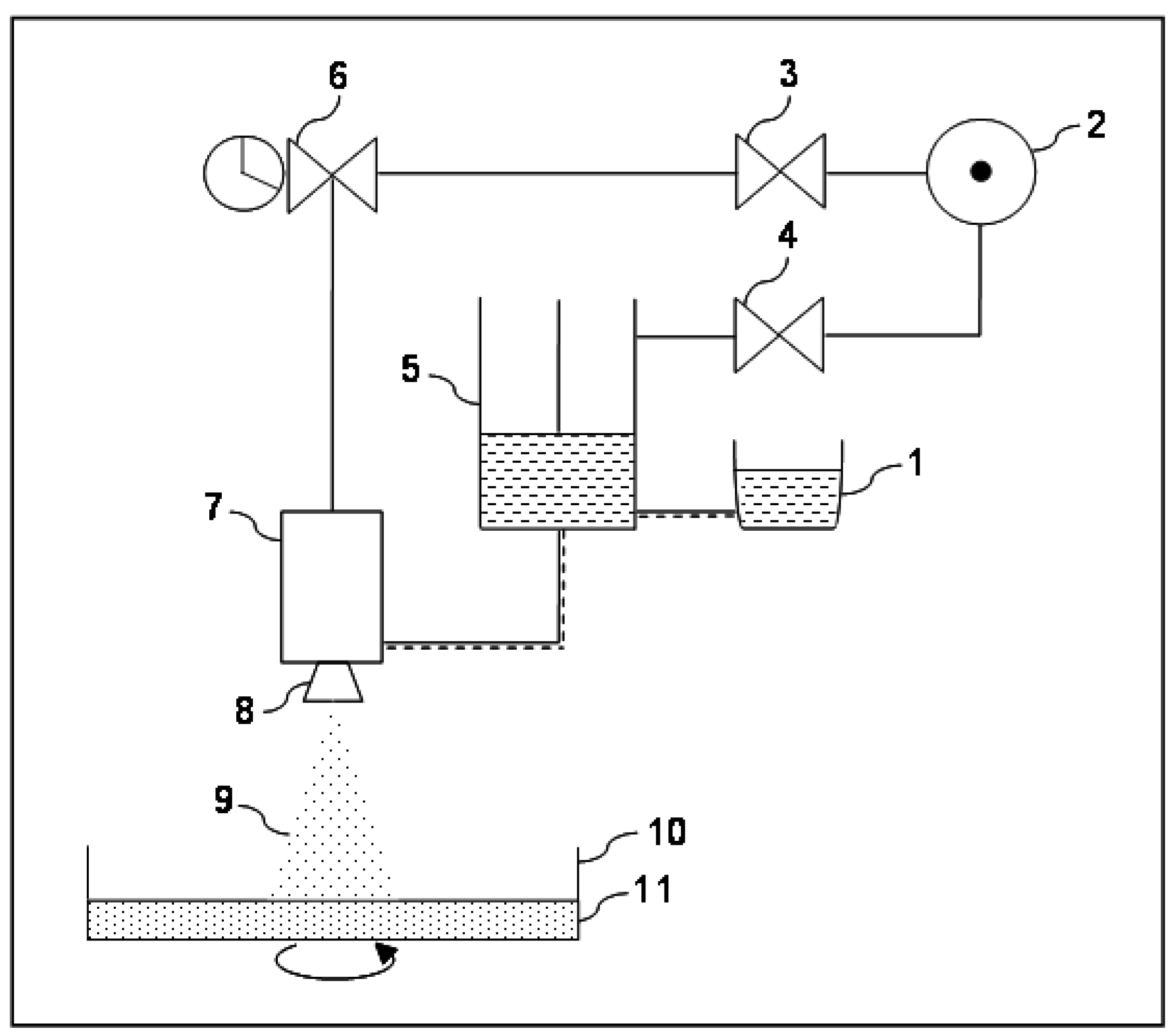
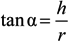2.4. Rheological Analysis
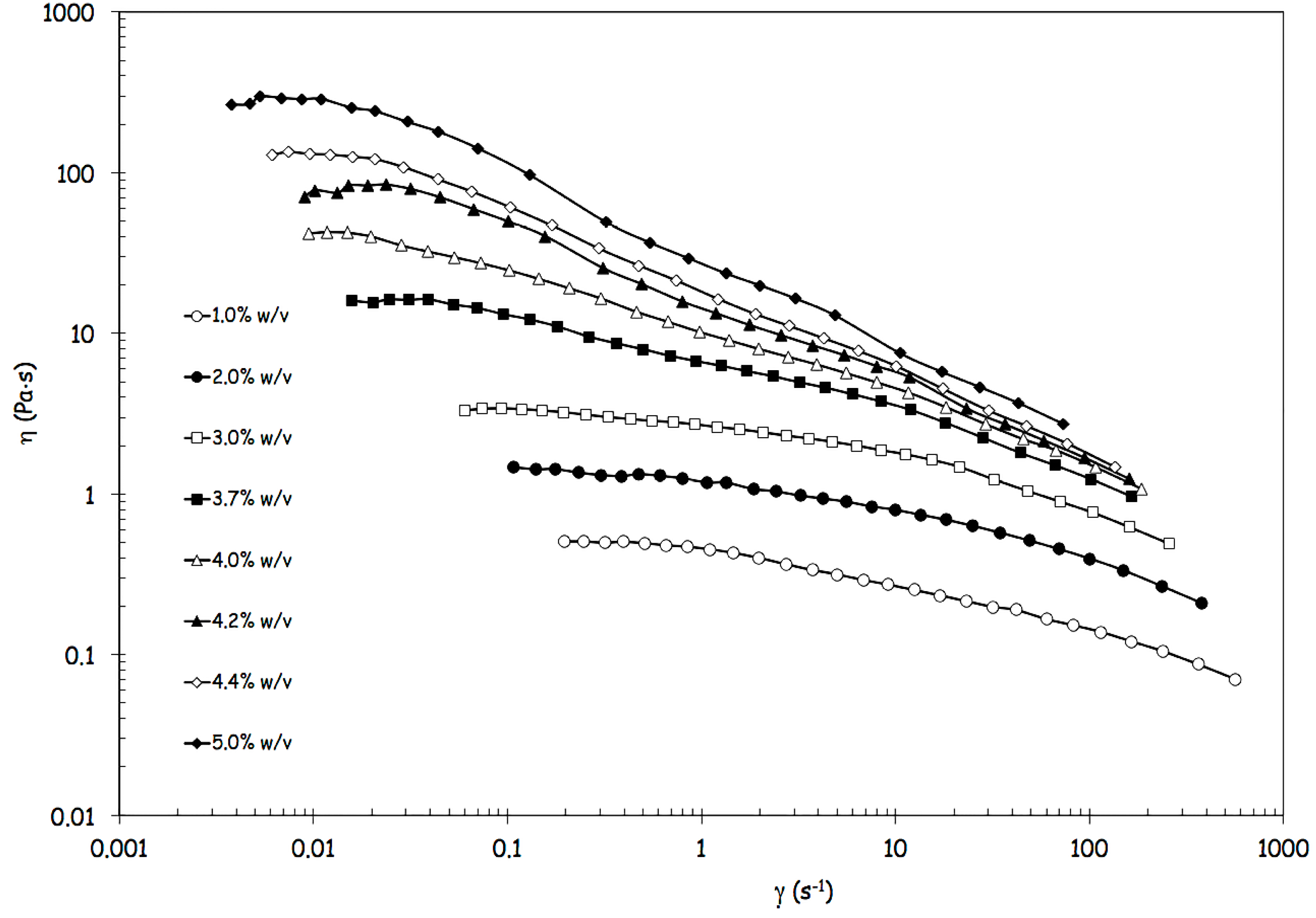
Since the prototype was designed to process highly viscous FSs and, in preliminary experiments, alginate concentration showed to influence the production yield, a deep rheological characterization was performed on the different FSs. The flow curves of FSs, showed in
Figure 2, are reported in terms of viscosity
versus shear rate.
Figure 2.
Flow curves of feed solutions containing alginate in a range from 1% w/v to 5% w/v, in terms of viscosity versus shear rate.
Figure 2.
Flow curves of feed solutions containing alginate in a range from 1% w/v to 5% w/v, in terms of viscosity versus shear rate.
Data of
Figure 2 clearly show how the shear thinning behavior becomes increasingly evident with increasing alginate concentrations, because the increase of viscosity is more pronounced at the lower shear rates [
14]. In parallel, the critical shear rate marking the transition from the upper Newtonian plateau to the shear thinning region shifts at lower values (
Figure 2). The flow behavior can be satisfactorily described with the Carreau-Yasuda equation (Equation 1),
where the infinite shear viscosity η
∞ can be arbitrarily set equal to zero in the absence of experimental data at sufficiently high shear conditions. Therefore, the effects of alginate concentration can be synthetically analyzed through the model parameters, in particular, through the variations of the zero shear viscosity η
0 and the critical shear rate
![Jfb 04 00312 i005]() c
c, derived from model parameters (a, b and p) in correspondence with η = η
0/2.
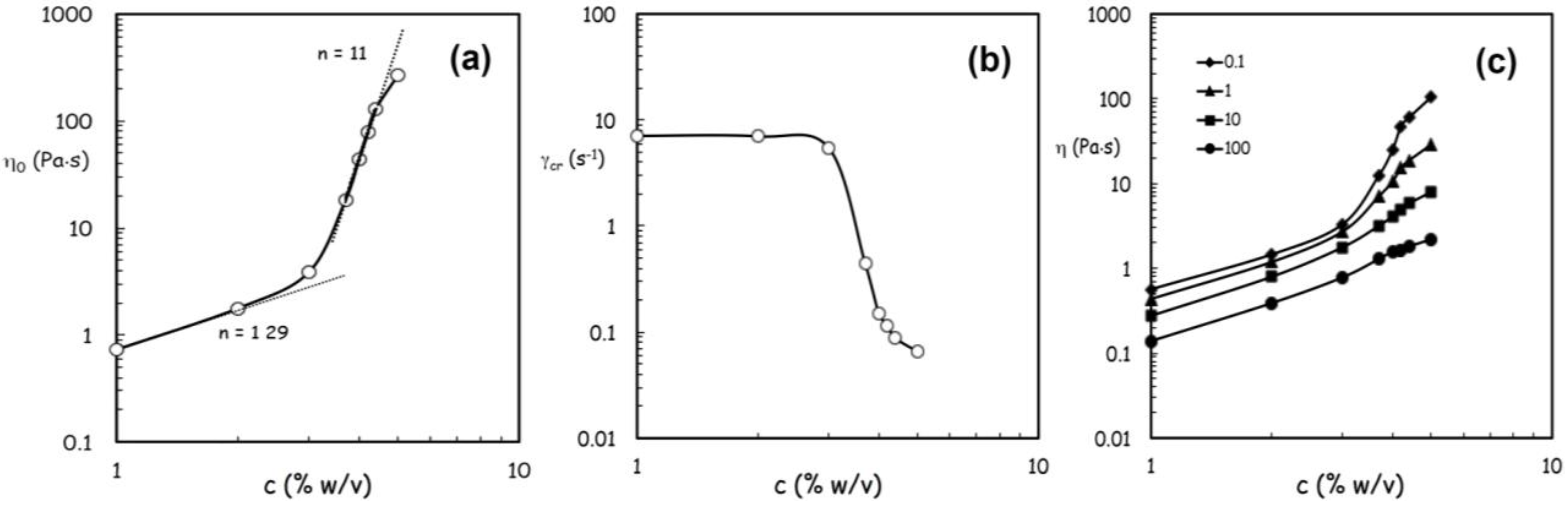
An evident transition occurs in the concentration dependence of both parameters around 3% w/v, as illustrated in
Figure 3a,b. At low concentration the rate of increase in zero shear viscosity η
0 falls in the typical range of dilute solutions while it is much higher at concentrations above 3% w/v. Such a high scaling law exponent, exceeding that of ordinary concentrated polymer solutions, can be attributed to structural interactions between alginate and (hydroxypropyl)methyl cellulose (HPMC) promoted by the co-presence of lysozyme and ethanol [
15,
16].
Figure 3.
Flow behavior of feed solutions containing alginate in a range from 1% w/v to 5% w/v according to Carreau-Yasuda equation. Panel (
a): effect of alginate concentration on the variations of the zero shear viscosity η
0; Panel (
b): effect of alginate concentration and on the critical shear rate
![Jfb 04 00312 i005]() c
c, at infinite shear viscosity η
∞ set equal to zero; and Panel (
c): effects produced by an increase of alginate concentration above 3% w/von viscosity values at different reference shear conditions (0.1, 1, 10, 100 s
−1).
Figure 3.
Flow behavior of feed solutions containing alginate in a range from 1% w/v to 5% w/v according to Carreau-Yasuda equation. Panel (
a): effect of alginate concentration on the variations of the zero shear viscosity η
0; Panel (
b): effect of alginate concentration and on the critical shear rate
![Jfb 04 00312 i005]() c
c, at infinite shear viscosity η
∞ set equal to zero; and Panel (
c): effects produced by an increase of alginate concentration above 3% w/von viscosity values at different reference shear conditions (0.1, 1, 10, 100 s
−1).
The pronounced drop in the critical shear rate c occurring above 3% w/v underlines how much the extension of the upper Newtonian plateau is confined at very low shear rates at higher alginate concentrations owing to the remarkable role played by inter-polymeric associative interactions on the rheological properties of concentrated FSs.
By comparing the viscosity values at different reference shear conditions (0.1, 1, 10, 100 s
−1), it can clearly be noticed that the effects produced by an increase of alginate concentration above 3% becomes more and more remarkable at lower shear rates (
Figure 3c).
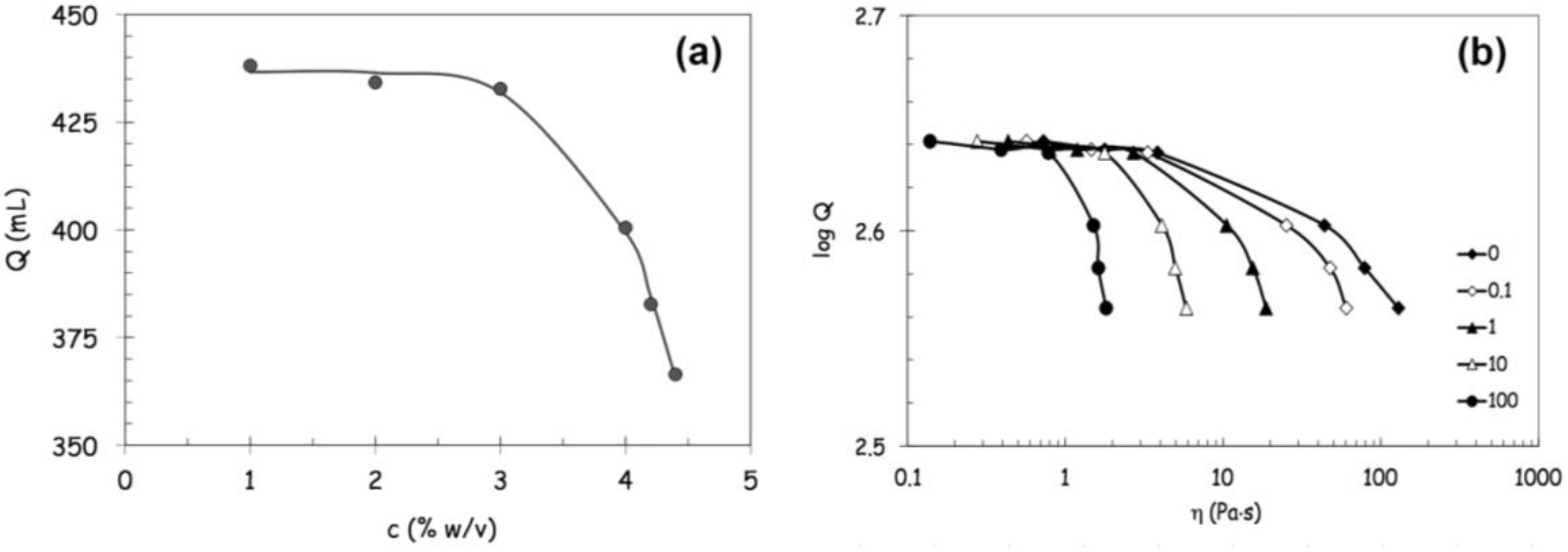
Figure 4a shows that the throughput
Q begins to decrease more significantly above 3% w/v with a profile that can be described by the following stretched exponential relation (Equation 2):
where
c is the alginate concentration.
The correlations between throughput and viscosity (the zero-shear viscosity η
0 and the viscosity values calculated at different reference shear rates from the Carreau-Yasuda equation, Equation 1) are illustrated in
Figure 4b, where the throughput decays again show stretched exponential profiles. The throughput began to decrease more rapidly when the viscosity exceeded a critical threshold value, which depended on the shear rate considered as reference value for flow conditions. Furthermore, the decay increased with increasing shear rate and tended to become even more pronounced at the highest shear rates, tending to approach the process flow conditions encountered within the nozzle. The poor process ability of the most concentrated FS (5% w/v) appears to be better correlated with its higher viscosity values displayed with high shear conditions.
Figure 4.
Prototype throughput analysis. Panel (a): prototype throughput Q as a function of alginate concentration in feed solutions; and Panel (b): correlation between throughput and viscosity. The zero-shear viscosity η0 and the viscosity values are calculated, at different reference shear rates, from the Carreau-Yasuda equation.
Figure 4.
Prototype throughput analysis. Panel (a): prototype throughput Q as a function of alginate concentration in feed solutions; and Panel (b): correlation between throughput and viscosity. The zero-shear viscosity η0 and the viscosity values are calculated, at different reference shear rates, from the Carreau-Yasuda equation.
Notwithstanding the satisfactory correlations found between throughput and viscosity values, it must be emphasized that the flow conditions experienced by the fluid in the prototype are intermittent and characterized by significant extensional components within various segments of the process, particularly in sudden contraction regions, and then far from the steady conditions established for the shear viscosity measurement.
A more comprehensive rheological characterization should include transient flow conditions and time-dependent viscoelastic responses as well as extensional viscosity measurements, so providing the experimental data basis necessary for testing the numerical simulation of the macroscopic behavior exhibited in the process configuration. Such a formidable task is outside of the aim of the present work.
2.5. Microparticle Characterization
Four of the preliminary batches (n. 1, 3, 6, and 8) were re-prepared in order to characterize the products obtained (
Table 4) in terms of particle size (also after swelling), morphology, LZ content and release in different media. Two FSs characterized by a different viscosity (high viscosity, H, and low viscosity, L), corresponding to the previously described “alginate 4% w/v” and “alginate 2% w/v”, with viscosities of about 100 Pa∙s and 1 Pa∙s respectively, were employed. LZ was added to each FS up to a final concentration of 4.7 g/L (
Table 4). A 15% w/v CaCl
2 was used as gelation fluid. Nebulization, gelation and further processing were done as previously described.
Table 4.
Operative setting for the batches extensively characterized.
Table 4.
Operative setting for the batches extensively characterized.
| Batch n. | % w/w | Viscosity of FSs: (H, high; L, low) | Nozzle size |
|---|
| Alginate | HPMC | LZ |
|---|
| 6 | 72.8 | 8.7 | 18.5 | H | Small |
| 1 | 72.8 | 8.7 | 18.5 | Large |
| 8 | 57.3 | 13.6 | 29.1 | L | Small |
| 3 | 57.3 | 13.6 | 29.1 | Large |
Mean particle size and distribution have been analyzed on the particle fraction smaller than 400 μm after batch sieving. The fraction with the largest particles (> 400 μm) was very tiny for the batches n. 1, 3, and 6 with 5.0% w/w, 7.1% w/w, and 3.0% w/w, respectively, while the batch n. 8 had a large particle fraction of about 18% w/w. The volume mean diameter (Vmd) and the particle size distribution of the fraction < 400 μm are reported in
Table 5 and illustrated in
Figure 5, respectively. Batches n. 1 and 6, both prepared with high viscosity solutions but with the small and large size nozzles, respectively, presented a smaller Vmd than batches n. 3 and 8, prepared with low viscosity solutions (
Table 5). Even though only the first two batches seemed to have a symmetric Gaussian distribution, particle distributions were similar for all the batches (
Figure 5).
Scanning electron microscopy (SEM) pictures, reported in
Figure 5, show that particles from batchn. 6 have a morphology different from the others. In fact, these particles were very irregular with crumpled surfaces and a non-spherical shape.
Figure 5.
Particle size distribution (bluish, native particle distribution; green, particle distribution after swelling in phosphate buffer saline) and scanning electron microscopy photographs of the four batches produced for characterization.
Figure 5.
Particle size distribution (bluish, native particle distribution; green, particle distribution after swelling in phosphate buffer saline) and scanning electron microscopy photographs of the four batches produced for characterization.
On the contrary, particles of the other batches appear to be formed by the aggregation of smaller spherical particles that probably occurred during the impact with the gelation solution (
Figure 5). This observation is of interest to foresee the potentiality of this novel airless spray-gun microencapsulation system. In fact, it can be speculated that there is the possibility to produce particles of a smaller size (20–50 µm) than those presented here (
Figure 6). This could be of interest to produce powders with a particle size closer to that generally obtained using spray-drying technology.
Table 5.
Volume mean diameter of microparticles before and after swelling.
Table 5.
Volume mean diameter of microparticles before and after swelling.
| Batch n. | Native Vmd (µm) | After swelling in H2O | After swelling in PBS |
|---|
| 1 | 158.7 ± 108.8 | 144.7 ± 105.3 | 153.6 ± 136.3 |
| 3 | 184.5 ± 122.7 | 189.8 ± 123.5 | 274.4 ± 131.7 |
| 6 | 68.9 ± 61.1 | 51.5 ± 45.3 | 137.5 ± 125.5 |
| 8 | 190.8 ± 132.2 | 175.9 ± 125.5 | 199.0 ± 142.0 |
Since the airless spray-gun apparatus has been conceived for large scale production of microparticulate systems starting from very viscous and bulky materials, powder characteristics like flowability was investigated. The angles of repose of the four batches investigated are reported in
Table 6. Powders from batches n. 1, 3, and 6, had an angle of repose that was found lower than 35° while the powder of the batch n. 8 showed a value slightly higher than 35°. Generally, an angle of repose smaller than 35° indicates a good flowability with no concerns for the industrial processes [
17].
Table 6.
Angle of repose.
Table 6.
Angle of repose.
| Measure | Batchn. 1 | Batch n. 3 | Batch n. 6 | Batch n. 8 |
|---|
| 1 | 32.62 | 32.21 | 31.80 | 36.87 |
| 2 | 32.21 | 32.21 | 32.21 | 38.31 |
| 3 | 33.02 | 33.42 | 31.38 | 37.23 |
| Mean ± S.D. | 32.6 ± 0.41 | 32.6 ± 0.70 | 31.8 ± 0.42 | 37.5 ± 0.75 |
Figure 6.
SEM photographs of microparticle surface morphology (batch n. 1). (a): Magnification, 329×; and (b): magnification, 2634×.
Figure 6.
SEM photographs of microparticle surface morphology (batch n. 1). (a): Magnification, 329×; and (b): magnification, 2634×.
Counterintuitively, in this specific case, the larger particle size corresponded to the higher angle of repose. Generally, it is believed that the larger is the particle size, the better is the flowability but this was not the case. Obviously, the particle size and distribution are not the only characteristics that influence the flowability and particle morphology, porosity, and surface area should be taken into account as well. By analyzing the surface morphology at higher magnification, particles seemed to derive from the stable aggregation of smaller particles as previously stated. However, the building blocks (the small particles) are not spherical like those of the batch n. 1 (
Figure 6) showing a collapsed and shriveled shape, probably due to their impact with the gelation solution. Even whether these characteristics are shared with batch n. 3, overall particle surface of the batch n. 8 is smoother (data not shown) and this may confer the larger surface for particle-particle interaction, conferring a lower flowability.
Vmd and particle size distributions before and after swelling are reported in
Table 5 and illustrated in
Figure 5. As expected, all the investigated particles, but those of batch n. 1, swollen after incubation in PBS because of the calcium extraction operated by the phosphate. On the contrary, water was able to swell and increase the Vmd only of the formulation n. 3 (
Table 5).
The effect of phosphate on calcium alginate is known and reported in literature [
18,
19] and it is ascribed to the affinity between phosphate and calcium ions, leading to a de-gelling of the system. In this specific case, the presence of an outer chitosan coating does not allow for fast particle destruction [
19]. The effect of water (reduction of Vmd) can be explained in a different way. Generally, calcium alginate MPs, especially when stabilized by a polycation outer layer, are stable in distilled water for months. The peculiar particle structure (agglomerates of smaller particles as shown in
Figure 6) might allow the detachment of small particles from the surface, reducing effectively the Vmd of the powder. However, the decrease of the particle optical density [
20] leading to minor light obscuration when travelling into the measure cells [
21] cannot be excluded. If this was the case, particles even larger (swollen) than the original ones (dry) would have been seen smaller and the recorded Vmd reduction was only apparent.
Finally, MP release behaviors were characterized to assess the possibility to use LZ-embedded MPs as a prolonged delivery system in fish farming. To this aim, release studies were performed at room temperature and not at 37 °C, even though fish body temperature may be lower. In fact, fresh water fishes are poikilothermic animals and their body temperature fluctuates as a consequence of variation in the ambient environmental temperature [
22].
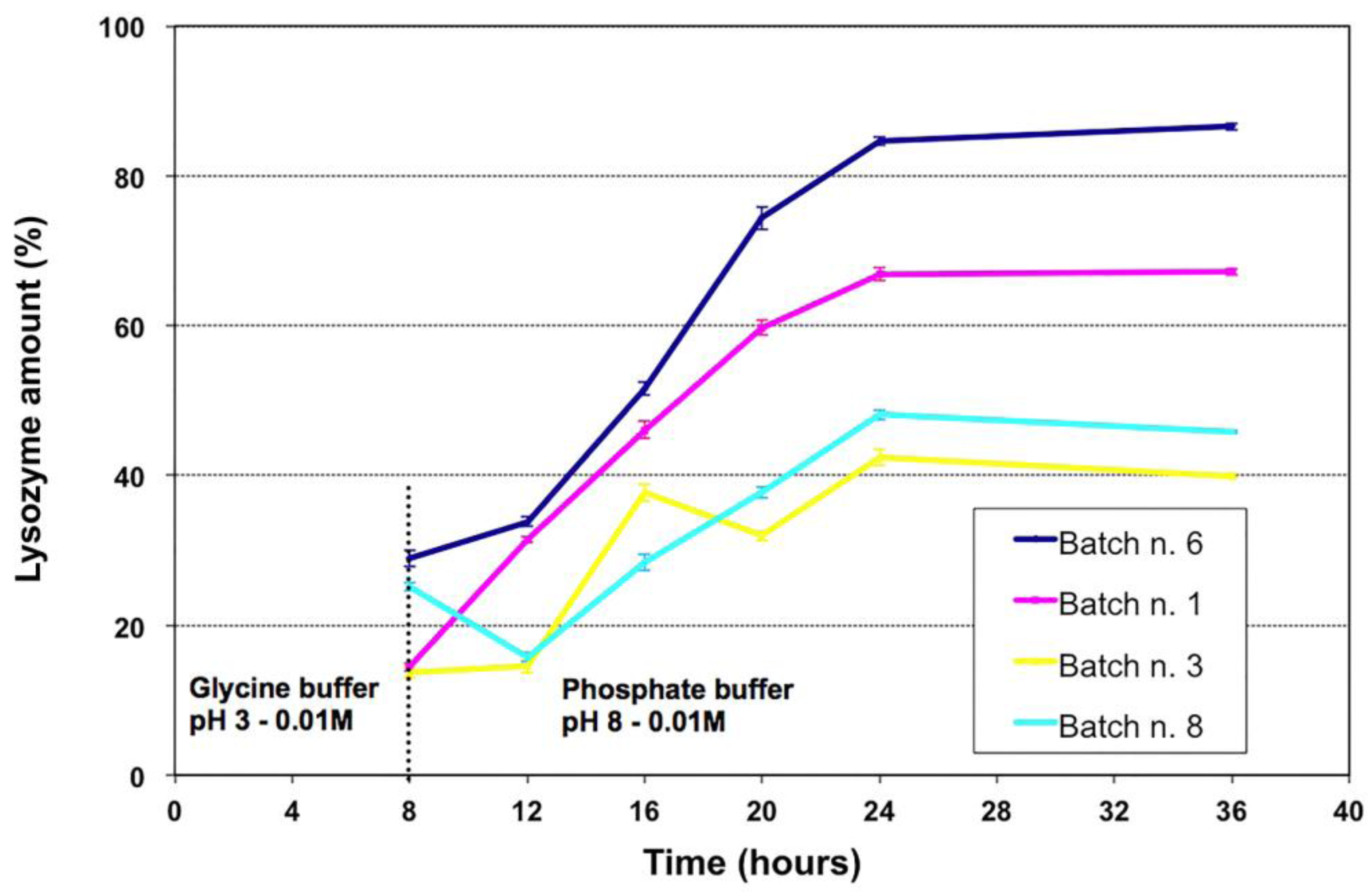
Figure 7 shows LZ release from MPs when incubated 8 hours in glycine buffer (0.01 M, pH 3), to mimic gastric environment, and subsequently in PBS (0.01 M, pH 8). MPs released about 15%–30% of the loaded LZ in the acidic environment with the batches n. 1 and 8 releasing around 15%. The resistance to gastric environment is generally due to the outer chitosan coating and similar behavior has been previously observed with chitosan-coated alginate MPs [
23,
24].
After removing the acidic buffer and submitting MPs to conditions mimicking intestinal environment, larger differences were observed between the four batches (
Figure 7). MPs from the batches n.3 and 8 released less than 50% of their content while batches n. 1 and 6 released ~70% and ~90%, respectively.
Figure 7.
Lysozyme release patterns at pH 3 and at pH 8 of the four selected batches. Microparticles were incubated 8 hours in glycine buffer (pH 3) to mimic gastric environment and, successively, in phosphate buffer saline (pH 8) to mimic small bowel’s environment.
Figure 7.
Lysozyme release patterns at pH 3 and at pH 8 of the four selected batches. Microparticles were incubated 8 hours in glycine buffer (pH 3) to mimic gastric environment and, successively, in phosphate buffer saline (pH 8) to mimic small bowel’s environment.
Batch n. 6 seems the most promising because of the gradual and almost complete release, even though about 30% of the content was released in the acidic environment. However, the time of acidic incubation has been exacerbated due to the variation of the gastric transit time for different fish species.
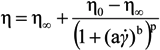
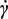 c, derived from model parameters (a, b and p) in correspondence with η = η0/2.
c, derived from model parameters (a, b and p) in correspondence with η = η0/2.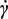 c, at infinite shear viscosity η∞ set equal to zero; and Panel (c): effects produced by an increase of alginate concentration above 3% w/von viscosity values at different reference shear conditions (0.1, 1, 10, 100 s−1).
c, at infinite shear viscosity η∞ set equal to zero; and Panel (c): effects produced by an increase of alginate concentration above 3% w/von viscosity values at different reference shear conditions (0.1, 1, 10, 100 s−1).
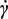 c, at infinite shear viscosity η∞ set equal to zero; and Panel (c): effects produced by an increase of alginate concentration above 3% w/von viscosity values at different reference shear conditions (0.1, 1, 10, 100 s−1).
c, at infinite shear viscosity η∞ set equal to zero; and Panel (c): effects produced by an increase of alginate concentration above 3% w/von viscosity values at different reference shear conditions (0.1, 1, 10, 100 s−1).