 2 diabetes: A 1-year, double-blind, randomized, controlled trial. Diabetes Care 2012, 35, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Acquaviva, R.; Campisi, A.; Sorrenti, V.; Giacomo, C.D.; Virgata, G.; Barcellona, M.L.; Vanella, A. Bioflavonoids as antiradicals, antioxidants and DNA cleavage protectors. Cell Biol. Toxicol. 2000, 16, 91–98. [Google Scholar] [CrossRef] [PubMed]
- De Souza, R.F.; de Giovani, F.W. Antioxidant properties of complexes of flavonoids with metal ions. Redox Rep. 2004, 9, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Torreggiani, A.; Tamba, M.; Trinchero, A. Copper(II)-Quercetin complexes in aqueous solutions: Spectroscopic and kinetic properties. J. Mol. Struct. 2005, 44, 759–766. [Google Scholar] [CrossRef]
- Kang, J.; Zhou, L.; Lu, X.; Liu, H.; Zhang, M.; Wu, H. Electrochemical Investigation on Interaction between DNA with Quercetin and Eu–Qu3 Complex. J. Inorg. Biochem. 2004, 98, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Krishnamachari, V.; Levine, L.H.; Pareá, P.W. Flavonoid oxidation by the radical generator AIBN: A unified mechanism for quercetin radical scavenging. J. Agric. Food Chem. Am. Chem. Soc. USA 2002, 50, 4357–4363, ISSN: 0021-8561. [Google Scholar] [CrossRef]
- Das, A.; Wang, J.H.; Lien, E.J. Carcinogenicity, mutagenicity and cancer preventing activities of flavonoids: A structure-system-activity relationship (SSAR) analysis. Prog. Drug Res. 1994, 42, 133–166. [Google Scholar] [CrossRef] [PubMed]
- Duthie, S.J.; Johnson, W.; Dobson, V.L. The effect of dietary flavonoids on DNA damage (strand breaks and oxidised pyrimidines) and growth in human cells. Mutat. Res. 1997, 390, 141–151. [Google Scholar] [CrossRef]
- Jun, T.; Liancai, Z.; Bochu, W. Effects of quercetin on DNA damage induced by copper ion. Int. J. Pharmacol. 2007, 3, 19–26. [Google Scholar] [CrossRef]
- Galaris, D.; Evangelou, A. The role of oxidative stress in mechanisms of metal-induced carcinogenesis. Crit. Rev. Oncol. Hematol. 2002, 42, 93–103. [Google Scholar] [CrossRef]
- Paur, I.; Carlsen, M.H.; Halvorsen, B.L.; Blomhoff, R. Antioxidants in herbs and spices. Roles in oxidative stress and redox signaling. In Herbal Medicine: Biomolecular and Clinical AspectsChapter 2, 2nd ed.; Benzie, I.F.F., Wachtel-Galor, S., Eds.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2011. [Google Scholar]
- Peto, R.; Doll, R.; Buckley, J.D.; Sporn, M.B. Can dietary beta carotene materially reduce human cancer rates? Nature 1981, 290, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, T. A large scale cohort study on cancer risks by diet with special reference to the risk reducing effects of green-yellow vegetable consumption. In Diet, Nutrition and Cancer; Hayashi, Y., Nagao, M., Sugimura, T., Takayama, S., Tomatis, L., Wattenberg, L.W., Wogan, G., Eds.; Japanese Science Society Press, Tokyo/VNU Science Press: Utrecht, The Netherlands, 1986; pp. 41–53. [Google Scholar]
- Lee, H.P.; Gourley, L.; Duffy, S.W.; Estéve, J.; Lee, J.; Day, N.E. Dietary effects on breast-cancer risk in Singapore. Lancet 1991, 337, 1197–1200. [Google Scholar] [CrossRef]
- Boyle, P.; Maisonneuve, P. Lung cancer and tobacco smoking. Lung Cancer 1995, 12, 167–181. [Google Scholar] [CrossRef]
- Krinsky, N.I. Effects of carotenoids in cellular and animal systems. Am. J. Clin. Nutr. 1991, 53 (Suppl. S1), 238S–246S. [Google Scholar] [PubMed]
- Singh, R.B.; Niaz, M.A.; Ghosh, S.; Beegum, R.; Bishnoi, I.; Agarwal, P.; Agarwal, A. Dietary intake and plasma levels of antioxidant vitamins in health and disease. A hospital based case control study. J. Nutr. Environ. Med. 1995, 5, 235–242. [Google Scholar] [CrossRef]
- Gallicchio, L.; Boyd, K.; Matanoski, G.; Tao, X.G.; Chen, L.; Lam, T.K.; Shiels, M.; Hammond, E.; Robinson, K.A.; Caulfield, L.E.; et al. Carotenoids and the risk of developing lung cancer: A systematic review. Am. J. Clin. Nutr. 2008, 88, 372–383. [Google Scholar] [PubMed]
- Eliassen, A.H.; Hendrickson, S.J.; Brinton, L.A.; Buring, J.E.; Campos, H.; Dai, Q.; Dorgan, J.F.; Franke, A.A.; Gao, Y.T.; Goodman, M.T.; et al. Circulating carotenoids and risk of breast cancer: Pooled analysis of eight prospective studies. J. Natl. Cancer Inst. 2012, 104, 1905–1916. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Spiegelman, D.; Baglietto, L.; Bernstein, L.; Boggs, D.A.; van den Brandt, P.A.; Buring, J.E.; Gapstur, S.M.; Giles, G.G.; Giovannucci, E.; et al. Carotenoid intakes and risk of breast cancer defined by estrogen receptor and progesterone receptor status: A pooled analysis of 18 prospective cohort studies. Am. J. Clin. Nutr. 2012, 95, 713–725. [Google Scholar] [CrossRef] [PubMed]
- Eliassen, A.H.; Liao, X.; Rosner, B.; Tamimi, R.M.; Tworoger, S.S.; Hankinson, S.E. Plasma carotenoids and risk of breast cancer over 20 year of follow-up. Am. J. Clin. Nutr. 2015, 101, 1197–1205. [Google Scholar] [CrossRef] [PubMed]
- Bolhassani, A. Cancer chemoprevention by natural carotenoids as an efficient strategy. Anti-Cancer Agents Med. Chem. 2015, 15, 1026–1031. [Google Scholar] [CrossRef]
- Chen, J.; O’Donoghue, A.; Deng, Y.F.; Zhang, B.; Kent, F.; O’Hare, T. The effect of lycopene on the PI3K/Akt signalling pathway in prostate cancer. Anti-Cancer Agents Med. Chem. 2014, 14, 800–805. [Google Scholar]
- Khuda-Bukhsh, A.R.; Das, S.; Saha, S.K. Molecular approaches toward targeted cancer prevention with some food plants and their products: Inflammatory and other signal pathways. Nutr. Cancer 2014, 66, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Puglia, C.D.; Powell, S.R. Inhibition of cellular antioxidants: A possible mechanism of toxic cell injury. Environ. Health Perspect. 1984, 57, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.; Frei, B. Does vitamin C act as a pro-oxidant under physiological conditions? FASEB J. 1999, 13, 1007–1024. [Google Scholar] [PubMed]
- Linder, M.C. The relationship of copper to DNA damage and damage prevention in humans. Mutat. Res. 2012, 733, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A.; Shahabuddin; Hadi, S.M.; Parish, J.H.; Ainley, K. Strand scission in DNA induced by quercetin and Cu(II): Role of Cu(I) and oxygen free radicals. Carcinogenesis 1989, 10, 1833–1839. [Google Scholar] [CrossRef] [PubMed]
- Fukuhara, K.; Miyata, N. Resveratrol as a new type of DNA-cleaving agent. Bioorg. Med. Chem. Lett. 1998, 8, 3187–3192. [Google Scholar] [CrossRef]
- Zheng, L.F.; Wei, Q.Y.; Cai, Y.J.; Fang, J.G.; Zhou, B.; Yang, L.; Liu, Z.L. DNA damage induced by resveratrol and its synthetic analogues in the presence of Cu (II) ions: Mechanism and structure-activity relationship. Free Radic. Biol. Med. 2006, 41, 1807–1816. [Google Scholar] [CrossRef] [PubMed]
- Hadi, S.M.; Asad, S.F.; Singh, S.; Ahmad, A. Putative mechanism for anticancer and apoptosis-inducing properties of plant-derived polyphenolic compounds. IUBMB Life 2000, 50, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.C.; Wang, Y.H.; Liou, C.C.; Lin, Y.C.; Huang, H.; Liu, Y.C. Induction of oxidative DNA damage by flavonoids of propolis: Its mechanism and implication about antioxidant capacity. Chem. Res. Toxicol. 2012, 25, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Azmi, A.S.; Bhat, S.H.; Hanif, S.; Hadi, S.M. Plant polyphenols mobilize endogenous copper in human peripheral lymphocytes leading to oxidative DNA breakage: A putative mechanism for anticancer properties. FEBS Lett. 2006, 580, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Azmi, A.S.; Sarkar, F.H.; Hadi, S.M. Pro-oxidant activity of dietary chemopreventive agents: An under-appreciated anti-cancer property. Version 1. F1000Research 2013, 2, 135. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hackshaw-McGeagh, L.E.; Perry, R.E.; Leach, V.A.; Qandil, S.; Jeffreys, M.; Martin, R.M.; Lane, J.A. A systematic review of dietary, nutritional, and physical activity interventions for the prevention of prostate cancer progression and mortality. Cancer Causes Control 2015, 26, 1521–1550. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Banerjee, S.; Sil, P.C. The beneficial role of curcumin on inflammation, diabetes and neurodegenerative disease: A recent update. Food Chem. Toxicol. 2015, 83, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Kadakol, A.; Shasthrula, P.K.; Mundhe, N.A.; Jamdade, V.S.; Barua, C.C.; Gaikwad, A.B. Curcumin as an adjuvant to breast cancer treatment. Anti-Cancer Agents Med. Chem. 2015, 15, 647–656. [Google Scholar] [CrossRef]
- Surh, Y.J.; Chun, K.S. Cancer chemopreventive effects of curcumin. Adv. Exp. Med. Biol. 2007, 595, 149–172. [Google Scholar] [CrossRef] [PubMed]
- Miriyala, S.; Panchatcharam, M.; Rengarajulu, P. Cardioprotective effects of curcumin. Adv. Exp. Med. Biol. 2007, 595, 359–377. [Google Scholar] [CrossRef] [PubMed]
- Krishnaswamy, K. Traditional Indian spices and their health significance. Asian Pac. J. Clin. Nutr. 2008, 17 (Suppl. S1), 265–268. [Google Scholar]
- Oyagbemi, A.A.; Saba, A.B.; Ibraheem, A.O. Curcumin: From food spice to cancer prevention. Asian Pac. J. Cancer Prev. APJCP 2009, 10, 963–967. [Google Scholar] [PubMed]
- Gerhauser, C. Cancer chemoprevention and nutriepigenetics: State of the art and future challenges. Top. Curr. Chem. 2013, 329, 73–132. [Google Scholar] [CrossRef] [PubMed]
- Henning, S.M.; Zhang, Y.; Seeram, N.P.; Lee, R.P.; Wang, P.; Bowerman, S.; Heber, D. Antioxidant capacity and phytochemical content of herbs and spices in dry, fresh and blended herb paste form. Int. J. Food Sci. Nutr. 2011, 62, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Henning, S.M.; Zhang, Y.; Zerlin, A.; Li, L.; Gao, K.; Lee, R.-P.; Karp, H.; Thames, G.; Bowerman, S.; et al. Antioxidant-rich spice added to hamburger meat during cooking results in reduced meat, plasma, and urine malondialdehyde concentrations. Am. J. Clin. Nutr. 2010, 91, 1180–1184. [Google Scholar] [CrossRef] [PubMed]
- Marnett, L.J. Lipid peroxidation-DNA damage by malondialdehyde. Mutat. Res. 1999, 424, 83–95. [Google Scholar] [CrossRef]
- Gorelik, S.; Ligumsky, M.; Kohen, R.; Kanner, J. The stomach as a “bioreactor”: When red meat meets red wine. J. Agric. Food Chem. 2008, 56, 5002–5007. [Google Scholar] [CrossRef] [PubMed]
- Gorelik, S.; Lapidot, T.; Shaham, I.; Granit, R.; Ligumsky, M.; Kohen, R.; Kanner, J. Lipid peroxidation and coupled vitamin oxidation in simulated and human gastric fluid inhibited by dietary polyphenols: Health implications. J. Agric. Food Chem. 2005, 53, 3397–3402. [Google Scholar] [CrossRef] [PubMed]
- Gorelik, S.; Kohen, R.; Ligumsky, M.; Kanner, J. Saliva plays a dual role in oxidation process in stomach medium. Arch. Biochem. Biophys. 2007, 458, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Gorelik, S.; Ligumsky, M.; Kohen, R.; Kanner, J. A novel function of red wine polyphenols in humans: Prevention of absorption of cytotoxic lipid peroxidation products. FASEB J. 2008, 22, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Viuda-Martos, M.; Ruiz-Navajas, Y.; Fernández-López, J.; Pérez-Álvarez, J.A. Spices as functional foods. Crit. Rev. Food Sci. Nutr. 2010, 51, 13–28. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, B.B.; Sundaram, C.; Prasad, S.; Reuter, S.; Kannappan, R.; Yadav, V.R.; Park, B.; Kim, J.H.; Gupta, S.C.; Phromnoi, K.; et al. Chronic diseases caused by chronic inflammation require chronic treatment: The anti-inflammatory lifestyle. In Inflammation, Lifestyle and Chronic Diseases: The Silent Link; Aggarwal, B.B., Krishnan, S., Guha, S., Eds.; CRC Press, Taylor and Francis Group: Boca Raton, FL, USA, 2012; Chapter 14. [Google Scholar]
- Prasad, S.; Aggarwal, B.B. Chronic diseases caused by chronic Inflammation require chronic treatment: Anti-inflammatory role of dietary spices. J. Clin. Cell. Immunol. 2014, 5, 238. [Google Scholar] [CrossRef]
- Marchiani, A.; Rozzo, C.; Fadda, A.; Delogu, G.; Ruzza, P. Curcumin and curcumin-like molecules: From spice to drugs. Curr. Med. Chem. 2014, 21, 204–222. [Google Scholar] [CrossRef] [PubMed]
- Sandur, S.K.; Ichikawa, H.; Pandey, M.K.; Kunnumakkara, A.B.; Sung, B.; Sethi, G.; Aggarwal, B.B. Role of pro-oxidants and antioxidants in the anti-inflammatory and apoptotic effects of curcumin (diferuloylmethane). Free Radic. Biol. Med. 2007, 43, 568–580. [Google Scholar] [CrossRef] [PubMed]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef] [PubMed]
- Hungin, A.P.S.; Mulligan, C.; Pot, B.; Whorwell, P.; Agréus, L.; Fracasso, P.; Lionis, C.; Mendive, J.; Philippart, D.E.; Foy, J.M.; et al. Systematic review: Probiotics in the management of lower gastrointestinal symptoms in clinical practice—An evidence-based international guide. Aliment. Pharmacol. Ther. 2013, 38, 864–886. [Google Scholar] [CrossRef] [PubMed]
- Gut Microflora for Health. World Summit 2016. Available online: http://summit-registration.gutmicrobiotaforhealth.com/ (accessed on 27 July 2016).
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Griffiths, K.; Aggarwal, B.B.; Singh, R.B.; Buttar, H.S.; Wilson, D.; De Meester, F. Food Antioxidants and Their Anti-Inflammatory Properties: A Potential Role in Cardiovascular Diseases and Cancer Prevention. Diseases 2016, 4, 28. https://doi.org/10.3390/diseases4030028
Griffiths K, Aggarwal BB, Singh RB, Buttar HS, Wilson D, De Meester F. Food Antioxidants and Their Anti-Inflammatory Properties: A Potential Role in Cardiovascular Diseases and Cancer Prevention. Diseases. 2016; 4(3):28. https://doi.org/10.3390/diseases4030028
Chicago/Turabian StyleGriffiths, Keith, Bharat B. Aggarwal, Ram B. Singh, Harpal S. Buttar, Douglas Wilson, and Fabien De Meester. 2016. "Food Antioxidants and Their Anti-Inflammatory Properties: A Potential Role in Cardiovascular Diseases and Cancer Prevention" Diseases 4, no. 3: 28. https://doi.org/10.3390/diseases4030028
APA StyleGriffiths, K., Aggarwal, B. B., Singh, R. B., Buttar, H. S., Wilson, D., & De Meester, F. (2016). Food Antioxidants and Their Anti-Inflammatory Properties: A Potential Role in Cardiovascular Diseases and Cancer Prevention. Diseases, 4(3), 28. https://doi.org/10.3390/diseases4030028




