Er3+/Ho3+-Codoped Fluorotellurite Glasses for 2.7 µm Fiber Laser Materials
Abstract
:1. Introduction
2. Experimental Section
3. Results and Discussion
3.1. Absorption Spectra and Judd-Ofelt Analyses

| Absorption | Wavelength (nm) | Oscillator strength (10−6) | |
|---|---|---|---|
| Measured | calculated | ||
| 4I15/2→4I11/2 | 975 | 0.752 | 0.882 |
| 4I15/2→4I9/2 | 799 | 0.344 | 0.496 |
| 4I15/2→2H11/2 | 521 | 7.448 | 9.269 |
| 4I15/2→4F7/2 | 488 | 3.784 | 3.139 |
| 4I15/2→2G11/2 | 378 | 20.337 | 16.597 |
| Ωt(10−20 cm2) | TF | Fluoride | Phosphate | Germanate | Silicate | |
|---|---|---|---|---|---|---|
| Er3+ | Ω2 | 3.83 | 2.98 | 6.65 | 5.81 | 4.23 |
| Ω4 | 2.00 | 1.40 | 1.52 | 0.85 | 1.04 | |
| Ω6 | 1.37 | 1.04 | 1.11 | 0.28 | 0.61 | |
| Ref. | This work | [16] | [17] | [17] | [17] | |
| Ho3+ | Ω2 | 3.80 | 1.86 | 5.60 | 6.66 | 5.84 |
| Ω4 | 3.28 | 1.90 | 2.72 | 6.06 | 2.38 | |
| Ω6 | 1.10 | 1.32 | 1.87 | 2.26 | 1.75 | |
| Ref. | This work | [18] | [19] | [20] | [21] |
| Transitions | Aed (s−1) | Amd (s−1) | Er3+/Ho3+ | τ (ms) |
| β (%) | ||||
| 4I13/2→4I15/2 | 331.21 | 79.48 | 100.00 | 3.02 |
| 4I11/2→4I15/2 | 317.20 | 0 | 83.47 | 2.63 |
| 4I13/2 | 62.80 | 22.51 | 16.53 | |
| 4I9/2→4I15/2 | 319.89 | 0 | 74.63 | 2.33 |
| 4I13/2 | 104.17 | 0 | 24.30 | |
| 4I11/2 | 4.55 | 4.55 | 1.06 | |
| 4F9/2→4I15/2 | 3,488.20 | 0 | 91.48 | 0.26 |
| 4I13/2 | 175.72 | 0 | 4.61 | |
| 4I11/2 | 144.22 | 0 | 3.78 | |
| 4I9/2 | 4.92 | 0 | 0.13 | |
| 4S3/2→4I15/2 | 2,627.95 | 0 | 62.97 | 0.24 |
| 4I13/2 | 1,329.76 | 0 | 31.86 | |
| 4I11/2 | 82.77 | 0 | 1.98 | |
| 4I9/2 | 132.94 | 0 | 3.19 | |
| 2H11/2→4I15/2 | 11,737.54 | 0 | - | |
| 4F7/2→4I15/2 | 6,764.75 | 0 | - | |
| 4F5/2→4I15/2 | 1,638.04 | 0 | - | |
| 2H9/2→4I15/2 | 3,161.88 | 0 | 40.52 | 0.13 |
| 4I13/2 | 3,480.09 | 0 | 44.60 | |
| 4I11/2 | 1,042.72 | 0 | 13.36 | |
| 4I9/2 | 41.44 | 0 | 0.53 | |
| 4F9/2 | 77.59 | 0 | 0.99 |
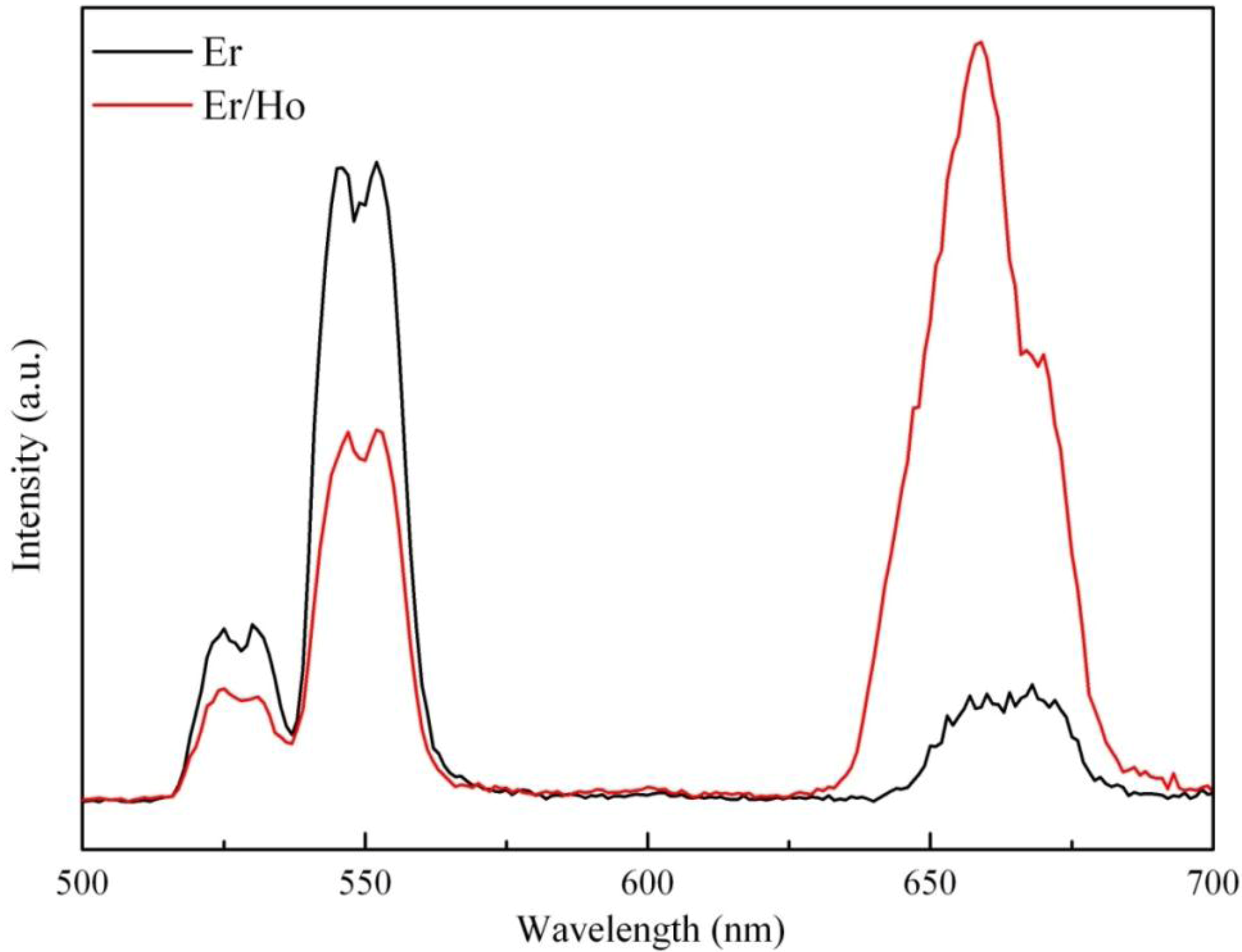
3.2. Fluorescence Spectra Analyses and Energy Transfer Mechanisms
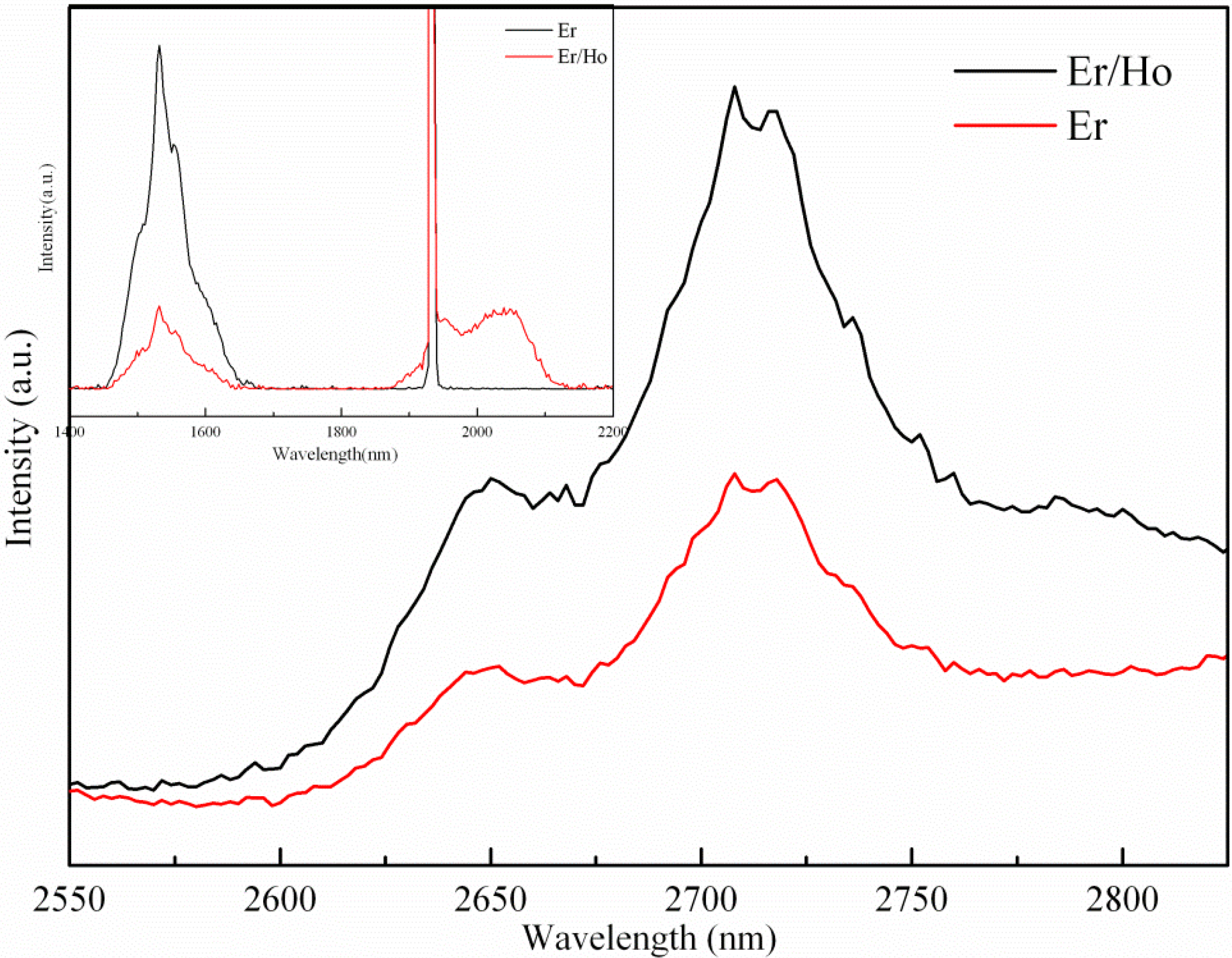
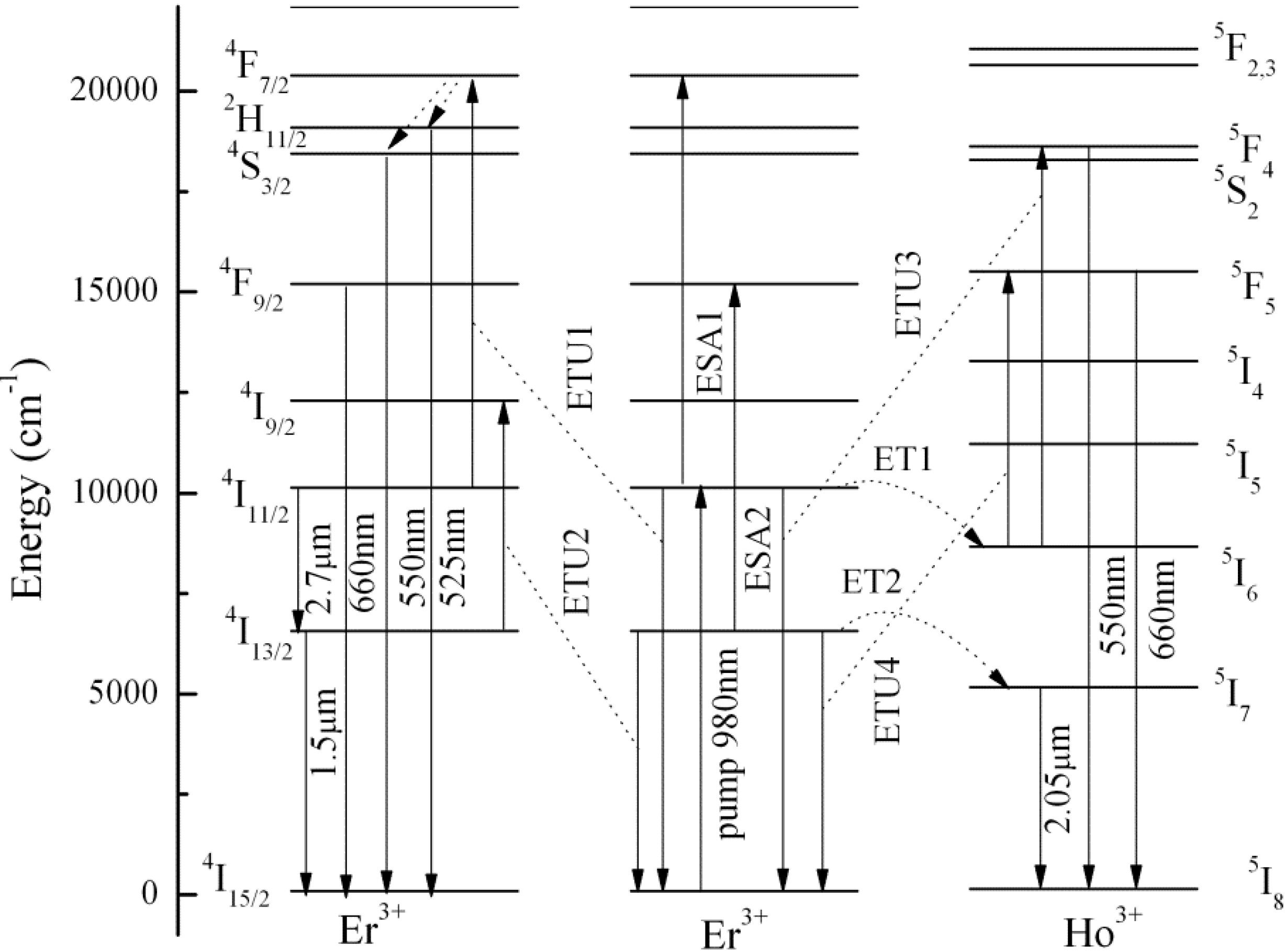
3.3. Fluorescence Spectra Analyses and Energy Transfer Mechanisms
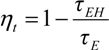
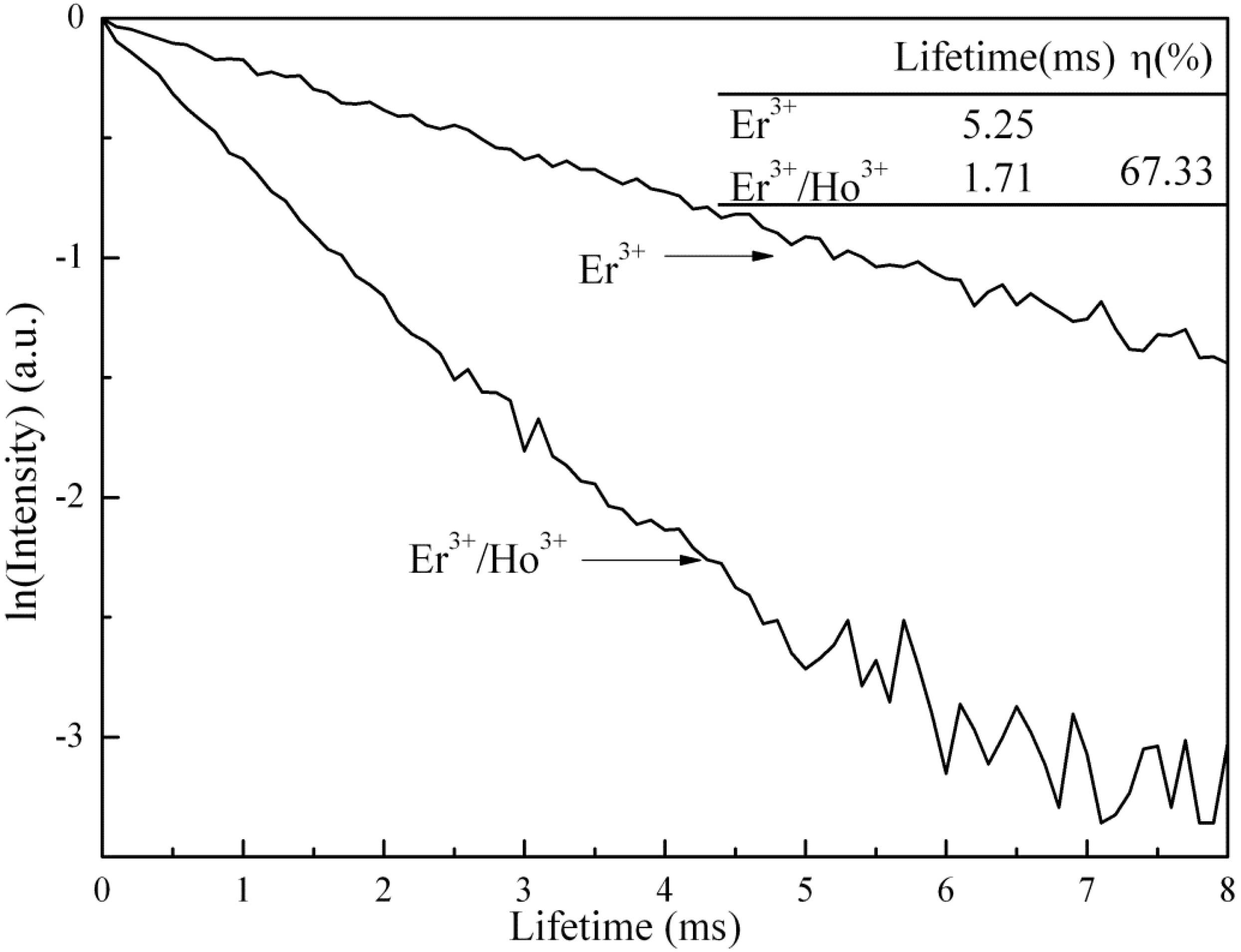
3.4. Cross-Sections Analyses
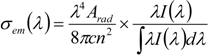
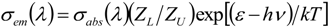

4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Zhu, X.; Jain, R. 10W level diode-pumped compact 2.78 µm ZBLAN fiber laser. Opt. Lett. 2007, 32, 26–28. [Google Scholar] [CrossRef]
- Sanghera, J.S.; Shaw, L.B.; Aggarwal, I.D. Chalcogenide Glass-Fiber-Based Mid-IR Sources and Applications. IEEE J. Sel. Top. Quant. 2009, 15, 114–119. [Google Scholar] [CrossRef]
- Jackson, S.D.; King, T.A.; Pollnau, A.M. Diode-pumped 1.7 W erbium 3 µm fiber laser. Opt. Lett. 1999, 24, 1133–1135. [Google Scholar] [CrossRef]
- Tokita, S.; Murakami, M.; Shimizu, S.; Hashida, M.; Sakabe, S. Liquid-cooled 24 W mid-infrared Er3+:ZBLAN fiber laser. Opt. Lett. 2009, 34, 3062–3064. [Google Scholar] [CrossRef]
- Wang, J.S.; Vogel, E.M.; Snitzer, E. Tellurite glass: a new candidate for fiber devices. Opt. Mater. 1994, 3, 187–203. [Google Scholar] [CrossRef]
- Richards, B.; Tsang, Y.; Binks, D.; Lousteau, J.; Jha, A. Efficient ~2um Tm3+-doped tellurite fiber laser. Opt. Lett. 2008, 33, 402–404. [Google Scholar] [CrossRef]
- Mori, A.; Ohishi, Y.; Sudo, S. Erbium-doped tellurite glass fibre laser and amplifier. Electron. Lett. 1997, 33, 863–864. [Google Scholar] [CrossRef]
- Golding, P.S.; Jackson, S.D.; King, T.A.; Pollnau, M. Energy transfer processes in Er3+-doped and Er3+,Pr3+-codoped ZBLAN glasses. Phys. Rev. B 2000, 62, 856–864. [Google Scholar] [CrossRef]
- Chou, Y.G.; Kim, K.H.; Lee, B.J.; Shin, Y.B.; Kim, Y.S.; Heo, J. Emission properties of the Er3+:4I11/2–4I13/2 transition in Er3+ and Er3+/Tm3+-codoped Ge-Ga-As-S glasses. J. Non-Cryst. Solids 2000, 278, 137–144. [Google Scholar] [CrossRef]
- Zhong, H.; Chen, B.; Ren, G.; Cheng, L.; Yao, L.; Sun, J. 2.7 μm emission of Nd3+, Er3+codoped tellurite glass. J. Appl. Phys. 2009, 106, 083114. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, Z.; Tian, Y.; Zhang, J.; Hu, L. Comparative investigation on the 2.7 μm emission in Er3+/Ho3+ codoped fluorophosphate glass. J. Appl. Phys. 2011, 110, 093106. [Google Scholar] [CrossRef]
- O’Donnell, M.D.; Miller, C.A.; Furniss, D.; Tikhomirov, V.K.; Seddon, A.B. Fluorotellurite glasses with improved mid-infrared transmission. J. Non-Cryst. Solids 2003, 331, 48–57. [Google Scholar] [CrossRef]
- Ofelt, S.G. Intensities of Crystal Spectra of Rare-Earth Ions. J. Chem. Phys. 1962, 37, 511. [Google Scholar] [CrossRef]
- Judd, B.R. Optical Absorption Intensities of Rare-Earth Ions. Phys.l Rev. 1962, 127, 750–761. [Google Scholar]
- Xu, R.R.; Tian, Y.; Wang, M.; Hu, L.L.; Zhang, J.J. Spectroscopic properties of 1.8 μm emission of thulium ions ingermanate glass. Appl. Phys. B 2010, 102, 109–116. [Google Scholar]
- Ivanova, S.; Pelle, F. Strong 1.53 μm to NIR-VIS-UV upconversion in Er-doped fluoride glass for high-efficiency solar cells. J. Opt. Soc. Am. B 2009, 26, 1930–1938. [Google Scholar] [CrossRef]
- Zou, X.; Izumitani, T. Spectroscopic properties and mechanisms of excited state absorption and energy transfer upconversion for Er3+-doped glasses. J. Non-Cryst. Solids 1993, 162, 68–80. [Google Scholar] [CrossRef]
- Peng, B.; Izumitani, T. Optical properties, fluorescence mechanisms and energy transfer in Tm3+, Ho3+ and Tm3+-Ho3+ doped near-infrared laser glasses, sensitized by Yb3+. Opt. Mater. 1995, 4, 797–810. [Google Scholar] [CrossRef]
- Reisfeld, R.; Hormadaly, J. Optical intensities of holmium in tellurite, calibo, and phosphate glasses. J. Chem. Phys. 1976, 64, 3207–3212. [Google Scholar] [CrossRef]
- Xu, R.; Pan, J.; Hu, L.; Zhang, J. 2.0 μm emission properties and energy transfer processes of Yb3+/Ho3+ codoped germanate glass. J. Appl. Phys. 2010, 108, 043522. [Google Scholar] [CrossRef]
- Feng, L.; Wang, J.; Tang, Q.; Liang, L.; Liang, H.; Su, Q. Optical properties of Ho3+-doped novel oxyfluoride glasses. J. Lumin. 2007, 124, 187–194. [Google Scholar] [CrossRef]
- Zhang, X.D.; Xu, T.F.; Dai, S.X.; Nie, Q.H.; Shen, X.; Lu, L.; Zhang, X.H. Investigation of energy transfer and frequency upconversionin Er3+/Ho3+ co-doped tellurite glasses. J. Alloy Compd. 2008, 450, 306–309. [Google Scholar] [CrossRef]
- Pollnan, M. The route toward a diode-pumped 1 W erbium 3 μm fiber laser. IEEE J. Sel. Top. Quant. 1997, 33, 1982–1990. [Google Scholar] [CrossRef]
- Carnall, W.T.; Fields, P.R.; Rajnak, K. Spectral Intensities of the Trivalent Lanthanides and Actinides in Solution. II. Pm3+, Sm3+, Eu3+, Gd3+, Tb3+, Dy3+, and Ho3+. J. Chem. Phys. 1968, 49, 4412–4423. [Google Scholar] [CrossRef]
- Stephen, A.P.; Chase, L.L.; Smith, L.K.; Kway, W.L.; Krupke, W.F. Infrared Cross—Section Measurements for Crystals Doped with Er3+, Tm3+, and Ho3+. IEEE J. Quantum Elect. 1992, 28, 2619–2630. [Google Scholar]
- McCumber, D.E. Theory of Phonon Terminated Optical Masers. Phy. Rev. 1964, 134, A299–A306. [Google Scholar] [CrossRef]
- Guo, Y.; Tian, Y.; Zhang, L.; Hu, L.; Chen, N.K.; Zhang, J. Pr3+-sensitized Er3+-doped bismuthate glass for generating high inversion rates at 2.7 μm wavelength. Opt. Lett. 2012, 37, 3387–3389. [Google Scholar] [CrossRef]
- Tian, Y.; Xu, R.; Zhang, L.; Hu, L.; Zhang, J. Observation of 2.7 μm emission from diode-pumped Er3+/Pr3+-codoped fluorophosphate glass. Opt. Lett. 2011, 36, 109–111. [Google Scholar] [CrossRef]
- Tikhomirov, V.K.; Méndez-Ramos, J.; Rodríguez, V.D.; Furniss, D.; Seddon, A.B. Laser and gain parameters at 2.7 μm of Er3+-doped oxyfluoride transparent glass–ceramics. Opt. Mater. 2006, 28, 1143–1146. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ma, Y.; Huang, F.; Hu, L.; Zhang, J. Er3+/Ho3+-Codoped Fluorotellurite Glasses for 2.7 µm Fiber Laser Materials. Fibers 2013, 1, 11-20. https://doi.org/10.3390/fib1020011
Ma Y, Huang F, Hu L, Zhang J. Er3+/Ho3+-Codoped Fluorotellurite Glasses for 2.7 µm Fiber Laser Materials. Fibers. 2013; 1(2):11-20. https://doi.org/10.3390/fib1020011
Chicago/Turabian StyleMa, Yaoyao, Feifei Huang, Lili Hu, and Junjie Zhang. 2013. "Er3+/Ho3+-Codoped Fluorotellurite Glasses for 2.7 µm Fiber Laser Materials" Fibers 1, no. 2: 11-20. https://doi.org/10.3390/fib1020011
APA StyleMa, Y., Huang, F., Hu, L., & Zhang, J. (2013). Er3+/Ho3+-Codoped Fluorotellurite Glasses for 2.7 µm Fiber Laser Materials. Fibers, 1(2), 11-20. https://doi.org/10.3390/fib1020011




