Implementation of a Clinical Decision Support Alert for the Management of Clostridium difficile Infection
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Design
2.2. Institutional Clinical Decision Support for C. difficile
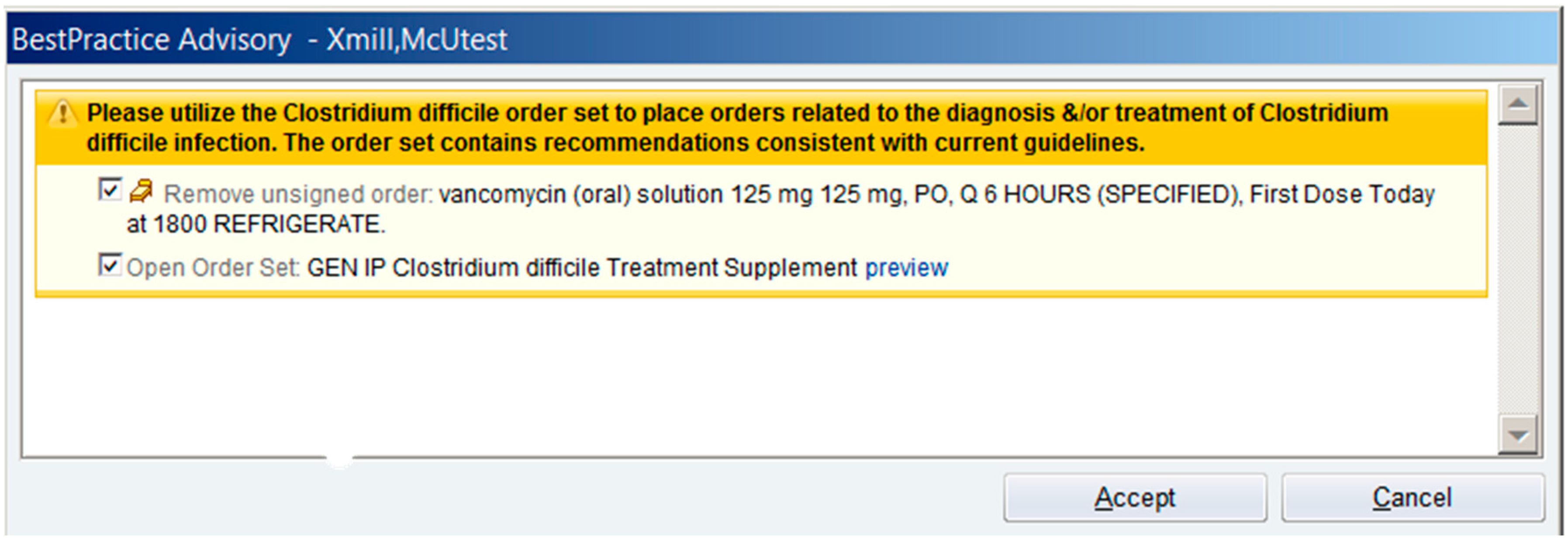
2.3. Outcomes
2.4. Statistical Analysis
3. Results and Discussion
3.1. Results

| Demographics | Historical Group (n = 165) | Intervention Group (n = 146) | p Value |
|---|---|---|---|
| Age, y, mean ± SD | 60.7 ± 17.3 | 58.6 ± 14.9 | 0.184 |
| Male sex, no. (%) | 79 (47.9) | 69 (47.3) | 0.292 |
| BMI, mean ± SD | 29.5 ± 10.1 | 29.3 ± 8.7 | 0.970 |
| Albumin, mean ± SD | 3.2 ± 0.8 | 3.4 ± 0.7 | 0.029 |
| Mild/moderate infection, no. (%) | 120 (72.7) | 99 (67.8) | 0.384 |
| Severe infection, no. (%) | 45 (27.3) | 47 (32.2) | 0.384 |
| Complicated infection, no (%) | 25 (15.2) | 29 (19.9) | 0.296 |
| ICU admission when NAAT positive, no. (%) | 29 (17.6) | 46 (31.5) | 0.005 |
| Hospital-acquired, no. (%) | 66 (40.0) | 70 (47.9) | 0.176 |
| Infectious diseases consult, no. (%) | 22 (13.3) | 21 (14.4) | 0.870 |
| Patients with recurrent CDIs, no. (%) | 34 (20.6) | 41 (28.1) | 0.144 |
| Historical Group n = 165 | Intervention Group n = 146 | p Value | Intervention Group With Use of Order Set n = 45 | Intervention Group Without Use of Order Set n = 101 | p Value | |
|---|---|---|---|---|---|---|
| Order set utilization | 2 (1.2) | 45 (30.8) | <0.001 | NA | NA | NA |
| Treatment compliant with guideline | 115 (69.7) | 104 (71.2) | 0.605 | 36 (80.0) | 68 (67.3) | 0.165 |
| Reasons for Non-Compliance | ||||||
| Given metronidazole for severe infection | 10/50 (20.0) | 20/45 * (44.4) | 0.015 | 4/9 (44.4) | 16/36 * (44.4) | 1.000 |
| Given vancomycin for mild/moderate infection | 12/50 (24.0) | 5/45 (11.1) | 0.116 | 1/9 (11.1) | 4/36 (11.1) | 1.000 |
| Combination therapy given but not needed | 6/50 (12.0) | 7/45 (15.6) | 0.767 | 3/9 (33.3) | 4/36 (11.1) | 0.131 |
| Patient required combination therapy but did not receive | 17/50 (34.0) | 4/45 (8.9) | 0.006 | 0/9 (0.0) | 4/36 (11.1) | 0.569 |
| Vancomycin taper indicated but not given | 2/50 (4.0) | 4/45 (8.9) | 0.418 | 1/9 (11.1) | 3/36 (8.3) | 1.000 |
| Other ** | 3/50 (6.0) | 5/45 (11.1) | 0.470 | 0/9 (0.0) | 5/36 (13.9) | 0.566 |
| Initial Treatment Regimen Included Vancomycin n = 48 | Initial Treatment Regimen Did Not Include Vancomycin n = 98 | p value | |
|---|---|---|---|
| Order set utilization, no. (%) | 30/48 (62.5) | 15/98 (15.3) | <0.001 |
| Treatment compliant with guideline | 29/48 (60.4) | 71/98 (74.4) | 0.184 |
| Guideline compliance when order set utilized | 22/30 (73.3) | 11/15 (73.3) | 1.000 |
| Guideline compliance when order set not utilized | 7/18 (38.9) | 60/83 (72.3) | 0.012 |
3.2. Discussion
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Lassa, F.C.; Mu, Y.; Bamberg, W.M.; Beldavs, Z.G.; Dumyati, G.K.; Dunn, J.R.; Farley, M.M.; Holzbauer, S.M.; Meek, J.I.; Phipps, E.C.; et al. Burden of Clostridium difficile infection in the United States. N. Engl. J. Med. 2015, 372, 825–834. [Google Scholar]
- Campbell, R.; Dean, B.; Nathanson, B.; Haidar, T.; Strauss, M.; Thomas, S. Length of stay and hospital costs among high-risk patients with hospital-origin Clostridium difficile-associated diarrhea. J. Med. Econ. 2013, 16, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Reveles, K.R.; Lee, G.C.; Boyd, N.K.; Frei, C.R. The rise in Clostridium difficile infection incidence among hospitalized adults in the United States: 2001–2010. Am. J. Infect. Control. 2014, 42, 1028–1032. [Google Scholar] [CrossRef] [PubMed]
- Zimlichman, E.; Henderson, D.; Tamir, O.; Franz, C.; Song, P.; Yamin, C.K.; Keohane, C.; Denham, C.R.; Bates, D.W. Health-care associated infections. JAMA Intern. Med. 2013, 173, 2039–2046. [Google Scholar] [CrossRef] [PubMed]
- Maroo, S.; LaMont, J.T. Recurrent Clostridium difficile. Gastroenterology 2006, 130, 1311–1316. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.H.; Gerding, D.N.; Johnson, S.; Kelly, C.P.; Loo, V.G.; McDonald, L.C.; Pepin, J.; Wilcox, M.H. Clinical practice guidelines for Clostridium difficile infection in adults: 2010 update by the Society for Healthcare Epidemiology of America and the Infectious Diseases Society of America. Infect. Control. Hosp. Epidemol. 2010, 31, 431–455. [Google Scholar] [CrossRef] [PubMed]
- Surawicz, C.M.; Brandt, L.J.; Binion, D.G.; Ananthakrishnan, A.N.; Curry, S.R.; Gilligan, P.H.; McFarland, L.V.; Mellow, M.; Zuckerbraun, B.S. Guidelines for diagnosis, treatment, and prevention of Clostridium difficile infections. Am. J. Gastroenterol. 2013, 108, 478–498. [Google Scholar] [CrossRef] [PubMed]
- Dellit, T.H.; Owens, R.C.; McGowan, J.E., Jr.; Gerding, D.N.; Weinstein, R.A.; Burke, J.P.; Huskins, C.; Paterson, D.L.; Fishman, N.O.; Carpenter, C.F.; et al. Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America guidelines for developing an institutional program to enhance antimicrobial stewardship. Clin. Infect. Dis. 2007, 44, 159–177. [Google Scholar] [CrossRef] [PubMed]
- Braxton, C.C.; Gerstenberger, P.A.; Cox, G.G. Improving antibiotic stewardship: Order sest implementation to improve prophylactic antimicrobial prescribing in the outpatient surgical setting. J. Ambul. Care Manage. 2010, 33, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Hall, A.B.; Montero, J.; Cobian, J.; Regan, T. The effects of an electronic order set on vancomycin dosing in the ED. Am. J. Emerg. Med. 2015, 33, 92–94. [Google Scholar] [CrossRef] [PubMed]
- Pestotnik, S.L. Expert clinical decision support systems to enhance antimicrobial stewardship programs: Insights from the Society of Infectious Diseases Pharmacists. Pharmacotherapy 2005, 25, 1116–1125. [Google Scholar] [CrossRef] [PubMed]
- Zar, F.A.; Bakkanagari, S.R.; Moorthi, K.M.L.S.T.; Davis, M.B. A comparison of vancomycin and metronidazole for the treatment of Clostridium difficile-associated diarrhea, stratified by disease severity. Clin. Infect. Dis. 2007, 45, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.; Louie, T.J.; Gerding, D.N.; Cornely, O.A.; Chasan-Taber, S.; Fitts, D.; Gelone, S.P.; Broom, C.; Davidson, D.M.; for the Polymer Alternative for CDI Treatment (PACT) Investigators. Vancomycin, metronidazole, or tolevamer for Clostridium difficile infection: Results from two multinational, randomized, controlled trials. Clin. Infect. Dis. 2014, 59, 345–354. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Revolinski, S. Implementation of a Clinical Decision Support Alert for the Management of Clostridium difficile Infection. Antibiotics 2015, 4, 667-674. https://doi.org/10.3390/antibiotics4040667
Revolinski S. Implementation of a Clinical Decision Support Alert for the Management of Clostridium difficile Infection. Antibiotics. 2015; 4(4):667-674. https://doi.org/10.3390/antibiotics4040667
Chicago/Turabian StyleRevolinski, Sara. 2015. "Implementation of a Clinical Decision Support Alert for the Management of Clostridium difficile Infection" Antibiotics 4, no. 4: 667-674. https://doi.org/10.3390/antibiotics4040667





