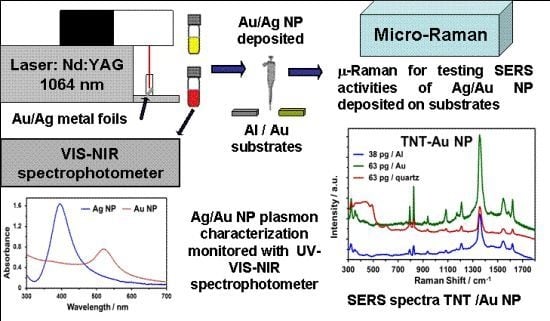
Surface Enhanced Raman Scattering (SERS) Studies of Gold and Silver Nanoparticles Prepared by Laser Ablation
Abstract
:1. Introduction
2. Experimental Section
2.1. Laser Ablation Synthesis
2.2. Characterization of NPs Suspensions
2.3. Effect of pH on the Synthesis of NPs
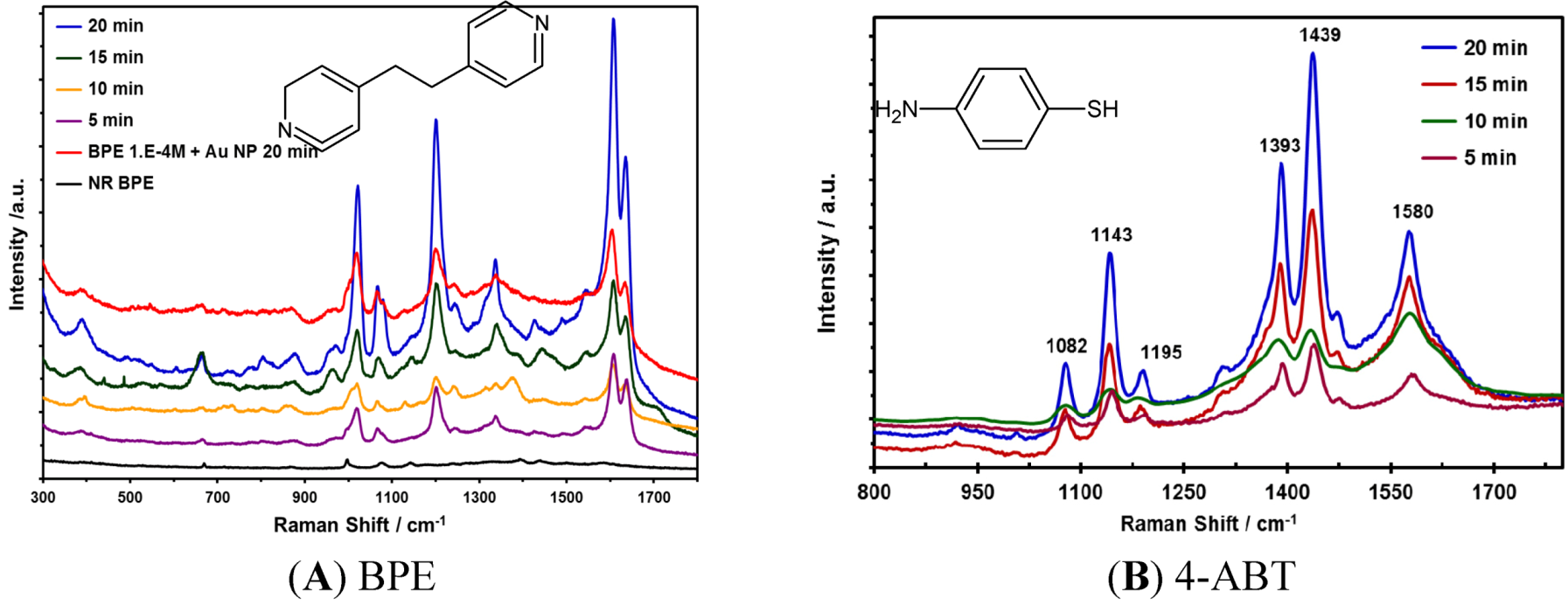
2.4. Evaluation of SERS Activity
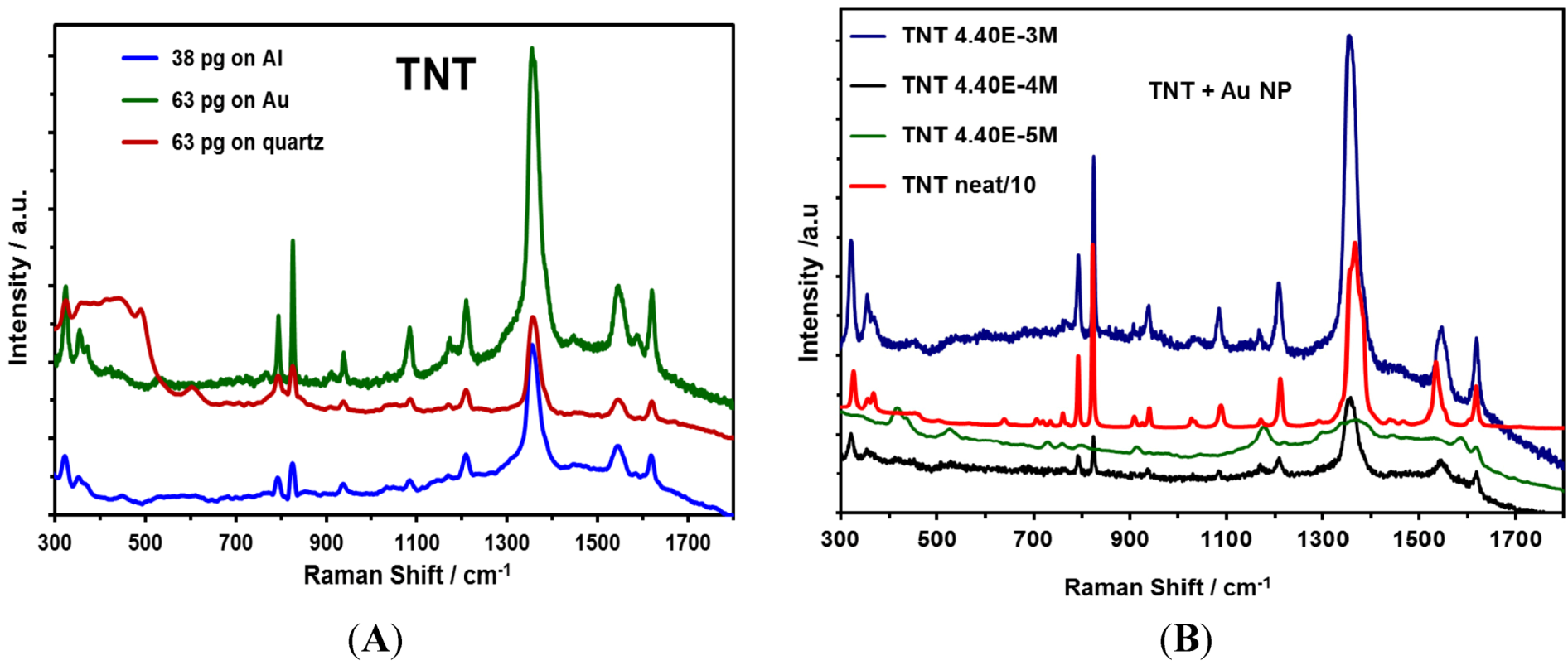
2.5. Detection of TNT Using Au NP/Au Substrates
3. Results and Discussion
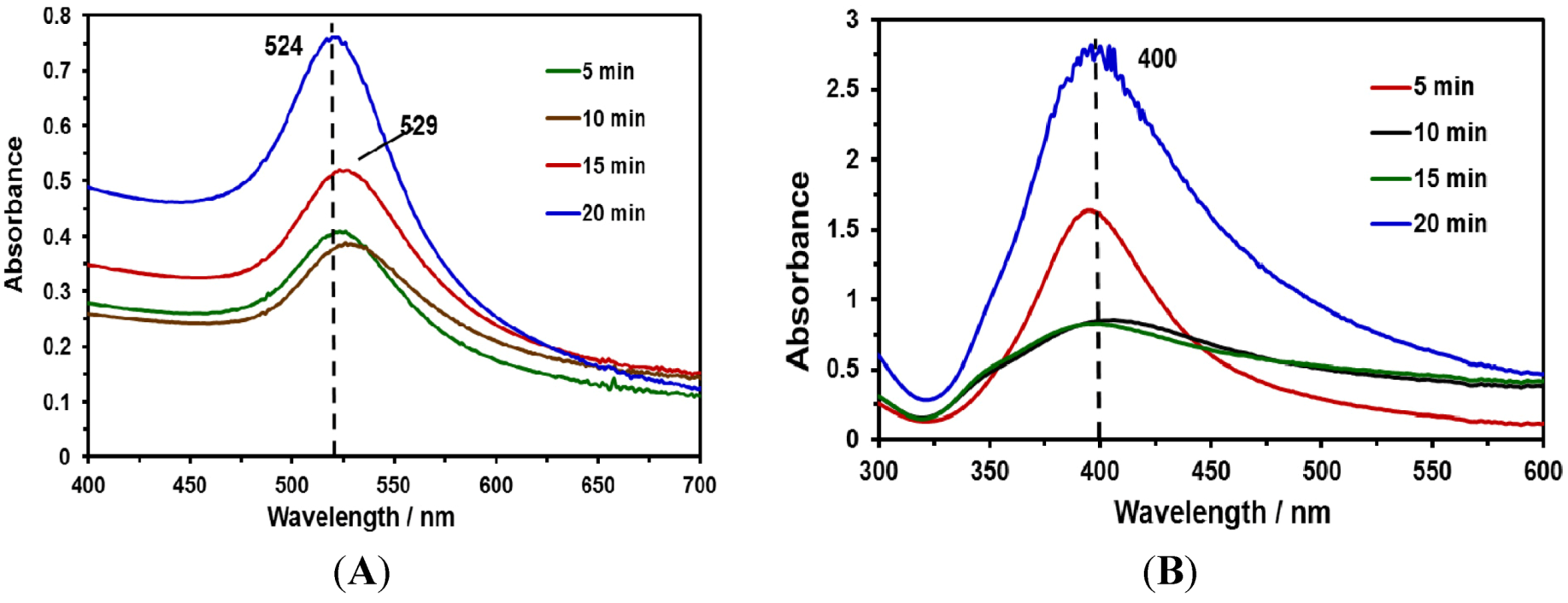

| Nanoparticle Type | Irradiation Time (min) | Average Size (Z size, nm) |
|---|---|---|
| Au | 5 | 96 |
| Au | 10 | 82 |
| Au | 15 | 71 |
| Au | 20 | 75 |
| Ag | 5 | 77 |
| Ag | 20 | 66 |

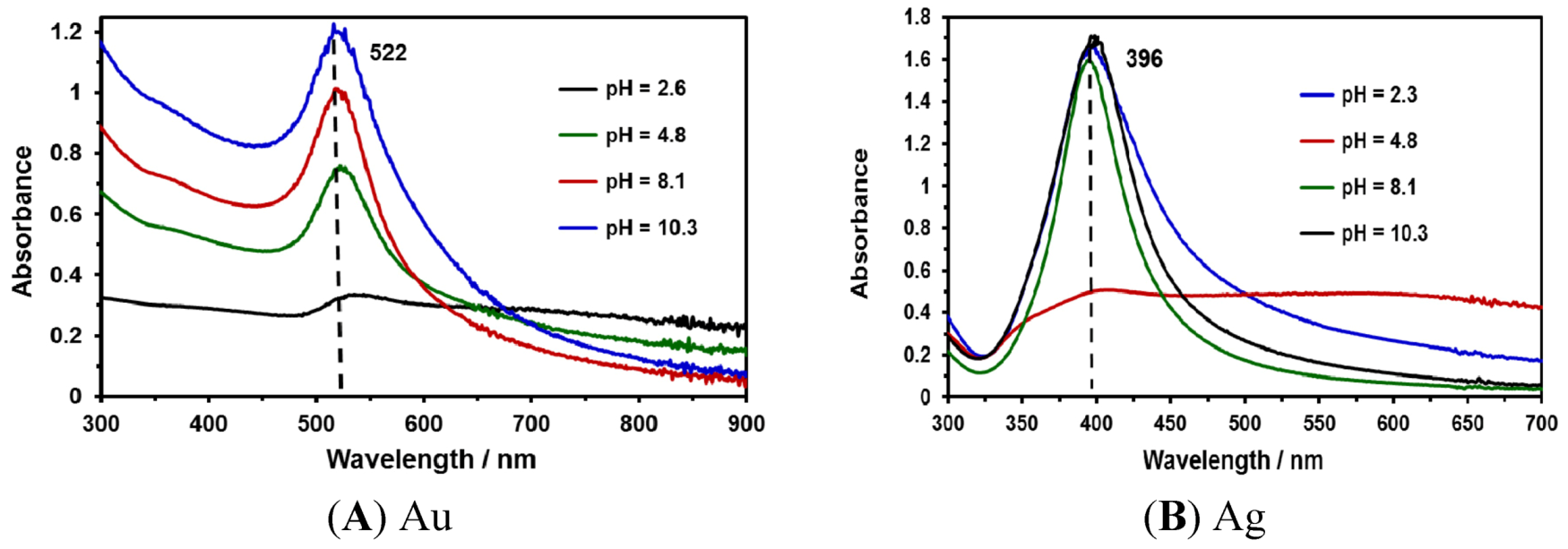
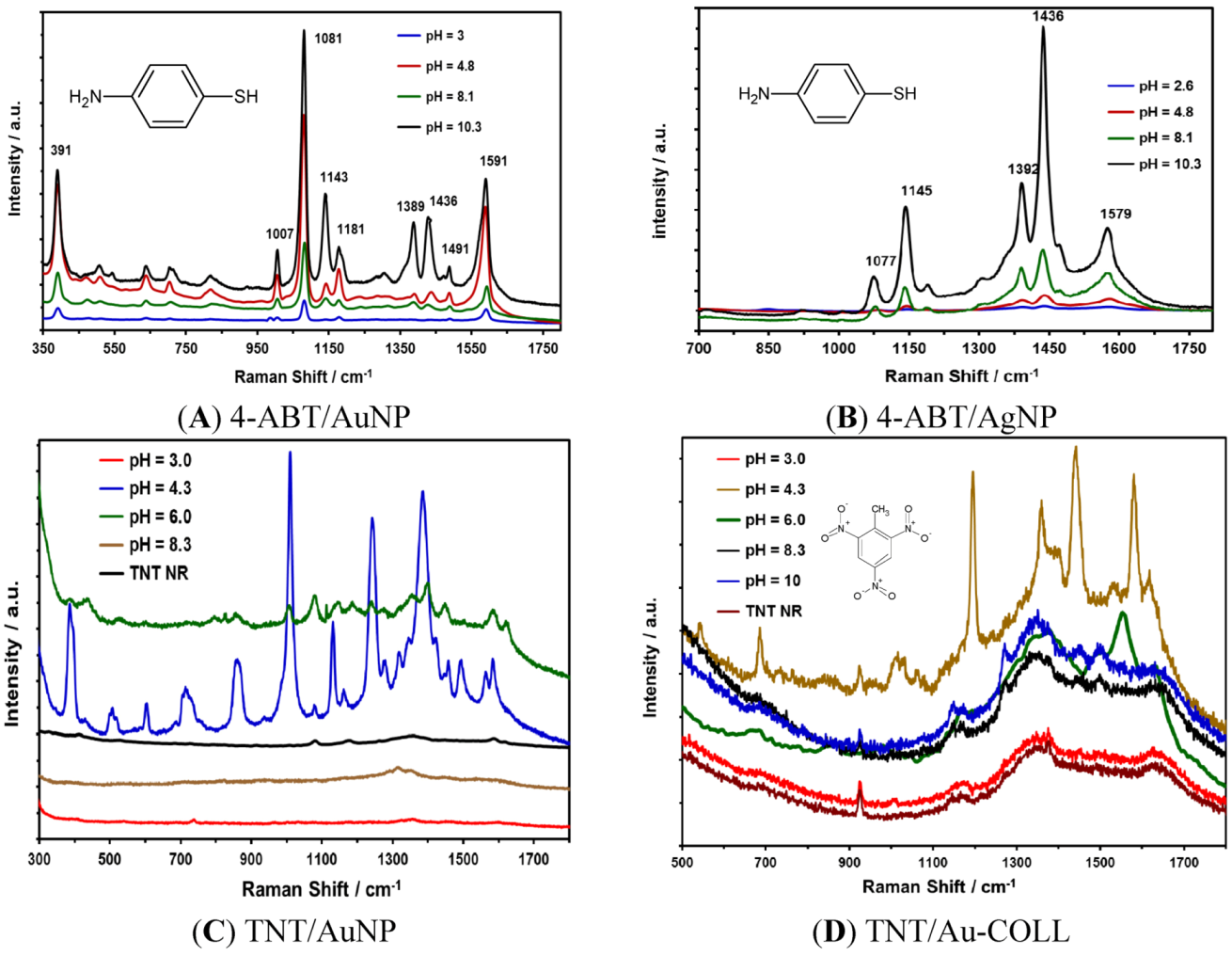
| NP Type | pH | Average Size (nm) | Z Potential (mV) |
|---|---|---|---|
| Ag | 4.2 | 96 | −18.0 |
| Ag | 6.0 | 90 | −21.8 |
| Ag | 8.3 | 87 | −24.1 |
| Ag | 10.0 | 72 | −26.2 |
| Ag | 10.8 | 70 | −29.1 |
| Au | 4.3 | 93 | −11.1 |
| Au | 6.0 | 75 | −39.2 |
| Au | 8.3 | 73 | −3.7 |
| Au | 10.0 | 47 | −5.2 |
4. Conclusions
Acknowledgments
References
- Hamouda, I.M. Current perspectives of nanoparticles in medical and dental biomaterials. J. Biomed. Res. 2012, 26, 143–151. [Google Scholar] [CrossRef]
- Albrecht, M.; Janke, V.; Sievers, S.; Siegner, U.; Schüler, D.; Heyen, U. Scanning force microscopy study of biogenic nanoparticles for medical applications. J. Magn. Magn. Mater. 2005, 290–291, 269–271. [Google Scholar]
- Rai, M.; Yadav, A.; Gade, A. Silver nanoparticles as a new generation of antimicrobials. Biotechnol. Adv. 2009, 27, 76–83. [Google Scholar] [CrossRef]
- Piao, M.J.; Kang, K.A.; Lee, I.K.; Kim, H.S.; Kim, S.; Choi, J.Y.; Choi, J.; Hyun, J.W. Silver nanoparticles induce oxidative cell damage in human liver cells through inhibition of reduced glutathione and induction of mitochondria-involved apoptosis. Toxicol. Lett. 2011, 201, 92–100. [Google Scholar] [CrossRef]
- Chen, X.; Schluesener, H.J. Nanosilver: A nanoproduct in medical application. Toxicol. Lett. 2008, 176, 1–12. [Google Scholar] [CrossRef]
- Wei, D.; Ye, Y.; Jia, X.; Yuan, C.; Qian, W. Chitosan as an active support for assembly of metal nanoparticles and application of the resultant bioconjugates in catalysis. Carbohydr. Res. 2010, 345, 74–81. [Google Scholar] [CrossRef]
- Lee, K.Y.; Hwang, J.; Lee, Y.W.; Kim, J.; Han, S.W. One-step synthesis of gold nanoparticles using azacryptand and their applications in SERS and catalysis. J. Colloid Interface Sci. 2007, 316, 476–481. [Google Scholar] [CrossRef]
- Hutchings, G.J.; Edwards, J.K. Application of Gold Nanoparticles in Catalysis. Chapter 6. In Frontiers of Nanoscience; Roy, L.J., Wilcoxon, J.P., Eds.; Elsevier: Amsterdam, Netherlands, 2012; Volume 3, pp. 249–293. [Google Scholar]
- Niu, Y.; Crooks, R.M. Dendrimer-encapsulated metal nanoparticles and their applications to catalysis. Comptes Rendus Chimie 2003, 6, 1049–1059. [Google Scholar] [CrossRef]
- Chang, C.-C.; Yang, K.-H.; Liu, Y.-C.; Hsu, T.-C. New pathway to prepare gold nanoparticles and their applications in catalysis and surface-enhanced Raman scattering. Colloids Surf. B 2012, 93, 169–173. [Google Scholar] [CrossRef]
- Yang, J.; Pan, J. Hydrothermal synthesis of silver nanoparticles by sodium alginate and their applications in surface-enhanced Raman scattering and catalysis. Acta Mater. 2012, 60, 4753–4758. [Google Scholar] [CrossRef]
- Eriksson, S.; Nylén, U.; Rojas, S.; Boutonnet, M. Preparation of catalysts from microemulsions and their applications in heterogeneous catalysis. Appl. Catal. A 2004, 265, 207–219. [Google Scholar] [CrossRef]
- Haruta, M. Catalysis and applications of gold nanoparticles. In Studies in Surface Science and Catalysis; Masakazu Anpo, M.O., Hiromi, Y., Eds.; Elsevier: Amsterdam, Netherlands, 2003; Volume 145, pp. 31–38. [Google Scholar]
- Chen, X.; Zhu, H. 3.01—Catalysis by Supported Gold Nanoparticles. In Comprehensive Nanoscience and Technology; David, L.A., Gregory, D.S., Gary, P.W., Eds.; Academic Press, Elsevier: Amsterdam, Netherlands, 2011; pp. 1–11. [Google Scholar]
- El-Rafie, M.H.; Shaheen, T.I.; Mohamed, A.A.; Hebeish, A. Bio-synthesis and applications of silver nanoparticles onto cotton fabrics. Carbohydr. Polym. 2012, 90, 915–920. [Google Scholar] [CrossRef]
- Jung, S.-H.; Kim, K.-I.; Ryu, J.-H.; Choi, S.-H.; Kim, J.-B.; Moon, J.-H.; Jin, J.-H. Preparation of radioactive core-shell type 198Au@SiO2 nanoparticles as a radiotracer for industrial process applications. Appl. Radiat. Isot. 2010, 68, 1025–1029. [Google Scholar] [CrossRef]
- Yu, C.H.; Oduro, W.; Tam, K.; Tsang, E.S.C. Chapter 10. Some Applications of Nanoparticles. In Handbook of Metal Physics; John, A.B., Ed.; Elsevier: Amsterdam, Netherlands, 2008; Volume 5, pp. 365–380. [Google Scholar]
- Ganesh, S.; Amit, S. Perspectives on the Science and Technology of Nanoparticle Synthesis. In Nanomaterials Handbook; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Lawrence, D.Ä.; Ryan, R. Synthesis of Metal Colloids. In Catalyst Preparation; CRC Press: New York, NY, USA, 2006; pp. 93–137. [Google Scholar]
- Sergeev, G.B. 2—Synthesis and Stabilization of Nanoparticles. In Nanochemistry; Elsevier Science: Amsterdam, Netherlands, 2006; pp. 7–36. [Google Scholar]
- Takeshi, T. Preparation of Nanoparticles Using Laser Ablation in Liquids. In Laser Ablation in Liquids; Pan Stanford Publishing: Singapore, 2012; pp. 207–268. [Google Scholar]
- Haibo, Z.; Shikuan, Y.; Weiping, C. Formation of Nanoparticles under Laser Ablation of Solids in Liquids. In Laser Ablation in Liquids; Pan Stanford Publishing: Singapore, 2012; pp. 327–396. [Google Scholar]
- Stratakisa, E.; Shafeevb, G. Nanostructures? Formation under Laser Ablation of Solids in Liquids. In Laser Ablation in Liquids; Pan Stanford Publishing: Singapore, 2012; pp. 815–854. [Google Scholar]
- López-Tocón, I.; Centeno, S.P.; Otero, J.C.; Marcos, J.I. Selection rules for the charge transfer enhancement mechanism in SERS: Dependence of the intensities on the L-matrix. J. Mol. Struct. 2001, 565–566, 369–372. [Google Scholar]
- Luo, Z.; Fang, Y. Investigation of the mechanism of influence of colloidal gold/silver substrates in nonaqueous liquids on the surface enhanced Raman spectroscopy (SERS) of fullerenes C60 (C70). J. Colloid Interface Sci. 2006, 301, 184–192. [Google Scholar] [CrossRef]
- Primera-Pedrozo, O.M.; Rodríguez, G.D.M.; Castellanos, J.; Felix-Rivera, H.; Resto, O.; Hernández-Rivera, S.P. Increasing surface enhanced Raman spectroscopy effect of RNA and DNA components by changing the pH of silver colloidal suspensions. Spectrochim. Acta A 2012, 87, 77–85. [Google Scholar] [CrossRef]
- Hou, X.; Zhang, X.; Chen, S.; Fang, Y.; Li, N.; Zhai, X.; Liu, Y. Size-controlled synthesis of Au nanoparticles and nanowires and their application as SERS substrates. Colloids Surf. A 2011, 384, 345–351. [Google Scholar]
- Kundu, S.; Mandal, M.; Ghosh, S.K.; Pal, T. Photochemical deposition of SERS active silver nanoparticles on silica gel and their application as catalysts for the reduction of aromatic nitro compounds. J. Colloid Interface Sci. 2004, 272, 134–144. [Google Scholar] [CrossRef]
- Photopoulos, P.; Boukos, N.; Panagopoulou, M.; Meintanis, N.; Pantiskos, N.; Raptis, Y.; Tsoukalas, D. Size control of Ag nanoparticles for SERS sensing applications. Procedia. Eng. 2011, 25, 280–283. [Google Scholar] [CrossRef]
- Vlckova, B.; Pavel, I.; Sladkova, M.; Siskova, K.; Slouf, M. Single molecule SERS: Perspectives of analytical applications. J. Mol. Struct. 2007, 834–836, 42–47. [Google Scholar]
- Primera-Pedrozo, O.M.; Jerez-Rozo, J.I.; De La Cruz-Montoya, E.; Luna-Pineda, T.; Pacheco-Londoño, L.C.; Hernandez-Rivera, S.P. Nanotechnology-based detection of explosives and biological agents simulants. Sens. J. 2008, 8, 963–973. [Google Scholar]
- Burda, C.; Chen, X.; Narayanan, R.; El-Sayed, M.A. Chemistry and properties of nanocrystals of different shapes. Chem. Rev. 2005, 105, 1025–1102. [Google Scholar]
- Kim, K.; Shin, D.; Choi, J.-Y.; Kim, K.L.; Shin, K.S. Surface-enhanced Raman scattering characteristics of 4-aminobenzenethiol derivatives adsorbed on silver. J. Phys. Chem. C 2011, 115, 24960–24966. [Google Scholar]
- Kim, K.; Lee, H.B.; Choi, J.-Y.; Kim, K.L.; Shin, K.S. Surface-enhanced Raman scattering of 4-aminobenzenethiol in nanogaps between a planar Ag substrate and Pt nanoparticles. J. Phys. Chemistry C 2011, 115, 13223–13231. [Google Scholar] [CrossRef]
- Kim, K.; Kim, K.L.; Shin, D.; Choi, J.-Y.; Shin, K.S. Surface-enhanced Raman scattering of 4-aminobenzenethiol on Ag and Au: pH dependence of b2-type bands. J. Phys. Chem. C 2012, 116, 4774–4779. [Google Scholar]
- Kim, K.; Kim, K.L.; Lee, H.B.; Shin, K.S. Similarity and dissimilarity in surface-enhanced Raman scattering of 4-aminobenzenethiol, 4,4'-dimercaptoazobenzene, and 4,4'-dimercaptohydrazobenzene on Ag. J. Phys. Chem. C 2012, 116, 11635–11642. [Google Scholar]
- Kim, K.; Yoon, J.K.; Lee, H.B.; Shin, D.; Shin, K.S. Surface-enhanced Raman scattering of 4-aminobenzenethiol in Ag sol: Relative intensity of a1- and b2-type bands invariant against aggregation of Ag nanoparticles. Langmuir 2011, 27, 4526–4531. [Google Scholar] [CrossRef]
- Kim, A.; Ou, F.S.; Ohlberg, D.A.A.; Hu, M.; Williams, R.S.; Li, Z. Study of molecular trapping inside gold nanofinger arrays on surface-enhanced Raman substrates. J. American Chem. Soc. 2011, 133, 8234–8239. [Google Scholar]
- Baladi, A.; Sarraf Mamoory, R. Investigation of different liquid media and ablation times on pulsed laser ablation synthesis of aluminum nanoparticles. Appl. Surf. Sci. 2010, 256, 7559–7564. [Google Scholar] [CrossRef]
- Mahfouz, R.; Cadete Santos Aires, F.J.; Brenier, A.; Jacquier, B.; Bertolini, J.C. Synthesis and physico-chemical characteristics of nanosized particles produced by laser ablation of a nickel target in water. Appl. Surf. Sci. 2008, 254, 5181–5190. [Google Scholar]
- Tsuji, T.; Iryo, K.; Watanabe, N.; Tsuji, M. Preparation of silver nanoparticles by laser ablation in solution: Influence of laser wavelength on particle size. Appl. Surf. Sci. 2002, 202, 80–85. [Google Scholar] [CrossRef]
- Riabinina, D.; Zhang, J.; Chaker, M.; Margot, J.; Ma, D. Size control of gold nanoparticles synthesized by laser ablation in liquid media. ISRN Nanotechnol. 2012, 2012, 5. [Google Scholar]
- Cañamares, M.V.; Garcia-Ramos, J.V.; Gómez-Varga, J.D.; Domingo, C.; Sanchez-Cortes, S. Comparative study of the morphology, aggregation, adherence to glass, and surface-enhanced Raman scattering activity of silver nanoparticles prepared by chemical reduction of Ag+ using citrate and hydroxylamine. Langmuir 2005, 21, 8546–8553. [Google Scholar]
- Schatz, S.H.G. Synthesis and Analysis of SILVER/GOLD Nanoparticles. Available online: http://nanohub.org/topics/GeneralChemistry/File:Silvergold_module.pdf (accessed on 10 December 2012).
- Kim, K.; Lee, H.S. Effect of Ag and Au nanoparticles on the SERS of 4-aminobenzenethiol assembled on powdered copper. J. Phys. Chem. B 2005, 109, 18929–18934. [Google Scholar] [CrossRef]
- Kim, K. Surface-Enhanced Raman Scattering Characteristics of 4-Aminobenzenethiol Derivatives Adsorbed on Silver. Available online: http://ipc.iisc.ernet.in/~umalab/icors2012/Mo_109.pdf (accessed on 10 December 2012).
- Liu, X.; Zhao, L.; Shen, H.; Xu, H.; Lu, L. Ordered gold nanoparticle arrays as surface-enhanced Raman spectroscopy substrates for label-free detection of nitroexplosives. Talanta 2011, 83, 1023–1029. [Google Scholar] [CrossRef]
- Kawaguchi, T.; Shankaran, D.R.; Kim, S.J.; Matsumoto, K.; Toko, K.; Miura, N. Surface plasmon resonance immunosensor using Au nanoparticle for detection of TNT. Sens. Actuators B 2008, 133, 467–472. [Google Scholar] [CrossRef]
- Kneipp, K.; Wang, Y.; Dasari, R.R.; Feld, M.S.; Gilbert, B.D.; Janni, J.; Steinfeld, J.I. Near-infrared surface-enhanced Raman scattering of trinitrotoluene on colloidal gold and silver. Spectrochim. Acta A 1995, 51, 2171–2175. [Google Scholar] [CrossRef]
- Lin, D.; Liu, H.; Qian, K.; Zhou, X.; Yang, L.; Liu, J. Ultrasensitive optical detection of trinitrotoluene by ethylenediamine-capped gold nanoparticles. Anal. Chim. Acta 2012, 744, 92–98. [Google Scholar] [CrossRef]
- Izake, E.L. Forensic and homeland security applications of modern portable Raman spectroscopy. Forensic Sci. Int. 2010, 202, 1–8. [Google Scholar] [CrossRef]
- Caygill, J.S.; Davis, F.; Higson, S.P.J. Current trends in explosive detection techniques. Talanta 2012, 88, 14–29. [Google Scholar] [CrossRef] [Green Version]
- Fierro-Mercado, P.; Renteria-Beleño, B.; Hernández-Rivera, S.P. Preparation of SERS-active substrates using thermal inkjet technology. Chem. Phys. Lett. 2012, 552, 108–113. [Google Scholar] [CrossRef]
- Fierro-Mercado, P.; Hernández-Rivera, S.P. Highly sensitive filter paper substrate for SERS trace explosives detection. Int. J. Spectrosc. 2012, 2012. [Google Scholar] [CrossRef]
- Jerez Rozo, J.I.; Chamoun, A.M.; Peña, S.L.; Hernández-Rivera, S.P. Enhanced Raman scattering of TNT on nanoparticle substrates: Ag colloids prepared by reduction with hydroxylamine hydrochloride and sodium citrate. Proc. SPIE 2007, 6538, 653824–653835. [Google Scholar]
- Cosgrove, T. Colloid Science Principles, Methods and Applications; Blackwell Publishing Ltd: Department of Chemistry, University of Bristol, Bristol, UK, 2005. [Google Scholar]
- Muniz-Miranda, M.; Pergolese, B.; Bigotto, A.; Giusti, A. Stable and efficient silver substrates for SERS spectroscopy. J. Colloid Interface Sci. 2007, 314, 540–544. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Herrera, G.M.; Padilla, A.C.; Hernandez-Rivera, S.P. Surface Enhanced Raman Scattering (SERS) Studies of Gold and Silver Nanoparticles Prepared by Laser Ablation. Nanomaterials 2013, 3, 158-172. https://doi.org/10.3390/nano3010158
Herrera GM, Padilla AC, Hernandez-Rivera SP. Surface Enhanced Raman Scattering (SERS) Studies of Gold and Silver Nanoparticles Prepared by Laser Ablation. Nanomaterials. 2013; 3(1):158-172. https://doi.org/10.3390/nano3010158
Chicago/Turabian StyleHerrera, Gloria M., Amira C. Padilla, and Samuel P. Hernandez-Rivera. 2013. "Surface Enhanced Raman Scattering (SERS) Studies of Gold and Silver Nanoparticles Prepared by Laser Ablation" Nanomaterials 3, no. 1: 158-172. https://doi.org/10.3390/nano3010158
APA StyleHerrera, G. M., Padilla, A. C., & Hernandez-Rivera, S. P. (2013). Surface Enhanced Raman Scattering (SERS) Studies of Gold and Silver Nanoparticles Prepared by Laser Ablation. Nanomaterials, 3(1), 158-172. https://doi.org/10.3390/nano3010158