Pipeline of Known Chemical Classes of Antibiotics
Abstract
:1. Introduction
2. Experimental
- Identification of companies doing research and development into new antibiotics around the world;
- Mapping of the antibiotic development pipelines of the companies identified.
2.1. Identification of Companies Doing R&D into New Antibiotics
2.2. Mapping the Companies’ New Antibiotics Pipeline
3. Pipeline of New Antibiotics in Chemical Classes Known on the Market
| Class | Compound (Subclass) | Company (Country) | Development Phase | Bacteria | Chemical structure | Source |
|---|---|---|---|---|---|---|
| Quinolones | ||||||
| Nemonoxacin | TaiGen Biotechnology (Taiwan) | Phase III | Broad-spectrum |  | [13,14,15,16,17,18] | |
| Ozenoxacin (topical use) | Ferrer Internacional (Spain) | Phase III | Gram-positive |  | [19,20,21] | |
| Zabofloxacin (fluoroquinolone) | Dong Wha Pharmaceutical (South Korea) | Phase III | Broad-spectrum |  | [22,23,24,25] | |
| Delafloxacin (fluoroquinolone) | Rib-X Pharmaceuticals (USA) | Phase III | Broad-spectrum |  | [26,27,28] | |
| Avarofloxacin (fluoroquinolone) | Furiex Pharmaceuticals (USA) | Phase II completed | Broad-spectrum |  | [29,30,31,32,33,34,35] | |
| Finafloxacin (fluoroquinolone) | MerLion Pharmaceuticals (Singapore) | Phase II | H. pylori, Broad-Spectrum |  | [36,37,38,39] | |
| WCK 2349 a prodrug of WCK 771 a,b (fluoroquinolone) | Wockhardt ( India) | Phase II | Gram-positive |  S-(−)-nadifloxacin b | [40,41,42,43] | |
| KRP-AM1977X (oral) | Kyorin (Japan) | Phase I | Respiratory infections | ND | [44,45] | |
| KRP-AM1977Y | Kyorin | Phase I | MRSA | ND | [44,45] | |
| DS-8587 (fluoroquinolone) | Daiichi Sankyo (Japan) | Phase I | Broad-spectrum |  | [46,47] | |
| ACH-702 (Isothiazoloquinolone) | Achillion Pharmaceuticals (USA) | Pre-Clinical | Broad-spectrum | 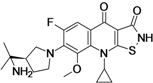 | [48,49] | |
| Aminoglycosides | ||||||
| Plazomicin | Achaogen (USA) | Phase II completed | Broad-spectrum |  | [50,51,52,53,54,55,56] | |
| Neomycin analogs c | Achaogen (USA) | Discovery | Broad-spectrum |  | [57,58,59,60,61,62,63] | |
| FY-901 | Changzhou Fangyuan Pharmaceutical (China) | Pre-Clinical | S. Aureus | ND | [64] | |
| FY-902 | Changzhou Fangyuan Pharmaceutical | Pre-Clinical | M. tuberculosis | ND | [64] | |
| New compounds | Changzhou Fangyuan Pharmaceutical | Discovery | ND | ND | [65] | |
| Macrolides | ||||||
| Solithromycin (2-fluoroketolide) | Cempra Pharmaceuticals (USA) | Phase III | S. pneumoniae; N. gonorrhoeae |  | [66,67,68,69,70,71,72] | |
| New Macrolides d | Cempra Pharmaceuticals | Discovery | ND |  | [73,74] | |
| EDP-788 (bicyclolide-bridged bicyclic macrolide) | Enanta Pharmaceuticals (USA) | Pre-Clinical | Gram-positive | ND | [75,76] | |
| new bicyclolides c | Enanta Pharmaceuticals | Discovery | Gram-positive |  | [77,78,79,80] | |
| WCK 4873 (Ketolide) | Wockhardt | Pre-Clinical | MDR pneumoccocus, Strep, H. influenzae | ND | [81,82] | |
| RX-02 program | Rib-X Pharmaceuticals | Discovery | Gram-positive | ND | [83] | |
| Oxazolidinones | ||||||
| Tedizolid | Cubist Pharmaceuticals (USA) | Phase III | Gram-positive |  | [84,85,86,87,88] | |
| Cadazolid | Actelion (Switzerland) | Phase II completed | Clostridium difficile |  | [89,90,91,92] | |
| Radezolid | Rib-X Pharmaceuticals | Phase II completed | Gram-positive | 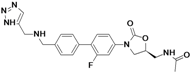 | [93,94,95,96,97,98] | |
| AZD5847 | AstraZeneca (UK) | Phase II | M. tuberculosis |  | [99,100,101,102] | |
| LCB01-0371 | LegoChem Biosciences (South Korea) | Phase I | Gram-positive |  | [103,104] | |
| MRX-I | MicuRx Pharmaceuticals (USA) | Phase I | Gram-positive |  | [105,106] | |
| MRX-II | MicuRx Pharmaceuticals | Pre-Clinical | Gram-positive | ND | [106] | |
| 2nd Generation Oxazolidinones | Wockhardt | Pre-Clinical | Gram-positive | ND | [81] | |
| Tetracyclines | ||||||
| Omadacycline (aminomethylcycline) | Paratek (USA) | Phase III | Broad-spectrum |  | [93,107,108] | |
| Aminomethylcyclines | Paratek | Discovery | Clostridium difficile | ND | [107] | |
| Eravacycline (fluorocycline) | Tetraphase Pharmaceuticals (USA) | Phase II (cIAI) | Gram-negative |  | [109,110,111,112] | |
| TP-271 | Tetraphase Pharmaceuticals | Pre-Clinical | bacterial biothreats, CABP pathogens |  | [109,113,114] | |
| TP-834 | Tetraphase Pharmaceuticals | Pre-Clinical | CABP pathogens | ND | [109] | |
| New tetracycline derivatives c | Tetraphase Pharmaceuticals | Discovery | Gram-negative |  | [115,116,117,118] | |
| Pleuromutilins | ||||||
| BC-3781 | Nabriva (Austria) | Phase II Completed | Gram-positive, including MRSA | 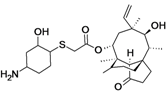 | [119,120,121,122,123,124] | |
| BC-7013 (topical) | Nabriva | Phase I | Gram-positive |  | [119,124] | |
| Discovery/development of pleuromutilins | Nabriva | Discovery | Broad spectrum |  | [119,125,126,127] | |
| Beta-lactams | ||||||
| Ceftolozane + tazobactam (CXA-201) | Cubist Pharmaceuticals | Phase III | Gram-negative |  (ceftolozane) | [6,128,129,130,131,132] | |
| BAL30072 (sulfactam) | Basilea Pharmaceutica (Switzerland) | Phase I | Gram-negative |  | [133,134,135] | |
| S-649266 (GSK2696266) (Cephalosporin) | Shionogi (Japan)/ GlaxoSmithKline (UK) | Phase I | Gram-negative | ND | [136,137,138] | |
| CB-027 (Cephalosporin) | Cubist Pharmaceuticals | Pre-Clinical | Broad-spectrum | ND | [139] | |
| FSI-1671 (Carbapenem) | FOB Synthesis, Inc. (USA) | Pre-Clinical | Gram-negative |  | [140,141] | |
| Beta-lactamase inhibitors—Diazabicyclooctanes (DBOs) | ||||||
| Ceftazidime/Avibactam (CAZ AVI) e | Forest Laboratories (USA)/Astrazeneca | Phase III | Gram-negative |  (avibactam) | [142,143,144,145,146,147,148] | |
| Ceftaroline/Avibactam (CXL) e | Forest Laboratories/ Astrazeneca | Phase II | Gram-negative |  (avibactam) | [120,143,147,148,149,150,151] | |
| MK-7655 + Imipenem/Cilastatin | Merck & Co., Inc. (USA) | Phase II | Gram-negative |  (MK-7655) | [6,147,148,152,153,154,155] | |
| FPI-1465 | Fedora Pharmaceuticals Inc. (USA) | Pre-Clinical | Gram-negative |  | [156,157] | |
| Beta-lactamase inhibitors—Boronate β-lactamase inhibitor | ||||||
| Carbavance (biapenem + RPX7009) | Rempex Pharmaceuticals (Sweden) | Phase I | Gram-negative |  (RPX7009) | [158,159,160,161] | |
| Beta-lactamase inhibitors—phosphonate-based beta-lactamase inhibitor (BLI) | ||||||
| MG96077 | Mirati Therapeutics (USA) | Pre-Clinical | Gram-negative | 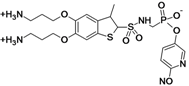 | [162,163] | |
| Lipoglycopeptides | ||||||
| Dalbavancin | Durata Therapeutics (USA) | Phase III completed | Gram-positive |  | [164,165,166,167,168,169,170,171,172,173] | |
| Oritavancin Diphosphate | The Medicines Company (USA) | Phase III completed | Gram-positive |  | [7,169,170,173,174] | |
| Polymyxins | ||||||
| NAB739 | Northern Antibiotics (Finland) | Pre-Clinical | Gram-negative |  | [89,175,176,177,178,179] | |
| NAB7061 | Northern Antibiotics | Pre-Clinical | Gram-negative |  | [176,177,178,179] | |
| NAB741 | Northern Antibiotics | Pre-Clinical | Gram-negative |  | [176,177,178,179] | |
| Cyclic lipopeptides | ||||||
| Surotomycin | Cubist Pharmaceuticals | Phase III | C. difficile |  | [180,181,182,183] | |
| WAP-8294A2 (lotilibcin) | Green Cross (South Korea) | Phase I | S. aureus | ND | [184,185,186] | |
4. Quinolones
5. Aminoglycosides
6. Macrolides
7. Oxazolidinones
8. Tetracyclines
9. Pleuromutilins
10. Beta-Lactams
11. Beta-Lactamase Inhibitors—Diazabicyclooctanes (DBOs)
12. Beta-Lactamase Inhibitors—Boronate β-Lactamase Inhibitor
13. Beta-Lactamase Inhibitors—Non-beta-Lactam Phosphonate-Based
14. Glycopeptides (Lipoglycopeptides)
15. Polymyxins
16. Cyclic Lipopeptides
17. Final Remarks
Conflicts of Interest
References
- Fischbach, M.; Walsh, C. Antibiotics for emerging pathogens. Science 2009, 325, 1089–1093. [Google Scholar] [CrossRef]
- Walsh, C.; Wencewicz, T. Prospects for new antibiotics: A molecule-centered perspective. J. Antibiot. 2013. [Google Scholar] [CrossRef]
- Ziemska, J.; Rajnisz, A.; Solecka, J. New perspectives on antibacterial drug research. Cent. Eur. J. Biol. 2013, 8, 943–957. [Google Scholar] [CrossRef]
- Boucher, H.W.; Talbot, G.H.; Benjamin, D.K.; Bradley, J.; Guidos, R.J.; Jones, R.N.; Murray, B.E.; Bonomo, R.A.; Gilbert, D. 10 × '20 Progress—Development of new drugs active against Gram-negative bacilli: An update from the infectious diseases society of America. Clin. Infect. Dis. 2013, 56, 1685–1694. [Google Scholar]
- Bassetti, M.; Merelli, M.; Temperoni, C.; Astilean, A. New antibiotics for bad bugs: Where are we? Ann. Clin. Microbiol. Antimicrob. 2013, 12. [Google Scholar] [CrossRef]
- Karras, G.; Giannakaki, V.; Kotsis, V.; Miyakis, S. Novel antimicrobial agents against multi-drug-resistant Gram-negative bacteria: An overview. Recent Pat. Antiinfect. Drug Discov. 2012, 7, 175–181. [Google Scholar] [CrossRef]
- Giannakaki, V.; Miyakis, S. Novel antimicrobial agents against multi-drug-resistant Gram-positive bacteria: An overview. Recent Pat. Antiinfect. Drug Discov. 2012, 7, 182–188. [Google Scholar] [CrossRef]
- Bush, K. Improving known classes of antibiotics: An optimistic approach for the future. Curr. Opin. Pharmacol. 2012, 12, 527–534. [Google Scholar] [CrossRef]
- Butler, M.S.; Blaskovich, M.A.; Cooper, M.A. Antibiotics in the clinical pipeline in 2013. J. Antibiot. 2013. [Google Scholar] [CrossRef]
- Collier, J.; Iheanacho, I. The pharmaceutical industry as an informant. Lancet 2002, 360, 1405–1409. [Google Scholar] [CrossRef]
- World Health Organization. WHO International Clinical Trials Registry Platform (ICTRP). Available online: http://www.who.int/ictrp/en/ (accessed on 30 December 2012).
- Dialog LLC. Chemical Business NewsBase (CBNB)—Bluesheet. Available online: http://library.dialog.com/bluesheets/html/bl0319.html (accessed on 17 February 2013).
- Beining, G.; Xiaojie, W.; Yingyuan, Z.; Yaoguo, S.; Jicheng, Y.; Guoying, C.; Jing, Z. Safety and clinical pharmacokinetics of nemonoxacin, a novel non-fluorinated quinolone, in healthy Chinese volunteers following single and multiple oral doses. Clin. Drug Investig. 2012, 32, 475–486. [Google Scholar] [CrossRef]
- Chung, D.T.; Tsai, C.-Y.; Chen, S.-J.; Chang, L.-W.; King, C.-H.R.; Hsu, C.-H.; Chiu, K.-M.; Tan, H.-C.; Chang, Y.-T.; Hsu, M.-C. Multiple-dose safety, tolerability, and pharmacokinetics of oral nemonoxacin (TG-873870) in healthy volunteers. Antimicrob. Agents Chemother. 2010, 54, 411–417. [Google Scholar] [CrossRef]
- TaiGen Biotechnology. Study to evaluate the efficacy and safety of oral administration with nemonoxacin and levofloxacin in patients with CAP. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01529476 (accessed on 15 April 2013).
- TaiGen Biotechnology. Safety and efficacy study of TG-873870 (Nemonoxacin) in diabetic foot infections. Available online: http://www.clinicaltrials.gov/ct2/show/NCT00685698 (accessed on 15 July 2013).
- TaiGen Biotechnology. Nemonoxacin: Once-a-day oral and IV, with excellent activity toward MRSA and VRSA. Available online: http://www.taigenbiotech.com.tw/research.html (accessed on 15 April 2013).
- TaiGen Biotechnology. TaiGen Biotechnology announces submission of new drug application for nemonoxacin in Taiwan and China. Available online: http://www.taigenbiotech.com.tw/news.html#25 (accessed on 15 July 2013).
- Ferrer Group. Key development projects. Available online: http://www.ferrergrupo.com/Innovation_Innovacion-Pipeline-de-proyectos-ENG (accessed on 15 April 2013).
- Yamakawa, T.; Mitsuyama, J.; Hayashi, K. In vitro and in vivo antibacterial activity of T-3912, a novel non-fluorinated topical quinolone. J. Antimicrob. Chemother. 2002, 49, 455–465. [Google Scholar] [CrossRef]
- Ferrer Internacional. Efficacy and safety of ozenoxacin 1% cream versus placebo in the treatment of patients with impetigo. Available online: http://clinicaltrials.gov/ct2/show/NCT01397461 (accessed on 13 April 2013).
- Kosowska-Shick, K.; Credito, K.; Pankuch, G.A.; Lin, G.; Bozdogan, B.; McGhee, P.; Dewasse, B.; Choi, D.-R.; Ryu, J.M.; Appelbaum, P.C. Antipneumococcal activity of DW-224a, a new quinolone, compared to those of eight other agents. Antimicrob. Agents Chemother. 2006, 50, 2064–2071. [Google Scholar] [CrossRef]
- Park, H.-S.; Kim, H.-J.; Seol, M.-J.; Choi, D.-R.; Choi, E.-C.; Kwak, J.-H. In vitro and in vivo antibacterial activities of DW-224a, a new fluoronaphthyridone. Antimicrob. Agents Chemother. 2006, 50, 2261–2264. [Google Scholar] [CrossRef]
- Dong Wha Pharmaceutical Co. Ltd. A study to evaluate efficacy and safety profile of Zabofloxacin tablet 400 mg and moxifloxacin tablet 400 mg. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01658020 (accessed on 15 July 2013).
- Dong Wha Pharmaceutical Co. Ltd. A new quinolone antibiotic. Available online: http://www.dong-wha.co.kr/english/rnd/rnd02_03.asp (accessed on 15 April 2013).
- Remy, J.M.; Tow-Keogh, C.A.; McConnell, T.S.; Dalton, J.M.; DeVito, J.A. Activity of delafloxacin against methicillin-resistant Staphylococcus aureus: Resistance selection and characterization. J. Antimicrob. Chemother. 2012, 67, 2814–2820. [Google Scholar] [CrossRef]
- Rib-X Pharmaceuticals. Delafloxacin. Available online: http://www.rib-x.com/pipeline/delafloxacin.php (accessed on 15 April 2013).
- Otsuka Pharmaceutical. Delafloxacin versus vancomycin and aztreonam for the treatment of acute bacterial skin and skin structure infections. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01811732 (accessed on 15 April 2013).
- Furiex Pharmaceuticals. Efficacy and safety study of JNJ-32729463 compared with moxifloxacin for the treatment of subjects requiring hospitalization for community-acquired bacterial pneumonia. Available online: http://clinicaltrials.gov/ct2/show/NCT01198626 (accessed on 13 April 2013).
- Furiex Pharmaceuticals. Efficacy and safety study of JNJ-32729463 for treating complicated skin and skin structure infections compared to linezolid (zyvox). Available online: http://www.clinicaltrials.gov/ct2/show/NCT01128530 (accessed on 22 July 2013).
- Furiex. JNJ-Q2 best-in-class, broad-spectrum fluoroquinolone with potent MRSA coverage. Available online: http://www.furiex.com/wp-content/uploads/2011/07/JNJ-Q2_Info-Sheet-JUNE-2011.pdf (accessed on 13 April 2013).
- Furiex. Furiex receives qualified infectious disease product and fast track designations from the U.S. Food and Drug Administration for Avarofloxacin (JNJ-Q2). Available online: http://investor.furiex.com/releasedetail.cfm?ReleaseID=742926 (accessed on 13 April 2013).
- Biedenbach, D.J.; Farrell, D.J.; Flamm, R.K.; Liverman, L.C.; McIntyre, G.; Jones, R.N. Activity of JNJ-Q2, a new fluoroquinolone, tested against contemporary pathogens isolated from patients with community-acquired bacterial pneumonia. Int. J. Antimicrob. Agents 2012, 39, 321–325. [Google Scholar] [CrossRef]
- Covington, P.; Davenport, J.M.; Andrae, D.; O’Riordan, W.; Liverman, L.; McIntyre, G.; Almenoff, J. Randomized, double-blind, phase II, multicenter study evaluating the safety/ tolerability and efficacy of JNJ-Q2, a novel fluoroquinolone, compared with linezolid for treatment of acute bacterial skin and skin structure infection. Antimicrob. Agents Chemother. 2011, 55, 5790–5797. [Google Scholar] [CrossRef]
- Morrow, B.J.; He, W.; Amsler, K.M.; Foleno, B.D.; Macielag, M.J.; Lynch, A.S.; Bush, K. In vitro antibacterial activities of JNJ-Q2, a new broad-spectrum fluoroquinolone. Antimicrob. Agents Chemother. 2010, 54, 1955–1964. [Google Scholar] [CrossRef]
- Durata Therapeutics, Inc. Finafloxacin for the treatment of cUTI and/or acute pyelonephritis. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01928433 (accessed on 28 September 2013).
- Merlion Pharma. A multi-dose, double-blind, double-dummy, active control, randomized clinical (Phase II) study of two dosing regimens of finafloxacin for the treatment of cUTI and/or acute pyelonephritis. Available online: http://www.clinicaltrialsregister.eu/ctr-search/trial/2011–006041–14/PL/ (accessed on 14 April 2013).
- Pharma, M. FDA Grants Qualified Infectious Disease Product Designation and Fast Track Status for MerLion Pharma’s Lead Antibacterial Candidate Finafloxacin; Merlion Pharma: Singapore, 2013; Volume 2013. [Google Scholar]
- Lemaire, S.; van Bambeke, F.; Tulkens, P.M. Activity of finafloxacin, a novel fluoroquinolone with increased activity at acid pH, towards extracellular and intracellular Staphylococcus aureus, Listeria monocytogenes and Legionella pneumophila. Int. J. Antimicrob. Agents 2011, 38, 52–59. [Google Scholar] [CrossRef]
- Appelbaum, P.C. 2012 and beyond: Potential for the start of a second pre-antibiotic era? J. Antimicrob. Chemother. 2012, 67, 2062–2068. [Google Scholar] [CrossRef]
- Wockhardt Limited. Phase II open label, multi-centeric clinical trial of oral WCK 2349 (1000 mg bid and 1200 mg bid) to evaluate safety and efficacy in the treatment of complicated skin and soft tissue infections caused by gram positive bacteria. Available online: http://www.ctri.nic.in/Clinicaltrials/pmaindet2.php?trialid=1797 (accessed on 15 April 2013).
- Wockhardt. New chemical entity pipeline. Available online: http://www.wockhardt.com/how-we-touch-lives/new-drug-discover.aspx (accessed on 15 April 2013).
- Peric, M.; Jacobs, M.R.; Appelbaum, P.C. Antianaerobic activity of a novel fluoroquinolone, WCK 771, compared to those of nine other agents. Antimicrob. Agents Chemother. 2004, 48, 3188–3192. [Google Scholar] [CrossRef]
- Kyorin. Kyorin—Main R&D Activities-1 (4 February 2013 Release). Available online: http://www.kyorin-pharm.co.jp/en/business/pdf/main_rd_activities_20130204_en.pdf (accessed on 4 February 2013).
- Kyorin. Drug discovery, development, and lcm with medical professionals and patients in mind. Available online: http://www.kyorin-gr.co.jp/en/business/gensen/r_and_d.shtml (accessed on 11 April 2013).
- Higuchi, S.; Onodera, Y.; Chiba, M.; Hoshino, K.; Gotoh, N. Potent in vitro antibacterial activity of DS-8587, a new generation of broad spectrum quinolone, against Acinetobacter baumannii. Antimicrob. Agents Chemother. 2013. [Google Scholar] [CrossRef]
- Daiichi Sankyo. Major R&D pipeline as of July, 2013. Available online: http://www.daiichisankyo.com/rd/pipeline/pdf/Pipeline_EN.pdf (accessed on 28 September 2013).
- Achillion Pharmaceuticals. About ACH-702. Available online: http://www.achillion.com/PL/pdf/04_ach_702_bg.pdf (accessed on 2 May 2013).
- Pucci, M.J.; Podos, S.D.; Thanassi, J.A.; Leggio, M.J.; Bradbury, B.J.; Deshpande, M. In vitro and in vivo profiles of ACH-702, an isothiazoloquinolone, against bacterial pathogens. Antimicrob. Agents Chemother. 2011, 55, 2860–2871. [Google Scholar] [CrossRef]
- Achaogen. Study for the treatment of complicated urinary tract infection and acute pyelonephritis. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01096849 (accessed on 11 April 2013).
- Zhanel, G.G.; Lawson, C.D.; Zelenitsky, S.; Findlay, B.; Schweizer, F.; Adam, H.; Walkty, A.; Rubinstein, E.; Gin, A.S.; Hoban, D.J.; et al. Comparison of the next-generation aminoglycoside plazomicin to gentamicin, tobramycin and amikacin. Expert Rev. Anti-Infect. Ther. 2012, 10, 459–473. [Google Scholar] [CrossRef]
- Endimiani, A.; Hujer, K.M.; Hujer, A.M.; Armstrong, E.S.; Choudhary, Y.; Aggen, J.B.; Bonomo, R.A. ACHN-490, a neoglycoside with potent in vitro activity against multidrug-resistant Klebsiella pneumoniae isolates. Antimicrob. Agents Chemother. 2009, 53, 4504–4507. [Google Scholar]
- Achaogen. Achaogen pipeline. Available online: http://www.achaogen.com (accessed on 30 August 2012).
- Achaogen. Achaogen Awarded $60M Contract Option by BARDA for the Clinical Development of Plazomicin. Available online: http://www.achaogen.com/news/151/15 (accessed on 19 June 2013).
- Achaogen. Achaogen announces all objectives met in Phase 2 Plazomicin complicated urinary tract infections study and start of first-in-human study with ACHN-975. Available online: http://www.achaogen.com/uploads/news/id148/Achaogen_PressRelease_2012–05–15.pdf (accessed on 10 April 2013).
- Achaogen. Achaogen Announces Agreement with FDA on a Special Protocol Assessment for a Phase 3 Clinical Trial of Plazomicin to Treat Infections Caused by Carbapenem-Resistant Enterobacteriaceae (CRE); Achaogen: San Francisco, CA, USA, 2013. [Google Scholar]
- Achaogen. Next-generation aminoglycosides. Available online: http://www.achaogen.com/pipeline/next-generation-aminoglycosides (accessed on 6 December 2012).
- Maianti, J.P.; Aggen, J.B.; Dozzo, P.; Goldblum, A.A.; Hildebrandt, D.J.; Kane, T.R.; Gliedt, M.J.; Linsell, M.S.; Hanessian, S.; Giguere, A. Antibacterial Aminoglycoside Analogs. WO2011044498A1, 14 April 2011. [Google Scholar]
- Dozzo, P.; Goldblum, A.A.; Aggen, J.B. Antibacterial Aminoglycoside Analogs. WO2011044502A1, 14 April 2011. [Google Scholar]
- Aggen, J.B.; Goldblum, A.A.; Dozzo, P.; Hildebrandt, D.J.; Kane, T.R.; Gliedt, M.J.; Linsell, M.S. Antibacterial Aminoglycoside Analogs. WO2011044503A1, 14 April 2011. [Google Scholar]
- Aggen, J.B.; Dozzo, P.; Goldblum, A.A.; Hildebrandt, D.J.; Kane, T.R.; Gliedt, M.J.; Linsell, M.S. Antibacterial Aminoglycoside Analogs. WO2011044538A1, 14 April 2011. [Google Scholar]
- Aggen, J.B.; Dozzo, P.; Goldblum, A.A.; Hildebrandt, D.J.; Kane, T.R.; Gliedt, M.J.; Linsell, M.S. Antibacterial Aminoglycoside Analogs. WO2011044501A2, 14 April 2011. [Google Scholar]
- Goldblum, A.A.; Dozzo, P.; Kane, T.R.; Aggen, J.B.; Linsell, M.S.; Hildebrandt, D.J.; Gliedt, M.J. Antibacterial Aminoglycoside Analogs. US20120122809A1, 17 May 2012. [Google Scholar]
- Changzhou Fangyuan Pharmaceutical. Achievements Introduced. Available online: http://s2.shenjian.net/Cn_about_ID13.html (accessed on 7 April 2013).
- Changzhou Fangyuan Pharmaceutical. Jiangsu a semi-sinthetic antibiotic engeenering technology research center. Available online: http://s2.shenjian.net/Cn_about_ID13.html (accessed on 6 December 2012).
- Cempra Pharmaceuticals. Solithromycin (CEM-101). Available online: http://www.cempra.com/products/Solithromycin-cem-101/ (accessed on 15 April 2013).
- Cempra Pharmaceuticals. Efficacy and safety study of oral solithromycin (CEM-101) compared to oral moxifloxacin in treatment of patients with community-acquired bacterial pneumonia. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01756339 (accessed on 15 April 2013).
- Cempra Pharmaceuticals. Safety and efficacy study of single-dose oral CEM-101 in patients with uncomplicated urogenital gonorrhea. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01591447 (accessed on 15 April 2013).
- Lemaire, S.; van Bambeke, F.; Tulkens, P.M. Cellular accumulation and pharmacodynamic evaluation of the intracellular activity of CEM-101, a novel fluoroketolide, against Staphylococcus aureus, Listeria monocytogenes, and Legionella pneumophila in Human THP-1 macrophages. Antimicrob. Agents Chemother. 2009, 53, 3734–3743. [Google Scholar]
- McGhee, P.; Clark, C.; Kosowska-Shick, K.M.; Nagai, K.; Dewasse, B.; Beachel, L.; Appelbaum, P.C. In vitro activity of CEM-101 against Streptococcus pneumoniae and Streptococcus pyogenes with defined macrolide resistance mechanisms. Antimicrob. Agents Chemother. 2010, 54, 230–238. [Google Scholar] [CrossRef]
- Fernandes, P.; Pereira, D.; Jamieson, B.; Keedy, K. Solithromycin: Macrolide antibiotic. Drugs Future 2011, 36, 751–758. [Google Scholar]
- Oldach, D.; Clark, K.; Schranz, J.; Das, A.; Craft, J.C.; Scott, D.; Jamieson, B.D.; Fernandes, P. Randomized, double-blind, multicenter phase 2 study comparing the efficacy and safety of oral solithromycin (CEM-101) to those of oral levofloxacin in the treatment of patients with community-acquired bacterial pneumonia. Antimicrob. Agents Chemother. 2013, 57, 2526–2534. [Google Scholar] [CrossRef]
- Cempra Pharmaceuticals. Preclinical programs—About macrolides. Available online: http://www.cempra.com/preclinical-programs/about-macrolides/ (accessed on 6 December 2012).
- Pereira, D.E. Hydrogen Bond Forming Fluoro Ketolides for Treating Diseases. WO2012034058A1, 15 March 2012. [Google Scholar]
- Enanta Pharmaceuticals. EDP-788/EDP-322. Available online: http://www.enanta.com/research/antibiotics/ (accessed on 11 April 2013).
- Enanta Pharmaceuticals. Enanta Pharmaceuticals Announces National Institute of Allergy and Infectious Diseases (NIAID) Will Fund Further Development of a Novel Bicyclolide; Business Wire: Watertown, NY, USA, 2013. [Google Scholar]
- Enanta Pharmaceuticals. Bicyclolide antibiotics. Available online: http://www.enanta.com/research/antibiotics/ (accessed on 6 December 2012).
- Enanta Pharmaceuticals. Enanta reports new macrolide-related drug class, bicyclolides, to combat hospital and community acquired superbug infections, including MRSA. Available online: http://www.prnewswire.com/news-releases/enanta-reports-new-macrolide-related-drug-class-bicyclolides-to-combat-hospital-and-community-acquired-superbug-infections-including-mrsa-57361732.html (accessed on 6 December 2012).
- Wang, G.; Xu, G.; Or, Y.S.; Gang, L.; Niu, D.; Fu, H.; Qui, Y.; Wang, Y.; Basayek, M.; Peng, Y.; et al. New 6–11 Bicyclic Macrolide Derivative Useful in Pharmaceutical Composition for Preventing and/or Treating Bacterial Infection in Humans. CN101955508-A, 26 January 2011. [Google Scholar]
- Kim, I.J.; Liu, T.; Long, J.; Wang, G.; Qiu, Y.; Kim, H.; Wang, Y.; Phan, L.T.; Or, Y.S. 6,11-Bridged Biaryl Macrolides. US2013165641-A1, 27 June 2013. [Google Scholar]
- Wockhardt. New chemical entities pipeline strategic focus on sepsis and antiinfectives. Available online: http://www.wockhardt.com/how-we-touch-lives/new-drug-discover.aspx (accessed on 4 April 2013).
- Wockhardt. Wockhardt annual report 2011–2012. Available online: http://www.wockhardt.com/pdf/Annual-Report-2011–2012-f932f.pdf (accessed on 4 April 2013).
- Rib-X Pharmaceuticals. Discovery programs—RX-02. Available online: http://www.rib-x.com/pipeline/discovery-programs.php (accessed on 12 July 2012).
- Vera-Cabrera, L.; Gonzalez, E.; Rendon, A.; Ocampo-Candiani, J.; Welsh, O.; Velazquez-Moreno, V.M.; Hak Choi, S.; Molina-Torres, C. In vitro activities of DA-7157 and DA-7218 against Mycobacterium tuberculosis and Nocardia brasiliensis. Antimicrob. Agents Chemother. 2006, 50, 3170–3172. [Google Scholar] [CrossRef]
- Prokocimer, P.; Bien, P.; DeAnda, C.; Pillar, C.M.; Bartizal, K. In vitro activity and microbiological efficacy of Tedizolid (TR-700) against Gram-positive clinical isolates from a phase 2 study of oral Tedizolid phosphate (TR-701) in patients with complicated skin and skin structure infections. Antimicrob. Agents Chemother. 2012, 56, 4608–4613. [Google Scholar] [CrossRef]
- Prokocimer, P.; de Anda, C.; Fang, E.; Mehra, P.; Das, A. Tedizolid phosphate vs. linezolid for treatment of acute bacterial skin and skin structure infections: The establish-1 randomized trial. JAMA 2003, 309, 559–569. [Google Scholar] [CrossRef]
- Trius Therapeutics. TR-701 FA vs. linezolid for the treatment of acute bacterial skin and skin structure infections. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01421511 (accessed on 15 April 2013).
- Smith, L. Trius: Clinical trials show important differentiation of tedizolid from zyvox (TSRX, $6.48). Available online: http://smithonstocks.com/trius-clinical-trials-show-important-differentiation-of-tedizolid-from-zyvox-tsrx-6–48/ (accessed on 15 April 2013).
- Shapiro, S. Speculative strategies for new antibacterials: All roads should not lead to Rome. J. Antibiot. (Tokyo) 2013, 66, 371–386. [Google Scholar] [CrossRef]
- Actelion Pharmaceuticals. Cadazolid. Available online: http://www1.actelion.com/en/scientists/development-pipeline/phase-3/cadazolid.page (accessed on 10 August 2013).
- Chilton, C.H.; Crowther, G.S.; Baines, S.D.; Todhunter, S.L.; Freeman, J.; Locher, H.H.; Athanasiou, A.; Wilcox, M.H. In vitro activity of cadazolid against clinically relevant Clostridium difficile isolates and in an in vitro gut model of C. difficile infection. J. Antimicrob. Chemother. 2013. [Google Scholar] [CrossRef]
- Rempex Pharmaceuticals. ACT-179811 in patients with Clostridium difficile infection (CDI). Available online: http://www.clinicaltrials.gov/ct2/show/NCT01222702 (accessed on 15 August 2013).
- Sutcliffe, J.A. Antibiotics in development targeting protein synthesis. Ann. NY Acad. Sci. 2011, 1241, 122–152. [Google Scholar] [CrossRef]
- Rib-X. Radezolid. Available online: http://www.rib-x.com/pipeline/radezolid.php#development (accessed on 14 April 2013).
- Rib-X Pharmaceuticals, Inc. Safety and efficacy study of oxazolidinone to treat pneumonia. Available online: http://www.clinicaltrials.gov/ct2/show/NCT00640926 (accessed on 14 April 2013).
- Rib-X Pharmaceuticals, Inc. Safety and efficacy study of oxazolidinones to treat uncomplicated skin infections. Available online: http://www.clinicaltrials.gov/ct2/show/NCT00646958 (accessed on 14 April 2013).
- Shaw, K.J.; Barbachyn, M.R. The oxazolidinones: Past, present, and future. Ann. NY Acad. Sci. 2011, 1241, 48–70. [Google Scholar] [CrossRef]
- Skripkin, E.; McConnell, T.S.; DeVito, J.; Lawrence, L.; Ippolito, J.A.; Duffy, E.M.; Sutcliffe, J.; Franceschi, F. Rχ-01, a new family of oxazolidinones that overcome ribosome-based linezolid resistance. Antimicrob. Agents Chemother. 2008, 52, 3550–3557. [Google Scholar] [CrossRef]
- AstraZeneca. New tuberculosis drug trial begins in South Africa. Available online: http://www.astrazeneca.com/Research/news/Article/20121210--new-tuberculosis-drug-trial-begins-in-south-africa (accessed on 12 April 2013).
- Working Group on New TB Drugs. AZD5847 oxazolidinone. Available online: http://www.newtbdrugs.org/project.php?id=174 (accessed on 12 April 2013).
- National Institute of Allergy and Infectious Diseases (NIAID). Phase 2a EBA trial of AZD5847. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01516203 (accessed on 12 April 2013).
- Villemagne, B.; Crauste, C.; Flipo, M.; Baulard, A.R.; Déprez, B.; Willand, N. Tuberculosis: The drug development pipeline at a glance. Eur. J. Med. Chem. 2012, 51, 1–16. [Google Scholar] [CrossRef]
- Jeong, J.-W.; Jung, S.-J.; Lee, H.-H.; Kim, Y.-Z.; Park, T.-K.; Cho, Y.-L.; Chae, S.-E.; Baek, S.-Y.; Woo, S.-H.; Lee, H.-S.; et al. In vitro and In vivo activities of LCB01–0371, a new oxazolidinone. Antimicrob. Agents Chemother. 2010, 54, 5359–5362. [Google Scholar] [CrossRef]
- LegoChem Biosciences. Multiple ascendoing dose study for LCB01–0371. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01842516 (accessed on 15 August 2013).
- MicuRx Pharmaceuticals, Inc. MicuRx and Shanghai Zhangjiang biomedical industry venture capital partner to develop next-generation antibiotic MRX-I for Chinese market. Available online: http://www.micurx.com/doc/10-24-12%20JV-FINAL.doc (accessed on 11 April 2013).
- MicuRx Pharmaceuticals. Discovery and development. Available online: http://www.micurx.com/d1.htm (accessed on 12 December 2012).
- Paratek. About Paratek. Available online: http://www.paratekpharm.com/ (accessed on 15 January 2013).
- Paratek. Paratek Pharmaceuticals announces FDA Grant of Qualified Infectious Disease Product (QIDP) designation for its lead product candidate, Omadacycline—QIDP status designated for both intravenous and oral formulations. Available online: http://www.paratekpharm.com/press/010313%20Paratek%20QIDP%20Press%20Release.pdf (accessed on 15 April 2013).
- Tetraphase Pharmaceuticals. Tetraphase pipeline—Overview. Available online: http://tphase.com/pipeline/overview/default.cfm#tp271 (accessed on 10 April 2013).
- Tetraphase Pharmaceuticals. Study to compare TP-434 and Ertapenem in CA complicated intra-abdominal infections. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01265784 (accessed on 14 April 2013).
- Grossman, T.H.; Starosta, A.L.; Fyfe, C.; O’Brien, W.; Rothstein, D.M.; Mikolajka, A.; Wilson, D.N.; Sutcliffe, J.A. Target- and resistance-based mechanistic studies with TP-434, a novel fluorocycline antibiotic. Antimicrob. Agents Chemother. 2012, 56, 2559–2564. [Google Scholar] [CrossRef]
- Ronn, M.; Zhu, Z.; Hogan, P.C.; Zhang, W.-Y.; Niu, J.; Katz, C.E.; Dunwoody, N.; Gilicky, O.; Deng, Y.; Hunt, D.K.; et al. Process R&D of Eravacycline: The first fully synthetic fluorocycline in clinical development. Org. Process Res. Dev. 2013, 17, 838–845. [Google Scholar]
- Cynamon, M.; Jureller, J.; Desai, B.; Ramachandran, K.; Sklaney, M.; Grossman, T.H. In vitro activity of TP-271 against Mycobacterium abscessus, Mycobacterium fortuitum, and Nocardia Species. Antimicrob. Agents Chemother. 2012, 56, 3986–3988. [Google Scholar] [CrossRef]
- Grossman, T.; Hunt, D.; Heine III, H.; Sutcliffe, J. TP-271, a novel oral fluorocycline for community-acquired respiratory and biothreat pathogens. In Proceedings of 50th International Interscience Conference on Antimicrobial Agents and Chemotherapy, Boston, MA, USA, 12–15 September 2010.
- Tetraphase. Our technology. Available online: http://tphase.com/technology/our_technology/default.cfm (accessed on 11 April 2013).
- Chen, C.-L.; Clark, R.B.; Deng, Y.; Plamondon, L.; Sun, C.; Xiao, X.-Y. Tetracycline Analogs. WO2012021712A1, 16 February 2012. [Google Scholar]
- Xiao, X.-Y.; Clark, R.; Hunt, D.; Roenn, M.; Plamondon, L.; He, M.; Sutcliffe, J.; Grossman, T. Polycyclic Tetracycline Compounds. WO2011123536A1, 6 October 2011. [Google Scholar]
- Deng, Y.; Plamondon, L.; Sun, C.; Xiao, X.-Y.; Zhou, J.; Sutcliffe, J.A.; Ronn, M.P. Tetracycline Compounds. WO2011025982A2, 3 March 2011. [Google Scholar]
- Nabriva. Pleuromutilins. Available online: http://www.nabriva.com/programs/pleuromutilins/ (accessed on 7 December 2012).
- Forest Laboratories. Our pipeline: Solid, and set for further growth. Available online: http://www.frx.com/research/pipeline.aspx (accessed on 13 April 2013).
- Sader, H.S.; Biedenbach, D.J.; Paukner, S.; Ivezic-Schoenfeld, Z.; Jones, R.N. Antimicrobial activity of the investigational pleuromutilin compound BC-3781 tested against Gram-positive organisms commonly associated with acute bacterial skin and skin structure infections. Antimicrob. Agents Chemother. 2012, 56, 1619–1623. [Google Scholar] [CrossRef]
- Sader, H.S.; Paukner, S.; Ivezic-Schoenfeld, Z.; Biedenbach, D.J.; Schmitz, F.J.; Jones, R.N. Antimicrobial activity of the novel pleuromutilin antibiotic BC-3781 against organisms responsible for community-acquired respiratory tract infections (CARTIs). J. Antimicrob. Chemother. 2012, 67, 1170–1175. [Google Scholar] [CrossRef]
- Nabriva Therapeutics AG. Study comparing the safety and efficacy of two doses of BC-3781 vs. vancomycin in patients with acute bacterial skin and skin structure infection (ABSSSI). Available online: http://www.clinicaltrials.gov/ct2/show/NCT01119105 (accessed on 13 April 2013).
- Novak, R. Are pleuromutilin antibiotics finally fit for human use? Ann. NY Acad. Sci. 2011, 1241, 71–81. [Google Scholar] [CrossRef]
- Riedl, R.; Thirring, K.; Heilmayer, W. Pleuromutilin Derivatives for Use in the Treatment of Diseases Mediated by Microbes. WO2012031307A1, 15 March 2012. [Google Scholar]
- Mang, R.; Thirring, K.; Heilmayer, W. Pleuromutilin Derivatives for Use in the Treatment of Diseases Mediated by Microbes. EP2433926A1, 28 March 2012. [Google Scholar]
- Heilmayer, W.; Mang, R.; Badegruber, R.; Stickmann, D.; Novak, R.; Ferencic, M.; Bulusu, A.R.C.M. Pleuromutilin Derivatives for the Treatment of Diseases Mediated by Microbes. MX2010009451A, 27 February 2012. [Google Scholar]
- García-Sánchez, J.E.; García-Merino, E.; Martín-del-Rey, Á.; García-Sánchez, E. Antibiotheraphy in the 21st century, antibacterials for the second decade. Posibilities or realities in the future? Rev. Esp. Quimioter. 2012, 25, 100–121. [Google Scholar]
- Cubist Pharmaceuticals. Safety and efficacy of intravenous CXA-201 and intravenous meropenem in complicated intraabdominal infections. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01445665 (accessed on 15 April 2013).
- Cubist Pharmaceuticals. Study comparing the safety and efficacy of intravenous CXA-201 and intravenous levofloxacin in complicated urinary tract infection, including pyelonephritis. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01345955 (accessed on 15 April 2013).
- Cubist Pharmaceuticals. Study comparing the safety and efficacy of intravenous CXA-201 and intravenous meropenem in complicated intraabdominal infections. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01445678 (accessed on 15 April 2013).
- Cubist Pharmaceuticals. Ceftolozane/tazobactam overview. Available online: http://www.cubist.com/products/cxa_201 (accessed on 15 August 2013).
- Basilea Pharmaceutica. Basilea Awarded Contract by BARDA of Up to USD 89 Million for the Development of Its Novel Antibiotic BAL30072; Basilea Pharmaceutica Ltd.: Basel, Switzerland, 2013; Volume 2013. [Google Scholar]
- Page, M.G.P.; Dantier, C.; Desarbre, E. In vitro properties of BAL30072, a novel siderophore sulfactam with activity against multiresistant Gram-negative bacilli. Antimicrob. Agents Chemother. 2010, 54, 2291–2302. [Google Scholar] [CrossRef]
- Basilea Pharmaceutica. Basilea Pharmaceutica Ltd. Annual Report 2012. Available online: http://annualreport.basilea.com/downloads/Basilea_AR12_E.pdf (accessed on 11 April 2013).
- Shionogi. Pipeline (as of August 2013). Available online: http://www.shionogi.co.jp/en/company/pmrltj0000000u4v-att/e_kaihatsu.pdf (accessed on 4 August 2013).
- Shionogi. Research and development at Shionogi. Available online: http://www.shionogi.com/pdf/RDmeeting2012.pdf (accessed on 12 April 2013).
- GlaxoSmithKline. GlaxoSmithKline product development pipeline 2013. Available online: http://www.gsk.com/content/dam/gsk/globals/documents/pdf/GSK%202013%20Pipeline.pdf (accessed on 11 April 2013).
- Cubist Pharmaceuticals. Cubist to Feature Antibiotic Development Programs at 52nd Annual Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC). Available online: http://www.businesswire.com/news/home/20120906005268/en/Cubist-Feature-Antibiotic-Development-Programs-52nd-Annual (accessed on 8 April 2013).
- Joo, H.; Choi, W.-B.; Kim, D.-I.; Kowalik, S.; Mao, E.; Li, Y.; Liu, S.; Hager, M.W. FSI-1671, a Novel Anti-Acinetobacter Carbapenem; in vitro Activity of FSI-1671 and FSI-1671/Sulbactam against MDR-A.baumannii. In Proceedings of 53rd International Interscience Conference on Antimicrobial Agents and Chemotherapy, Denver, CO, USA, 11 September 2013.
- Joo, H.; Choi, W.-B.; Kim, D.-I.; Kowalik, E.; Hager, M.W.; Mao, S.; Li, Y.; Liu, S. FSI-1671, a Novel Anti-Acinetobacter Carbapenem; in vivo Efficacy against Carbapenem-Resistance Gram-Negative Bacterial Infection. In Proceedings of 53rd International Interscience Conference on Antimicrobial Agents and Chemotherapy, Denver, CO, USA, 11 September 2013.
- AstraZeneca. Compare ceftazidime-avibactam and doripenem followed by oral therapy for hospitalized adults with complicated UTIs (Urinary Tract Infections). Available online: http://www.clinicaltrials.gov/ct2/show/NCT01599806 (accessed on 15 April 2013).
- AstraZeneca. Our Pipeline—Development pipeline as at 31 December 2012. Available online: http://www.astrazeneca.com/Research/Our-pipeline-summary (accessed on 12 April 2013).
- AstraZeneca; Cerexa, Inc. Ceftazidime-Avibactam for the treatment of infections due to ceftazidime resistant pathogens. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01644643 (accessed on 15 April 2013).
- AstraZeneca; Cerexa, Inc. Ceftazidime-Avibactam compared with doripenem followed by oral therapy for hospitalized adults with complicated UTIs (Urinary Tract Infections). Available online: http://www.clinicaltrials.gov/ct2/show/NCT01595438 (accessed on 15 April 2013).
- AstraZeneca; Cerexa, Inc. A study comparing Ceftazidime-Avibactam + Metronidazole versus Meropenem in adults with complicated intra-abdominal infections. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01500239 (accessed on 15 April 2013).
- Coleman, K. Diazabicyclooctanes (DBOs): A potent new class of non-beta-lactam beta-lactamase inhibitors. Curr. Opin. Microbiol. 2011, 14, 550–555. [Google Scholar] [CrossRef]
- Shlaes, D.M. New β-lactam-β-lactamase inhibitor combinations in clinical development. Ann. NY Acad. Sci. 2013, 1277, 105–114. [Google Scholar] [CrossRef]
- Cerexa, Inc. Comparative study of coadministered ceftaroline fosamil and NXL104 vs. intravenous doripenem in adult subjects with complicated Urinary Tract Infections. Available online: http://clinicaltrials.gov/ct2/show/NCT01281462 (accessed on 13 April 2013).
- Riccobene, T.A.; Su, S.F.; Rank, D. Single- and multiple-dose study to determine the safety, tolerability, and pharmacokinetics of ceftaroline fosamil in combination with avibactam in healthy subjects. Antimicrob. Agents Chemother. 2013, 57, 1496–1504. [Google Scholar] [CrossRef]
- Castanheira, M.; Sader, H.S.; Farrell, D.J.; Mendes, R.E.; Jones, R.N. Activity of ceftaroline-avibactam tested against Gram-negative organism populations, including strains expressing one or more β-lactamases and methicillin-resistant Staphylococcus aureus carrying various Staphylococcal cassette chromosome mec types. Antimicrob. Agents Chemother. 2012, 56, 4779–4785. [Google Scholar] [CrossRef]
- Merck. Merck pipeline. Available online: http://www.merck.com/research/pipeline/home.html (accessed on 22 February 2013).
- Merck & Co. Inc. Study of the safety, tolerability, and efficacy of MK-7655 + Imipenem/ Cilastatin versus Imipenem/Cilastatin Alone for the treatment of complicated Urinary Tract Infection (cUTI) (MK-7655–003 AM4). Available online: http://www.clinicaltrials.gov/ct2/show/NCT01505634 (accessed on 14 April 2013).
- Merck & Co. Inc. Study of the Safety, Tolerability, and Efficacy of MK-7655 + Imipenem/ Cilastatin versus Imipenem/Cilastatin Alone to Treat Complicated Intra-Abdominal Infection [cIAI] (MK-7655–004 AM2). Available online: http://www.clinicaltrials.gov/ct2/show/NCT01506271 (accessed on 14 April 2013).
- Hirsch, E.B.; Ledesma, K.R.; Chang, K.T.; Schwartz, M.S.; Motyl, M.R.; Tam, V.H. In vitro activity of MK-7655, a novel β-lactamase inhibitor, in combination with imipenem against carbapenem-resistant Gram-negative bacteria. Antimicrob. Agents Chemother. 2012, 56, 3753–3757. [Google Scholar] [CrossRef]
- Mendes, R.E.; Rhomberg, P.R.; Becker, H.K.; Jones, R.N. Activity of β-Lactam agents tested in combination with novel β-lactamase inhibitor compounds against enterobacteriaceae producing extended-spectrum β-Lactamases. In Proceedings of 53nd International Interscience Conference on Antimicrobial Agents and Chemotherapy, Denver, CO, USA, 12 September 2013.
- Fedora Pharmaceuticals. Fedora Pharmaceuticals Demonstrates that FPI-1465 Increases Activity of Certain Antibiotics against Drug-Resistant Bacteria; Fedora Pharmaceuticals: Edmonton, Canada, 2013. [Google Scholar]
- Shlaes, D.M. Antibiotics, ICAAC and Bronchitis. Available online: http://antibiotics-theperfectstorm.blogspot.com.br/2012/09/antibiotics-icaac-and-bronchitis.html (accessed on 12 April 2013).
- Rempex Abstracts were Published as Part of the 52nd ICAAC, September 2012, San Francisco, CA, USA. Available online: http://www.rempexpharma.com/pubs/icaac2012abstracts (accessed on 11 April 2013).
- Rempex Pharmaceuticals. Safety Study of Intravenous Biapenem (RPX2003) and RPX7009 Given Alone and in Combination. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01772836 (accessed on 12 April 2013).
- Rempex Pharmaceuticals. Safety, tolerability, pharmacokinetics of intravenous RPX7009 in healthy adult subjects. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01751269 (accessed on 12 April 2013).
- Martell, L.A.; Rahil, G.; Vaisburg, A.; Young, K.; Hickey, E.; Hermes, J.; Dininno, F.; Besterman, J.M. A Novel Beta-Lactamase Inhibitor Potentiates and Extends the Antibacterial Activity of Imipenem against β-Lactam-Resistant P. aeruginosa and K. pneumoniae. In Proceedings of 49th ICAAC Annual Meeting, San Francisco, CA, USA, 14 September 2009.
- Mirati Therapeutics. MG96077. Available online: http://mirati.com/other-pipeline-assets/mg96077 (accessed on 9 July 2013).
- Durata Therapeutics, Inc. Efficacy and safety of dalbavancin for the treatment of acute bacterial skin and skin structure infections. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01431339 (accessed on 13 April 2013).
- Durata Therapeutics. Product pipeline dalbavancin overview. Available online: http://www.duratatherapeutics.com/product-pipeline/dalbavancin/overview (accessed on 15 April 2013).
- Durata Therapeutics. Efficacy and Safety of Dalbavancin for the Treatment of ABSSSI. Available online: https://www.clinicaltrialsregister.eu/ctr-search/trial/2010-024042-30/DE/ (accessed on 14 April 2013).
- Durata Therapeutics. Durata therapeutics announces preliminary, topline Phase 3 clinical trial results for dalbavancin in the treatment of ABSSSI. Available online: http://content.stockpr.com/_news/duratatherapeutics/2013-02-25_Durata_Therapeutics_Announces_Preliminary_Topline_364.pdf (accessed on 15 April 2013).
- Durata Therapeutics. Durata therapeutics meets primary endpoints for EMA review in Phase 3 clinical trial. Available online: http://content.stockpr.com/_news/duratatherapeutics/2013-01-04_Durata_Therapeutics_Meets_Primary_Endpoints_for_EM_343.pdf (accessed on 15 April 2013).
- The Medicines Company. Evaluation of the effect of an investigational antibiotic (Oritavancin) in the treatment of skin and skin structure infections compared to a commonly used antibiotic. Available online: https://www.clinicaltrialsregister.eu/ctr-search/trial/2010-021720-10/GB/ (accessed on 15 April 2013).
- The Medicines Company. Oritavancin versus IV vancomycin for the treatment of patients with acute bacterial skin and skin structure infection (SOLO I). Available online: http://www.clinicaltrials.gov/ct2/show/NCT01252719 (accessed on 15 April 2013).
- The Medicines Company. Oritavancin versus IV vancomycin for the treatment of patients with acute bacterial skin and skin structure infection (SOLO II). Available online: http://www.clinicaltrials.gov/ct2/show/NCT01252732 (accessed on 15 April 2013).
- The Medicines Company. Product Development is Innovation. Available online: http://www.themedicinescompany.com/page/pipeline (accessed on 15 April 2013).
- Karaoui, L.R.; El-Lababidi, R.; Chahine, E.B. Oritavancin: An investigational lipoglycopeptide antibiotic. Am. J. Health Syst. Pharm. 2013, 70, 23–33. [Google Scholar] [CrossRef]
- Johnson, A.P. New antibiotics for selective treatment of gastrointestinal infection caused by Clostridium difficile. Expert Opin. Ther. Pat. 2010, 20, 1389–1399. [Google Scholar] [CrossRef]
- Vaara, M.; Sader, H.S.; Rhomberg, P.R.; Jones, R.N.; Vaara, T. Antimicrobial activity of the novel polymyxin derivative NAB739 tested against Gram-negative pathogens. J. Antimicrob. Chemother. 2013, 68, 636–639. [Google Scholar] [CrossRef]
- Northern Antibiotics. Northern antibiotics fights against multi-resistant gram-negatives. Available online: http://www.northernantibiotics.com/index2.php?pageid=3&lang=em (accessed on 9 April 2013).
- Vaara, M.; Vaara, T. Novel derivatives of polymyxin B, a review on the NAB compounds. Available online: http://www.northernantibiotics.com/kuvat/NAB%20Technical%20Info%20Sheet%20Nov%202010.pdf (accessed on 9 April 2013).
- Kadar, B.; Kocsis, B.; Nagy, K.; Szabo, D. The renaissance of polymyxins. Curr. Med. Chem. 2013, 20, 3759–3773. [Google Scholar] [CrossRef]
- Vaara, M. Novel derivatives of polymyxins. J. Antimicrob. Chemother. 2013, 68, 1213–1219. [Google Scholar] [CrossRef]
- Cubist Pharmaceuticals. Cubist products and pipeline. Available online: http://www.cubist.com/products/ (accessed on 15 April 2013).
- Cubist Pharmaceuticals. Study of CB-183,315 in patients with Clostridium difficile associated diarrhea. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01597505 (accessed on 15 April 2013).
- Cubist Pharmaceuticals. A study of CB-183,315 in patients with Clostridium difficile associated diarrhea. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01598311 (accessed on 15 April 2013).
- Mascio, C.T.M.; Mortin, L.I.; Howland, K.T.; van, P.A.D.G.; Zhang, S.; Arya, A.; Chuong, C.L.; Kang, C.; Li, T.; Silverman, J.A. In vitro and in vivo characterization of CB-183,315, a novel lipopeptide antibiotic for treatment of Clostridium difficile. Antimicrob. Agents Chemother. 2012, 56, 5023–5030. [Google Scholar] [CrossRef]
- Kato, A.; Nakaya, S.; Ohashi, Y.; Hirata, H.; Fujii, K.; Harada, K.-I. WAP-8294A2, a novel anti-MRSA antibiotic produced by Lysobacter sp. J. Am. Chem. Soc. 1997, 119, 6680–6681. [Google Scholar] [CrossRef]
- Kato, A.; Hirata, H.; Ohashi, Y.; Fujii, K.; Mori, K.; Harada, K.I. A new anti-MRSA antibiotic complex, WAP-8294A II. Structure characterization of minor components by ESI LCMS and MS/MS. J. Antibiot. (Tokyo) 2011, 64, 373–379. [Google Scholar] [CrossRef]
- Pharma Japan. aRigen licenses novel anti-MRSA to GCC in South Korea. Available online: http://www.pharma-japan.com/ (accessed on 15 April 2013).
- Emrich, N.-C.; Heisig, A.; Stubbings, W.; Labischinski, H.; Heisig, P. Antibacterial activity of finafloxacin under different pH conditions against isogenic strains of Escherichia coli expressing combinations of defined mechanisms of fluoroquinolone resistance. J. Antimicrob. Chemother. 2010, 65, 2530–2533. [Google Scholar] [CrossRef]
- Stubbings, W.; Leow, P.; Yong, G.C.; Goh, F.; Körber-Irrgang, B.; Kresken, M.; Endermann, R.; Labischinski, H. In vitro spectrum of activity of finafloxacin, a novel, pH-activated fluoroquinolone, under standard and acidic conditions. Antimicrob. Agents Chemother. 2011, 55, 4394–4397. [Google Scholar] [CrossRef]
- Bhagwat, S.S.; Mundkur, L.A.; Gupte, S.V.; Patel, M.V.; Khorakiwala, H.F. The anti-methicillin-resistant Staphylococcus aureus quinolone WCK 771 has potent activity against sequentially selected mutants, has a narrow mutant selection window against quinolone-resistant Staphylococcus aureus, and preferentially targets DNA gyrase. Antimicrob. Agents Chemother. 2006, 50, 3568–3579. [Google Scholar] [CrossRef]
- Bhagwat, S.S.; McGhee, P.; Kosowska-Shick, K.; Patel, M.V.; Appelbaum, P.C. In vitro activity of the quinolone WCK 771 against recent U.S. hospital and community-acquired Staphylococcus aureus pathogens with various resistance types. Antimicrob. Agents Chemother. 2009, 53, 811–813. [Google Scholar] [CrossRef]
- Zhu, D.; Hoshino, Y.K.K. DS-8587, A new generation of broad spectrum quinolone: In vitro antibacterial activities against Chinese clinical isolates. In Proceedings of 52nd International Interscience Conference on Antimicrobial Agents and Chemotherapy, San Francisco, CA, USA, 12 September 2012.
- Achillion Pharmaceuticals, Inc. SEC filling form 10-Q quarterly report filed August 7, 2013. Available online: http://ir.achillion.com/secfiling.cfm?filingID=1193125–13–324297 (accessed on 28 September 2013).
- Reyes, N.; Aggen, J.B.; Kostrub, C.F. In vivo efficacy of the novel aminoglycoside ACHN-490 in murine infection models. Antimicrob. Agents Chemother. 2011, 55, 1728–1733. [Google Scholar] [CrossRef]
- Cass, R.T.; Brooks, C.D.; Havrilla, N.A.; Tack, K.J.; Borin, M.T.; Young, D.; Bruss, J.B. Pharmacokinetics and safety of single and multiple doses of ACHN-490 injection administered intravenously in healthy subjects. Antimicrob. Agents Chemother. 2011, 55, 5874–5880. [Google Scholar] [CrossRef]
- Basilea Pharmaceutica. Exploratory programs. Available online: http://www.basilea.com/Research/Anti-infectives-research/ (accessed on 6 December 2012).
- Trius Therapeutics. TR-701 FA vs. linezolid for the treatment of acute bacterial skin and skin structure infections. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01170221 (accessed on 15 April 2013).
- Michalska, K.; Karpiuk, I.; Król, M.; Tyski, S. Recent development of potent analogues of oxazolidinone antibacterial agents. Biorg. Med. Chem. 2013, 21, 577–591. [Google Scholar] [CrossRef]
- Cubist Pharmaceuticals. Cubist Announces Submission of New Drug Application for Investigational Antibiotic Tedizolid for Treatment of Serious Skin Infections; Cubist Pharmaceuticals: Lexington, MA, USA.
- Noel, G.J.; Draper, M.P.; Hait, H.; Tanaka, S.K.; Arbeit, R.D. A randomized, evaluator-blind, phase 2 study comparing the safety and efficacy of omadacycline to those of linezolid for treatment of complicated skin and skin structure infections. Antimicrob. Agents Chemother. 2012, 56, 5650–5654. [Google Scholar] [CrossRef]
- Paratek Pharmaceuticals, Inc. Form S-1 Registration Statement under the Securities Act of 1933—Paratek Pharmaceuticals, Inc. Available online: http://www.sec.gov/Archives/edgar/data/1037643/000119312512406909/d386058ds1.htm#toc386058_10 (accessed on 5 August 2013).
- Tetraphase Pharmaceuticals. Efficacy and safety study of eravacycline compared with ertapenem in complicated intra-abdominal infections. Available online: http://www.clinicaltrials.gov/ct2/show/NCT01844856 (accessed on 10 August 2013).
- Tetraphase Pharmaceuticals. Tetraphase Outlines Plans for Phase 3 Clinical Trial of Eravacycline IV/Oral in Complicated Urinary Tract Infections; Tetraphase Pharmaceuticals: Watertown, MA, USA, 2013. [Google Scholar]
- Nabriva. Nabriva Therapeutics Retains BC-3781 Rights as Forest Collaboration Elapses; Nabriva: Vienna, Austria, 2013; Volume 2013. [Google Scholar]
- Hornsey, M.; Phee, L.; Stubbings, W.; Wareham, D.W. In vitro activity of the novel monosulfactam BAL30072 alone and in combination with meropenem versus a diverse collection of important Gram-negative pathogens. Int. J. Antimicrob. Agents 2013, 42, 343–346. [Google Scholar] [CrossRef]
- Sader, H.S.; Rhomberg, P.R.; Farrell, D.J.; Jones, R.N. Antimicrobial activity of CXA-101, a novel cephalosporin tested in combination with Tazobactam against enterobacteriaceae, Pseudomonas aeruginosa, and Bacteroides fragilis strains having various resistance phenotypes. Antimicrob. Agents Chemother. 2011, 55, 2390–2394. [Google Scholar] [CrossRef]
- Zhang, S.; Chuong, L.I.M.C.; Khang, I.C.; Alsup, A.; Li, T.; Arya, A.; He, Y.; Yin, N.; Rock, J.; Abel, C.; et al. In Vivo efficacy of CB-027 against methicillin-resistant Staphylococcus aureus, and ceftazidime-resistant Pseudomonas aeruginosa and Klebsiella pneumoniae infections in mice. In Proceedings of 52nd International Interscience Conference on Antimicrobial Agents and Chemotherapy, San Francisco, CA, USA, 10 September 2012.
- Lucasti, C.; Popescu, I.; Ramesh, M.K.; Lipka, J.; Sable, C. Comparative study of the efficacy and safety of ceftazidime/avibactam plus metronidazole versus meropenem in the treatment of complicated intra-abdominal infections in hospitalized adults: Results of a randomized, double-blind, Phase II trial. J. Antimicrob. Chemother. 2013, 68, 1183–1192. [Google Scholar] [CrossRef]
- Zhanel, G.; Lawson, C.; Adam, H.; Schweizer, F.; Zelenitsky, S.; Lagacé-Wiens, P.; Denisuik, A.; Rubinstein, E.; Gin, A.; Hoban, D.; et al. Ceftazidime-avibactam: A novel cephalosporin/ β-lactamase inhibitor combination. Drugs 2013, 73, 159–177. [Google Scholar] [CrossRef]
- Pucci, M.J.; Bush, K. Investigational antimicrobial agents of 2013. Clin. Microbiol. Rev. 2013, 26, 792–821. [Google Scholar] [CrossRef]
- Livermore, D.M.; Warner, M.; Mushtaq, S. Activity of MK-7655 combined with imipenem against Enterobacteriaceae and Pseudomonas aeruginosa. J. Antimicrob. Chemother. 2013, 68, 2286–2290. [Google Scholar]
- Maiti, S.N.; Ling, D.N.R.; Ha, C.M.; Ganguly, B.; Ou, L.; Shan, R.; Singh, R.; Kully, M.; Khan, J.; Salama, S.M.; et al. Design, synthesis and structure activity relationship of novel substituted amides containing diaza bicyclic heterocyclic compounds as broad-spectrum β-lactamase inhibitors. In Proceedings of 53nd International Interscience Conference on Antimicrobial Agents and Chemotherapy, Denver, CO, USA, 12 September 2013.
- Zhanel, G.G.; Calic, D.; Schweizer, F.; Zelenitsky, S.; Adam, H.; Lagacé-Wiens, P.R.S.; Rubinstein, E.; Gin, A.S.; Hoban, D.J.; Karlowsky, J.A. New lipoglycopeptides: A comparative review of Dalbavancin, Oritavancin and Telavancin. Drugs 2010, 70, 859–886. [Google Scholar] [CrossRef]
- The Medicines Company. FDA Grants QIDP designation to the medicines company’s investigational antibiotic oritavancin. Available online: http://ir.themedicinescompany.com/phoenix.zhtml?c=122204&p=irol-newsArticle&ID=1875172&highlight= (accessed on 12 November 2013).
- Goodman, L.S.; Gilman, A. Goodman and Gilman’s The Pharmacological Basis of Therapeutics, 12th ed.; McGraw-Hill Professional: Nova York, NY, USA, 2011; Volume 2, p. 1808. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
De Souza Mendes, C.D.; De Souza Antunes, A.M. Pipeline of Known Chemical Classes of Antibiotics. Antibiotics 2013, 2, 500-534. https://doi.org/10.3390/antibiotics2040500
De Souza Mendes CD, De Souza Antunes AM. Pipeline of Known Chemical Classes of Antibiotics. Antibiotics. 2013; 2(4):500-534. https://doi.org/10.3390/antibiotics2040500
Chicago/Turabian StyleDe Souza Mendes, Cristina D'Urso, and Adelaide Maria De Souza Antunes. 2013. "Pipeline of Known Chemical Classes of Antibiotics" Antibiotics 2, no. 4: 500-534. https://doi.org/10.3390/antibiotics2040500
APA StyleDe Souza Mendes, C. D., & De Souza Antunes, A. M. (2013). Pipeline of Known Chemical Classes of Antibiotics. Antibiotics, 2(4), 500-534. https://doi.org/10.3390/antibiotics2040500




