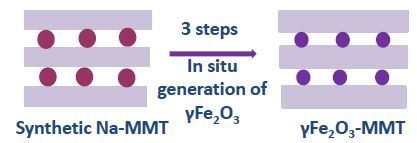
Maghemite Intercalated Montmorillonite as New Nanofillers for Photopolymers
Abstract
:1. Introduction
2. Results and Discussion
2.1. Nanofillers Preparation (γFe2O3-MMT)
2.1.1. Ion Exchange Reaction
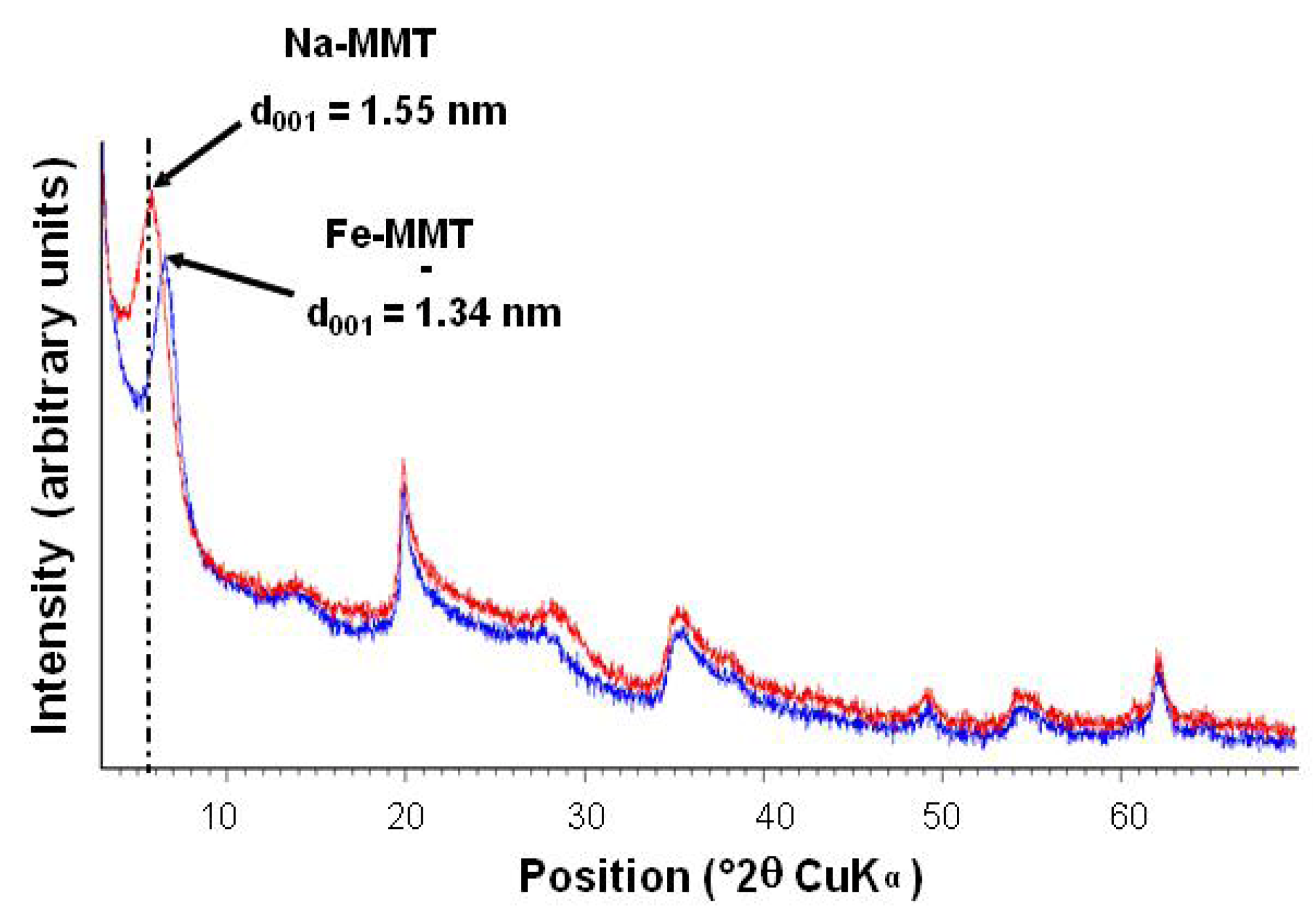
2.1.2. Formation of Goethite
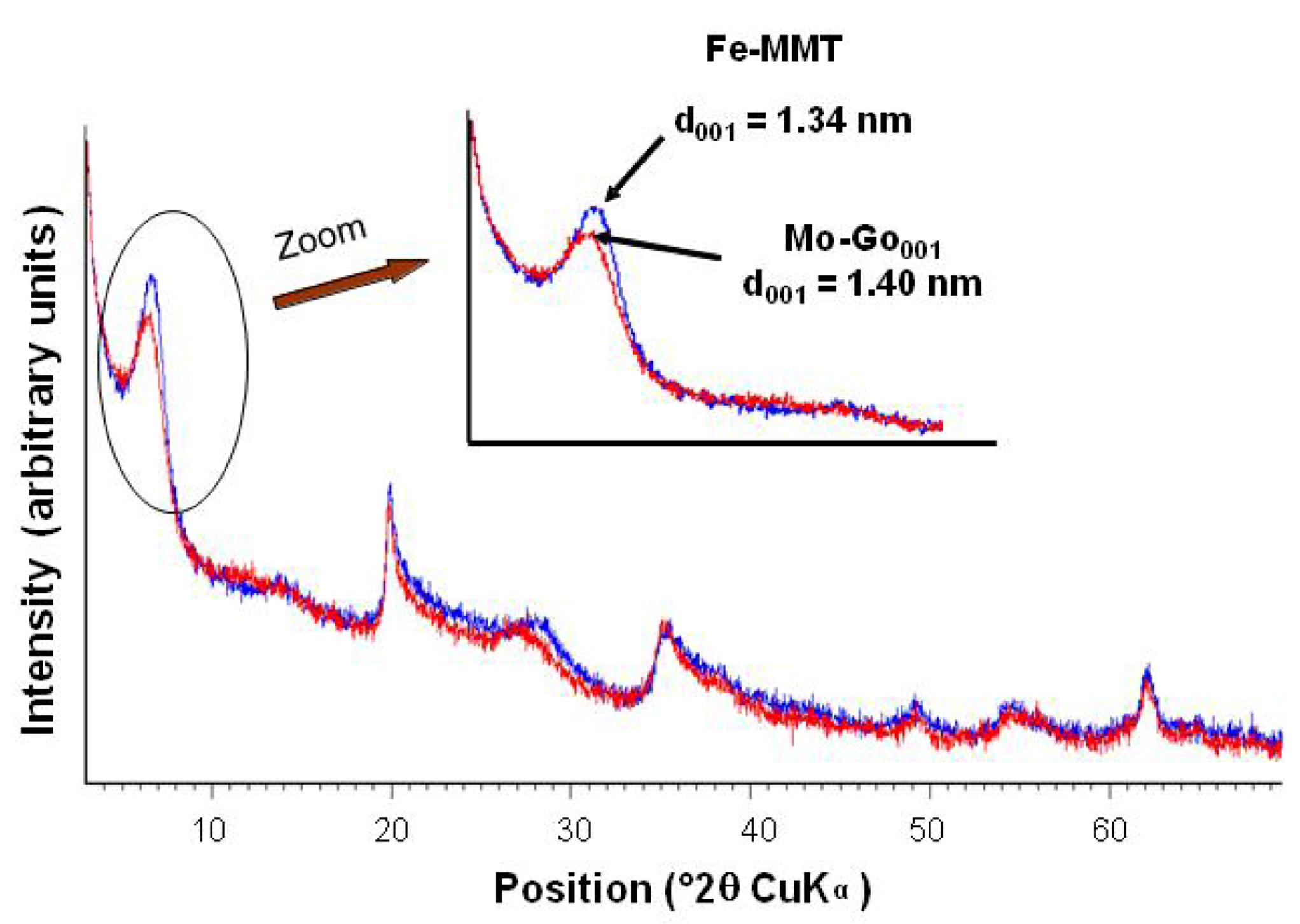
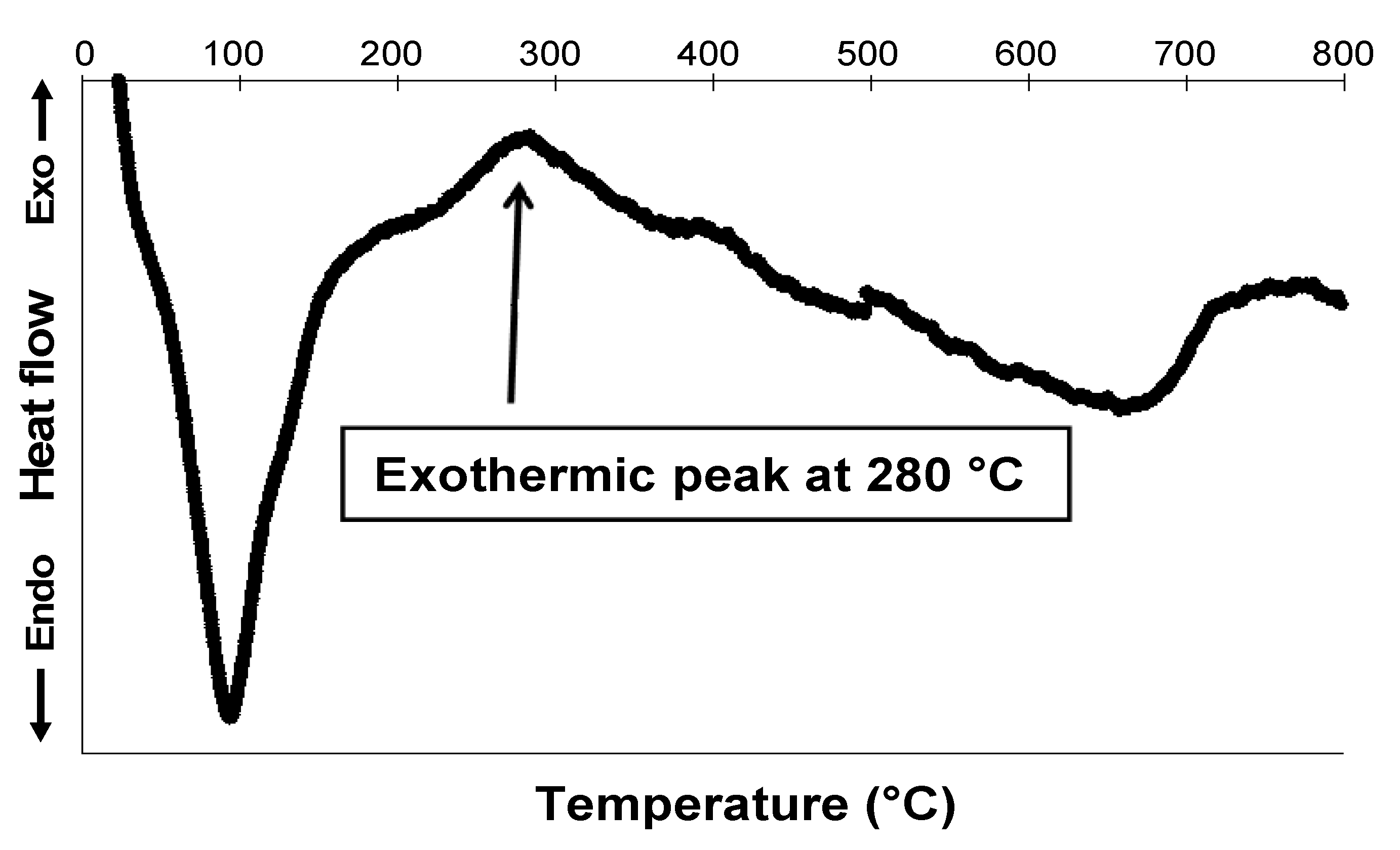
2.1.3. Thermal Solid-State Transformations

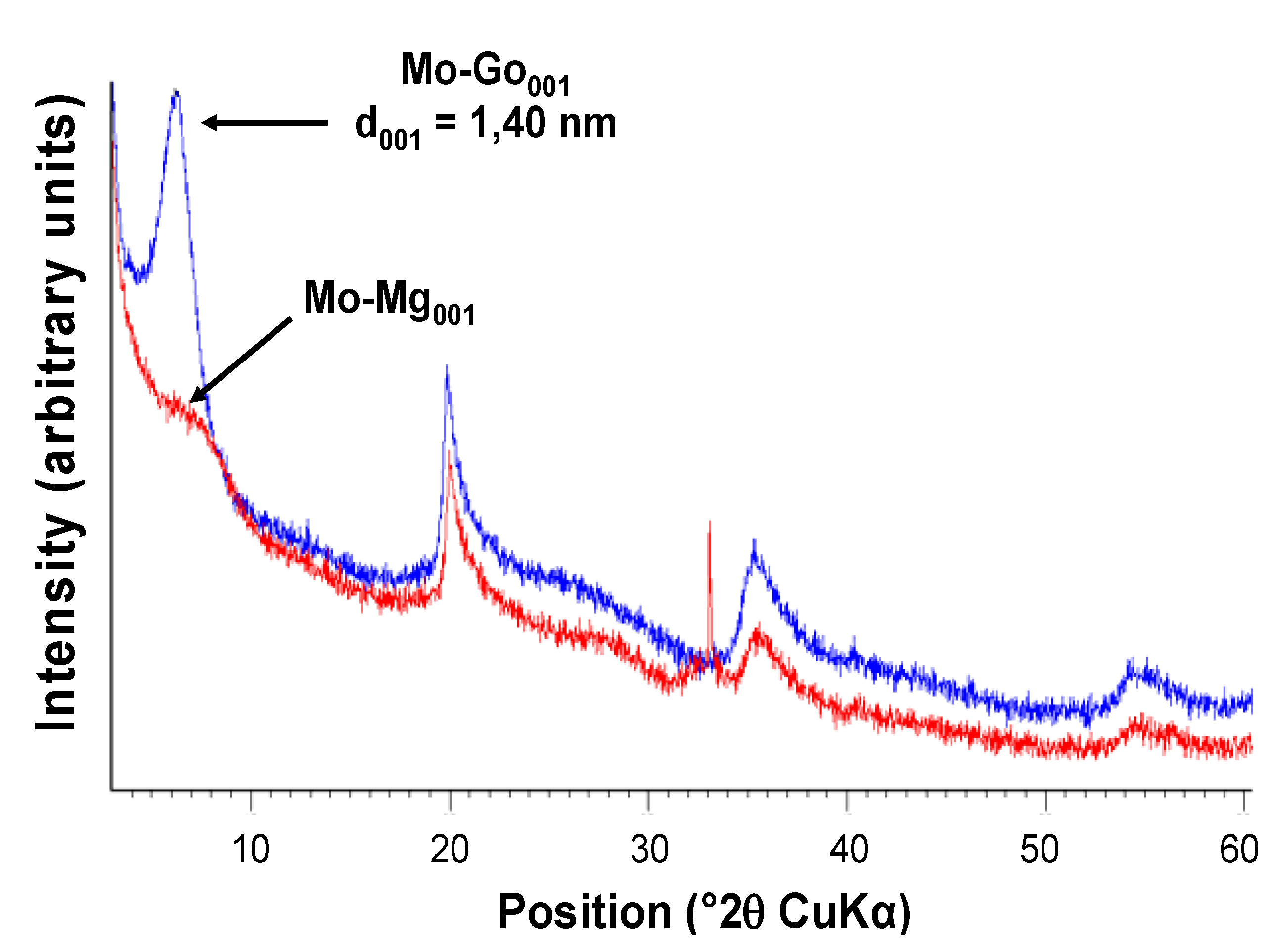
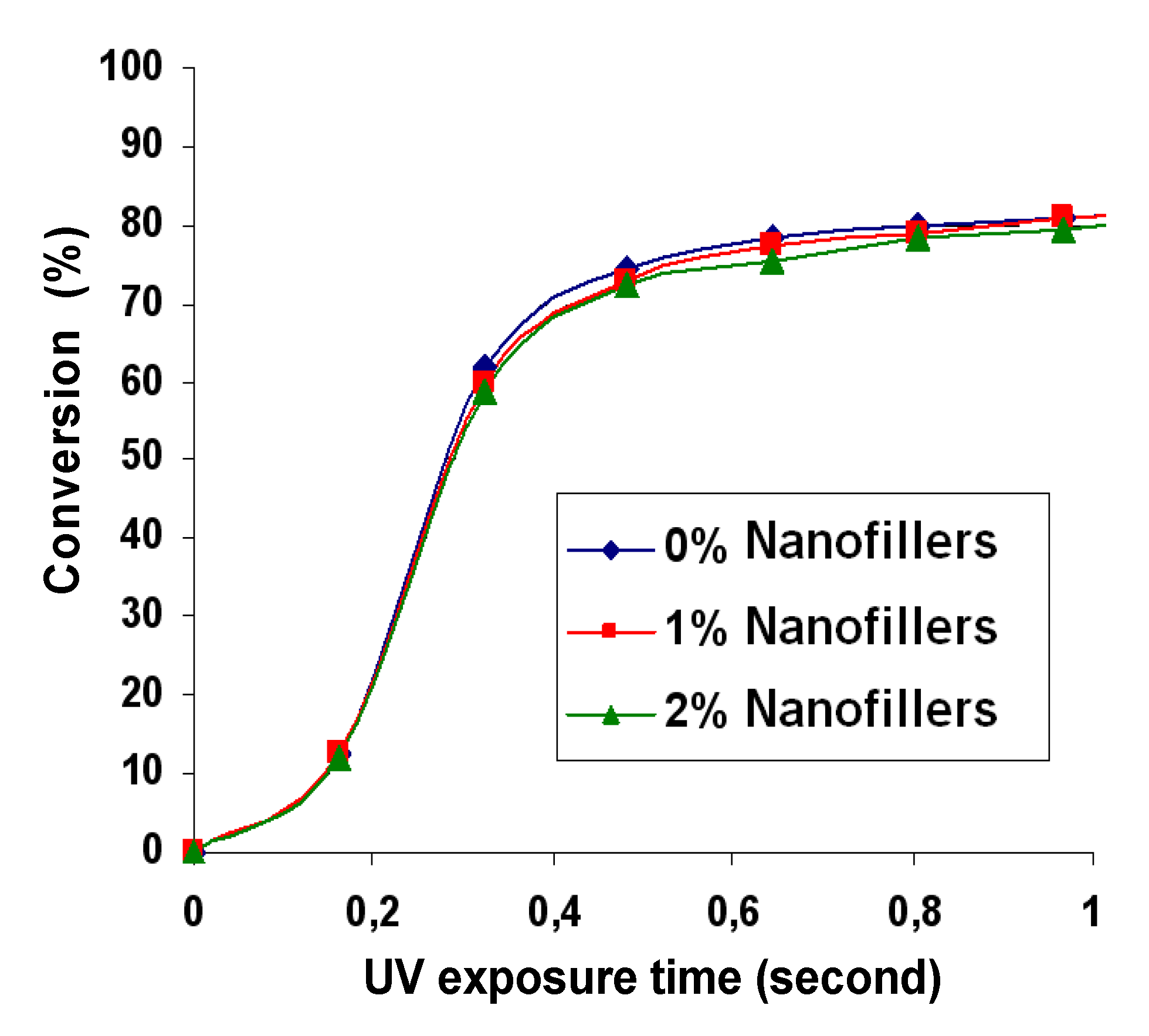
2.2. Kinetics of Photopolymerization
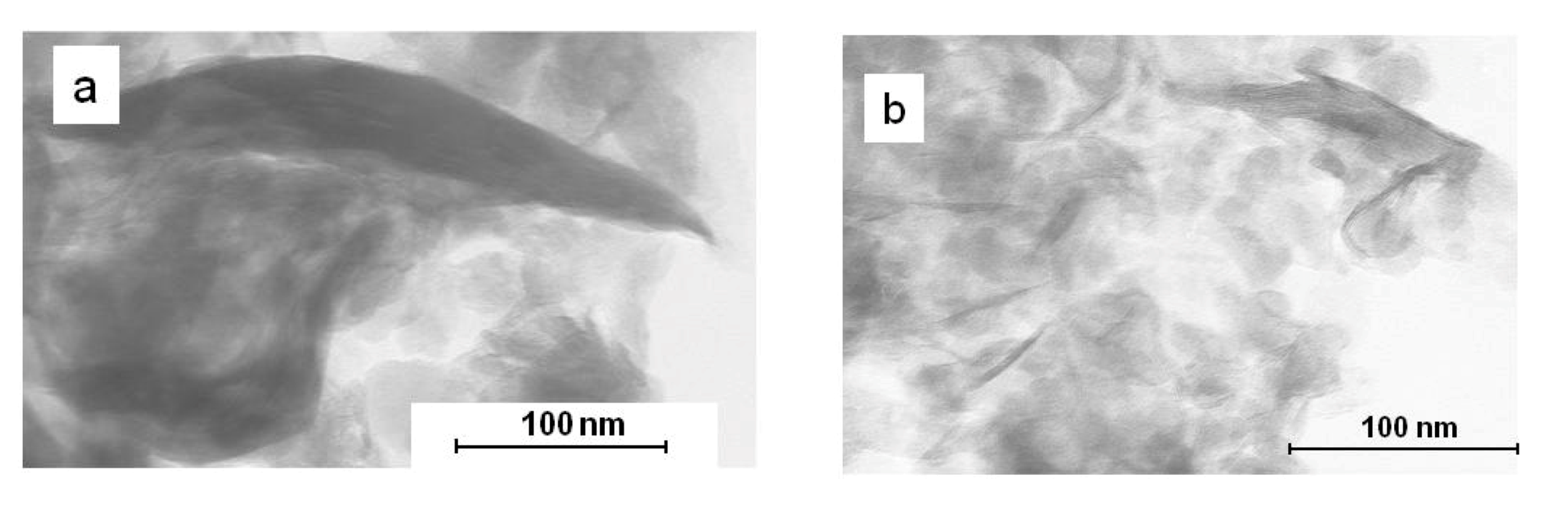
2.3. Nanocomposite Morphology
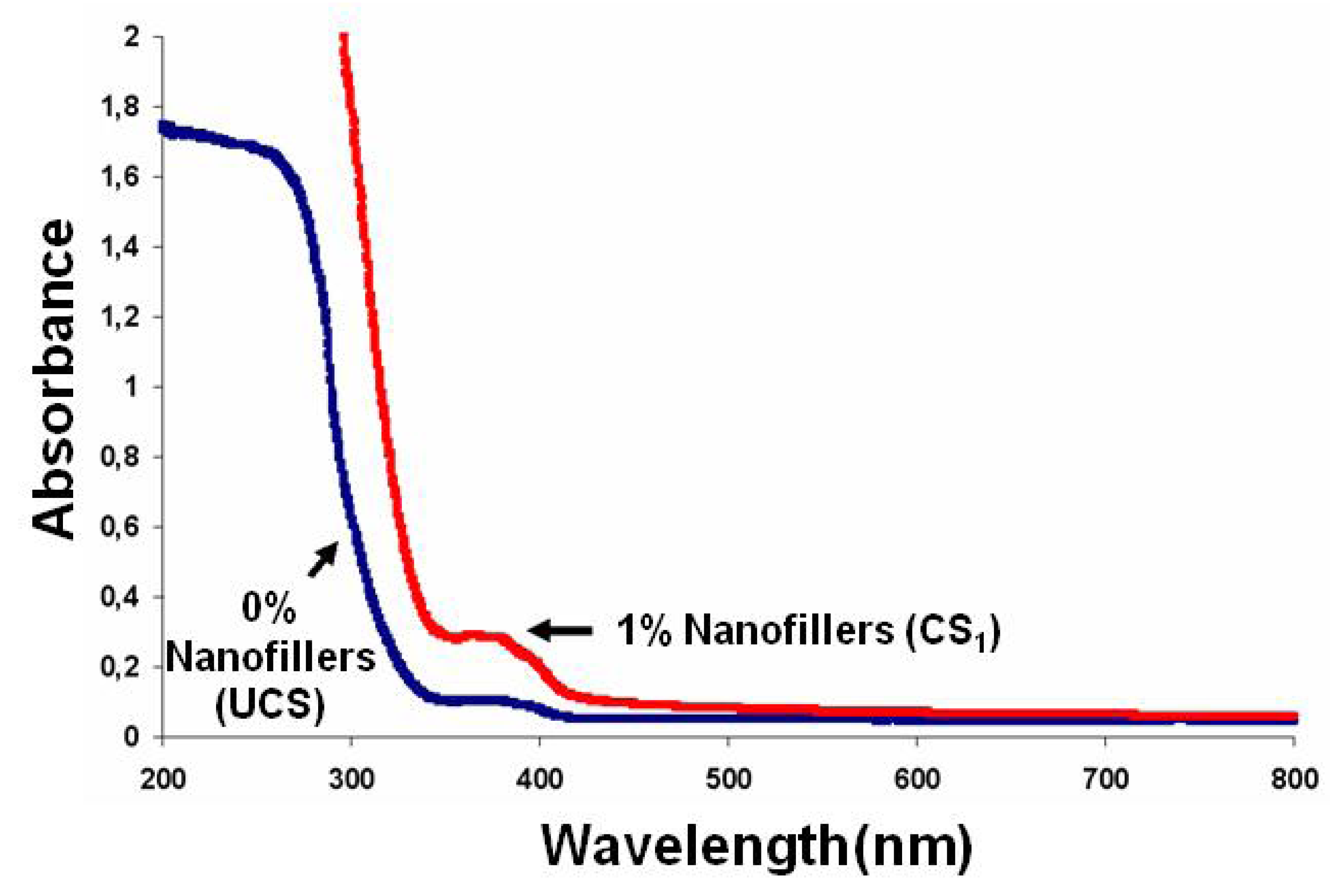

2.4. Properties of Nanocomposite Films
2.4.1. Viscoelastic Properties
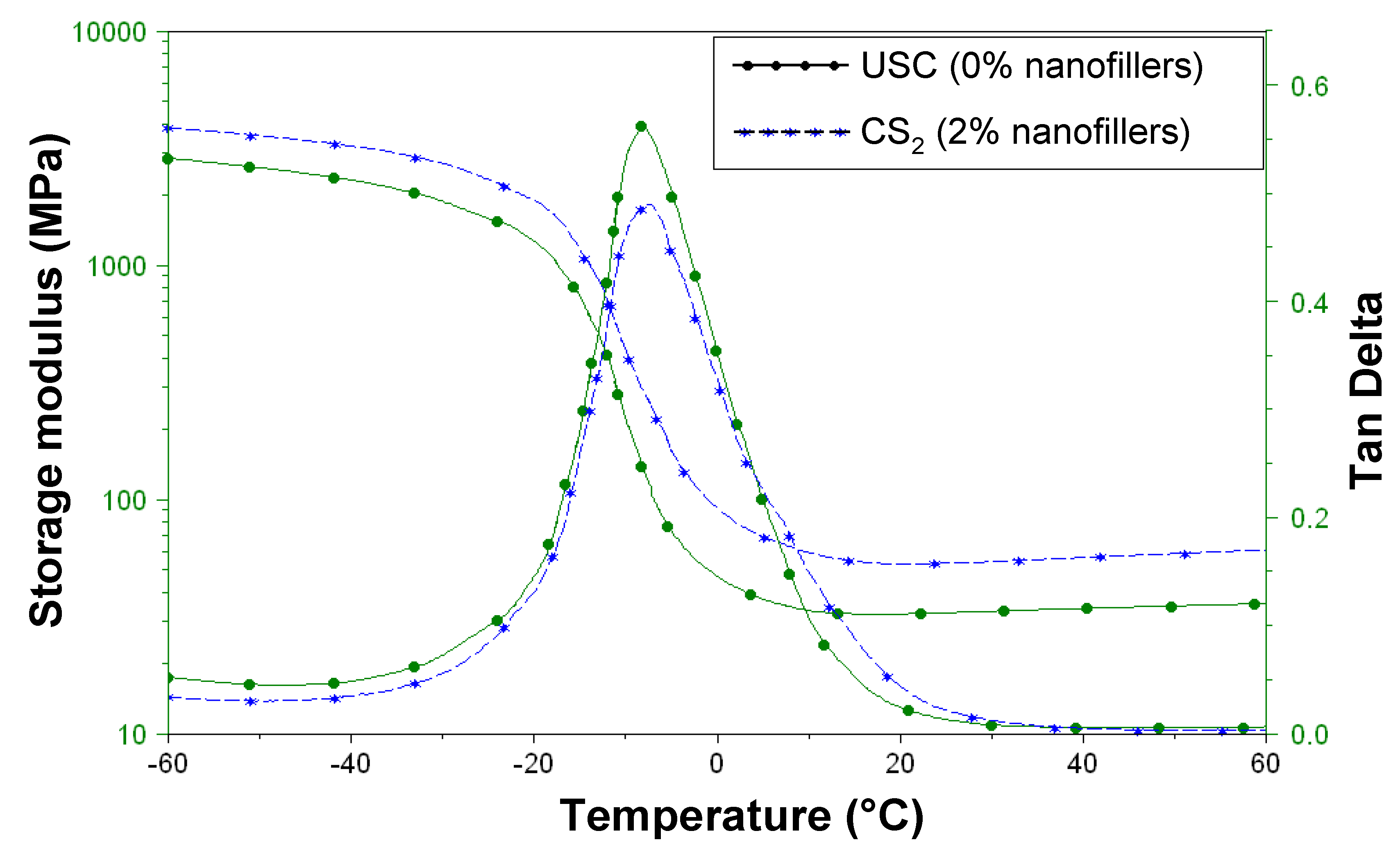
| Sample | Tg (°C) | Youngs modulus (MPa) at 25 °C |
|---|---|---|
| crude polymer (UCS) | –7.1 | 32 |
| CS2 | –7.3 | 53 |
2.4.2. Hardness Properties
| Sample | Hardness (s) | Gloss at 20° (%) |
|---|---|---|
| crude polymer (UCS) | 195 ± 10 | 100 |
| CS1 | 190 ± 10 | 91 |
| CS2 | 192 ± 10 | 83 |
2.4.3. Gloss Properties
3. Experimental Section
3.1. Materials
| Symbol | Name | Structure |
|---|---|---|
| HDDA | 1,6-Hexanediol diacrylate |  |
| SR 344 | Polyethylene glycol (400) diacrylate |  |
3.2. Procedures
3.2.1. Preparation of Montmorillonite
3.2.2. Preparation of γFe2O3-MMT
- Ion Exchange
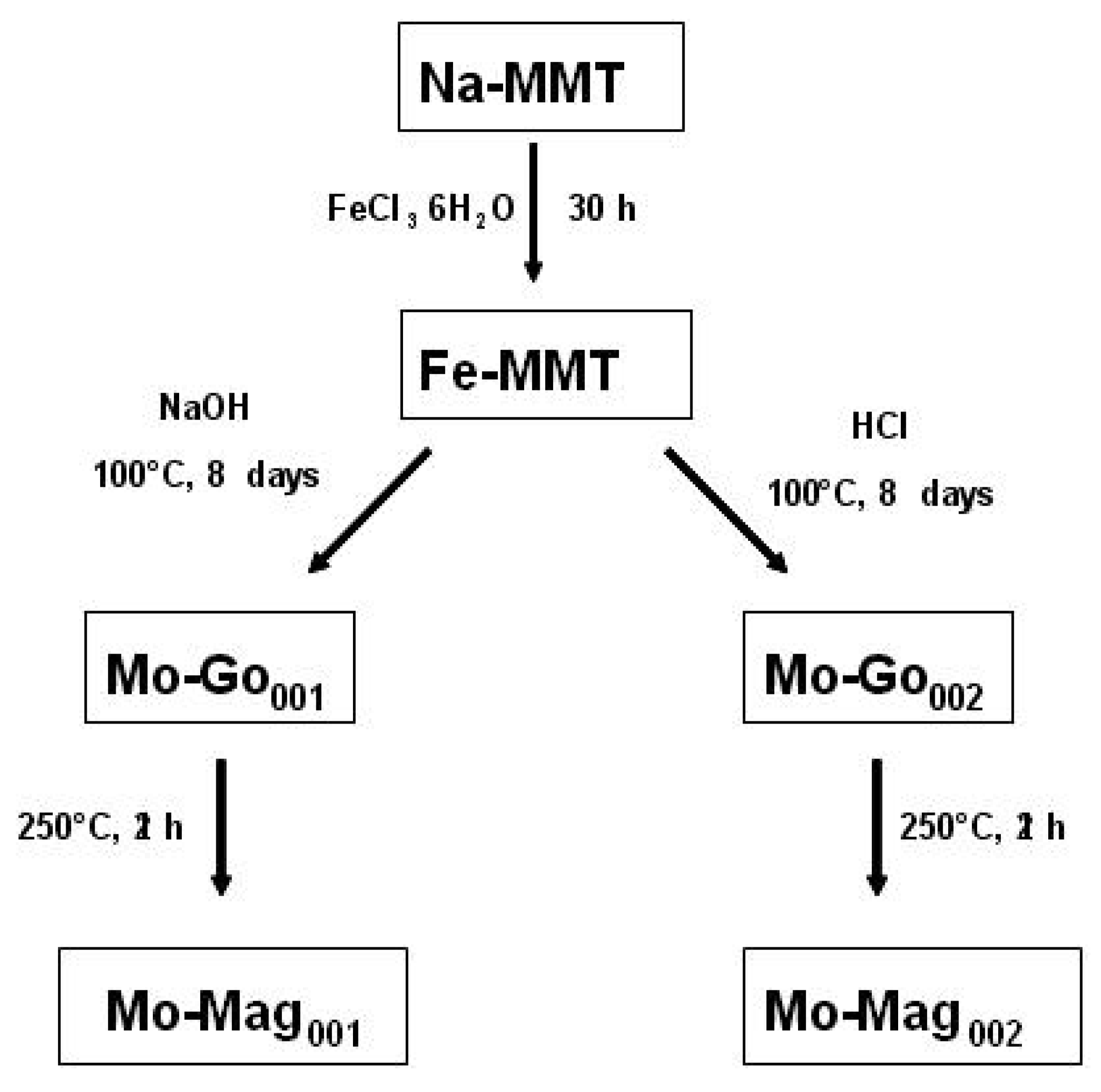
- Goethite Formation
- -
- The basic route [35]: 1 g of Fe-MMT was dispersed in 11 mL of water. Then 9.3 mL of a 5.4 M sodium hydroxide aqueous solution were added dropwise. The OH- to Fe3+ molar ratio and the pH were respectively 5.4 and 12.3.
- -
- The acidic route [36]: 1 g of Fe-MMT was dispersed in 23 mL of a 0.01 M hydrochloric acid aqueous solution.
- Thermal Treatment
3.2.3. Preparation of the Formulations and Photopolymerization
- Preparation of CS1 and CS2 Formulations
- Photopolymerization
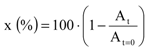
3.3. Characterization
4. Conclusions
References
- Rong, M.Z.; Zhang, M.Q.; Zheng, Y.X.; Zeng, H.M. Improvement of tensile properties of nano-SiO2/PP composites in relation to percolation mechanism. Polymer 2001, 42, 3301–3304. [Google Scholar] [CrossRef]
- Li, Y.; Yu, J.; Guo, Z. The influence of interphase on nylon-6/nano-SiO2 composite materials obtained from in situ polymerization. Polym. Int. 2003, 52, 981–986. [Google Scholar] [CrossRef]
- Novakova, A.; Smirnovb, E.V.; Gendler, T.S. Magnetic anisotropy in Fe3O4-PVA nanocomposites as a result of Fe3O4-nanoparticles chains formation. J. Magn. Magn. Mater. 2006, 300, e354–e358. [Google Scholar]
- Ali-zade, R.A. Structure and magnetic properties of polymer microspheres filled with magnetite nanoparticles. Inorg. Mater. 2004, 40, 509–515. [Google Scholar] [CrossRef]
- Dumont, M.J.; Reyna-Valencia, A.; Emond, J.P.; Bousmina, M. Barrier properties of polypropylene/organoclaynanocomposites. J. Appl. Polym. Sci. 2007, 103, 618–625. [Google Scholar]
- Zhu, J.; He, Q.; Luo, Z.; Khasanov, A.; Li, Y.; Sun, L.; Wang, Q.; Wei, S.; Guo, Z. Property manipulated polypropylene–iron nanocomposites with maleic anhydride polypropylene. J. Mater. Chem. 2012, 22, 15928–15938. [Google Scholar]
- Gass, J.; Poddar, P.; Almand, J.; Srinath, S.; Srikanth, H. Superparamagnetic polymer nanocomposites with uniform Fe3O4 nanoparticle dispersions. Adv. Funct. Mater. 2006, 16, 71–75. [Google Scholar]
- Qiu, G.; Wang, Q.; Nie, M. Polyaniline/Fe3O4 magnetic nanocomposite prepared by ultrasonic irradiation. J. Appl. Polym. Sci. 2006, 102, 2107–2111. [Google Scholar] [CrossRef]
- Schmidt, A.M. Electromagnetic activation of shape memory polymer networks containing magnetic nanoparticles. Macromol. Rapid Commun. 2006, 27, 1168–1172. [Google Scholar]
- He, Q.; Yuan, T.; Zhu, J.; Luo, Z.; Haldolaarachchige, N.; Sun, L.; Khasanov, A.; Li, Y.; Young, D.P.; Wei, S.; et al. Magnetic high density polyethylene nanocomposites reinforced with in situ synthesized Fe@FeO core-shell nanoparticles. Polymer 2012, 53, 3642–3652. [Google Scholar] [CrossRef]
- Alexandre, M.; Dubois, P. Polymer-layered silicate nanocomposites: Preparation, properties and uses of a new class of materials. Mater. Sci. Eng. Rev. 2000, 28, 1–63. [Google Scholar] [CrossRef]
- Zhu, J.; Wei, S.; Haldolaarachchige, N.; Young, D.P.; Guo, Z. Electromagnetic field shielding polyurethane nanocomposites reinforced with core–shell Fe–Silica nanoparticles. J. Phys. Chem. C 2011, 115, 15304–15310. [Google Scholar]
- Roco, M.C. Nanoparticles and nanotechnology research. J. Nanopart. Res. 1999, 1, 1–6. [Google Scholar]
- Kronmüller, H.; Fischer, R.; Bachmann, M.; Leineweber, T. Magnetization processes in small particles and nanocrystalline materials. J. Magn. Magn. Mater. 1999, 203, 12–17. [Google Scholar] [CrossRef]
- Barbic, M. Single domain magnets in bio-medical applications. Eur. J. Cells Mater. 2002, 3, 132–134. [Google Scholar]
- Ciobanu, C.C.; Iconaru, S.L.; Gyorgy, E.; Radu, M.; Costache, M.; Dinischiotu, A.; Le Coustumer, P.; Lafdi, K.; Predoi, D. Biomedical properties and preparation of iron oxide-dextran nanostructure by MAPLE technique. Chem. Cent. J. 2012, 6, 1–12. [Google Scholar] [CrossRef]
- Neamtu, J.; Verga, N. Magnetic nanoparticles for magneto-resonance imaging and targeted drug delivery. Dig. J. Nanomater. Biostruct. 2011, 6, 969–978. [Google Scholar]
- Mahoudi, M.; Simchi, A.; Imani, M.; Hafali, U.O. Superparamagnetic iron oxide nanoparticles with rigid cross-linked polyethylene glycol fumarate coating for application in imaging and drug delivery. J. Phys. Chem. C 2009, 113, 8124–8131. [Google Scholar]
- Massart, R. Préparation de ferrofluides aqueux en l’absence de surfactant, comportement en fonction du pH et de la nature des ions présents en solution. C. R. Acad. Sci. Paris 1980, C1, t291. [Google Scholar]
- Meneghetti, P.; Qutubuddin, S. Synthesis, thermal properties and applications of polymer-clay nanocomposites. Thermochim. Acta 2006, 442, 74–77. [Google Scholar] [CrossRef]
- Betega de Paiva, L.; Morales, A.R.; Valenzuela Diaz, F.R. Organoclays: Properties, preparation and applications. Appl. Clay Sci. 2008, 42, 8–24. [Google Scholar]
- Jaber, M.; Miehe-Brendle, J. Organoclays: Preparation, Properties and Applications. In Ordered Porous Solids; Valtchev, V., Mintova, S., Tsapatsis, M., Eds.; Elsevier: London, UK, 2008; pp. 31–49. [Google Scholar]
- Godowsky, D.Y.; Varfolomeev, A.V.; Efremova, G.D.; Cherepanov, V.M.; Kapustin, G.A.; Volkov, A.V.; Moskvina, M.A. Magnetic properties of polyvinyl alcohol-based composites containing iron oxide nanoparticles. Adv. Mater. Opt. Electron. 1999, 9, 87–91. [Google Scholar]
- Zhitomirsky, I.; Niewczas, M.; Petric, A. Electrodeposition of hybrid organic-inorganic films containing iron oxide. Mater. Lett. 2003, 57, 1045–1050. [Google Scholar] [CrossRef]
- Yrkov, G.Y.; Gubin, S.P.; Pankratov, D.A.; Koksharov, Y.A.; Kozinkin, A.V.; Spichkin, Y.I.; Nedoseikina, T.I.; Pirog, I.V.; Vlasenko, V.G. Iron (III) oxide nanoparticles in a polyethylene matrix. Inorg. Mater. 2002, 38, 137–145. [Google Scholar] [CrossRef]
- Wilson, J.L.; Poddar, P.; Frey, N.A.; Srikanth, H.; Mohomed, K.; Harmon, J.P.; Kotha, S.; Wachsmuth, J. Synthesis and magnetic properties of polymer nanocomposites with embedded iron nanoparticles. J. Appl. Phys. 2004, 95, 1439–1443. [Google Scholar]
- Peeterbroeck, S.; Alexandre, M.; Dubois, P. Processing of Polymer Nanocomposites: New Developments and Challenges. In Recent Advances in Polymer Nanocomposites: Synthesis and Characterisation; Thomas, S., Valsaraj, S.V., Meera, A.P., Zaikov, G., Eds.; CRC Press: Boca Raton, FL, USA, 2010; pp. 19–47. [Google Scholar]
- Zhang, Y.; Evans, J.R.G. Approaches to the manufacture of layered nanocomposites. Appl. Surf. Sci. 2012, 258, 2098–2102. [Google Scholar]
- Bitinis, N.; Hernandez, M.; Verdejo, R.; Kenny, J.M.; Lopez-Manchado, M.A. Recent advances in clay/polymer nanocomposites. Adv. Mater. 2011, 23, 5229–5236. [Google Scholar] [CrossRef]
- Akbari, A.; Talebanfard, S.; Hassan, A. The effect of the structure of clay and clay modifier on polystyrene-clay nanocomposite morphology: A review. Polym.-Plast. Technol. Eng. 2010, 49, 1433–1444. [Google Scholar] [CrossRef]
- Flesch, C.; Unterfinger, Y.; Bourgeat-Lami, E.; Duguet, E.; Delaite, C.; Dumas, P. Poly(ethylene glycol) surface coated magnetic particles. Macromol. Rapid Commun. 2005, 26, 1494–1498. [Google Scholar] [CrossRef]
- Azadmajiri, J.; Hojati-Talemi, P.; Simon, G.P.; Suzuki, K.; Selomulya, C. Synthesis and electromagnetic interference shielding properties of iron oxide/polypyrrolenanocomposites. Polym. Eng. Sci. 2011, 51, 247–253. [Google Scholar]
- Agarwal, T.; Gupta, K.A.; Alam, S.; Zaidi, M.G.H. Fabrication and characterization of iron oxide filled polyvinylpyrrolidone nanocomposites. Int. J. Compos. Mater. 2012, 2, 17–21. [Google Scholar]
- Laachachi, A.; Leroy, E.; Cochez, M.; Ferriol, M.; Cuesta, J.M.L. Use of oxide nanoparticles and organoclays to improve thermal stability and fire retardancy of poly(methyl methacrylate). Polym. Degrad. Stab. 2005, 89, 344–352. [Google Scholar]
- Mamedov, A.; Ostrander, J.; Aliev, F.; Kotov, N.A. Stratified assemblies of magnetite nanoparticles and montmorillonite prepared by the layer-by-layer assembly. Langmuir 2000, 16, 3941–3949. [Google Scholar] [CrossRef]
- Vassilios, T.; Georgi, B.; Vassilios, T.; Georgia, B.; Aristides, B.; Costas, G.; Hadjipanayis, H.; Mao, D.; Niarchos, G.; Hadjipanayis, C.; et al. Immobilization of magnetic iron oxide nanoparticles on laponite discs—An easy way to biocompatible ferrofluids and ferrogels. J. Mater. Chem. 2010, 20, 5418–5428. [Google Scholar]
- Sugimoto, T.; Muramatsu, A.; Sakata, K.; Shindo, D. Characterization of hematite particles of different shapes. J. Colloid Interface Sci. 1993, 158, 420–428. [Google Scholar] [CrossRef]
- Bailey, J.K.; Brinker, C.J.; Mercartney, M.L. Growth mechanisms of iron oxide particles of differing morphologies from the forced hydrolysis of ferric chloride solutions. J. Colloid Interface Sci. 1993, 157, 1–13. [Google Scholar] [CrossRef]
- Fang, J.; Kumbhar, A.; Zhou, W.L.; Stokes, K.L. Nanoneedles of maghemite iron oxide prepared from a wet chemical route. Mater. Res. Bull. 2003, 38, 461–467. [Google Scholar] [CrossRef]
- Cudennec, Y.; Lecerf, A. Topotactic transformations of goethite and lepidocrocite into hematite and maghemite. Solid State Sci. 2005, 7, 520–529. [Google Scholar] [CrossRef]
- Mazo-Zuluaga, J.; Barrero, C.A.; Diaz-Teran, J.; Jerez, A. Thermally induced magnetite–haematite transformation. Hyperfine Interact. 2003, 148–149, 153–161. [Google Scholar] [CrossRef]
- Keller, L.; Decker, C.; Zahouily, K.; Benfarhi, S.; Meins, J.M.L.; Miehe-Brendle, J. Synthesis of polymer nanocomposites by UV-curing of organoclay–acrylic resins. Polymer 2004, 45, 7437–7441. [Google Scholar] [CrossRef]
- Decker, C.; Keller, L.; Zahouily, K.; Benfarhi, S. Synthesis of nanocomposite polymers by UV-radiation curing. Polymer 2005, 46, 6640–6648. [Google Scholar]
- Reinholdt, M.; Miehe-Brendle, J.; Delmotte, L.; Tuilier, M.-H.; le Dred, R.; Cortes, R.; Flank, A.-M. Fluorine route synthesis of montmorillonites containing Mg or Zn and characterization by XRD, thermal analysis, MAS NMR, and EXAFS spectroscopy. Eur. J. Inorg. Chem. 2001, 11, 2831–2841. [Google Scholar]
- Reinholdt, M.; Miehe-Brendle, J.; Delmotte, L.; le Dred, R.; Tuilier, M.-H. Synthesis and characterization of montmorillonite-type phyllosilicates in a fluoride medium. Clay Miner. 2005, 40, 177–190. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tarablsi, B.; Delaite, C.; Brendle, J.; Croutxe-Barghorn, C. Maghemite Intercalated Montmorillonite as New Nanofillers for Photopolymers. Nanomaterials 2012, 2, 413-427. https://doi.org/10.3390/nano2040413
Tarablsi B, Delaite C, Brendle J, Croutxe-Barghorn C. Maghemite Intercalated Montmorillonite as New Nanofillers for Photopolymers. Nanomaterials. 2012; 2(4):413-427. https://doi.org/10.3390/nano2040413
Chicago/Turabian StyleTarablsi, Bassam, Christelle Delaite, Jocelyne Brendle, and Celine Croutxe-Barghorn. 2012. "Maghemite Intercalated Montmorillonite as New Nanofillers for Photopolymers" Nanomaterials 2, no. 4: 413-427. https://doi.org/10.3390/nano2040413
APA StyleTarablsi, B., Delaite, C., Brendle, J., & Croutxe-Barghorn, C. (2012). Maghemite Intercalated Montmorillonite as New Nanofillers for Photopolymers. Nanomaterials, 2(4), 413-427. https://doi.org/10.3390/nano2040413