Enhanced Upconversion Luminescence in Yb3+/Tm3+-Codoped Fluoride Active Core/Active Shell/Inert Shell Nanoparticles through Directed Energy Migration
Abstract
:1. Introduction
2. Results and Discussion
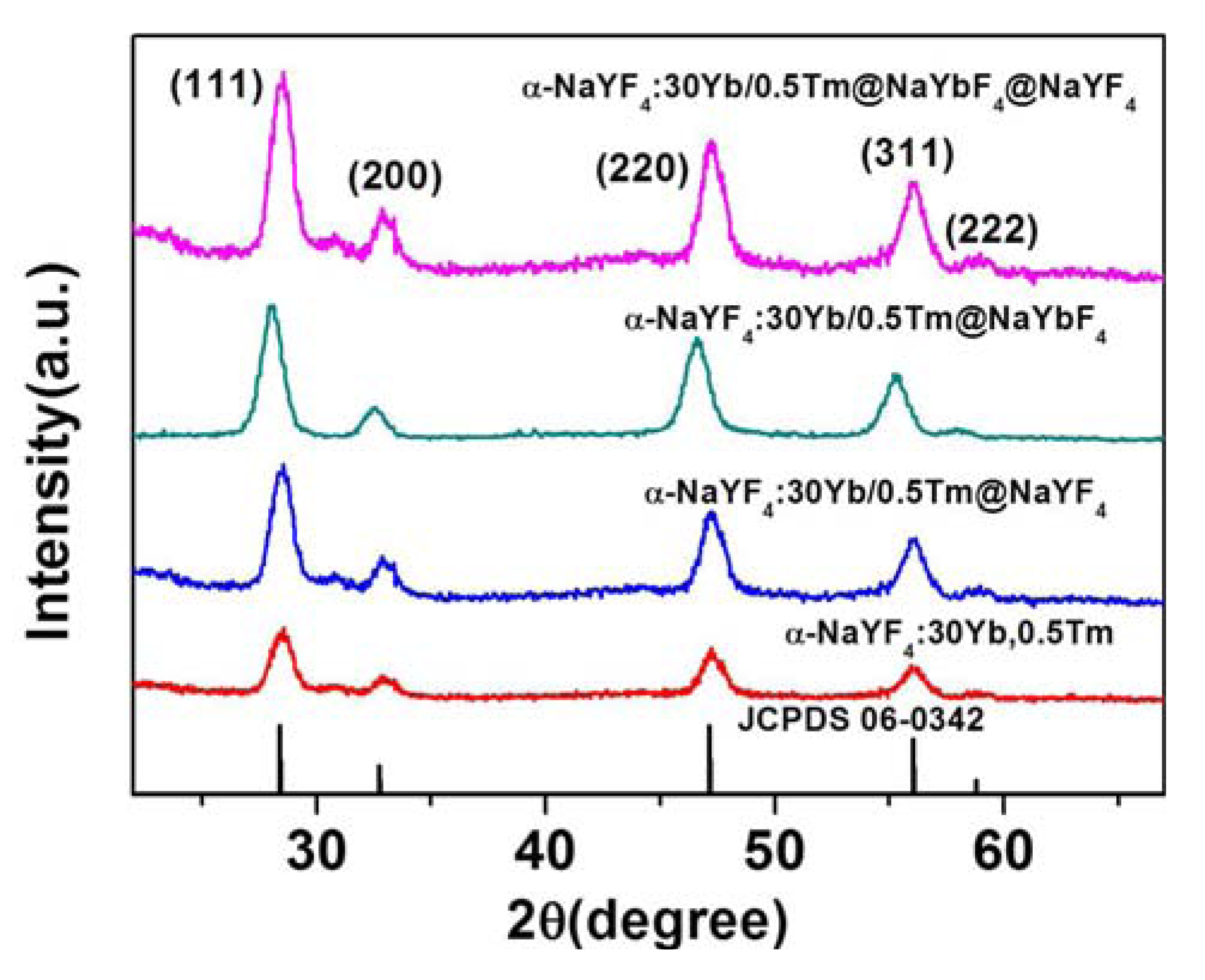
2.1. Characterizations of Morphology and Crystal Structure
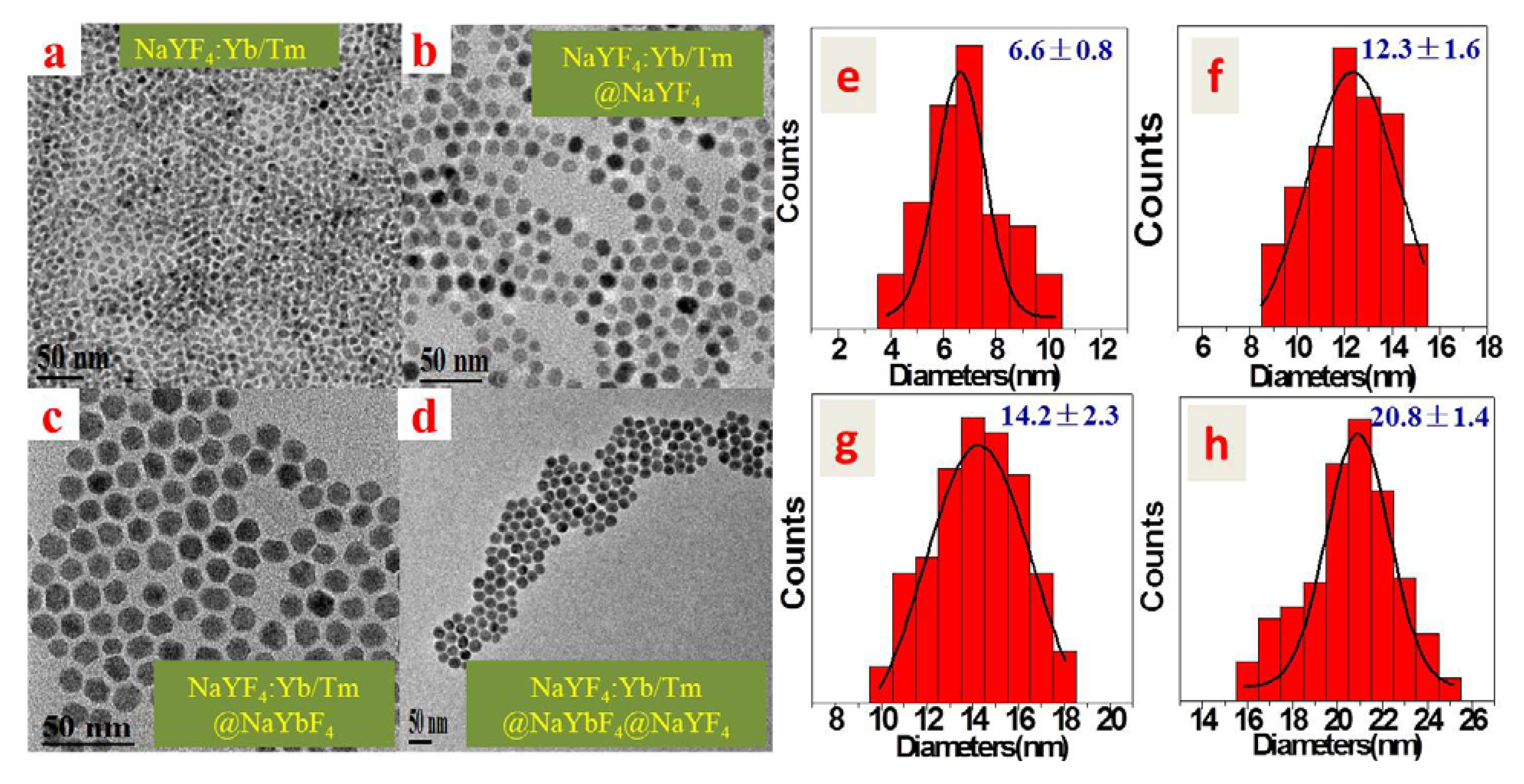
2.2. Upconversion Luminescence
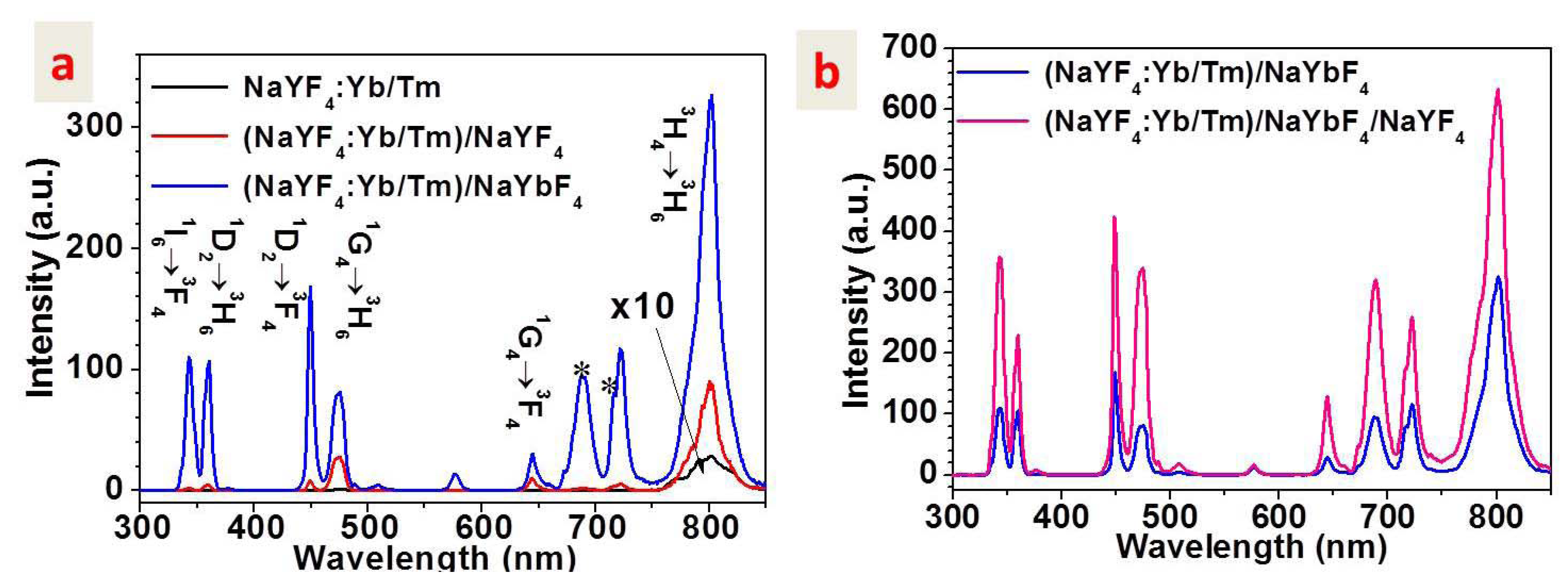
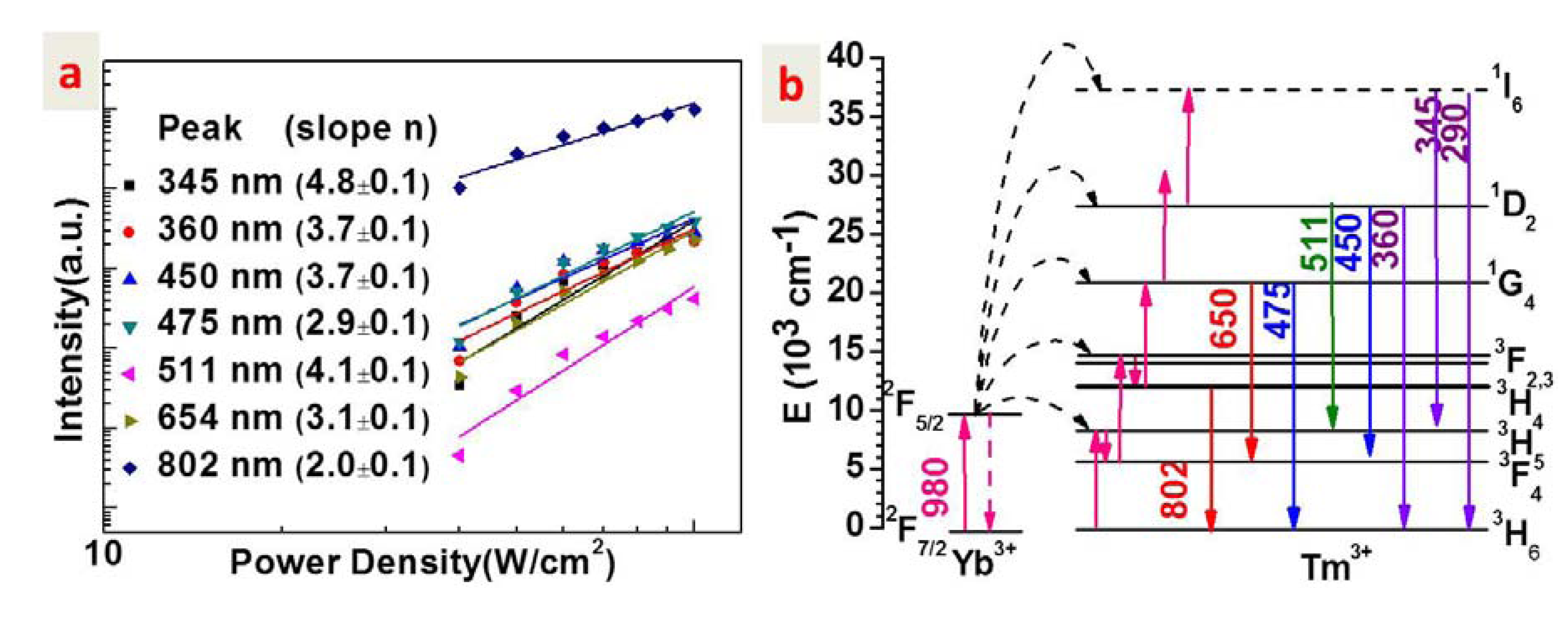
2.3. Upconversion Mechanism
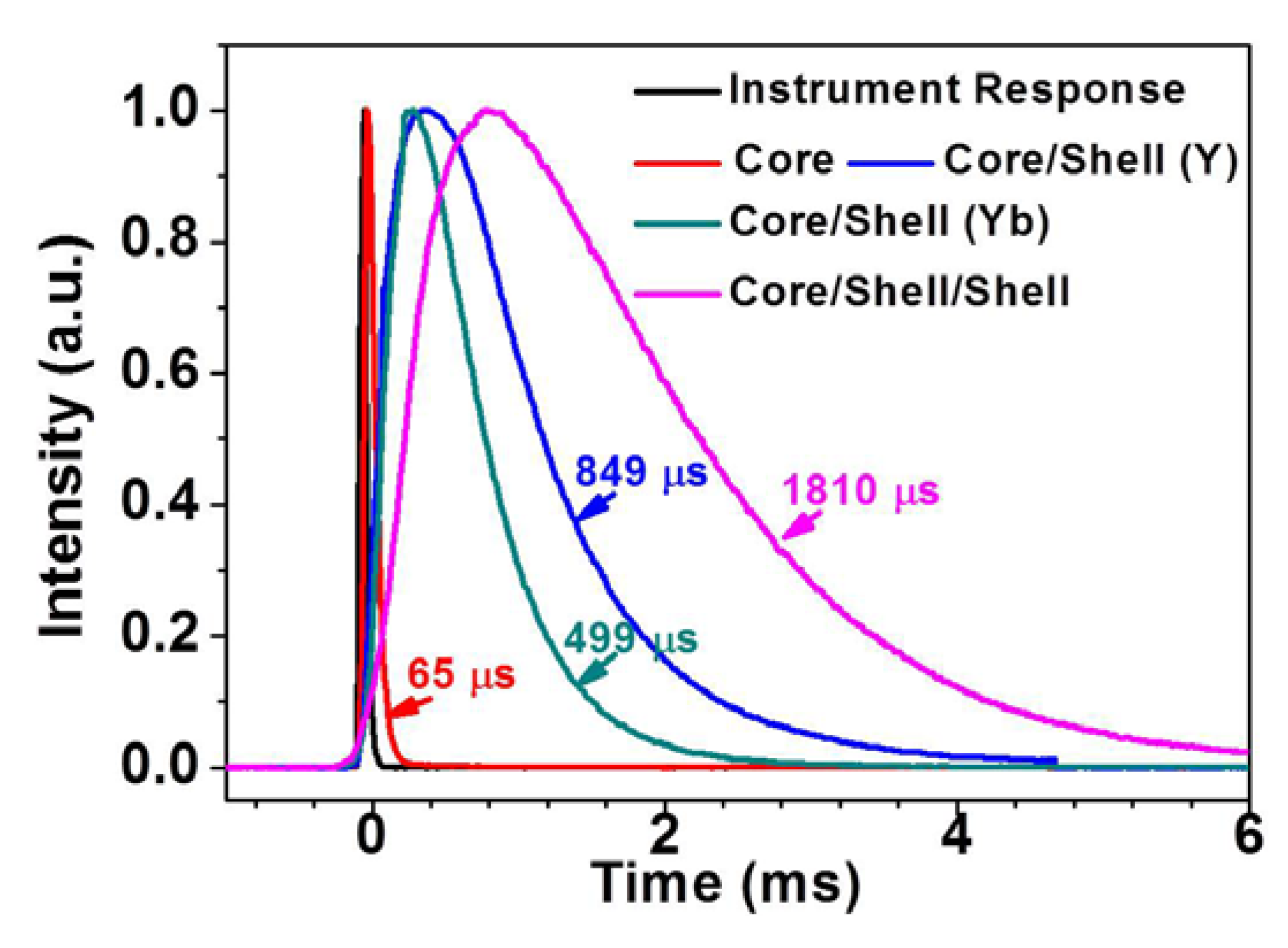
2.4. Decay of Upconversion Luminescence

2.5. Quenching and Enhancing Mechanisms
3. Experimental Section
3.1. Synthesis of NaYF4: Yb3+30%/Tm3+0.5% Core Nanoparticles
3.2. Synthesis of (NaYF4: Yb3+30%/Tm3+0.5%)/NaYbF4 Active Core/Active Shell Nanoparticles
3.3. Synthesis of (NaYF4: Yb3+30%/Tm3+0.5%)/NaYF4 Active Core/Inert Shell Nanoparticles
3.4. Synthesis of (NaYF4: Yb3+30%/Tm3+0.5%)/NaYbF4/NaYF4 Active Core/Active Shell/Inert Shell Nanoparticles
3.5. Instruments
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Auzel, F. Upconversion and anti-stokes processes with f and d ions in solids. Chem. Rev. 2004, 104, 139–174. [Google Scholar] [CrossRef]
- Wang, F.; Liu, X.G. Recent advances in the chemistry of lanthanide-doped upconversion nanocrystals. Chem. Soc. Rev. 2009, 38, 976–989. [Google Scholar] [CrossRef]
- Naccache, R.; Rodríguez, E.M.; Bogdan, N.; Sanz-Rodríguez, F.; Cruz, M.C.I.; Fuente, J.; Vetrone, F.; Jaue, D.; Solé, J.G.; Capobianco, J.A. High resolution fluorescence imaging of cancers using lanthanide ion-doped upconverting nanoparticles. Cancers 2012, 4, 1067–1105. [Google Scholar] [CrossRef]
- Chen, G.; Yang, C.; Prasad, P.N. Nanophotonics and nanochemistry: Controlling the excitation dynamics for frequency upconversion and downconversion in lanthanide-doped nanoparticles. Acc. Chem. Res. 2013, 46, 1474–1486. [Google Scholar] [CrossRef]
- Chen, G.; Seo, J.W.; Yang, C.; Prasad, P.N. Nanochemistry and nanomaterials for photovoltaics. Chem. Soc. Rev. 2013, 42, 8304–8338. [Google Scholar] [CrossRef]
- Mai, H.X.; Zhang, Y.W.; Si, R.; Yan, Z.G.; Sun, L.D.; You, L.P.; Yan, C.H. High-quality sodium rare-earth fluoride nanocrystals: Controlled synthesis and optical properties. J. Am. Chem. Soc. 2006, 128, 6426–6436. [Google Scholar]
- Boyer, J.C.; Cuccia, L.A.; Capobianco, J.A. Syntheis of colloidal upconverting NaYF4:Er3+/Yb3+ and Tm3+/Yb3+ monodisperse nanocrystals. Nano Lett. 2007, 7, 847–852. [Google Scholar]
- Ehlert, O.; Thomann, R.; Darbandi, M.; Nann, T. A four-color colloidal multiplexing nanoparticle system. ACS Nano 2008, 2, 120–124. [Google Scholar] [CrossRef]
- Wang, F.; Liu, X.G. Upconversion multicolor fine-tuning: Visible to near-infrared emission from lanthanide-doped NaYF4 nanocrystals. J. Am. Chem. Soc. 2008, 130, 5642–5643. [Google Scholar] [CrossRef]
- Chen, G.; Qiu, H.; Fan, R.; Hao, S.; Tan, S.; Yang, C.; Han, G. Lanthanide-doped ultrasmall yttrium fluoride nanoparticles with enhanced multicolor upconversion photoluminescence. J. Mater. Chem. 2012, 22, 20190–20196. [Google Scholar] [CrossRef]
- Krämer, K.W.; Biner, D.; Frei, G.; Güdel, H.U.; Hehlen, M.P.; Lüthi, S.R. Hexagonal sodium yttrium fluoride based green and blue emitting upconversion phosphors. Chem. Mater. 2004, 16, 1244–1251. [Google Scholar] [CrossRef]
- Zhao, J.; Jin, D.; Schartner, E.P.; Lu, Y.; Liu, Y.; Zvyagin, A.; Zhang, L.; Dawes, J.M.; Xi, P.; Piper, J.A.; et al. Single-nanocrystal sensitivity achieved by enhanced upconversion luminescence. Nat. Nanotechnol. 2013, 8, 729–734. [Google Scholar] [CrossRef]
- Chen, G.; Liu, H.; Liang, H.; Somesfalean, G.; Zhang, Z. Upconversion emission enhancement in Yb3+/Er3+-codoped Y2O3 nanocrystals by tridoping with Li+ ions. J. Phys. Chem. C 2008, 112, 12030–12036. [Google Scholar] [CrossRef]
- Chen, G.; Liu, H.; Liang, H.; Somesfalean, G.; Zhang, Z. Enhanced multiphoton ultraviolet and blue upconversion emission in Y2O3:Er3+ nanocrystals by codoping with Li+ ions. Solid State Commun. 2008, 148, 96–100. [Google Scholar] [CrossRef]
- Schietinger, S.; Aichele, T.; Wang, H.; Nann, T.; Benson, O. Plasmon-enhanced upconversion in Single NaYF4:Yb3+/Er3+ codoped nanocrystals. Nano Lett. 2010, 10, 134–138. [Google Scholar] [CrossRef]
- Vetrone, F.; Naccache, R.; Mahalingam, V.; Morgan, C.G.; Capobianco, J.A. The active-core/active-shell approach: A strategy to enhance the upconversion luminescence in lanthanide-doped nanoparticles. Adv. Funct. Mater. 2009, 19, 2924–2929. [Google Scholar] [CrossRef]
- Yang, D.; Li, C.; Li, G.; Shang, M.; Kang, X.; Lin, J. Colloidal synthesis and remarkable enhancement of the upconversion luminescence of BaGdF5:Yb3+/Er3+ nanoparticles by active-shell modification. J. Mater. Chem. 2011, 21, 5923–5927. [Google Scholar] [CrossRef]
- Wu, F.; Liu, X.; Kong, X.; Zhang, Y.; Tu, L.; Liu, K.; Song, S.; Zhang, H. The real role of active-shell in enhancing the luminescence of lanthanides doped nanomaterials. Appl. Phys. Lett. 2013, 102, 243104:1–243104:4. [Google Scholar]
- Chen, G.; Ohulchanskyy, T.Y.; Kumar, R.; Ågren, H.; Prasad, P.N. Ultrasmall monodisperse NaYF4:Yb3+/Tm3+ nanocrystals with enhanced near-infrared to near-infrared upconversion photoluminescence. ACS Nano 2010, 4, 3163–3168. [Google Scholar] [CrossRef]
- Chen, G.; Shen, J.; Ohulchanskyy, T.Y.; Patel, N.J.; Kuikov, A.; Li, Z.; Song, J.; Pandey, R.K.; Ågren, H.; Prasad, P.N. (α-NaYbF4:Tm3+)/CaF2 core/shell nanoparticles with efficient near-infrared to near-infrared upconversion for high-contrast deep tissue bioimaging. ACS Nano 2012, 6, 8280–8287. [Google Scholar] [CrossRef]
- Shen, J.; Chen, G.; Ohulchanskyy, T.Y.; Kesseli, S.J.; Buchholz, S.; Li, Z.; Ågren, H.; Prasad, P.N.; Han, G. Tunable near infrared to ultraviolet upconversion luminescence enhancement in (α-NaYF4:Yb,Tm)/CaF2 core/shell nanoparticles for in situ real-time recorded biocompatible photoactivation. Small 2013, 9, 3213–3217. [Google Scholar]
- Chen, X.; Zhao, Z.; Jiang, M.; Que, D.; Shi, S.; Zheng, N. Preparation and photodynamic therapy application of NaYF4:Yb,Tm-NaYF4:Yb,Er multifunctional upconverting nanoparticles. New J. Chem. 2013, 37, 1782–1788. [Google Scholar] [CrossRef]
- Yi, G.S.; Chow, G.M. Water-soluble NaYF4:Yb,Er(Tm)/NaYF4/polymer core/shell/shell nanoparticles with significant enhancement of upconversion fluorescence. Chem. Mater. 2007, 19, 341–343. [Google Scholar] [CrossRef]
- Wang, G.; Qin, W.; Wang, L.; Wei, G.; Zhu, P.; Kim, R. Intense ultraviolet upconversion luminescence from hexagonal NaYF4:Yb3+/Tm3+ microcrystals. Opt. Express 2008, 16, 11907–11914. [Google Scholar] [CrossRef]
- Chen, G.; Ohulchanskyy, T.Y.; Law, W.C.; Ågren, H.; Prasad, P.N. Monodisperse NaYbF4:Tm3+/NaGdF4 core/shell nanocrystals with near-infrared to near-infrared upconversion photoluminescence and magnetic resonance properties. Nanoscale 2011, 3, 2003–2008. [Google Scholar]
- Wang, F.; Liu, X. Direct evidence of a surface quenching effect on size-dependent luminescence of upconversion nanoparticles. Angew. Chem. 2010, 49, 7456–7460. [Google Scholar] [CrossRef]
- Zhang, F.; Che, R.; Li, X.; Yao, C.; Yang, J.; Shen, D.; Hu, P.; Li, W.; Zhao, D. Direct imaging the upconversion nanocrystal core/shell structure at the subnanometer level: Shell thickness dependence in upconverting optical properties. Nano Lett. 2012, 12, 2852–2858. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Qiu, H.; Yang, C.; Shao, W.; Damasco, J.; Wang, X.; Ågren, H.; Prasad, P.N.; Chen, G. Enhanced Upconversion Luminescence in Yb3+/Tm3+-Codoped Fluoride Active Core/Active Shell/Inert Shell Nanoparticles through Directed Energy Migration. Nanomaterials 2014, 4, 55-68. https://doi.org/10.3390/nano4010055
Qiu H, Yang C, Shao W, Damasco J, Wang X, Ågren H, Prasad PN, Chen G. Enhanced Upconversion Luminescence in Yb3+/Tm3+-Codoped Fluoride Active Core/Active Shell/Inert Shell Nanoparticles through Directed Energy Migration. Nanomaterials. 2014; 4(1):55-68. https://doi.org/10.3390/nano4010055
Chicago/Turabian StyleQiu, Hailong, Chunhui Yang, Wei Shao, Jossana Damasco, Xianliang Wang, Hans Ågren, Paras N. Prasad, and Guanying Chen. 2014. "Enhanced Upconversion Luminescence in Yb3+/Tm3+-Codoped Fluoride Active Core/Active Shell/Inert Shell Nanoparticles through Directed Energy Migration" Nanomaterials 4, no. 1: 55-68. https://doi.org/10.3390/nano4010055




