The Development of Advanced Optical Fibers for Long-Wave Infrared Transmission
Abstract
:1. Introduction
2. Experimental Section
2.1. Material Preparation
2.2. Fiber Preparation
3. Results and Discussion
3.1. Tellurium-Germanium-Gallium Fibers
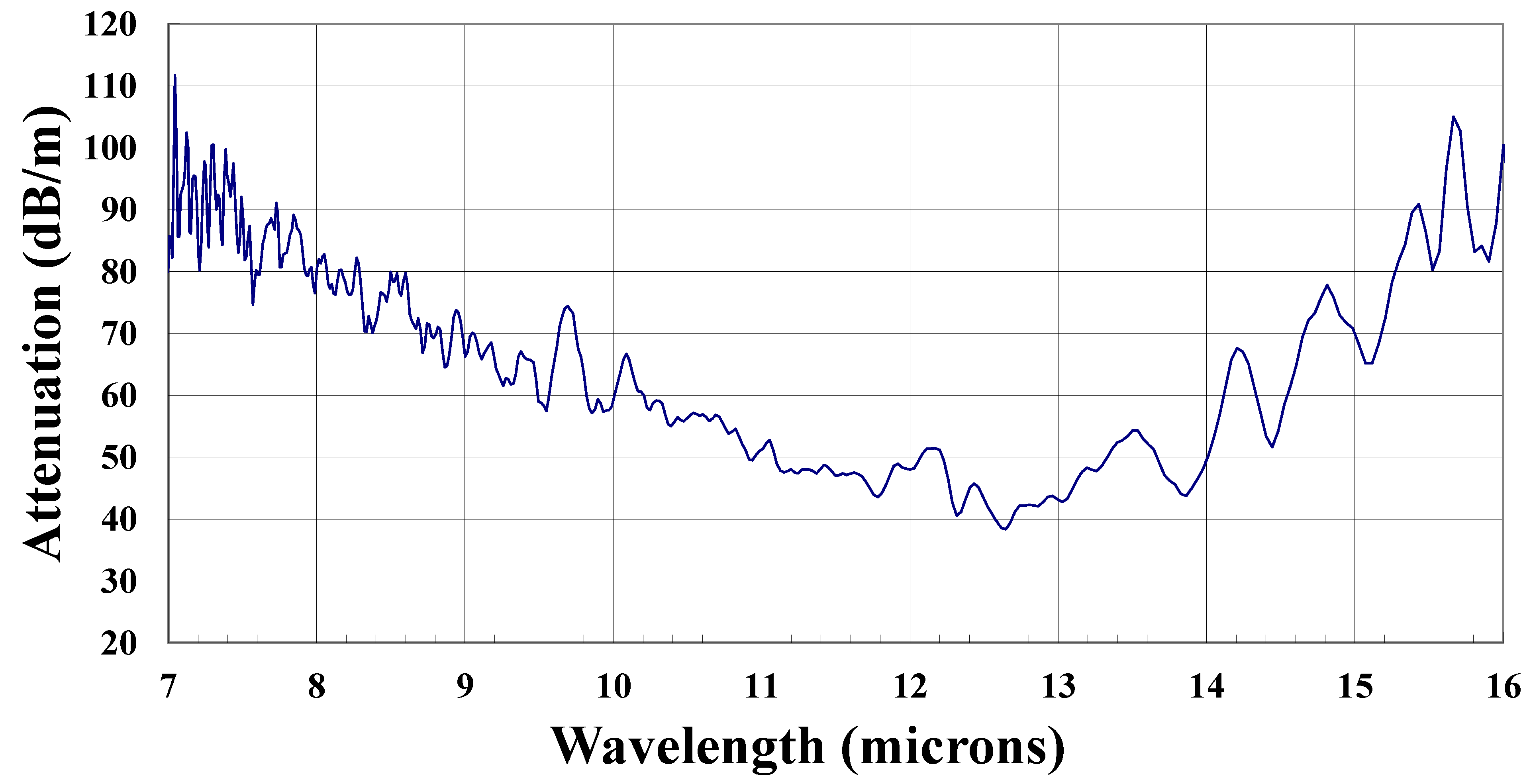
3.2. Tellurium Germanium Iodine Fibers
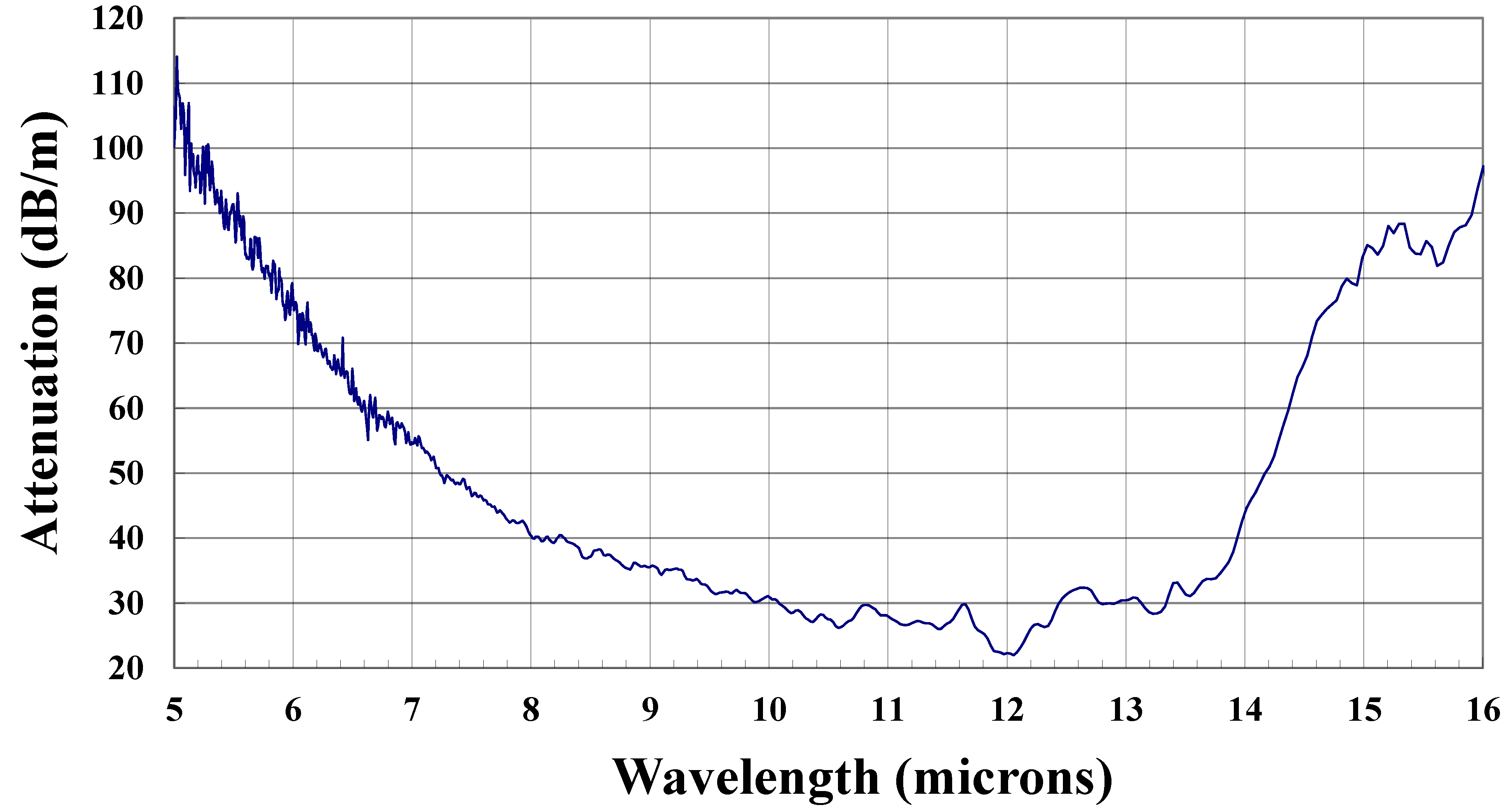
3.3. Tellurium Germanium Selenide Fibers
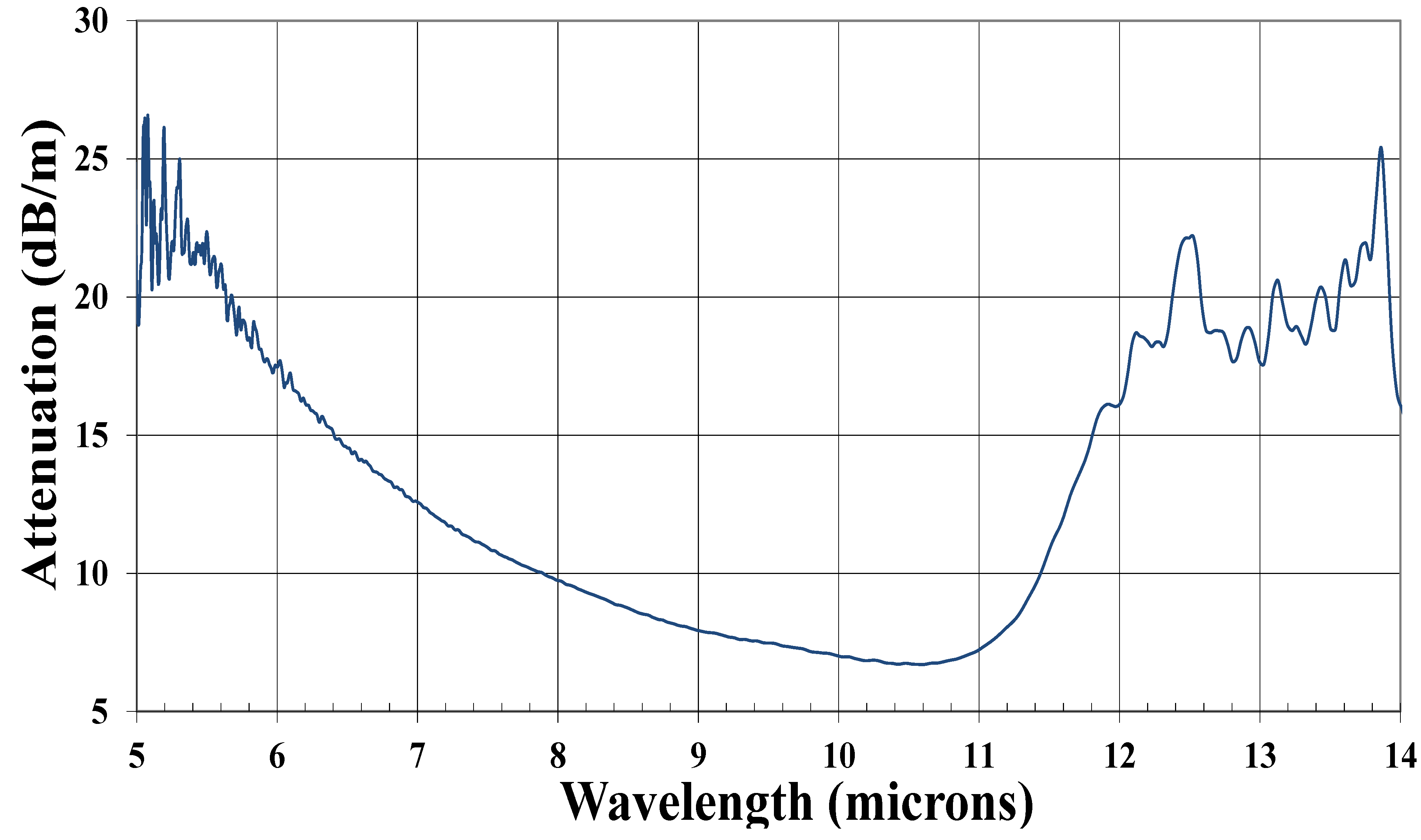
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Lucas, P.; Riley, M.R.; Boussard-Pledel, C.; Bureau, B. Advances in chalcogenide fiber evanescent wave biochemical sensing. Anal. Biochem. 2006, 351, 1–10. [Google Scholar] [CrossRef]
- Riley, M.R.; Lucas, P.; Le, C.D.; Juncker, C.; Boesewetter, D.E.; Collier, J.L.; DeRosa, D.M.; Katterman, M.E.; Boussard-Pledel, C.; Bureau, B. Lung cell fiber evanescent wave spectroscopic biosensing of inhalation health hazards. Biotechnol. Bioeng. 2006, 95, 599–612. [Google Scholar] [CrossRef]
- Keirsse, J.; Lahaye, E.; Bouter, A.; Dupont, V.; Boussard-Pledel, C.; Bureau, B.; Adam, J.-L.; Monbet, V.; Sire, O. Mapping bacterial surface population physiology in real-time: Infrared spectroscopy of proteus mirabilis swarm colonies. Appl. Spectrosc. 2006, 60, 584–591. [Google Scholar] [CrossRef]
- Michel, K.; Bureau, B.; Boussard-Plédel, C.; Jouan, T.; Adama, J.L.; Staubmann, K.; Baumannc, T. Monitoring of pollutant in waste water by infrared spectroscopy using chalcogenide glass optical fibers. Sens. Actuat. B 2004, 101, 252–259. [Google Scholar] [CrossRef]
- Michel, K.; Bureau, B.; Pouvreau, C.; Sangleboeuf, J.C.; Boussard-Pl edel, C.; Jouan, T.; Rouxel, T.; Adam, J.-L.; Staubmann, K.; Steinner, H.; et al. Development of a chalcogenide glass fiber device for in situ pollutant detection. J. Non-Cryst. Sol. 2003, 326, 434. [Google Scholar]
- Charpentier, F.; Bureau, B.; Troles, J.; Boussard-Pledel, C.; Michel-Le Pierres, K.; Smektala, F.; Adam, J.-L. Infrared monitoring of underground CO2 storage using chalcogenide glass fibers. Opt. Mat. 2009, 31, 496–500. [Google Scholar]
- Anne, M.L.; Le Lan, C.; Monbet, V.; Boussard-Pledel, C.; Ropert, M.; Sire, O.; Pouchard, M.; Jard, C.; Lucas, J.; Adam, J.L.; et al. Fiber evanescent wave spectroscopy using the mid-infrared provides useful fingerprints for metabolic profiling in humans. J. Biomed. Opt. 2009, 14, 054033. [Google Scholar] [CrossRef]
- Hocde, S.; Loreal, O.; Sire, O.; Boussard-Pledel, C.; Bureau, B.; Turlin, B.; Keirsse, J.; Leroyer, P.; Lucas, J. Metabolic imaging of tissues by infrared fiber-optic spectroscopy: An efficient tool for medical diagnosis. J. Biomed. Opt. 2004, 9, 404–407. [Google Scholar] [CrossRef]
- Lavi, Y.; Millo, A.; Katzir, A. Flexible ordered bundles of infrared transmitting silver-halide fibers: Design, fabrication, and optical measurement. Appl. Opt. 2006, 45, 5808–5814. [Google Scholar] [CrossRef]
- Lavi, Y.; Millo, A.; Katzir, A. Thin ordered bundles of infrared-transmitting silver halide fibers. Appl. Phys. Lett. 2005, 87, 241122. [Google Scholar] [CrossRef]
- Wilhelm, A.A.; Boussard-Pledel, C.; Coulombier, Q.; Lucas, J.; Bureau, B.; Lucas, P. Development of far-infrared-transmitting te based glasses suitable for carbon dioxide detection and space optics. Adv. Mater. 2007, 19, 3796–3800. [Google Scholar] [CrossRef]
- Houizot, P.; Boussard-Plédel, C.; Faber, A.J.; Cheng, L.K.; Bureau, B.; van Nijnatten, P.A.; Gielesen, W.L.M.; Carmo, J.P.d.; Lucas, J. Infrared single mode chalcogenide glass fiber for space. Opt. Express 2007, 15, 12529–12538. [Google Scholar]
- Danto, S.; Houizot, P.; Boussard-Pledel, C.; Zhang, X.H.; Smektala, F.; Lucas, J. A famiy of far infrared transmitting glasses in the Ga-Ge-Te system for space optics. Adv. Funct. Mat. 2006, 16, 1847. [Google Scholar] [CrossRef]
- Yang, Z.; Lucas, P. Tellurium-based far-infrared transmitting glasses. J. Am. Ceram. Soc. 2009, 92, 2920–2923. [Google Scholar] [CrossRef]
- Bureau, B.; Zhang, X.H.; Smektala, F.; Adam, J.-L.; Troles, J.; Ma, H.-L.; Boussard-Pledel, C.; Lucas, J.; Lucas, P.; Le, C.D.; et al. Recent advances in chalcogenide glasses. J. Non-Cryst. Solids 2004, 345&346, 276–283. [Google Scholar]
- Lucas, J. Infrared glasses. Curr. Opin. Sol. State Mater. Sci. 1999, 4, 181–187. [Google Scholar] [CrossRef]
- Lucas, P.; Yang, Z.; Fah, M.K.; Luo, T.; Jiang, S.; Boussard-Pledel, C.; Anne, M.-L.; Bureau, B. Telluride glasses for far infrared photonic applications. Opt. Mater. Express 2013, 3, 1049. [Google Scholar] [CrossRef]
- Maurugeon, S.; Bureau, B.; Boussard-Plédel, C.; Faber, A.J.; Lucas, P.; Zhang, X.H.; Lucas, J. Selenium modified GeTe4 based glasses optical fibers for far-infrared sensing. Opt. Mat. 2011, 33, 660–663. [Google Scholar] [CrossRef]
- Maurugeon, S.; Bureau, B.; Boussard-Plédel, C.; Troles, J.; Faber, A.J.; Lucas, P.; Zhang, X.H.; Lucas, J. Telluride glass step index fiber for the far infrared. J. Lightwave Tech. 2010, 28, 3358–3363. [Google Scholar]
- Yang, Z.; Luo, T.; Jiang, S.; Geng, J.; Lucas, P. Single-mode low-loss optical fibers for long-wave infrared transmission. Opt. Lett. 2010, 35, 3360–3362. [Google Scholar] [CrossRef]
- Conseil, C.; Shiryaev, V.S.; Cui, S.; Boussard-Pledel, C.; Troles, J.; Velmuzhov, A.P.; Potapov, A.M.; Suchkov, A.I.; Churbanov, M.F.; Bureau, B. Preparation of high purity Te-rich Ge-Te-Se fibers for 5–15 μm infrared range. J. Lightwave Technol. 2013, 31, 1703–1707. [Google Scholar] [CrossRef]
- Lucas, J.; Zhang, X.H. The tellurium halide glasses. J. Non-Cryst. Solids 1990, 125, 1. [Google Scholar] [CrossRef]
- Ma, H.L.; Zhang, X.H.; Lucas, J. “High Tg” tellurium selenium halide glasses. J. Non-Cryst. Solids 1991, 135, 49. [Google Scholar] [CrossRef]
- Zhang, X.H.; Ma, H.L.; Blanchetiere, C.; Lucas, J. Low loss optical fibres of the tellurium halide-based glasses, the tex glasses. J. Non-Cryst. Solids 1993, 161, 327. [Google Scholar] [CrossRef]
- Bureau, B.; Boussard-Pledel, C.; Lucas, P.; Zhang, X.; Lucas, J. Forming glasses from Se and Te. Molecules 2009, 14, 4337–4350. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Bureau, B.; Lucas, P.; Boussard-Pledel, C.; Lucas, J. Glasses for seeing beyond visible. Chemistry 2008, 14, 432–442. [Google Scholar] [CrossRef]
- Martin, R.M.; Lucovsky, G.; Helliwell, K. Intermolecular bonding and lattice dynamics of selenium and tellurium. Phys. Rev. B 1976, 13, 1383–1395. [Google Scholar] [CrossRef]
- Golovchak, R.; Calvez, L.; Bureau, B.; Jain, H. Structural evolution of Ga-Ge-Te glasses by combined exafs and xps analysis. J. Chem. Phys. 2013, 139, 054508. [Google Scholar] [CrossRef]
- Yang, Z.; Gulbiten, O.; Lucas, P.; Luo, T.; Jiang, S. Long-wave infrared-transmitting optical fibers. J. Am. Ceram. Soc. 2011, 94, 1761–1765. [Google Scholar] [CrossRef]
- Churbanov, M.F.; Shiryaev, V.S.; Smetanin, S.V.; Pimenov, V.G.; Zaitseva, E.A.; Kryukova, E.B.; Plotnichenko, V.G. Effect of oxygen impurity on the optical transmission of As2Se3.4 glass. Inorg. Mater. 2001, 37, 1188–1194. [Google Scholar] [CrossRef]
- Jovari, P.; Kaban, I.; Bureau, B.; Wilhelm, A.; Lucas, P.; Beuneu, B.; Zajac, D.A. Structure of Te-rich Te-Ge-X (X = I, Se, Ga) glasses. J. Phys.: Condens. Matter 2010, 22, 404207. [Google Scholar] [CrossRef]
- Calvez, L.; Ma, H.L.; Lucas, J.; Zhang, X.H. Selenium-based glasses and glass ceramics transmitting light from the visible to the far-IR. Adv. Mat. 2007, 19, 129. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lucas, P.; Boussard-Pledel, C.; Wilhelm, A.; Danto, S.; Zhang, X.-H.; Houizot, P.; Maurugeon, S.; Conseil, C.; Bureau, B. The Development of Advanced Optical Fibers for Long-Wave Infrared Transmission. Fibers 2013, 1, 110-118. https://doi.org/10.3390/fib1030110
Lucas P, Boussard-Pledel C, Wilhelm A, Danto S, Zhang X-H, Houizot P, Maurugeon S, Conseil C, Bureau B. The Development of Advanced Optical Fibers for Long-Wave Infrared Transmission. Fibers. 2013; 1(3):110-118. https://doi.org/10.3390/fib1030110
Chicago/Turabian StyleLucas, Pierre, Catherine Boussard-Pledel, Allison Wilhelm, Sylvain Danto, Xiang-Hua Zhang, Patrick Houizot, Sebastien Maurugeon, Clément Conseil, and Bruno Bureau. 2013. "The Development of Advanced Optical Fibers for Long-Wave Infrared Transmission" Fibers 1, no. 3: 110-118. https://doi.org/10.3390/fib1030110



