Insights into the Stress Response Triggered by Kasugamycin in Escherichia coli
Abstract
:1. Introduction
2. Results
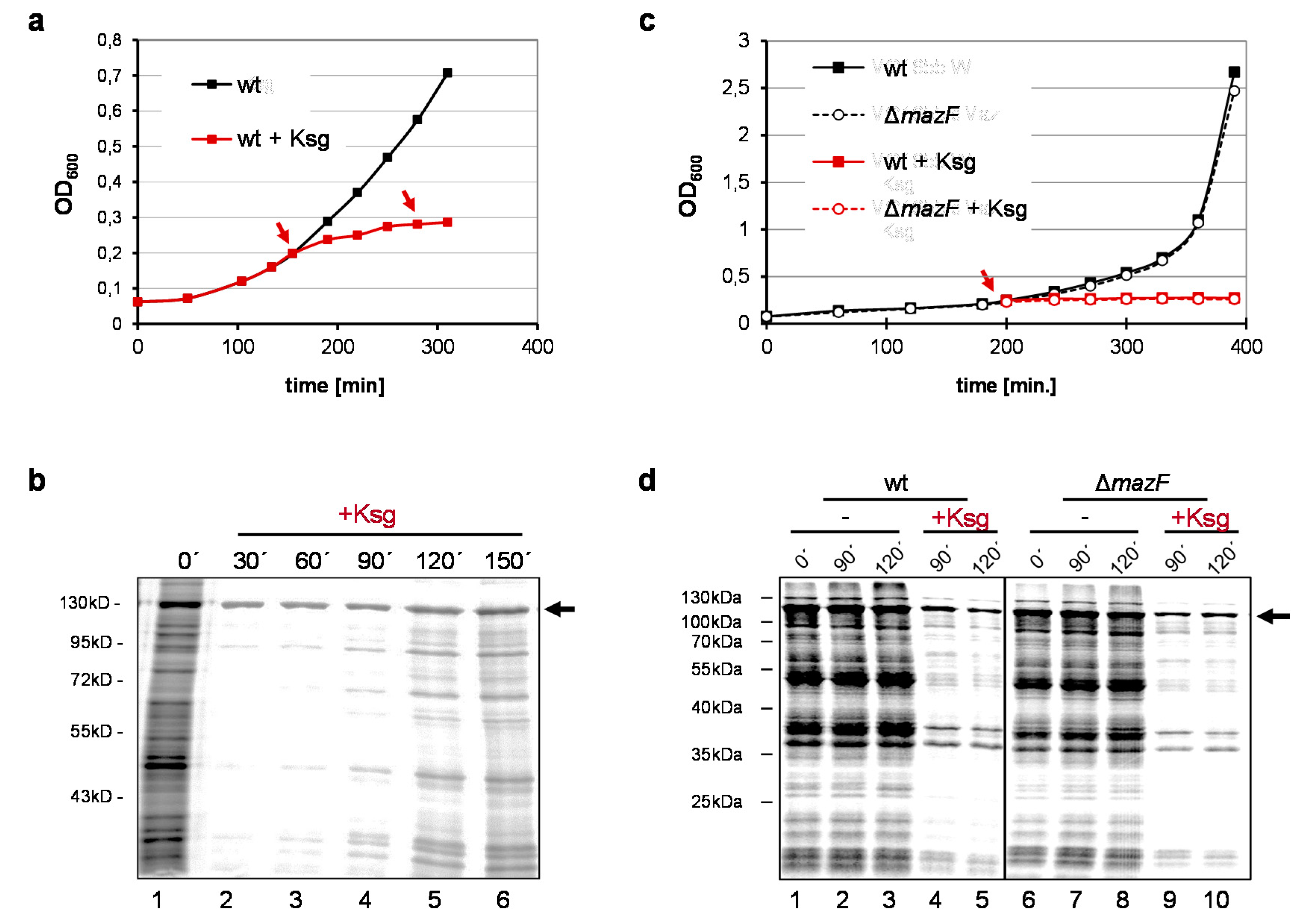
2.1. Selective Protein Synthesis after Prolonged Kasugamycin Treatment
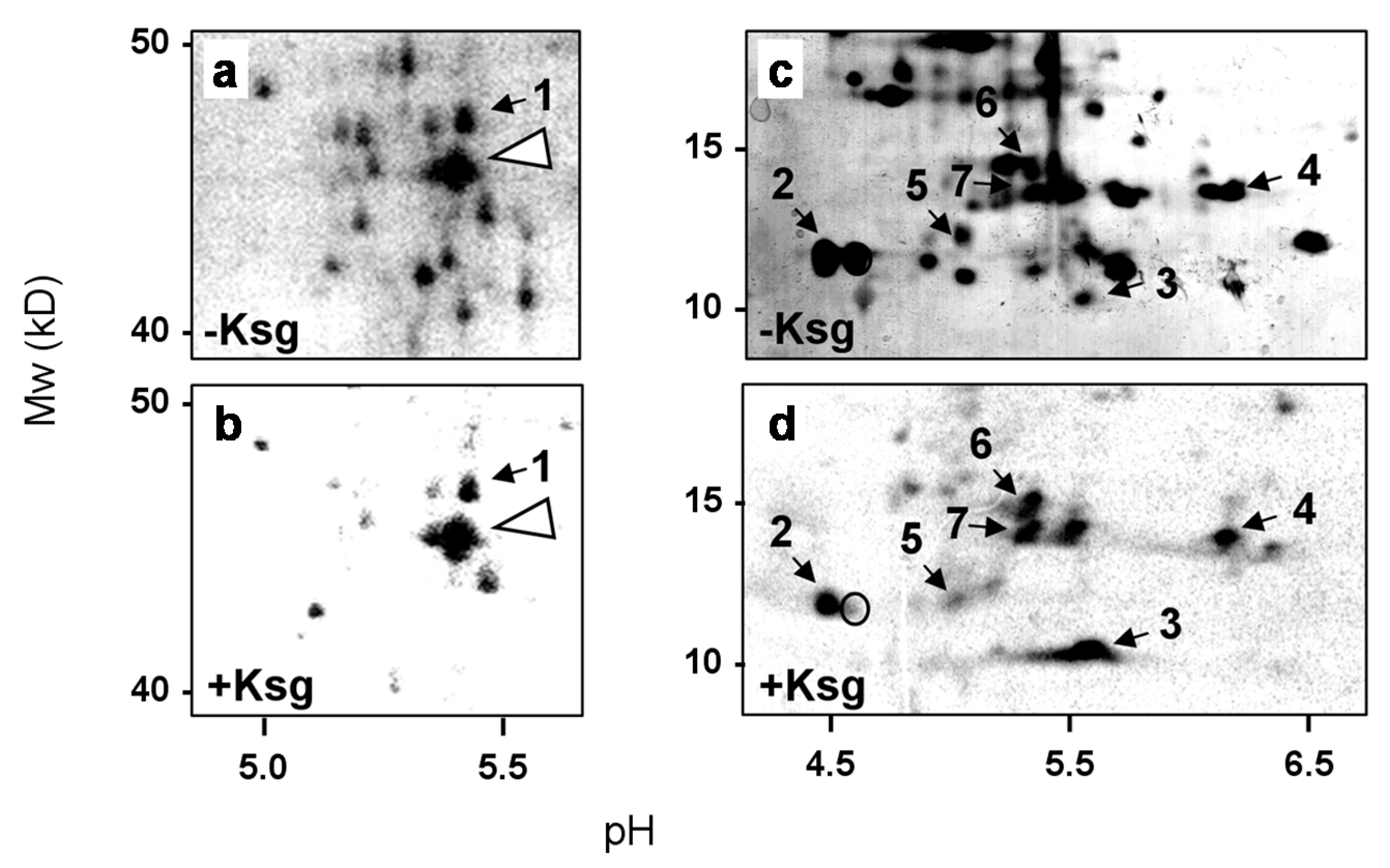
2.2. Identification of Proteins Selectively Synthesized in the Presence of Kasugamycin
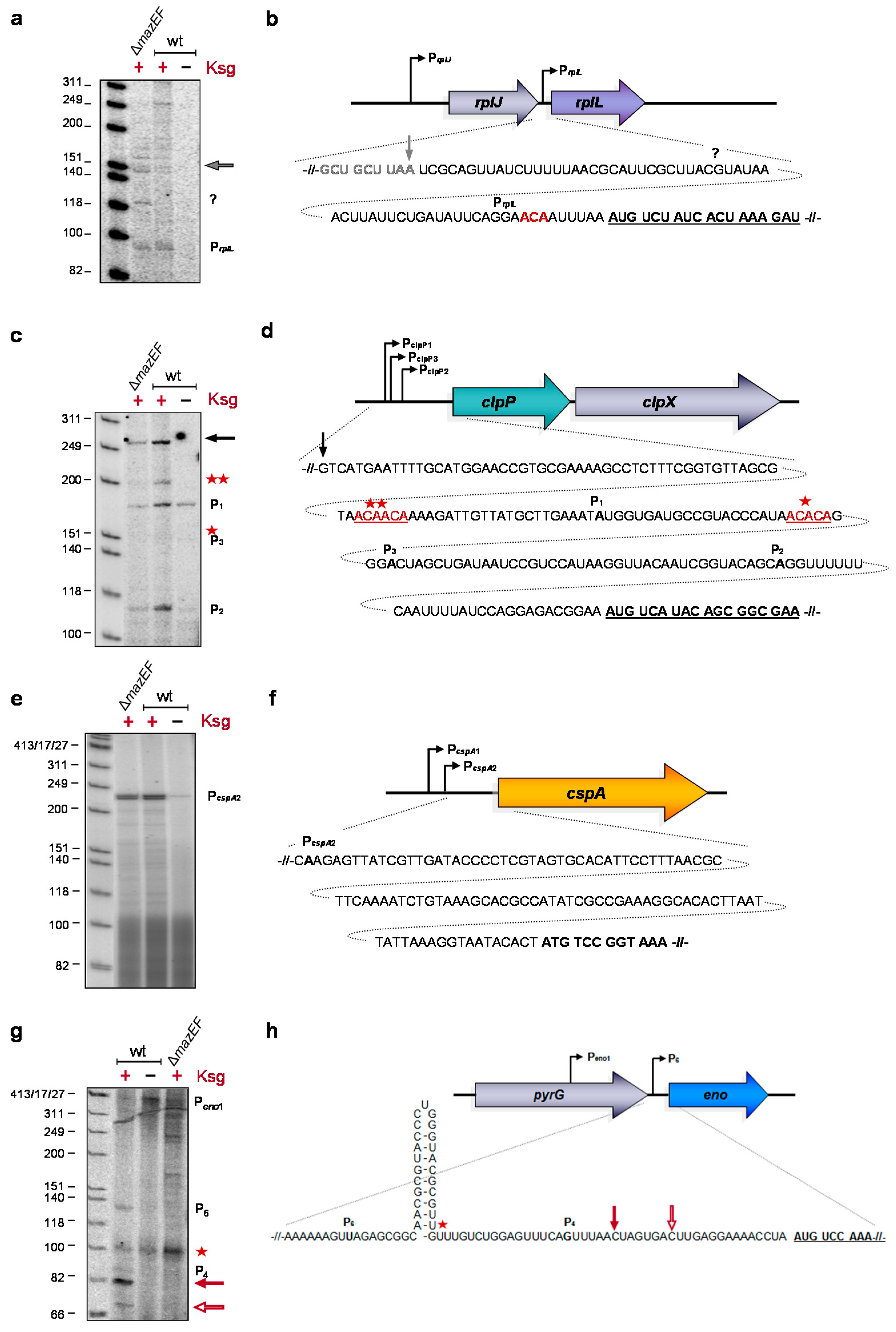
2.3. Formation of Short-leadered and lmRNAs upon Ksg Treatment
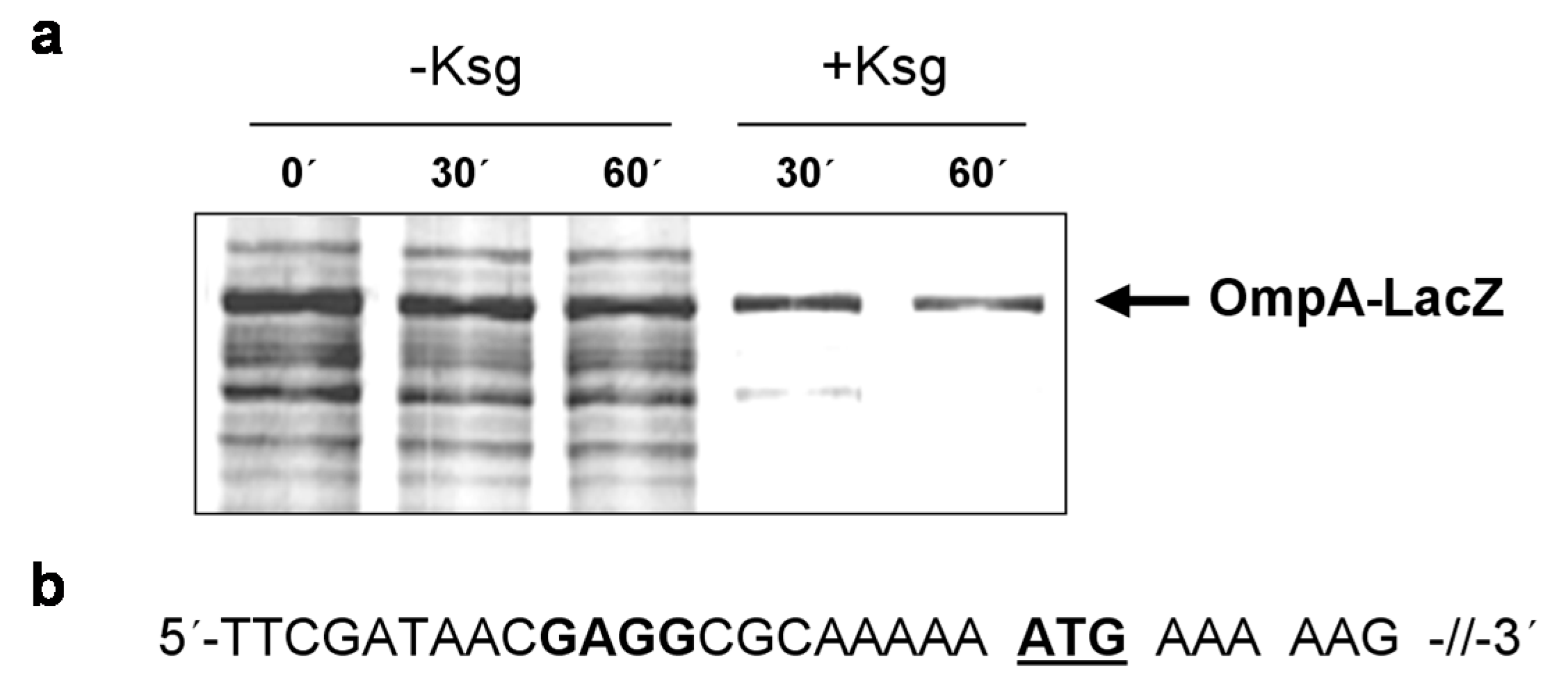
2.4. Selective Translation of mRNAs with Short Leaders upon Kasugamycin Treatment
2.5. Formation of RNA43 upon Kasugamycin Treatment
3. Discussion
3.1. Ribosome Heterogeneity Introduced by Kasugamycin
4. Materials and Methods
4.1. Bacterial Strains, Plasmids, and Oligonucleotides
4.2. Pulse Labeling Experiments
4.3. Proteome Analysis
4.4. Primer Extension Analysis
4.5. Northern Blot Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Okuyama, A.; Machiyama, N.; Kinoshita, T.; Tanaka, N. Inhibition by kasugamycin of initiation complex formation on 30s ribosomes. Biochem. Biophys. Res. Commun. 1971, 43, 196–199. [Google Scholar] [CrossRef]
- Kozak, M.; Nathans, D. Differential inhibition of coliphage MS2 protein synthesis by ribosome-directed antibiotics. J. Mol. Biol. 1972, 70, 41–55. [Google Scholar] [CrossRef]
- Poldermans, B.; Goosen, N.; Van Knippenberg, P.H. Studies on the function of two adjacent N6,N6-dimethyladenosines near the 3′ end of 16 S ribosomal RNA of Escherichia coli. I. The effect of kasugamycin on initiation of protein synthesis. J. Biol. Chem. 1979, 254, 9085–9089. [Google Scholar] [PubMed]
- Woodcock, J.; Moazed, D.; Cannon, M.; Davies, J.; Noller, H.F. Interaction of antibiotics with A- and P-site-specific bases in 16S ribosomal RNA. EMBO J. 1991, 10, 3099–3103. [Google Scholar] [PubMed]
- Schluenzen, F.; Takemoto, C.; Wilson, D.N.; Kaminishi, T.; Harms, J.M.; Hanawa-Suetsugu, K.; Szaflarski, W.; Kawazoe, M.; Shirouzu, M.; Nierhaus, K.H.; et al. The antibiotic kasugamycin mimics mRNA nucleotides to destabilize tRNA binding and inhibit canonical translation initiation. Nat. Struct. Mol. Biol. 2006, 13, 871–878. [Google Scholar] [CrossRef] [PubMed]
- Schuwirth, B.S.; Day, J.M.; Hau, C.W.; Janssen, G.R.; Dahlberg, A.E.; Cate, J.H.D.; Vila-Sanjurjo, A. Structural analysis of kasugamycin inhibition of translation. Nat. Struct. Mol. Biol. 2006, 13, 879–886. [Google Scholar] [CrossRef] [PubMed]
- Helser, T.L.; Davies, J.E.; Dahlberg, J.E. Change in methylation of 16s ribosomal RNA associated with mutation to kasugamycin resistance in Escherichia coli. Nat. New Biol. 1971, 233, 12–14. [Google Scholar] [CrossRef] [PubMed]
- Van Knippenberg, P.H.; Van Kimmenade, J.M.; Heus, H.A. Phylogeny of the conserved 3′-terminal structure of the RNA of small ribosomal subunits. Nucleic Acids Res. 1984, 12, 2595–2604. [Google Scholar] [CrossRef] [PubMed]
- Rife, J.P.; Moore, P.B. The structure of a methylated tetraloop in 16s ribosomal RNA. Structure 1998, 6, 747–756. [Google Scholar] [CrossRef]
- Hobartner, C.; Micura, R. Bistable secondary structures of small RNAs and their structural probing by comparative imino proton nmr spectroscopy. J. Mol. Biol. 2003, 325, 421–431. [Google Scholar] [CrossRef]
- Chin, K.; Shean, C.S.; Gottesman, M.E. Resistance of lambda cI translation to antibiotics that inhibit translation initiation. J. Bacteriol. 1993, 175, 7471–7473. [Google Scholar] [PubMed]
- Moll, I.; Bläsi, U. Differential inhibition of 30S and 70S translation initiation complexes on leaderless mRNA by kasugamycin. Biochem. Biophys. Res. Commun. 2002, 297, 1021–1026. [Google Scholar] [CrossRef]
- Pioletti, M.; Schlunzen, F.; Harms, J.; Zarivach, R.; Gluhmann, M.; Avila, H.; Bashan, A.; Bartels, H.; Auerbach, T.; Jacobi, C.; et al. Crystal structures of complexes of the small ribosomal subunit with tetracycline, edeine and IF3. EMBO J. 2001, 20, 1829–1839. [Google Scholar] [CrossRef] [PubMed]
- Moll, I.; Hirokawa, G.; Kiel, M.C.; Kaji, A.; Blasi, U. Translation initiation with 70S ribosomes: An alternative pathway for leaderless mRNAs. Nucleic Acids Res. 2004, 32, 3354–3363. [Google Scholar] [CrossRef] [PubMed]
- Kaberdina, A.C.; Szaflarski, W.; Nierhaus, K.H.; Moll, I. An unexpected type of ribosomes induced by kasugamycin: A look into ancestral times of protein synthesis? Mol. Cell 2009, 33, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, M.A.; Fricke, J.; Pedersen, S. Ribosomal protein S1 is required for translation of most, if not all, natural mRNAs in Escherichia coli in vivo. J. Mol. Biol. 1998, 280, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Moll, I.; Grill, S.; Grundling, A.; Blasi, U. Effects of ribosomal proteins S1, S2 and the DeaD/CsdA DEAD-box helicase on translation of leaderless and canonical mRNAs in Escherichia coli. Mol. Microbiol. 2002, 44, 1387–1396. [Google Scholar] [CrossRef] [PubMed]
- Moll, I.; Resch, A.; Blasi, U. Discrimination of 5′-terminal start codons by translation initiation factor 3 is mediated by ribosomal protein S1. FEBS Lett. 1998, 436, 213–217. [Google Scholar] [CrossRef]
- Moll, I.; Grill, S.; Gualerzi, C.O.; Blasi, U. Leaderless mRNAs in bacteria: Surprises in ribosomal recruitment and translational control. Mol. Microbiol. 2002, 43, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Engelberg-Kulka, H.; Sat, B.; Reches, M.; Amitai, S.; Hazan, R. Bacterial programmed cell death systems as targets for antibiotics. Trends Microbiol. 2004, 12, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Sat, B.; Hazan, R.; Fisher, T.; Khaner, H.; Glaser, G.; Engelberg-Kulka, H. Programmed cell death in Escherichia coli: Some antibiotics can trigger mazEFLethality. J. Bacteriol. 2001, 183, 2041–2045. [Google Scholar] [CrossRef] [PubMed]
- Hayes, F. Toxins-antitoxins: Plasmid maintenance, programmed cell death, and cell cycle arrest. Science 2003, 301, 1496–1499. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, J.; Hara, H.; Kato, I.; Inouye, M. Insights into the mRNA cleavage mechanism by MazF, an mRNA interferase. J. Biol. Chem. 2005, 280, 3143–3150. [Google Scholar] [CrossRef] [PubMed]
- Vesper, O.; Amitai, S.; Belitsky, M.; Byrgazov, K.; Kaberdina, A.C.; Engelberg-Kulka, H.; Moll, I. Selective translation of leaderless mRNAs by specialized ribosomes generated by MazF in Escherichia coli. Cell 2011, 147, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Sauert, M.; Wolfinger, M.T.; Vesper, O.; Muller, C.; Byrgazov, K.; Moll, I. The MazF-regulon: A toolbox for the post-transcriptional stress response in Escherichia coli. Nucleic Acids Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Sauert, M.; Temmel, H.; Moll, I. Heterogeneity of the translational machinery: Variations on a common theme. Biochimie 2015, 114, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Romero, D.A.; Hasan, A.H.; Lin, Y.F.; Kime, L.; Ruiz-Larrabeiti, O.; Urem, M.; Bucca, G.; Mamanova, L.; Laing, E.E.; van Wezel, G.P.; et al. A comparison of key aspects of gene regulation in streptomyces coelicolor and Escherichia coli using nucleotide-resolution transcription maps produced in parallel by global and differential RNA sequencing. Mol. Microbiol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, J.; Pollitt, N.S.; Inouye, M. Major cold shock protein of Escherichia coli. Proc. Natl. Acad. Sci. USA 1990, 87, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Chandran, V.; Luisi, B.F. Recognition of enolase in the Escherichia coli RNA degradosome. J. Mol. Biol. 2006, 358, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Ralling, G.; Linn, T. Relative activities of the transcriptional regulatory sites in the rplKAJLrpoBC gene cluster of Escherichia coli. J. Bacteriol. 1984, 158, 279–285. [Google Scholar] [PubMed]
- Sabina, J.; Dover, N.; Templeton, L.J.; Smulski, D.R.; Soll, D.; LaRossa, R.A. Interfering with different steps of protein synthesis explored by transcriptional profiling of Escherichia coli K-12. J. Bacteriol. 2003, 185, 6158–6170. [Google Scholar] [CrossRef] [PubMed]
- Gama-Castro, S.; Jimenez-Jacinto, V.; Peralta-Gil, M.; Santos-Zavaleta, A.; Penaloza-Spinola, M.I.; Contreras-Moreira, B.; Segura-Salazar, J.; Muniz-Rascado, L.; Martinez-Flores, I.; Salgado, H.; et al. Regulondb (version 6.0): Gene regulation model of Escherichia coli K-12 beyond transcription, active (experimental) annotated promoters and textpresso navigation. Nucleic Acids Res. 2008, 36, D120–D124. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhu, L.; Zhang, J.; Inouye, M. Characterization of ChpBK, an mRNA interferase from Escherichia coli. J. Biol. Chem. 2005, 280, 26080–26088. [Google Scholar] [CrossRef] [PubMed]
- VanBogelen, R.A.; Neidhardt, F.C. Ribosomes as sensors of heat and cold shock in Escherichia coli. Proc. Natl. Acad. Sci. USA 1990, 87, 5589–5593. [Google Scholar] [CrossRef] [PubMed]
- Tenson, T.; Mankin, A. Antibiotics and the ribosome. Mol. Microbiol. 2006, 59, 1664–1677. [Google Scholar] [CrossRef] [PubMed]
- Backendorf, C.; Ravensbergen, C.J.; Van der Plas, J.; van Boom, J.H.; Veeneman, G.; Van Duin, J. Basepairing potential of the 3′ terminus of 16S RNA: Dependence on the functional state of the 30S subunit and the presence of protein S21. Nucleic Acids Res. 1981, 9, 1425–1444. [Google Scholar] [CrossRef] [PubMed]
- Hazan, R.; Sat, B.; Engelberg-Kulka, H. Escherichia coli mazEF-mediated cell death is triggered by various stressful conditions. J. Bacteriol. 2004, 186, 3663–3669. [Google Scholar] [CrossRef] [PubMed]
- Deusser, E.; Wittmann, H.G. Ribosomal proteins: Variation of the protein composition in Escherichia coli ribosomes as function of growth rate. Nature 1972, 238, 269–270. [Google Scholar] [CrossRef] [PubMed]
- Helgstrand, M.; Mandava, C.S.; Mulder, F.A.A.; Liljas, A.; Sanyal, S.; Akke, M. The ribosomal stalk binds to translation factors IF2, EF-Tu, EF-G and RF3 via a conserved region of the L12 C-terminal domain. J. Mol. Biol. 2007, 365, 468–479. [Google Scholar] [CrossRef] [PubMed]
- Gordiyenko, Y.; Deroo, S.; Zhou, M.; Videler, H.; Robinson, C.V. Acetylation of L12 increases interactions in the Escherichia coli ribosomal stalk complex. J. Mol. Biol. 2008, 380, 404–414. [Google Scholar] [CrossRef] [PubMed]
- Byrgazov, K.; Vesper, O.; Moll, I. Ribosome heterogeneity: Another level of complexity in bacterial translation regulation. Curr. Opin. Microbiol. 2013, 16, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, W.V. Functional specialization of ribosomes? Trends Biochem. Sci. 2011, 36, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.H. Experiments in Molecular Genetics; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 1972. [Google Scholar]
- Baba, T.; Ara, T.; Hasegawa, M.; Takai, Y.; Okumura, Y.; Baba, M.; Datsenko, K.A.; Tomita, M.; Wanner, B.L.; Mori, H. Construction of Escherichia coli K-12 in-frame, single-gene knockout mutants: The keio collection. Mol. Syst. Biol. 2006, 2. [Google Scholar] [CrossRef] [PubMed]
- Sonnleitner, E.; Moll, I.; Blasi, U. Functional replacement of the Escherichia coli HFQ gene by the homologue of pseudomonas aeruginosa. Microbiology 2002, 148, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Pall, G.S.; Hamilton, A.J. Improved northern blot method for enhanced detection of small RNA. Nat. Protoc. 2008, 3, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. Nih image to imagej: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]

| # | Gene | Protein Function | Length of 5′-UTR | MazF Processing in the 5′-UTR |
|---|---|---|---|---|
| 1 | eno | Enolase, enzyme of glycolysis, phosphoprotein, component of the RNA degradosome | 76 nts | No |
| 2 | rplL | ribosomal protein L7/L12 | rplJ-rplL-operon | Yes |
| 3 | cspA | cold-shock protein A | 109 nts | No |
| 4 | rplI | ribosomal protein L9 | rpsF-rplI-operon | No |
| 5 | grcA | stress-induced alternate pyruvate formate-lyase subunit | 74 nts | Yes |
| 6 | groES | chaperone | 72 nts | No |
| 7 | uspA | universal stress protein A | 111 nts | No |
| 8 | dnaK | heat-shock chaperone | 115 nts | No |
| 9 | dnaJ | heat-shock chaperone | dnaK-dnaJ operon | No |
| 10 | rimL | ribosomal-protein-L12-serine acetyltransferase | 196 nts | No |
| 11 | clpP | Proteolytic subunit of ClpXP and ClpAP ATP-dependent proteases | 32 nts | Yes |
| 12 | groEL | chaperone | 409 nts | Yes |
| 13 | rbfA | ribosome binding factor required for processing of 16S rRNA | infB-operon | No |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Müller, C.; Sokol, L.; Vesper, O.; Sauert, M.; Moll, I. Insights into the Stress Response Triggered by Kasugamycin in Escherichia coli. Antibiotics 2016, 5, 19. https://doi.org/10.3390/antibiotics5020019
Müller C, Sokol L, Vesper O, Sauert M, Moll I. Insights into the Stress Response Triggered by Kasugamycin in Escherichia coli. Antibiotics. 2016; 5(2):19. https://doi.org/10.3390/antibiotics5020019
Chicago/Turabian StyleMüller, Christian, Lena Sokol, Oliver Vesper, Martina Sauert, and Isabella Moll. 2016. "Insights into the Stress Response Triggered by Kasugamycin in Escherichia coli" Antibiotics 5, no. 2: 19. https://doi.org/10.3390/antibiotics5020019





