Livestock-Associated MRSA: The Impact on Humans
Abstract
:1. Methicillin-Resistant Staphylococcus aureus
2. Host Specificity of S. aureus and of MRSA
| Clonal Lineages/Complexes | Human | Livestock | Companion Animals | Marine Mammals | Wildlife | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pig | Cattle | Sheep, Goat | Chicken | Turkey | Rabbit | Horse | Dog | Cat | Boar | Ruminants: Red Deer, Roe | Glires: Hare, Rat | Birds | |||
| Originally Known from Humans | |||||||||||||||
| CC1; ST1 | MSSA, HA-MRSA, CA-MRSA, PVL+ | MSSA 1, MRSA [37] | MRSA, subclinical mastitis [35,37] | MSSA | MRSA, [38] | ||||||||||
| CC5; ST5 | MSSA, HA-MRSA, CA-MRSA, PVL+ | MRSA [32] | MSSA [25] MRSA | MRSA [38] | |||||||||||
| CC5 | HA-MRSA (Europe) | MRSA [38] | MRSA | [39] | |||||||||||
| CC7; ST7 | MSSA, MRSA | MSSA | MSSA 1 | ||||||||||||
| CC8; ST8 | MSSA, HA-MRSA, CA-MRSA, PVL+ | CA-MRSA, PVL+ (USA) 2 | MRSA (Northern America, Europe) | MRSA, PVL+ 1 | MRSA [39] | MRSA [39] | |||||||||
| CC8; ST239 | HA-MRSA | MRSA (Belgium) | |||||||||||||
| CC8; ST254 | HA-MRSA | MRSA (Central Europe) | |||||||||||||
| CC15; ST15 | MSSA, HA-MRSA 2 | MSSA 1 | |||||||||||||
| CC22; ST22 | MSSA, HA-MRSA, CA-MRSA, PVL+ | MRSA 1 Own observation | MRSA [40] | MRSA [40] | |||||||||||
| CC30; ST30 | MSSA, CA-MRSA, PVL+ | MSSA (Germany) | |||||||||||||
| CC30; ST36 | HA-MRSA | HA-MRSA | HA-MRSA (New Zealand) | ||||||||||||
| CC45; ST45 | MSSA, HA-MRSA | MSSA | MSSA | ||||||||||||
| CC59; ST59 | MSSA, HA-MRSA, CA-MRSA | MRSA [31] | MRSA (Asia) | ||||||||||||
| CC398; ST398 human subpopulation | MSSA, MRSA, PVL+ [17,41] | ||||||||||||||
| CC398; ST398 Animal sub-population, LA-MRSA | MSSA, LA-MRSA 3 | MSSA, MRSA | MRSA | MRSA [42,43] | MRSA | MRSA | MRSA [44,45] | MRSA 4 | LA-MRSA | LA-MRSA | |||||
| Originally Known from Animals | |||||||||||||||
| CC9; ST9 | MSSA, LA-MRSA 4 | MSSA, LA-MRSA | LA-MRSA 1 | LA-MRSA | |||||||||||
| CC97; ST97 | MSSA 1, LA-MRSA 1 | MSSA [19] | MSSA, LA-MRSA 1 [19] | ||||||||||||
| CC130; ST130 | MRSA 1[37,46] | MRSA [47] | MRSA [47] | MRSA 1 (own observation) | MRSA [48] | MRSA [48] | MRSA [47] | MRSA [46,49] MSSA [49] | MRSA [46,47,49] | MRSA [50] | |||||
| ST425 | MRSA [51] | MRSA [51] | MSSA [52] | ||||||||||||
| CC133 | MSSA [53,54] | MSSA [52] | |||||||||||||
| CC522 | MSSA [53,54] | ||||||||||||||
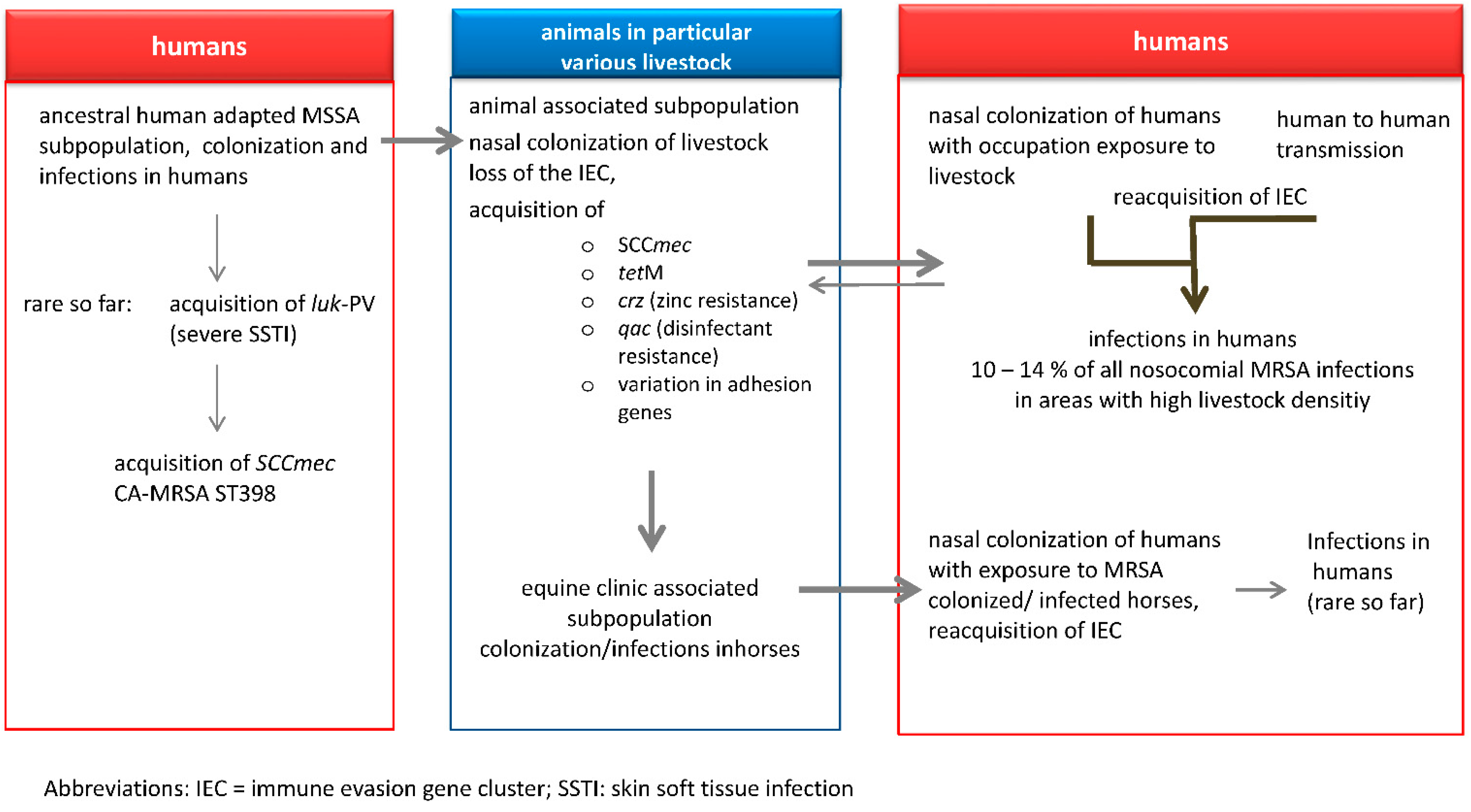
3. LA-MRSA in Livestock Animals
4. LA-MRSA CC398 in Other Animals besides Livestock
5. Transmission of LA-MRSA to Humans
6. LA-MRSA Infections in Humans
7. Outlook
Acknowledgments
Conflicts of Interest
References
- EARS net. Antimicrobial Resistance Surveillance in Europe 2013. Avaliable Online: http://ecdc.europa.eu/en/publications/_layouts/forms/Publication_DispForm.aspx?List=4f55ad51–4aed-4d32-b960-af70113dbb90&ID=1205#sthash.FzIwMeIt.dpuf (accessed on 1 November 2014).
- Köck, R.; Becker, K.; Cookson, B.; van Gemert-Pijnen, J.E.; Harbarth, S.; Kluytmans, J.; Mielke, M.; Peters, G.; Skov, R.L.; Struelens, M.J.; et al. Systematic literature analysis and review of targeted preventive measures to limit healthcare-associated infections by methicillin-resistant Staphylococcus aureus. Eurosurveillance 2014, 19. [Google Scholar] [CrossRef]
- Johnson, A.P.; Davies, J.; Guy, R.; Abernethy, J.; Sheridan, E.; Pearson, A.; Duckworth, G. Mandatory surveillance of methicillin-resistant Staphylococcus aureus (MRSA) bacteraemia in England: The first 10 years. J. Antimicrob. Chemother. 2012, 67, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Jurke, A.; Kock, R.; Becker, K.; Thole, S.; Hendrix, R.; Rossen, J.; Daniels-Haardt, I.; Friedrich, A. Reduction of the nosocomial methicillin-resistant Staphylococcus aureus incidence density by a region-wide search and follow-strategy in forty German hospitals of the EUREGIO, 2009 to 2011. Euro. Surveill. 2013, 18. doi:10.2807/1560-7917. [Google Scholar]
- Ciccolini, M.; Donker, T.; Köck, R.; Mielke, M.; Hendrix, R.; Jurke, A.; Rahamat-Langendoen, J.; Becker, K.; Niesters, H.G.; Grundmann, H.; et al. Infection prevention in a connected world: The case for a regional approach. Int. J. Med. Microbiol. 2013, 303, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Salgado, C.D.; Farr, B.M.; Calfee, D.P. Community-acquired methicillin-resistant Staphylococcus aureus: A meta-analysis of prevalence and risk factors. Clin. Infect. Dis. 2003, 36, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Li, J.; Qiao, Y.; Ning, X.; Zeng, T.; Shen, X. Prevalence and invasiveness of community-acquired methicillin-resistant Staphylococcus aureus: A meta-analysis. Indian J. Pathol. Microbiol. 2014, 57, 418–422. [Google Scholar] [PubMed]
- Otto, M. Community-associated MRSA: What makes them special? Int. J. Med. Microbiol. 2013, 303, 324–330. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Graber, C.J.; Karr, M.; Diep, B.A.; Basuino, L.; Schwartz, B.S.; Enright, M.C.; O’Hanlon, S.J.; Thomas, J.C.; Perdreau-Remington, F.; et al. A population-based study of the incidence and molecular epidemiology of methicillin-resistant Staphylococcus aureus disease in San Francisco, 2004–2005. Clin. Infect. Dis. 2008, 46, 1637–1646. [Google Scholar]
- Tavares, A.; Miragaia, M.; Rolo, J.; Coelho, C.; de Lencastre, H. CA-MRSA/MSSA working group. High prevalence of hospital-associated methicillin-resistant Staphylococcus aureus in the community in Portugal: Evidence for the blurring of community-hospital boundaries. Eur. J. Clin. Microbiol. Infect. Dis. 2013, 32, 1269–1283. [Google Scholar] [CrossRef] [PubMed]
- Huijsdens, X.W.; van Dijke, B.J.; Spalburg, E.; van Santen-Verheuvel, M.G.; Heck, M.E.; Pluister, G.N.; Voss, A.; Wannet, W.J.; de Neeling, A.J. Community-acquired MRSA and pig-farming. Ann. Clin. Microbiol. Antimicrob. 2006, 10, 5–26. [Google Scholar]
- Lewis, H.C.; Mølbak, K.; Reese, C.; Aarestrup, F.M.; Selchau, M.; Sørum, M.; Skov, R.L. Pigs as source of methicillin-resistant Staphylococcus aureus CC398 infections in humans, Denmark. Emerg. Infect. Dis. 2008, 14, 1383–1389. [Google Scholar] [CrossRef] [PubMed]
- Layer, F.; Cuny, C.; Strommenger, B.; Werner, G.; Witte, W. Aktuelle Daten und Trends zu Methicillin-resistenten Staphylococcus aureus (MRSA). Bundesgesundheitsbl 2012, 55, 1377–1386. [Google Scholar] [CrossRef] [PubMed]
- Feil, E.J.; Cooper, J.E.; Grundmann, H.; Robinson, D.A.; Enright, M.C.; Berendt, T.; Peacock, S.J.; Smith, J.M.; Murphy, M.; Spratt, B.G.; et al. How clonal is Staphylococcus aureus? J. Bacteriol. 2003, 185, 3307–3316. [Google Scholar] [CrossRef] [PubMed]
- Harmsen, D.; Claus, H.; Witte, W.; Rothgänger, J.; Claus, H.; Turnwald, D.; Vogel, U. Typing of methicillin-resistant Staphylococcus aureus in a university hospital setting by using novel software for spa repeat determination and database management. J. Clin. Microbiol. 2003, 41, 5442–5448. [Google Scholar] [CrossRef] [PubMed]
- Palavecino, E.L. Clinical, epidemiologic, and laboratory aspects of methicillin-resistant Staphylococcus aureus infections. Methods Mol. Biol. 2014, 1085, 1–24. [Google Scholar] [PubMed]
- Price, L.B.; Stegger, M.; Hasman, H.; Aziz, M.; Larsen, J.; Andersen, P.S.; Pearson, T.; Waters, A.E.; Foster, J.T.; Schupp, J.; et al. Staphylococcus aureus CC398: Host adaptation and emergence of methicillin resistance in livestock. MBio 2012, 3, 40305–40311. [Google Scholar] [CrossRef] [PubMed]
- Harrison, E.M.; Paterson, G.K.; Holden, M.T.; Larsen, J.; Stegger, M.; Larsen, A.R.; Petersen, A.; Skov, R.L.; Christensen, J.M.; Bak Zeuthen, A.; et al. Whole genome sequencing identifies zoonotic transmission of MRSA isolates with the novel mecA homologue mecC. EMBO Mol. Med. 2013, 5, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Spoor, L.E.; McAdam, P.R.; Weinert, L.A.; Rambaut, A.; Hasman, H.; Aarestrup, F.M.; Kearns, A.M.; Larsen, A.R.; Skov, R.L.; Fitzgerald, J.R. Livestock origin for a human pandemic clone of community-associated methicillin-resistant Staphylococcus aureus. MBio 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Meyer, W. A proposal for subdividing the species Staphylococcus aureus. Int. J. Syst. Bacteriol. 1967, 17, 387–389. [Google Scholar] [CrossRef]
- Cuny, C.; Friedrich, A.; Kozytska, S.; Layer, F.; Nübel, U.; Ohlsen, K.; Strommenger, B.; Walther, B.; Wieler, L.; Witte, W. Emergence of methicillin-resistant Staphylococcus aureus (MRSA) in different animal species. Int. J. Med. Microbiol. 2010, 300, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Pantosti, A. Methicillin-resistant Staphylococcus aureus associated with animals and its relevance to human health. Front. Microbiol. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, J.R. Livestock-associated Staphylococcus aureus: Origin, evolution and public health threat. Trends Microbiol. 2012, 20, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Espinosa-Gongora, C.; Moodley, A.; Lipinska, U.; Broens, E.M.; Hermans, K.; Butaye, P.; Devriese, LA.; Haesebrouck, F.; Guardabassi, L. Phenotypes and genotypes of old and contemporary porcine strains indicate a temporal change in the S. aureus population structure in pigs. PLoS ONE 2014, 9, e101988. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lowder, B.V.; Guinane, C.M.; Ben Zakour, N.L.; Weinert, L.A.; Conway-Morris, A.; Cartwright, R.A.; Simpson, A.J.; Rambaut, A.; Nübel, U.; Fitzgerald, J.R. Recent human-to-poultry host jump, adaptation, and pandemic spread of Staphylococcus aureus. Proc. Natl. Acad. Sci. USA 2009, 106, 19545–19550. [Google Scholar] [CrossRef] [PubMed]
- Walther, B.; Hermes, J.; Cuny, C.; Wieler, L.H.; Vincze, S.; Abou Elnaga, Y.; Stamm, I.; Kopp, P.A.; Kohn, B.; Witte, W.; et al. Sharing more than friendship—nasal colonization with coagulase—positive staphylococci (CPS) and co-habitation aspects of dogs and their owners. PLoS ONE 2012, 7, e35197. [Google Scholar] [CrossRef] [PubMed]
- Uhlemann, A.C.; Porcella, S.F.; Trivedi, S.; Sullivan, S.B.; Hafer, C.; Kennedy, A.D.; Barbian, K.D.; McCarthy, A.J.; Street, C.; Hirschberg, D.L.; et al. Identification of a highly transmissible animal-independent Staphylococcus aureus ST398 clone with distinct genomic and cell adhesion properties. MBio 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, A.J.; Witney, A.A.; Gould, K.A.; Moodley, A.; Guardabassi, L.; Voss, A.; Denis, O.; Broens, E.M.; Hinds, J.; Lindsay, J.A. The distribution of mobile genetic elements (MGEs) in MRSA CC398 is associated with both host and country. Genome Biol. Evol. 2011, 3, 1164–1174. [Google Scholar] [CrossRef] [PubMed]
- Utter, B.; Deutsch, D.R.; Schuch, R.; Winer, B.Y.; Verratti, K.; Bishop-Lilly, K.; Sozhamannan, S.; Fischetti, V.A. Beyond the chromosome: The prevalence of unique extra-chromosomal bacteriophages with integrated virulence genes in pathogenic Staphylococcus aureus. PLoS ONE 2014, 9, e100502. [Google Scholar] [CrossRef] [PubMed]
- Witte, W.; Strommenger, B.; Stanek, C.; Cuny, C. Methicillin-resistant Staphylococcus aureus ST398 in humans and animals, Central Europe. Emerg. Infect. Dis. 2007, 13, 255–258. [Google Scholar] [CrossRef] [PubMed]
- Chuang, Y.Y.; Huang, Y.C. Livestock-associated methicillin-resistant Staphylococcus aureus in Asia: An emerging issue? Int. J. Antimicrob. Agents 2015, 45, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Molla, B.; Byrne, M.; Abley, M.; Mathews, J.; Jackson, C.R.; Fedorka-Cray, P.; Sreevatsan, S.; Wang, P.; Gebreyes, W.A. Epidemiology and genotypic characteristics of methicillin-resistant Staphylococcus aureus strains of porcine origin. J. Clin. Microbiol. 2012, 50, 3687–3693. [Google Scholar] [CrossRef] [PubMed]
- Grundmann, H.; Schouls, L.M.; Aanensen, D.M.; Pluister, G.N.; Tami, A.; Chlebowicz, M.; Glasner, C.; Sabat, A.J.; Weist, K.; Heuer, O.; et al.; ESCMID Study Group on Molecular Epidemiological Markers; European Staphylococcal Reference Laboratory Working Group The dynamic changes of dominant clones of Staphylococcus aureus causing bloodstream infections in the European region: Results of a second structured survey. Euro Surveill. 2014, 19. [Google Scholar]
- Deurenberg, R.H.; Stobberingh, E.E. The molecular evolution of hospital- and community-associated methicillin-resistant Staphylococcus aureus. Curr. Mol. Med. 2009, 9, 100–115. [Google Scholar] [CrossRef] [PubMed]
- Juhasz-Kaszanyitzky, E.; Janosi, S.; Somogyi, P.; Dan, A.; van der Graaf, R.; van Bloois, L.; van Duijkeren, E.; Wagenaar, J.A. MRSA transmission between cows and humans. Emerg. Infect. Dis. 2007, 13, 630–632. [Google Scholar] [CrossRef] [PubMed]
- Cuny, C.; Strommenger, B.; Witte, W.; Stanek, C. Clusters of infections in horses with MRSA ST1, ST254, and ST398 in a veterinary hospital. Microb. Drug Res. 2008, 14, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Alba, P.; Feltrin, F.; Cordaro, G.; Porrero, M.C.; Kraushaar, B.; Argudín, M.A.; Nykäsenoja, S.; Monaco, M.; Stegger, M.; Aarestrup, F.M.; et al. Livestock-Associated Methicillin Resistant and Methicillin Susceptible Staphylococcus aureus Sequence Type (CC)1 in European Farmed Animals: High Genetic Relatedness of Isolates from Italian Cattle Herds and Humans. PLoS ONE 2015, 10, e0137143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loncaric, I.; Künzel, F.; Licka, T.; Simhofer, H.; Spergser, J.; Rosengarten, R. Identification and characterization of methicillin-resistant Staphylococcus aureus (MRSA) from Austrian companion animals and horses. Vet. Microbiol. 2014, 168, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Wardyn, S.E.; Kauffman, L.K.; Smith, T.C. Methicillin-resistant Staphylococcus aureus in central Iowa wildlife. J. Wildl. Dis. 2012, 48, 1069–1073. [Google Scholar] [CrossRef] [PubMed]
- Walther, B.; Wieler, L.H.; Friedrich, A.W.; Hanssen, A.M.; Kohn, B.; Brunnberg, L.; Lübke-Becker, A. Methicillin-resistant Staphylococcus aureus (MRSA) isolated from small and exotic animals at a university hospital during routine microbiological examinations. Vet. Microbiol. 2008, 127, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Cuny, C.; Abdelbary, M.; Layer, F.; Werner, G.; Witte, W. Prevalence of the immune evasion gene cluster in Staphylococcus aureus CC398. Vet. Microbiol. 2015, 177. [Google Scholar] [CrossRef] [PubMed]
- Loncaric, I.; Brunthaler, R.; Spergser, J. Suspected goat-to-human transmission of methicillin-resistant Staphylococcus aureus sequence type 398. J. Clin. Microbiol. 2013, 51, 625–626. [Google Scholar] [CrossRef] [PubMed]
- Pletinckx, L.J.; Verhegghe, M.; Crombé, F.; Dewulf, J.; de Bleecker, Y.; Rasschaert, G.; Butaye, P.; Goddeeris, B.M.; de Man, I. Evidence of possible methicillin-resistant Staphylococcus aureus ST398 spread between pigs and other animals and people residing on the same farm. Prev. Vet Med. 2013, 109, 293–303. [Google Scholar] [CrossRef] [PubMed]
- Agnoletti, F.; Mazzolini, E.; Bacchin, C.; Bano, L.; Berto, G.; Rigoli, R.; Muffato, G.; Coato, P.; Tonon, E.; Drigo, I. First reporting of methicillin-resistant Staphylococcus aureus (MRSA) ST398 in an industrial rabbit holding and in farm-related people. Vet. Microbiol. 2014, 170, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Loncaric, I.; Künzel, F. Sequence type 398 methicillin-resistant Staphylococcus aureus infection in a pet rabbit. Vet. Dermatol. 2013, 24, 70–72. [Google Scholar] [CrossRef] [PubMed]
- Cuny, C.; Layer, F.; Strommenger, B.; Witte, W. Rare occurrence of methicillin-resistant Staphylococcus aureus CC130 with a novel mecA homologue in humans in Germany. PLoS ONE 2011, 6, e24360. [Google Scholar] [CrossRef] [PubMed]
- Paterson, G.K.; Larsen, A.R.; Robb, A.; Edwards, G.E.; Pennycott, T.W.; Foster, G.; Mot, D.; Hermans, K.; Baert, K.; Peacock, S.J.; et al. The newly described mecA homologue, mecALGA251, is present in methicillin-resistant Staphylococcus aureus isolates from a diverse range of host species. Antimicrob. Chemother. 2012, 67, 2809–2813. [Google Scholar] [CrossRef] [PubMed]
- Walther, B.; Wieler, L.H.; Vincze, S.; Antão, E.M.; Brandenburg, A.; Stamm, I.; Kopp, P.A.; Kohn, B.; Semmler, T.; Lübke-Becker, A. MRSA variant in companion animals. Emerg. Infect. Dis. 2012, 18, 2017–2020. [Google Scholar] [CrossRef] [PubMed]
- Luzzago, C.; Locatelli, C.; Franco, A.; Scaccabarozzi, L.; Gualdi, V.; Viganò, R.; Sironi, G.; Besozzi, M.; Castiglioni, B.; Lanfranchi, P.; et al. Clonal diversity, virulence-associated genes and antimicrobial resistance profile of Staphylococcus aureus isolates from nasal cavities and soft tissue infections in wild ruminants in Italian Alps. Vet. Microbiol. 2014, 170, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Loncaric, I.; Kübber-Heiss, A.; Posautz, A.; Stalder, G.L.; Hoffmann, D.; Rosengarten, R.; Walzer, C. Characterization of methicillin-resistant Staphylococcus spp. carrying the mecC gene, isolated from wildlife. J. Antimicrob. Chemother. 2013, 68, 2222–2225. [Google Scholar] [CrossRef] [PubMed]
- García-Álvarez, L.; Holden, M.T.; Lindsay, J.; Webb, C.R.; Brown, D.F.; Curran, M.D.; Walpole, E.; Brooks, K.; Pickard, D.J.; Teale, C.; et al. Meticillin-resistant Staphylococcus aureus with a novel mecA homologue in human and bovine populations in the UK and Denmark: A descriptive study. Lancet Infect. Dis. 2011, 11, 595–603. [Google Scholar] [CrossRef]
- Meemken, D.; Blaha, T.; Hotzel, H.; Strommenger, B.; Klein, G.; Ehricht, R.; Monecke, S.; Kehrenberg, C. Genotypic and phenotypic characterization of Staphylococcus aureus isolates from wild boars. Appl. Environ. Microbiol. 2013, 79, 1739–1742. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.M.; Needs, P.F.; Manley, G.; Green, L.E. Global distribution and diversity of ovine-associated Staphylococcus aureus. Infect. Genet. Evol. 2014, 22, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Porrero, M.C.; Hasman, H.; Vela, A.I.; Fernández-Garayzábal, J.F.; Domínguez, L.; Aarestrup, F.M. Clonal diversity of Staphylococcus aureus originating from the small ruminants goats and sheep. Vet. Microbiol. 2012, 156, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Paterson, G.K.; Harrison, E.M.; Holmes, M.A. The emergence of mecC methicillin-resistant Staphylococcus aureus. Trends Microbiol. 2014, 22, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Espinosa-Gongora, C.; Harrison, E.M.; Moodley, A.; Guardabassi, L.; Holmes, M.A. MRSA carrying mecC in captive mara. J. Antimicrob. Chemother. 2015, 70, 1622–1624. [Google Scholar] [CrossRef] [PubMed]
- Loncaric, I.; Kübber-Heiss, A.; Posautz, A.; Stalder, G.L.; Hoffmann, D.; Rosengarten, R.; Walzer, C. mecC- and mecA-positive methicillin-resistant Staphylococcus aureus (MRSA) isolated from livestock sharing habitat with wildlife previously tested positive for mecC-positive MRSA. Vet. Dermatol. 2014, 25, 147–148. [Google Scholar] [CrossRef] [PubMed]
- Harrison, E.M.; Weinert, L.A.; Holden, M.T.; Welch, J.J.; Wilson, K.; Morgan, F.J.; Harris, S.R.; Loeffler, A.; Boag, A.K.; Peacock, S.J.; et al. A shared population of epidemic methicillin-resistant Staphylococcus aureus 15 circulates in humans and companion animals. MBio 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Walther, B.; Wieler, L.H.; Friedrich, A.W.; Kohn, B.; Brunnberg, L.; Lübke-Becker, A. Staphylococcus aureus and MRSA colonization rates among personnel and dogs in a small animal hospital: Association with nosocomial infections. Berl. Munch. Tierarztl. Wochenschr. 2009, 122, 178–185. [Google Scholar] [PubMed]
- Weese, J.S.; Rousseau, J.; Willey, B.M.; Archambault, M.; McGeer, A.; Low, D.E. Methicillin-resistant Staphylococcus aureus in horses at a veterinary teaching hospital: Frequency, characterization, and association with clinical disease. J. Vet. Intern. Med. 2006, 20, 182–186. [Google Scholar] [CrossRef] [PubMed]
- Faires, M.C.; Gehring, E.; Mergl, J.; Weese, J.S. Methicillin-resistant Staphylococcus aureus in marine mammals. Emerg. Infect. Dis. 2009, 15, 2071–2072. [Google Scholar] [CrossRef] [PubMed]
- Walther, B.; Monecke, S.; Ruscher, C.; Friedrich, A.W.; Ehricht, R.; Slickers, P.; Soba, A.; Wleklinski, C.G.; Wieler, L.H.; Lübke-Becker, A. Comparative molecular analysis substantiates zoonotic potential of equine methicillin-resistant Staphylococcus aureus. J. Clin. Microbiol. 2009, 47, 704–710. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). (2009) Analysis of the baseline survey on the prevalence of methicillin-resistant Staphylococcus aureus (MRSA) in holdings with breeding pigs in the EU, Part A: MRSA prevalence estimates. Eur. Food Saf. Auth. 2009, 7. [Google Scholar] [CrossRef]
- Chairat, S.; Gharsa, H.; Lozano, C.; Gómez-Sanz, E.; Gómez, P.; Zarazaga, M.; Boudabous, A.; Torres, C.; Ben Slama, K. Characterization of Staphylococcus aureus from Raw Meat Samples in Tunisia: Detection of Clonal Lineage ST398 from the African Continent. Foodborne Pathog. Dis. 2015, 12, 86–92. [Google Scholar]
- Groves, M.D.; O’Sullivan, M.V.; Brouwers, H.J.; Chapman, T.A.; Abraham, S.; Trott, D.J.; Al Jassim, R.; Coombs, G.W.; Skov, R.L.; Jordan, D. Staphylococcus aureus ST398 detected in pigs in Australia. J. Antimicrob. Chemother. 2014, 69, 1426–1428. [Google Scholar] [CrossRef] [PubMed]
- Graveland, H.; van Duijkeren, E.; van Nes, A.; Schoormans, A.; Broekhuizen-Stins, M.; Oosting-van Schothorst, I.; Heederik, D.; Wagenaar, J.A. Evaluation of isolation procedures and chromogenic agar media for the detection of MRSA in nasal swabs from pigs and veal calves. Vet. Microbiol. 2009, 139, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Nemati, M.; Hermans, K.; Lipinska, U.; Denis, O.; Deplano, A.; Struelens, M.; Devriese, L.A.; Pasmans, F.; Haesebrouck, F. Antimicrobial resistance of old and recent Staphylococcus aureus isolates from poultry: First detection of livestock-associated methicillin-resistant strain ST398. Antimicrob. Agents Chemother. 2008, 52, 3817–3819. [Google Scholar] [CrossRef] [PubMed]
- Vanderhaeghen, W.; Cerpentier, T.; Adriaensen, C.; Vicca, J.; Hermans, K.; Butaye, P. Methicillin-resistant Staphylococcus aureus (MRSA) ST398 associated with clinical and subclinical mastitis in Belgian cows. Vet. Microbiol. 2010, 144, 166–171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Richter, A.; Sting, R.; Popp, C.; Rau, J.; Tenhagen, B.A.; Guerra, B.; Hafez, H.M.; Fetsch, A. Prevalence of types of methicillin-resistant Staphylococcus aureus in turkey flocks and personnel attending the animals. Epidemiol. Infect. 2012, 140, 2223–2232. [Google Scholar] [CrossRef] [PubMed]
- Tenhagen, B.A.; Fetsch, A.; Stührenberg, B.; Schleuter, G.; Guerra, B.; Hammerl, J.A.; Hertwig, S.; Kowall, J.; Kämpe, U.; Schroeter, A.; et al. Prevalence of MRSA types in slaughter pigs in different German abattoirs. Vet. Rec. 2009, 165, 589–593. [Google Scholar] [CrossRef] [PubMed]
- Alt, K.; Fetsch, A.; Schroeter, A.; Guerra, B.; Hammerl, J.A.; Hertwig, S.; Senkov, N.; Geinets, A.; Mueller-Graf, C.; Braeunig, J.; et al. Factors associated with the occurrence of MRSA CC398 in herds of fattening pigs in Germany. BMC Vet. Res. 2011, 7. [Google Scholar] [CrossRef] [PubMed]
- Graveland, H.; Duim, B.; van Duijkeren, E.; Heederik, D.; Wagenaar, J.A. Livestock-associated methicillin-resistant Staphylococcus aureus in animals and humans. Int. J. Med. Microbiol. 2011, 301, 630–634. [Google Scholar] [CrossRef] [PubMed]
- Cuny, C.; Friedrich, A.W.; Witte, W. Absence of livestock-associated methicillin-resistant Staphylococcus aureus clonal complex CC398 as a nasal colonizer of pigs raised in an alternative system. Appl. Environ. Microbiol. 2012, 78, 1296–1297. [Google Scholar] [CrossRef] [PubMed]
- Van de Vijver, L.P.; Tulinski, P.; Bondt, N.; Mevius, D.; Verwer, C. Prevalence and molecular characteristics of methicillin-resistant Staphylococcus aureus (MRSA) in organic pig herds in The Netherlands. Zoonoses Public Health 2014, 61, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Slifierz, M.J.; Friendship, R.M.; Weese, J.S. Methicillin-resistant Staphylococcus aureus in commercial swine herds is associated with disinfectant and zinc usage. Appl. Environ. Microbiol. 2015, 81, 2690–2695. [Google Scholar] [CrossRef] [PubMed]
- Broens, E.M. MRSA CC398 in the pig production chain. Prev. Vet. Med. 2011, 98, 182–189. [Google Scholar] [CrossRef] [PubMed]
- De Boer, E.; Zwartkruis-Nahuis, J.T.; Wit, B.; Huijsdens, X.W.; de Neeling, A.J.; Bosch, T.; van Oosterom, R.A.; Vila, A.; Heuvelink, A.E. Prevalence of methicillin-resistant Staphylococcus aureus in meat. Int. J. Food Microbiol. 2009, 134, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Beneke, B.; Klees, S.; Stührenberg, B.; Fetsch, A.; Kraushaar, B.; Tenhagen, B.A. Prevalence of methicillin-resistant Staphylococcus aureus in a fresh meat pork production chain. J. Food Prot. 2011, 74, 126–129. [Google Scholar] [CrossRef] [PubMed]
- Vossenkuhl, B.; Brandt, J.; Fetsch, A.; Käsbohrer, A.; Kraushaar, B.; Alt, K.; Tenhagen, B.A. Comparison of spa Types, SCCmec Types and Antimicrobial Resistance Profiles of MRSA Isolated from Turkeys at Farm, Slaughter and from Retail Meat Indicates Transmission along the Production Chain. PLoS ONE 2014, 9, e96308. [Google Scholar] [CrossRef] [PubMed]
- Pu, S.; Han, F.; Ge, B. Isolation and characterization of methicillin-resistant Staphylococcus aureus strains from Louisiana retail meats. Appl. Environ. Microbiol. 2009, 75, 265–267. [Google Scholar] [CrossRef] [PubMed]
- Weese, J.S.; Reid-Smith, R.; Rousseau, J.; Avery, B. Methicillin-resistant Staphylococcus aureus (MRSA) contamination of retail pork. Can. Vet. J. 2010, 51, 749–752. [Google Scholar] [PubMed]
- Lin, J.; Yeh, K.S.; Liu, H.T.; Lin, J.H. Staphylococcus aureus isolated from pork and chicken carcasses in Taiwan: Prevalence and antimicrobial susceptibility. J. Food Prot. 2009, 72, 608–611. [Google Scholar] [PubMed]
- Argudín, M.A.; Tenhagen, B.A.; Fetsch, A.; Sachsenröder, J.; Käsbohrer, A.; Schroeter, A.; Hammerl, J.A.; Hertwig, S.; Helmuth, R.; Bräunig, J.; et al. Virulence and resistance determinants of German Staphylococcus aureus ST398 isolates from non human sources. Appl. Environ. Microbiol. 2011, 77, 3052–3060. [Google Scholar] [CrossRef] [PubMed]
- Kadlec, K.; Ehricht, R.; Monecke, S.; Steinacker, U.; Kaspar, H.; Mankertz, J.; Schwarz, S. Diversity of antimicrobial resistance pheno- and genotypes of methicillin-resistant Staphylococcus aureus ST398 from diseased swine. J. Antimicrob. Chemother. 2009, 64, 1156–1164. [Google Scholar] [CrossRef] [PubMed]
- Van Wamel, W.J.; Rooijakkers, S.H.; Ruyken, M.; van Kessel, K.P.; van Strijp, J.A. The innate immune modulators staphylococcal complement inhibitor and chemotaxis inhibitory protein of Staphylococcus aureus are located on beta-hemolysin-converting bacteriophages. J. Bacteriol. 2006, 188, 1310–1315. [Google Scholar] [CrossRef] [PubMed]
- Tenhagen, B.A.; Vossenkuhl, B.; Käsbohrer, A.; Alt, K.; Kraushaar, B.; Guerra, B.; Schroeter, A.; Fetsch, A. Methicillin-resistant Staphylococcus aureus in cattle food chains—Prevalence, diversity, and antimicrobial resistance in Germany. J. Anim. Sci. 2014, 92, 2741–2751. [Google Scholar] [CrossRef] [PubMed]
- Van Duijkeren, E.; Moleman, M.; Sloet van Oldruitenborgh-Oosterbaan, M.M.; Multem, J.; Troelstra, A.; Fluit, A.C.; van Wamel, W.J.; Houwers, D.J.; de Neeling, A.J.; Wagenaar, J. Methicillin-resistant Staphylococcus aureus in horses and horse personnel: An investigation of several outbreaks. Vet. Microbiol. 2010, 141, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Van den Eede, A.; Martens, A.; Lipinska, U.; Struelens, M.; Deplano, A.; Denis, O.; Haesebrouck, F.; Gasthuys, F.; Hermans, K. High occurrence of methicillin-resistant Staphylococcus aureus ST398 in equine nasal samples. Vet. Microbiol. 2009, 133, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Sieber, S.; Gerber, V.; Jandova, V.; Rossano, A.; Evison, J.M.; Perreten, V. Evolution of multidrug-resistant Staphylococcus aureus infections in horses and colonized personnel in an equine clinic between 2005 and 2010. Microb. Drug Res. 2011, 17, 47147–47148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vincze, S.; Stamm, I.; Kopp, P.A.; Hermes, J.; Adlhoch, C.; Semmler, T.; Wieler, L.H.; Lübke-Becker, A.; Walther, B. Alarming proportions of methicillin-resistant Staphylococcus aureus (MRSA) in wound samples from companion animals, Germany 2010–2012. PLoS ONE 2014, 9, e85656. [Google Scholar] [CrossRef] [PubMed]
- Abdelbary, M.M.; Wittenberg, A.; Cuny, C.; Layer, F.; Kurt, K.; Wieler, L.H.; Walther, B.; Skov, R.; Larsen, J.; Hasman, H.; et al. Phylogenetic analysis of Staphylococcus aureus CC398 reveals a sub-lineage epidemiologically associated with infections in horses. PLoS ONE 2014, 9, e88083. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vincze, S.; Brandenburg, A.; Espelage, W.; Stamm, I.; Wieler, L.H.; Kopp, P.A.; Lübke-Becker, A.; Walther, B. Risk factors for MRSA infection in companion animals: Results from a case-control study within Germany. Int. J. Med. Microbiol. 2014, 304, 787–793. [Google Scholar] [CrossRef] [PubMed]
- Schulz, J.; Friese, A.; Klees, S.; Tenhagen, B.A.; Fetsch, A.; Rösler, U.; Hartung, J. Longitudinal study of the contamination of air and of soil surfaces in the vicinity of pig barns by livestock-associated methicillin-resistant Staphylococcus aureus. Appl. Environ. Microbiol. 2012, 78, 5666–5671. [Google Scholar] [CrossRef] [PubMed]
- Bos, M.E.; Verstappen, K.M.; van Cleef, BA.; Dohmen, W.; Dorado-García, A.; Graveland, H.; Duim, B.; Wagenaar, J.A.; Kluytmans, J.A.; Heederik, D.J. Transmission through air as a possible route of exposure for MRSA. J. Expo. Sci. Environ. Epidemiol. 2014, 17. [Google Scholar] [CrossRef] [PubMed]
- Cuny, C.; Nathaus, R.; Layer, F.; Strommenger, B.; Altmann, D.; Witte, W. Nasal colonization of humans with methicillin-resistant Staphylococcus aureus (MRSA) CC398 with and without exposure to pigs. PLoS ONE 2009, 4, e6800. [Google Scholar] [CrossRef] [PubMed]
- Van den Broek, I.V.; van Cleef, B.A.; Haenen, A.; Broens, E.M.; van der Wolf, P.J.; van den Broek, M.J.; Huijsdens, X.W.; Kluytmans, J.A.; van de Giessen, A.W.; Tiemersma, E.W. Methicillin-resistant Staphylococcus aureus in people living and working in pig farms. Epidemiol. Infect. 2009, 137, 700–708. [Google Scholar] [CrossRef] [PubMed]
- Graveland, H.; Wagenaar, J.A.; Bergs, K.; Heesterbeek, H.; Heederik, D. Persistence of livestock associated MRSA CC398 in humans is dependent on intensity of animal contact. PLoS ONE 2011, 6, e16830. [Google Scholar] [CrossRef] [PubMed]
- Van Cleef, B.A.; Graveland, H.; Haenen, A.P.; van de Giessen, A.W.; Heederik, D.; Wagenaar, J.A.; Kluytmans, J.A. Persistence of livestock-associated methicillin-resistant Staphylococcus aureus in field workers after short-term occupational exposure to pigs and veal calves. J. Clin. Microbiol. 2011, 49, 1030–1033. [Google Scholar] [CrossRef] [PubMed]
- Köck, R.; Loth, B.; Köksal, M.; Schulte-Wülwer, J.; Harlizius, J.; Friedrich, A.W. Persistence of nasal colonization with livestock-associated methicillin-resistant Staphylococcus aureus in pig farmers after holidays from pig exposure. Appl. Environ. Microbiol. 2012, 78, 4046–4047. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Graells, C.; van Cleef, B.A.; Larsen, J.; Denis, O.; Skov, R.L.; Voss, A. Dynamic of livestock-associated methicillin-resistant Staphylococcus aureus CC398 in pig farm households: A pilot study. PLoS ONE 2013, 8, e65512. [Google Scholar] [CrossRef] [PubMed]
- Van Cleef, B.A.; van Benthem, B.H.; Verkade, E.J.; van Rijen, M.M.; Kluytmans, J.A.; van den Bergh, M.F.; Graveland, H.; Bosch, T.; Verstappen, K.M.; Wagenaar, J.A.; et al. Livestock-associated MRSA in household members of pig farmers: Transmission and dynamics of carriage, a prospective cohort study. PLoS ONE 2015, 10, e0127190. [Google Scholar] [CrossRef] [PubMed]
- Dorado-García, A.; Dohmen, W.; Bos, M.E.; Verstappen, K.M.; Houben, M.; Wagenaar, J.A.; Heederik, D.J. Dose-response relationship between antimicrobial drugs and livestock-associated MRSA in pig farming. Emerg. Infect. Dis. 2015, 21, 950–959. [Google Scholar] [CrossRef] [PubMed]
- Fang, H.W.; Chiang, P.H.; Huang, Y.C. Livestock-associated methicillin-resistant Staphylococcus aureus ST9 in pigs and related personnel in Taiwan. PLoS ONE 2014, 9, e88826. [Google Scholar] [CrossRef] [PubMed]
- Van Cleef, B.A.; Broens, E.M.; Voss, A. High prevalence of nasal MRSA carriage in slaughterhouse workers in contact with live pigs in The Netherlands. Epidemiol. Infect. 2010, 138, 756–763. [Google Scholar] [CrossRef] [PubMed]
- Mulders, M.N.; Haenen, A.P.; Geenen, P.L.; Vesseur, P.C.; Poldervaart, E.S.; Bosch, T.; Huijsdens, X.W.; Hengeveld, P.D.; Dam-Deisz, W.D.; Graat, E.A.; et al. Prevalence of livestock-associated MRSA in broiler flocks and risk factors for slaughterhouse personnel in The Netherlands. Epidemiol. Infect. 2010, 138, 743–755. [Google Scholar] [CrossRef] [PubMed]
- Hermes, J.; Witte, W.; Cuny, C.; Kleinkauf, N.; Jansen, A.; Eckmanns, T. Prevalence of MRSA nasal colonization over times in veterinarians and their household contacts in Germany. Intern. J. Med. Microbiol. 2012, 302, 146. [Google Scholar]
- Garcia-Graells, C.; Antoine, J.; Larsen, J.; Catry, B.; Skov, R.; Denis, O. Livestock veterinarians at high risk of acquiring methicillin-resistant Staphylococcus aureus ST398. Epidemiol. Infect. 2012, 140, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Verkade, E.; Kluytmans-van den Bergh, M.; van Benthem, B.; van Cleef, B.; van Rijen, M.; Bosch, T.; Schouls, L.; Kluytmans, J. Transmission of methicillin-resistant Staphylococcus aureus CC398 from livestock veterinarians to their household members. PLoS ONE 2014, 9, e100823. [Google Scholar] [CrossRef] [PubMed]
- Bosch, T.; Verkade, E.; van Luit, M.; Landman, F.; Kluytmans, J.; Schouls, L.M. Transmission and persistence of livestock-associated methicillin-resistant Staphylococcus aureus among veterinarians and their household members. Appl. Environ. Microbiol. 2015, 81, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Köck, R.; Schaumburg, F.; Mellmann, A.; Koksal, M.; Jurke, A.; Becker, K.; Friedrich, A.W. Livestock-associated methicillin-resistant Staphylococcus aureus (MRSA) as causes of human infection and colonization in Germany. PLoS ONE 2013, 8, e55040. [Google Scholar] [CrossRef] [PubMed]
- Feingold, B.J.; Silbergeld, E.K.; Curriero, F.C.; van Cleef, B.A.; Heck, M.E.; Kluytmans, J.A. Livestock density as risk factor for livestock-associated methicillin-resistant Staphylococcus aureus, the Netherlands. Emerg. Infect. Dis. 2012, 18, 1841–1849. [Google Scholar] [CrossRef] [PubMed]
- Friese, A.; Schulz, J.; Laube, H.; von Salviati, C.; Hartung, J.; Roesler, U. Faecal occurrence and emissions of livestock-associated methicillin-resistant Staphylococcus aureus (laMRSA) and ESbl/AmpC-producing E. coli from animal farms in Germany. Berl. Munch. Tierarztl. Wochenschr. 2013, 126, 175–180. [Google Scholar] [PubMed]
- Friese, A.; Schulz, J.; Zimmermann, K.; Tenhagen, B.A.; Fetsch, A.; Hartung, J.; Rösler, U. Occurrence of livestock-associated methicillin-resistant Staphylococcus aureus in Turkey and broiler barns and contamination of air and soil surfaces in their vicinity. Appl. Environ. Microbiol. 2013, 79, 2759–2766. [Google Scholar] [CrossRef] [PubMed]
- Loncaric, I.; Stalder, G.L.; Mehinagic, K.; Rosengarten, R.; Hoelzl, F.; Knauer, F.; Walzer, C. Comparison of ESBL- and AmpC producing Enterobacteriaceae and methicillin-resistant Staphylococcus aureus (MRSA) isolated from migratory and resident population of rooks (Corvus frugilegus) in Austria. PLoS ONE 2013, 8, e84048. [Google Scholar] [CrossRef] [PubMed]
- Bisdorff, B.; Scholhölter, J.L.; Claußen, K.; Pulz, M.; Nowak, D.; Radon, K. MRSA-ST398 in livestock farmers and neighbouring residents in a rural area in Germany. Epidemiol. Infect. 2012, 140, 1800–1808. [Google Scholar] [CrossRef] [PubMed]
- Casey, J.A.; Curriero, F.C.; Cosgrove, S.E.; Nachman, K.E.; Schwartz, B.S. High-density livestock operations, crop field application of manure, and risk of community-associated methicillin-resistant Staphylococcus aureus infection in Pennsylvania. JAMA Intern. Med. 2013, 173, 1980–1990. [Google Scholar] [CrossRef] [PubMed]
- Idelevich, E.A.; Lanckohr, C.; Horn, D.; Wieler, L.H.; Becker, K.; Köck, R. Multidrug-resistant bacteria in Germany: The impact of sources outside healthcare facilities. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz 2015, in press. [Google Scholar]
- Benito, D.; Lozano, C.; Rezusta, A.; Ferrer, I.; Vasquez, M.A.; Ceballos, S.; Zarazaga, M.; Revillo, M.J.; Torres, C. Characterization of tetracycline and methicillin resistant Staphylococcus aureus strains in a Spanish hospital: Is livestock-contact a risk factor in infections caused by MRSA CC398? Int. J. Med. Microbiol. 2014, 304, 1226–1232. [Google Scholar] [CrossRef] [PubMed]
- Deiters, C.; Günnewig, V.; Friedrich, A.W.; Mellmann, A.; Köck, R. Are cases of Methicillin-resistant Staphylococcus aureus clonal complex (CC) 398 among humans still livestock-associated? Int. J. Med. Microbiol. 2015, 305, 110–113. [Google Scholar] [CrossRef] [PubMed]
- Lekkerkerk, W.S.; van Wamel, W.J.; Snijders, S.V.; Willems, R.J.; van Duijkeren, E.; Broens, E.M.; Wagenaar, J.A.; Lindsay, J.A.; Vos, M.C. What Is the Origin of Livestock-Associated Methicillin-Resistant Staphylococcus aureus Clonal Complex 398 Isolates from Humans without Livestock Contact? An Epidemiological and Genetic Analysis. J. Clin. Microbiol. 2015, 53, 1836–1841. [Google Scholar] [CrossRef] [PubMed]
- Van Rijen, M.M.; Kluytmans-van den Bergh, M.F.; Verkade, E.J.; Ten Ham, P.B.; Feingold, B.J.; Kluytmans, J.A. Lifestyle-associated risk factors for community-acquired methicillin-resistant Staphylococcus aureus carriage in the Netherlands: An exploratory hospital-based case-control Study. PLoS ONE 2013, 8, e65594. [Google Scholar] [CrossRef] [PubMed]
- Van Cleef, B.A.; Monnet, D.L.; Voss, A.; Krziwanek, K.; Allerberger, F.; Struelens, M.; Zemlickova, H.; Skov, R.L.; Vuopio-Varkila, J.; Cuny, C.; et al. Livestock-associated methicillin-resistant Staphylococcus aureus in humans, Europe. Emerg. Infect. Dis. 2011, 17, 502–505. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Juretzek, M.; Armengol Porta, A.; Bühling, R.; Haubold, M.; Pohle, F.; Schwab, W.B. Epidemiology of methicillin-resistant Staphylococcus aureus at hospital admission in South Brandenburg, Germany. Abstract PV1. Intern. J. Med. Microbiol. 2011, 301, 100. [Google Scholar]
- Cuny, C.; Layer, F.; Werner, G.; Harmsen, D.; Daniels-Haardt, I.; Jurke, A.; Mellmann, A.; Witte, W.; Köck, R. State-wide surveillance of antibiotic resistance patterns and spa types of methicillin-resistant Staphylococcus aureus from blood cultures in North Rhine-Westphalia, 2011–2013. Clin. Microbiol. Infect. 2015, 21, 750–757. [Google Scholar] [CrossRef] [PubMed]
- Van de Sande-Bruinsma, N.; Leverstein van Hall, M.A.; Janssen, M.; Nagtzaam, N.; Leenders, S.; de Greeff, S.C.; Schneeberger, P.M. Impact of livestock-associated MRSA in a hospital setting. Antimicrob. Res. Infect. Control. 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Wulf, M.W.; Markestein, A.; van der Linden, F.T.; Voss, A.; Klaassen, C.; Verduin, C.M. First outbreak of methicillin-resistant Staphylococcus aureus ST398 in a Dutch hospital in June 2007. Euro Surveill. 2008, 13. [Google Scholar]
- Wassenberg, M.W.; Bootsma, M.C.; Troelstra, A.; Kluytmans, J.A.; Bonten, M.J. Transmissibility of livestock-associated methicillin-resistant Staphylococcus aureus (ST398) in Dutch hospitals. Clin. Microbiol. Infect. 2011, 17, 316–319. [Google Scholar] [CrossRef] [PubMed]
- Ward, M.J.; Gibbons, C.L.; McAdam, P.R.; van Bunnik, B.A.; Girvan, E.K.; Edwards, G.F.; Fitzgerald, J.R.; Woolhouse, M.E. Time-Scaled Evolutionary Analysis of the Transmission and Antibiotic Resistance Dynamics of Staphylococcus aureus Clonal Complex 398. Appl. Environ. Microbiol. 2014, 80, 7275–7282. [Google Scholar] [CrossRef] [PubMed]
- Witte, W.; Cuny, C. Emergence and spread of cfr-mediated multiresistance in staphylococci: An interdisciplinary challenge. Future Microbiol. 2011, 6, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Wendlandt, S.; Shen, J.; Kadlec, K.; Wang, Y.; Li, B.; Zhang, W.J.; Feßler, A.T.; Wu, C.; Schwarz, S. Multidrug resistance genes in staphylococci from animals that confer resistance to critically and highly important antimicrobial agents in human medicine. Trends Microbiol. 2015, 23, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Kehrenberg, C.; Schwarz, S. Distribution of florfenicol resistance genes fexA and cfr among chloramphenicol-resistant Staphylococcus isolates. Antimicrob. Agents Chemother. 2006, 50, 1156–1163. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.L.; Wei, H.K.; Wang, J.; Lin, D.C.; Liu, X.Q.; Liu, J.H. High prevalence of Cfr-producing Staphylococcus species in retail meat in Guangzhou, China. BMC Microbiol. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Mendes, R.E.; Flamm, R.K.; Hogan, P.A.; Ross, J.E.; Jones, R.N. Summary of linezolid activity and resistance mechanisms detected during the 2012 LEADER surveillance program for the United States. Antimicrob. Agents Chemother. 2014, 58, 1243–1247. [Google Scholar] [CrossRef] [PubMed]
- Bender, J.; Strommenger, B.; Steglich, M.; Zimmermann, O.; Fenner, I.; Lensing, C.; Dagwadordsch, U.; Kekulé, A.S.; Werner, G.; Layer, F. Linezolid resistance in clinical isolates of Staphylococcus epidermidis from German hospitals and characterization of two cfr-carrying plasmids. J. Antimicrob. Chemother. 2015, 70, 1630–1638. [Google Scholar] [CrossRef] [PubMed]
- Kehrenberg, C.; Cuny, C.; Strommenger, B.; Schwarz, S.; Witte, W. Methicillin-resistant and -susceptible Staphylococcus aureus strains of clonal lineages ST398 and ST9 from swine carry the multidrug resistance gene cfr. Antimicrob. Agents Chemother. 2009, 53, 779–781. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Wang, Y.; Schwarz, S. Presence and dissemination of the multiresistance gene cfr in Gram-positive and Gram-negative bacteria. J. Antimicrob. Chemother. 2013, 68, 1697–1706. [Google Scholar] [CrossRef] [PubMed]
- Mendes, R.E.; Deshpande, L.M.; Jones, R.N. Linezolid update: Stable in vitro activity following more than a decade of clinical use and summary of associated resistance mechanisms. Drug Res. Update 2014, 17, 1–12. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, A.J.; Lindsay, J.A. Staphylococcus aureus innate immune evasion is lineage-specific: A bioinformatics study. Infect. Genet. Evol. 2013, 19, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Jin, T.; Bokarewa, M.; Foster, T.; Mitchell, J.; Higgins, J.; Tarkowski, A. Staphylococcus aureus resists human defensins by production of staphylokinase, a novel bacterial evasion mechanism. J. Immunol. 2004, 172, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Cuny, C.; Layer, F.; Köck, R.; Werner, G.; Witte, W. Methicillin susceptible Staphylococcus aureus (MSSA) of clonal complex CC398, t571 from infections in humans are still rare in Germany. PLoS ONE 2013, 8, e8316. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Liu, Y.; Zhao, M.; Liu, Y.; Yu, Y.; Chen, H.; Sun, Q.; Chen, H.; Jiang, W.; Liu, Y.; et al. Characterization of community acquired Staphylococcus aureus associated with skin and soft tissue infection in Beijing high prevalence of PVL + ST398. PLoS ONE 2012, 7, e38577. [Google Scholar] [CrossRef] [PubMed]
- Welinder-Olsson, C.; Florén-Johansson, K.; Larsson, L.; Oberg, S.; Karlsson, L.; Ahrén, C. Infection with Panton-Valentine leukocidin-positive methicillin-resistant Staphylococcus aureus t034. Emerg. Infect. Dis. 2008, 14, 1271–1272. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuny, C.; Wieler, L.H.; Witte, W. Livestock-Associated MRSA: The Impact on Humans. Antibiotics 2015, 4, 521-543. https://doi.org/10.3390/antibiotics4040521
Cuny C, Wieler LH, Witte W. Livestock-Associated MRSA: The Impact on Humans. Antibiotics. 2015; 4(4):521-543. https://doi.org/10.3390/antibiotics4040521
Chicago/Turabian StyleCuny, Christiane, Lothar H. Wieler, and Wolfgang Witte. 2015. "Livestock-Associated MRSA: The Impact on Humans" Antibiotics 4, no. 4: 521-543. https://doi.org/10.3390/antibiotics4040521





