1. Introduction
Tape stripping is a safe and minimally invasive method for obtaining skin material from volunteers to test the efficacy of cosmetic formulations. When performing tape stripping, the outermost layer of the epidermis is removed, the more tape strips that are used on the same spot, the deeper into the epidermis the cells are harvested. The harvested material from the tape strips can then be extracted and the expression on both protein and RNA [
1,
2], as wells as lipids [
3] can be investigated, depending on the extraction conditions. The biomarkers investigated in this study are related to epidermal differentiation and barrier function, and include Keratin-6, Keratin-1,-10, Involucrin, Hornerin, Claudin-, Caspase-14 and Fibronectin.
The outermost part of the skin consists of a barrier that protects against transepidermal water loss and external damage, reviewed in [
4]. Involucrin and Loricrin are two of the main structural proteins forming the cornified envelope that constitutes the barrier. Involucrin is expressed as keratinocytes start to differentiate into corneocytes, reviewed in [
5]. Even if Involucrin is one of the major constituents of the cornified envelope, ablation of
Involucrin in mice does not change the basal skin barrier function but it does result in reduced barrier repair [
5]. In dry skin, Involucrin is prematurely expressed from the granular layers to the spinous layers [
6]. Levels of Involucrin are decreased in atopic dermatitis (AD) [
7], and it has been suggested that increased Involucrin may indicate thinner SC [
1].
On the inside of the corneocytes, keratin filaments interacting with Filaggrins are linked to the cornified envelope, reviewed by Proksch
et al. [
5]. Keratins are the major structural protein synthesized by Keratinocytes. Keratin-6 is induced by inflammation and elevated in wound healing and psoriasis [
8], hence increase in Keratin-6 may indicate increased keratinocyte repair [
1]. In addition, acute as well as chronic barrier disruption resulted in the induction of Keratin-6, and Keratin-10 whereas Keratin-1 was only induced after chronic barrier disruption [
9]; furthermore, dry skin has been associated with reduced Keratin-1 and Keratin-10 [
6].
Natural moisturising factors (NMFs) mainly from proteolysis of Filaggrin retain the hydration of the skin [
10]. Caspase-14 has been shown to be involved in the epidermal barrier formation, in the processing and degradation of pro-filaggrin [
11]. Degradation of pro-filaggrin results in the release of amino acids that acts as natural moisturizing factors that are essential for the skin hydration, reviewed in [
12]. Studies of
Caspase-14 knockout mice have also revealed an important function for Caspase-14 in the UVB protection of the stratum corneum [
11].
Hornerin is another component of the cornified envelope, and share features with filaggrin; moreover, it has been shown to be a substrate to transglutaminase-3 [
13]. The levels of Hornerin are upregulated after tape stripping [
13] and UVB-irradiation [
14] and suggested to be involved in the process of barrier repair. Both Filaggrin and Hornerin expression are reported to be decreased in patients with Atopic dermatitis (AD) [
15].
Tight junctions are semi permeable size- and ion-specific barriers within the epithelia important for water transport [
16]. Claudins are proteins in the tight junctions that control the specificity of the junction, they are also important in the formation of the tight junctions [
16]. There are several claudins, but Claudin-1 has been shown to be absolutely essential for hydration and water management [
17].
Fibronectin was also investigated in this study. Fibronectin increase may indicate repair of skin integrity as it is known to be a cross linker for keratins [
1].
2. Experimental Section
D-Squame tape strips (CuDerm Corporation, Dallas, TX, USA) were used in this clinical study to sample skin from the forearms of 10 volunteers. The study was approved by the Oriflame Research and Development Ethics Committee, Bray, Ireland, 2012. The study complied with the Guidelines for Medical Experiments in non-patient human volunteers, published by the Association of the British Pharmaceutical Industry (ABPI) and the World Medical Association’s Declaration of Helsinki (2000) concerning biomedical research involving human subjects. Each subject supplied written informed consent and received a copy of the INCI ingredient list for each product. The forearms were divided into three areas, where one area was left untreated and the other two areas were treated with two different product formulations Product A and Product B (
Table 1) once a day for 4 weeks. Both products are basic formula using different non-ionic emulsifier, with Product B containing Glycerin and the occlusive emollient Petrolatum, whereas Product A doesn’t contain an occlusive emollient, and a more biocompatible emulsifier.
Table 1.
Constituents of the formulations used in the clinical study.
Table 1.
Constituents of the formulations used in the clinical study.
| Product A | Product B |
|---|
| Aqua, Isopropyl Palmitate, Isostearyl Isostearate, Sorbitan Olivate, Cetearyl Glucoside, Dimethicone, Hydroxypropyl Starch Phosphate, Cetearyl Alcohol, Stearic Acid, Butyrospermum Parkii Butter, Caprylyl Glycol, Phenoxyethanol, Pentaerythrityl Distearate, Bis-Diglyceryl Polyacyladipate-2, Xanthan Gum, Piroctone Olamine | Aqua, Glycerin, Petrolatum, Stearic Acid, Glycol Stearate, Isopropyl Isostearate, Dimethicone, Tapioca starch, Glyceryl Stearate, Stearamide AMP, Cetyl alcohol, Triethanolamine, Magnesium Aluminium silicate, Carbomer, Disodium EDTA, Phenoxyethanol, Methylparaben, Propylparaben, CI 77891 |
Skin tape strips were collected before the start of treatment with product formulations (T0), at the end of the study after four weeks of product treatment (T4) and finally after one week of product treatment remission (T1R). For each area at the three time-points, eight consecutive tape strips were collected of which the first tape strip was discarded. Proteins were extracted from the remaining 7 tape strips, and pooled together. The extraction method was determined on the basis of the results from an extraction optimization study performed earlier in house. Single Enzyme-Linked ImmunoSorbent Assay (ELISA) techniques were used to measure Hornerin, Caspase-14, Claudin-1 (ABIN583822, ABIN511293, ABIN418089 all from Antibodies online). Multiplex ELISA techniques were used to measure Keratin-6, Involucrin, Keratin-1,-10 and Fibronectin (Milliplex map human Skin Panel SKIN-50K, Millipore, St. Charles, MO, USA). All biomarker levels were correlated to the total protein content of the tape strip extractions. Statistics were calculated using Wilcoxon signed-rank test.
3. Results
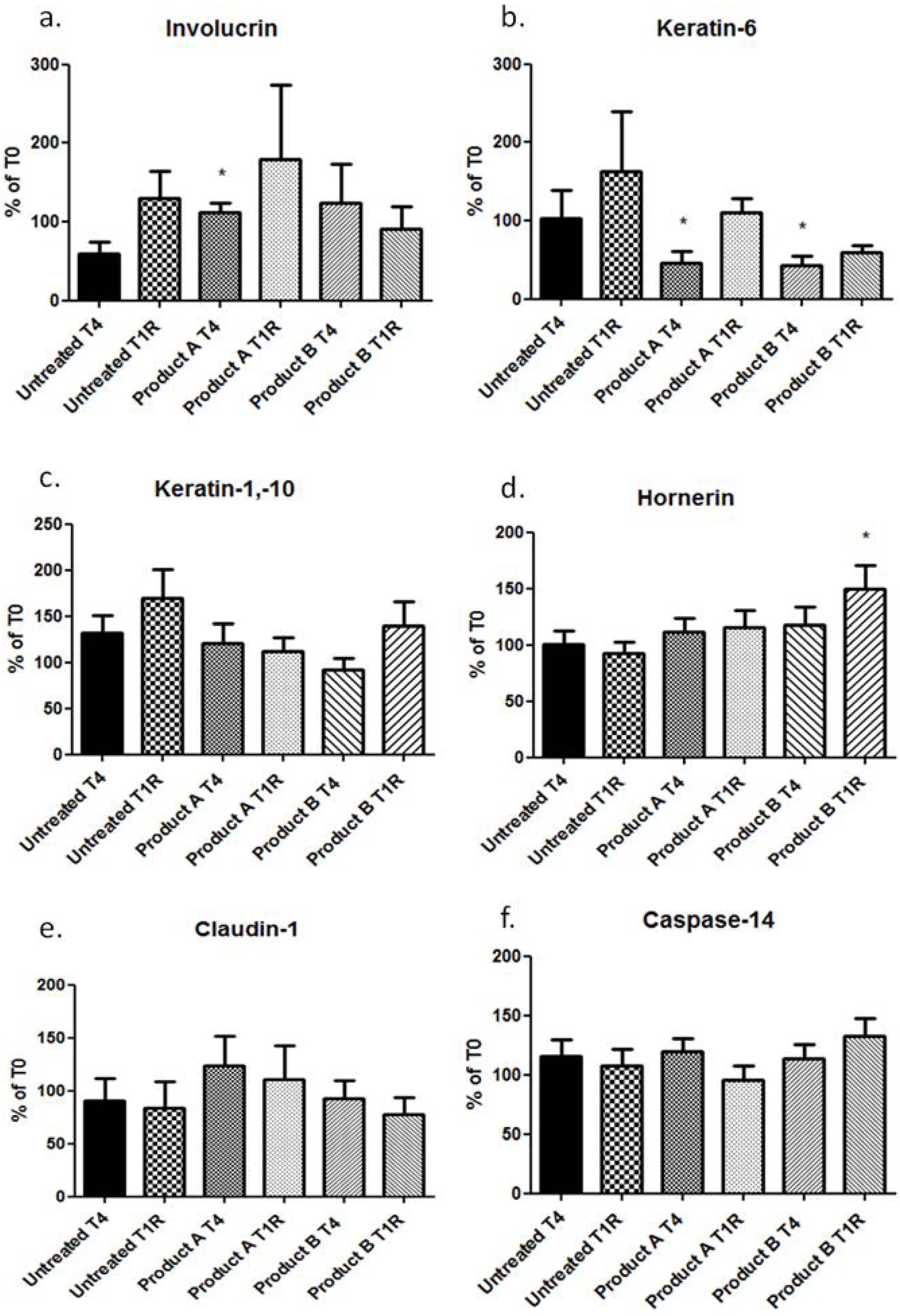
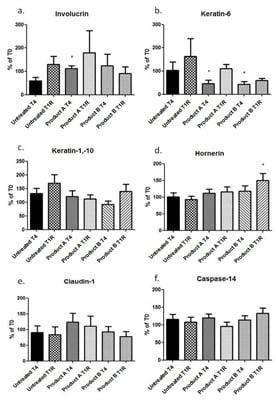
The levels of different biomarkers in tape strip protein extracts were investigated after four weeks of product applications and after one week of treatment remission. Treatment with Product A resulted in an increased level of Involucrin compared to untreated skin (
p = 0.039) (
Figure 1a). The same pattern was seen for Product B, but the increase did not reach significance. After one week of remission, the levels of Involucrin in Product A treated skin seemed to stayed elevated (although no significance was reached), whereas for Product B treatment, the levels declined again after treatment stopped. In addition, both treatments resulted in significant decreased levels of Keratin-6 (Product A
p = 0.019, Product B
p = 0.016) (
Figure 1b). This observed decrease in protein expression appeared to be prolonged after one week of remission compared to untreated samples (
p > 0.05). Both treatments resulted in a pattern of suppressed Keratin-1,-10 levels (
Figure 1c), with the greater reduction seen after Product B treatment, however the differences were not significant. At one week of remission the pattern was the same with even lower Keratin-1,-10 levels compared to untreated skin for Product A (
p = 0.10), whereas for Product B the levels were increasing again towards the levels of the untreated.
Treatment with Product A or B resulted in small but not significant increases of Hornerin (
Figure 1d). However, one week after remission the levels of Hornerin in Product B treated skin continued to increase and reached statistical significance (
p = 0.03). The levels of Claudin-1 were slightly but not significantly increased by Product A at both time points (
Figure 1e), whereas Product B had even less effect, neither directly after treatment nor after one week of remission. None of the treatments had any effect on the levels of Caspase-14 directly after treatment. One week after remission, there was a tendency for a small increase after treatment with Product B (
Figure 1f).
Levels of Fibronectin in this pilot study were below detection limits for all samples analyzed.
4. Conclusion
When evaluating product efficacy in vivo it is also important to understand the effects different products are having at a protein level to restore moisturization and barrier function.
In this study, through the use of tape strips we demonstrate the ability to detect key epidermal biomarkers from the upper layers of the epidermis.
Involucrin plays an important role in maintaining the epidermal barrier [
5], and an increased level has been suggested to improve barrier function [
1]. Both products resulted in increased levels of Involucrin after 4 weeks of treatments, suggesting a barrier strengthening effect. Interestingly, after one week of treatment remission, a trend is observed where the Involucrin levels continue to increase for Product A (
p > 0.05) whereas the Involucrin levels in Product B decrease after the treatment was completed.
Keratin-6 is expressed early in the differentiation process [
5], and increased levels of Keratin-6 suggest the skin is under some form of stress or undergoing repair mechanisms [
9], hence decreased levels would imply a healthy barrier, as we see after treatment with both Products A and B.
In addition to barrier function, biomarkers related to moisturization are also investigatedin this pilot study. Hornerin, with its connection to NMF, plays an essential role in the moisturization of the skin [
13]. The pattern observed of increased levels of Hornerin after treatments with Products A and B suggests that the treatments have a beneficial impact to improve the moisturization capacity within the skin. Claudin-1 is part of tight junctions and hence involved in both the maintenance of the barrier and water management [
17]. In addition, for this biomarker a pattern of increased levels were detected in this pilot study.
Figure 1.
The expression of (a) Involucrin; (b) Keratin-6; (c) Keratin-1,-10; (d) Hornerin; (e) Claudin; (f) Caspase-14 at the end of a four week treatment period (T4) and the after one week of remission (T1R). * Significant results compared to untreated at the same time point (p ˂ 0.05).
Figure 1.
The expression of (a) Involucrin; (b) Keratin-6; (c) Keratin-1,-10; (d) Hornerin; (e) Claudin; (f) Caspase-14 at the end of a four week treatment period (T4) and the after one week of remission (T1R). * Significant results compared to untreated at the same time point (p ˂ 0.05).
A pattern of decreased Keratin-1,-10 was seen for both treatments in this study. Decreased levels of Keratin-1,-10 are reported to be associated with dry skin [
6]. Our observed results of Hornerin and Claudin-1 expressions suggest a beneficial moisturization effect after treatment with Product A or Product B, therefore the observed Keratin-1,-10 decrease after product treatment warrants further investigation.
Tape stripping is sometimes used as a method to induce mild irritancy in skin [
18], and levels of prostaglandin E2 and Interleukin-1 have been shown to be increased after tape stripping. However, the increase is small and transient. In order to induce a clear inflammatory response, the level of prostaglandin E2 needs to exceed the tape strip induced elevation by up to 40 times [
18]. Moreover, it has been shown that the effect of tape stripping ceases after two days. In this pilot study, there was a four-week interval between the first (untreated T0 samples) and the second (T4 samples) tape stripping sessions and one week between the second (T4 samples) and the final (T1Rsamples) tape stripping sessions. The irritant effect caused by the tape stripping is hence not considered to have an effect on the levels of the different biomarkers investigated here. Nonetheless, comparisons were always made relative to the untreated site at all time points investigated. In this pilot study, only 10 volunteers were included. Due to large variability in the levels of the investigated biomarkers among the volunteers, sample size needs to be increased in future studies. However, the results obtained in this pilot study would suggest that with a larger sample size more significant results for some of the biomarkers tested would be achieved. Our results presented here show that we can detect differences in the expression of skin proteins that in addition, are influenced after treatment with different products.
We also found in this study that the two different formulations tested activated the epidermal biomarkers somewhat differently. Therefore, this method could be useful to differentiate the effect of different formulations targeting different processes, and also to compare a formulation with and without an active ingredient to provide in vivo claims for an active ingredient. Moreover, the ability to measure biomarkers from tape strips opens up the opportunity to with an even larger portfolio of biomarkers being able to investigate mechanisms targeting different skin needs, such as barrier function, moisturization, irritancy, and pigmentation, to mention a few. The results from such studies could support products targeting different skin needs.
Moreover, in this study, we show that skin sampling after one week of product treatment remission gives additional insights into the lasting effects of the product treatments on the biomarkers investigated.