Characterization of Porous TiO2 Surfaces Formed on 316L Stainless Steel by Plasma Electrolytic Oxidation for Stent Applications
Abstract
:1. Introduction
2. Experimental Section
2.1. Sample Preparation and PEO Treatment
2.2. Surface Characterization
2.3. Statistics
3. Results and Discussion
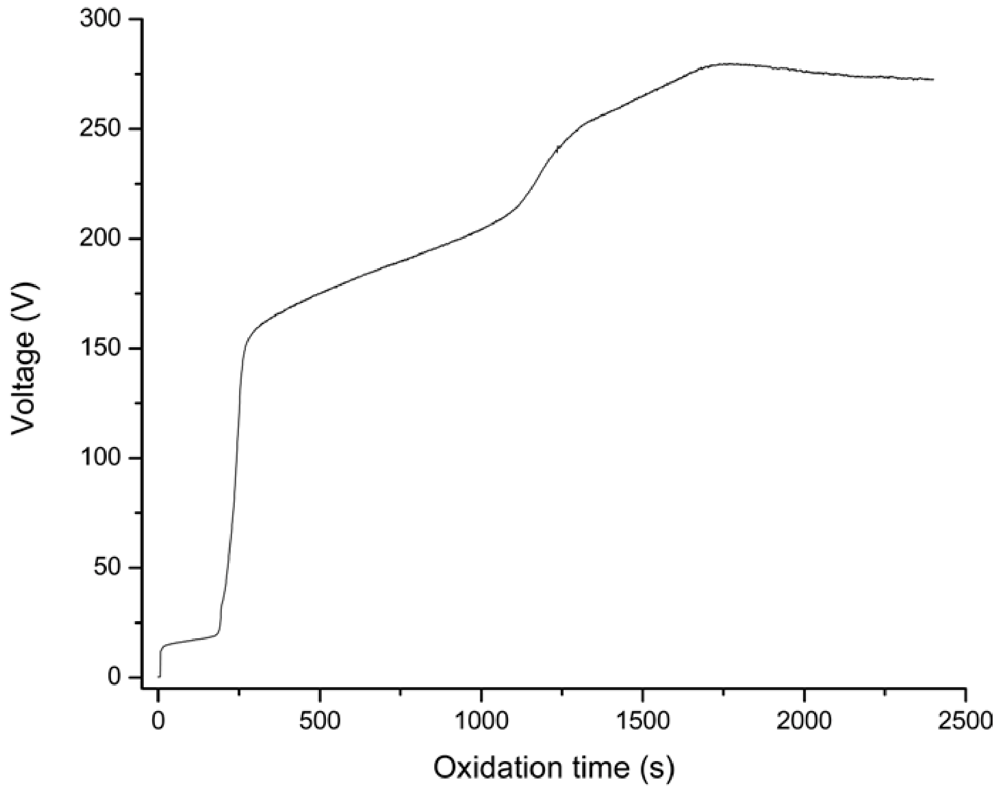
3.1. Voltage-Time Responses during the PEO Process
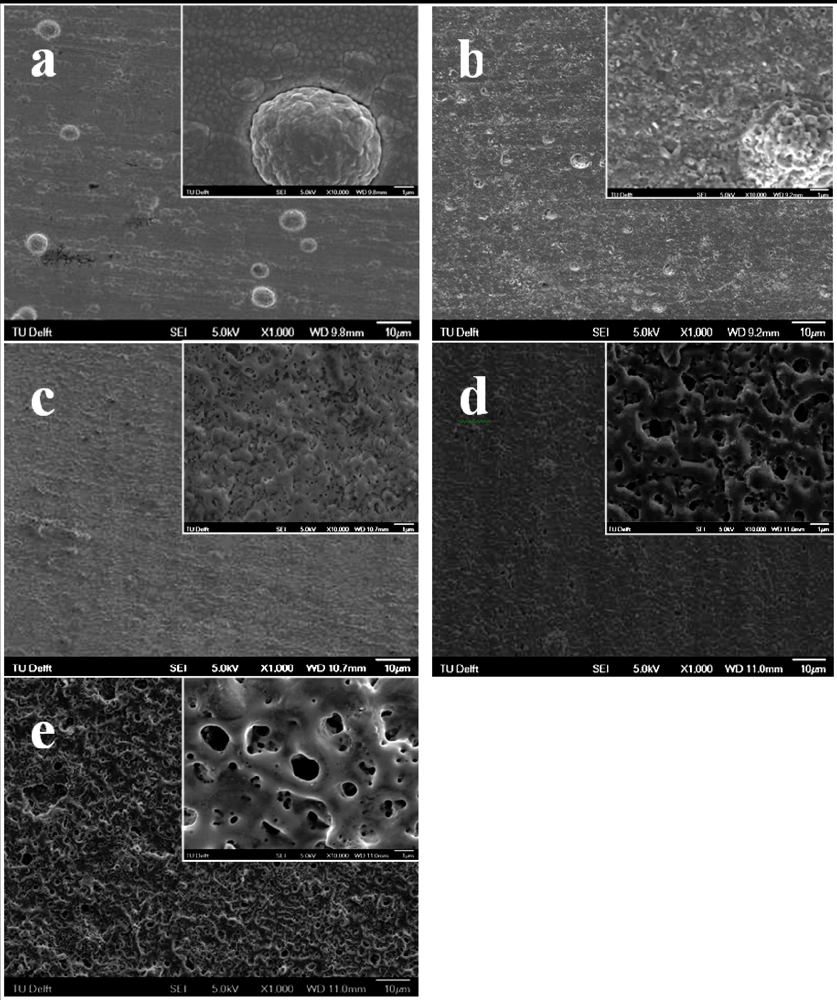
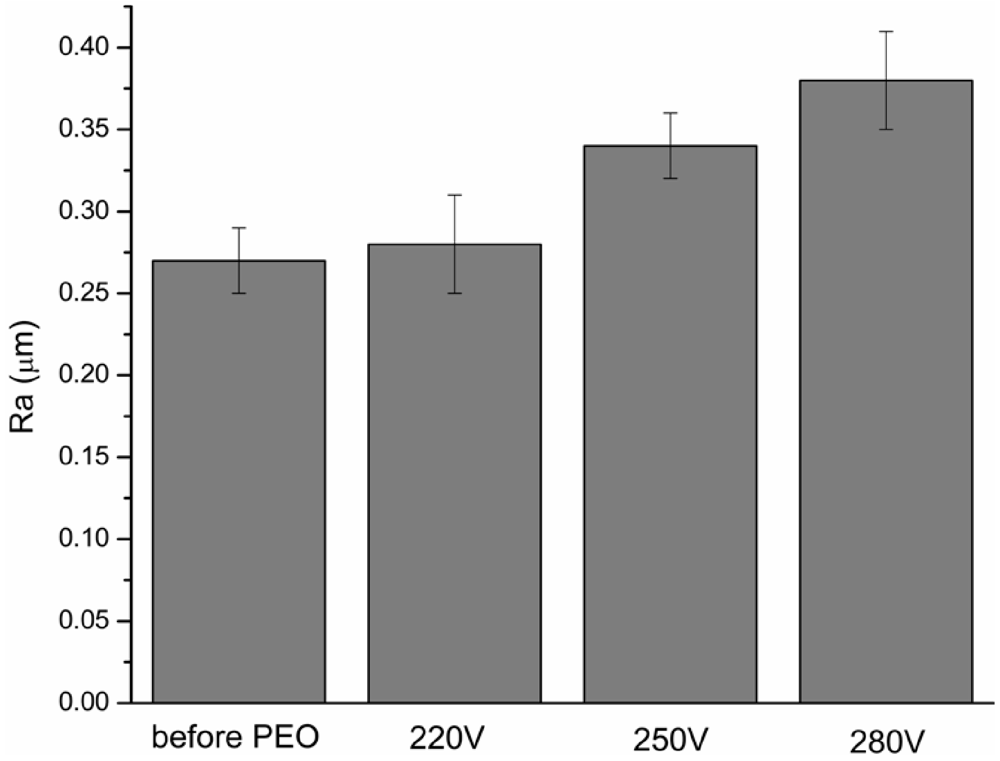
3.2. Surface Morphology and Chemical Composition of the PEO Layers
| Voltage during PEO | 220 V | 250 V | 280 V |
|---|---|---|---|
| Surface porosity (%) | 3.18 | 7.43 | 7.89 |
| Average pore size (µm) | 0.13 | 0.44 | 0.57 |
| Pore density (no. pores/mm2) | 2.49 × 106 | 5.10 × 105 | 2.97 × 105 |

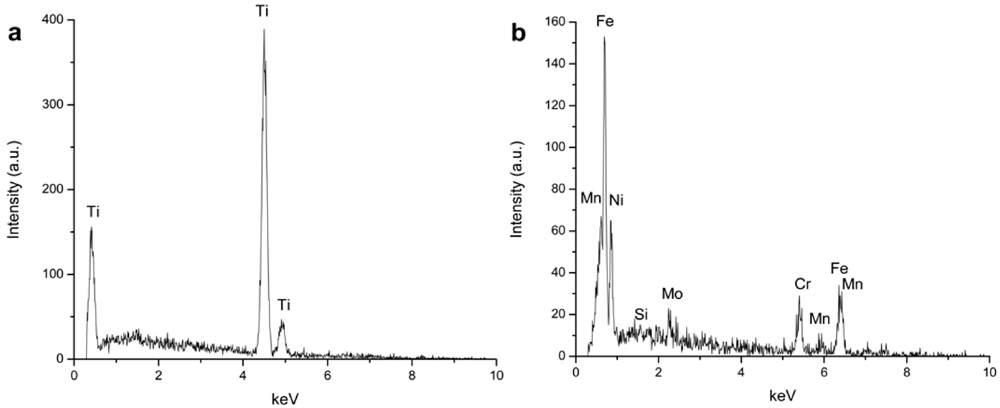
2.3. Morphology and Elemental Distribution across Oxide Layers Thickness

2.4. Wettability and Surface Free Energy
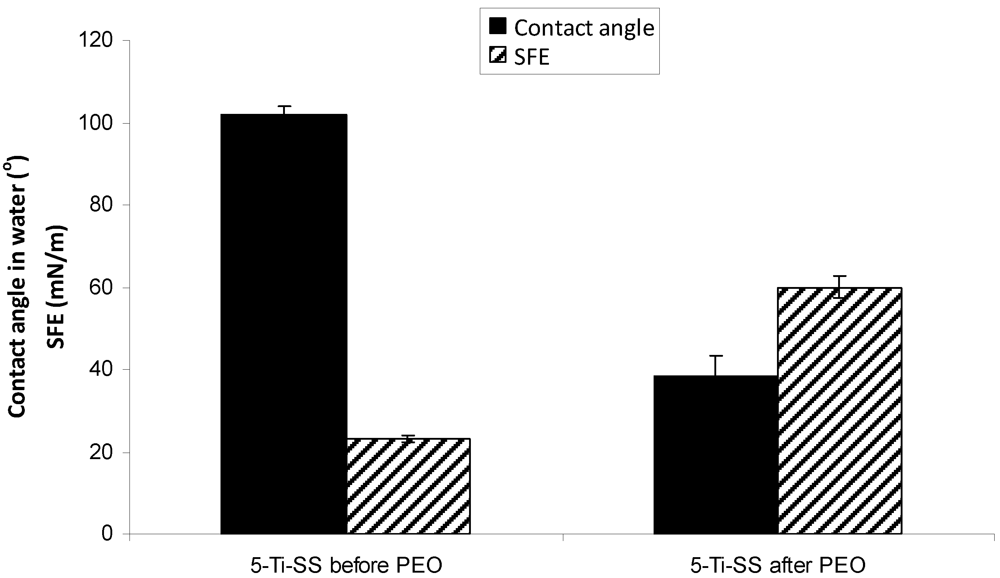
4. Conclusions
Acknowledgements
References
- Van de Hoeven, B.L.; Pires, N.M.M.; Warda, H.M.; Oemrawsingh, P.V.; Van Vlijmen, B.J.M.; Quax, P.H.A.; Schalij, M.J. Drug-eluting stents: Results, promises and problems. Int. J. Cardiol. 2005, 99, 9–17. [Google Scholar]
- Kukreja, N.; Onuma, Y.; Daemen, J.; Serruys, P.W. The future of drug eluting stents. Pharmacol. Res. 2008, 57, 171–180. [Google Scholar]
- Morice, M.C.; Serruys, P.W.; Sousa, J.E.; Fajadet, J.; Ban Hayashi, E.; Perin, M.; Colombo, A.; Schuler, G.; Barragan, P.; Guagliumi, G.; Molnar, F.; Falotico, R. A randomized comparison of a sirolimus-eluting stent with a standard stent for coronary revascularization. N. Engl. J. Med. 2002, 346, 1773–1780. [Google Scholar]
- Stone, G.W.; Ellis, S.G.; Cox, D.A.; Hermiller, J.; O’Shaughnessy, C.; Mann, J.T.; Turco, M.; Caputo, R.; Bergin, P.; Greenberg, J.; Popma, J.J.; Russell, M.E. A polymer based, paclitaxel-eluting stent in patients with coronary artery disease. N. Engl. J. Med. 2004, 350, 221–231. [Google Scholar]
- McFadden, E.P.; Stabile, E.; Regar, E.; Cheneau, E.; Ong, A.T.; Kinnaird, T.; Suddath, W.O.; Weissman, N.J.; Torguson, R.; Kent, K.M.; Pichard, A.D.; Satler, L.F.; Waksman, R.; Serruys, P.W. Late thrombosis in drug-eluting coronary stents after discontinuation of antiplatelet therapy. Lancet 2004, 364, 1519–1521. [Google Scholar]
- Wessely, R.; Kastrati, A.; Schömig, A. Late restenosis in patients receiving a polymer-coated sirolimus-eluting stent subsequently. Ann. Intern. Med. 2005, 143, 392–394. [Google Scholar]
- Hausleiter, J.; Kastrati, A.; Wessely, R.; Dibra, A.; Mehilli, J.; Schratzenstaller, T.; Graf, I.; Renke-Gluszko, M.; Behnisch, B.; Dirschinger, J.; Wintermantel, E.; Schömig, A. Prevention of restenosis by novel drug-eluting stent system with a dose-adjustable, polymer-free, on-site sent coating. Eur. Heart J. 2005, 26, 1475–1481. [Google Scholar]
- Zhang, F.; Zheng, Z.; Chen, Y.; Liu, X.; Chen, A.; Jiang, Z. In vivo investigation of blood compatibility of titanium oxide films. J. Biomed. Mater. Res. 1998, 42, 128–133. [Google Scholar]
- Williams, D.F. Titanium and titanium alloys. In Biocompatibility of Clinical Implant Materials; CRC Press: Boca Raton, FL, USA, 1981; Volume 1, pp. 9–44. [Google Scholar]
- Song, S.; Park, Y.J.; Cho, M.D.; Kim, J.H.; Jeong, M.H.; Kim, Y.S.; Cho, D.L. Preparation of a drug-eluting stent using a TiO2 film deposited by plasma enhanced chemical vapour deposition as a drug-combining matrix. J. Mater. Chem. 2010, 20, 4792–4801. [Google Scholar]
- Lazarev, V.B.; Sanygin, V.P.; Kvardakov, A.M.; Saakiyan, L.S.; Efremov, A.P.; Kutsev, A.V. Oxidized aluminum coatings on steel produced by microarc method. Inorg. Mater. 1991, 27, 614–619. [Google Scholar]
- Gu, W.-C.; Lv, G.-H.; Chen, H.; Chen, G.-L.; Feng, W.-R.; Yang, S.-Z. PEO protective coatings on inner surface of tubes. Surf. Coat. Technol. 2007, 201, 6619–6622. [Google Scholar] [CrossRef]
- Frauchiger, V.M.; Schlottig, F.; Gasser, B.; Textor, M. Anodic plasma-chemical treatment of CP titanium surfaces for biomedical applications. Biomaterials 2004, 25, 593–606. [Google Scholar]
- Yerokhin, A.L.; Nie, X.; Leyland, A.; Matthews, A.; Dowey, S.J. Plasma electrolysis for surface engineering. Surf. Coat. Technol. 1999, 122, 73–93. [Google Scholar]
- Apachitei, I.; Lonyuk, B.; Fratila-Apachitei, L.E.; Zhou, J.; Duszczyk, J. Fatigue response of porous coated titanium biomedical alloys. Scripta Mater. 2009, 61, 113–116. [Google Scholar]
- Necula, B.S.; Apachitei, I.; Tichelaar, F.D.; Fratila-Apachitei, L.E.; Duszczyk, J. An electron microscopical study on the growth of TiO2-Ag antibacterial coatings on Ti6Al7Nb biomedical alloy. Acta Biomater. 2011, 7, 2751–2757. [Google Scholar]
- Matykina, E.; Berkani, A.; Skeldon, P.; Thompson, G.E. Real-time imaging of coating growth during plasma electrolytic oxidation of titanium. Electrochim. Acta 2007, 53, 1987–1994. [Google Scholar]
- Montero, I.; Fernández, M.; Albella, J.M. Pore formation during the breakdown process in anodic Ta2O5 fims. Electrochim. Acta 1987, 32, 171–174. [Google Scholar]
- Afshar, A.; Vaezi, M.R. Evaluation of electrical breakdown of anodic films on titanium in phosphate-base solutions. Surf. Coat. Technol. 2004, 186, 398–404. [Google Scholar]
- Ryu, H.S.; Mun, S.J.; Lim, T.S.; Kim, H.C.; Shin, K.S.; Hong, S.H. Microstructure evolution during plasma electrolytic oxidation and its effects on the electrochemical properties of AZ91D Mg alloy. J. Electrochem. Soc. 2011, 158, C266–C273. [Google Scholar]
- Wang, Y.; Lei, T.; Jiang, B.; Guo, L. Growth, microstructure and mechanical properties of microarc oxidation coatings on titanium alloy in phosphate-containing solution. Appl. Surf. Sci. 2004, 233, 258–267. [Google Scholar]
- Rudnev, V.S.; Yarovaya, T.P.; Egorkin, V.S.; Sinebryukov, S.L.; Gnedenkov, S.V. Properties of coatings formed on titanium by plasma electrolytic oxidation in a phosphate-borate electrolyte. Russ. J. Appl. Chem. 2010, 83, 664–670. [Google Scholar]
- Parfenov, E.V.; Yerokhin, A.L.; Matthews, A. Frequency response studies for the plasma electrolytic oxidation process. Surf. Coat. Technol. 2007, 201, 8661–8670. [Google Scholar]
- Sundararajan, G.; Krishna, L.R. Mechanisms underlying the formation of thick alumina coatings through the MAO coating technology. Surf. Coat. Technol. 2003, 167, 269–277. [Google Scholar]
- Matykina, E.; Monfort, F.; Berkani, A.; Skeldon, P.; Thompson, G.E.; Gough, J. Characterization of sparks-anodized titanium for biomedical applications. J. Electrochem. Soc. 2007, 154, C279–C285. [Google Scholar]
- Ding, S.J.; Ju, C.P.; Lin, J.H. Characterization of hydroxyapatite and titanium coatings sputtered on Ti-6Al-4V substrate. J. Biomed. Mater. Res. 1999, 44, 266–279. [Google Scholar]
- Huang, P.; Wang, F.; Xu, K.; Han, Y. Mechanical properties of titania prepared by plasma electrolytic oxidation at different voltages. Surf. Coat. Technol. 2007, 201, 5168–5171. [Google Scholar]
- Otsuka, Y.; Chronos, N.A.; Apkarian, R.P.; Robinson, K.A. Scanning electron microscopic analysis of defects in polymer coatings of three commercially available stents: Comparison of BiodivYsio, Taxus and Cypher stents. J. Invasive Cardiol. 2007, 19, 71–76. [Google Scholar]
- Song, S.J.; Kim, K.S.; Kim, K.H.; Li, H.J.; Kim, J.H.; Jeong, M.H.; Kim, B.H.; Ko, Y.M.; Cho, D.L. Preparation of a biocompatible stent surface by plasma polymerization followed by chemical grafting of drug compounds. J. Mater. Chem. 2009, 19, 3248–3252. [Google Scholar]
- Díaz, M.; Sevilla, P.; Galán, A.M.; Escolar, G.; Engel, E.; Gil, F.J. Evaluation of ion release, cytotoxicity, and platelet adhesion of electrochemical anodized 316L stainless steel cardiovascular stents. J. Biomed. Mater. Res. Part B 2008, 87B, 555–561. [Google Scholar]
- Annarelli, C.C.; Fornazero, J.; Cohen, R.; Bert, J.; Besse, J.L. Collidal protein solutions as a new standard sensor for adhesive wettability measurements. J. Colloid Interface Sci. 1999, 213, 386–394. [Google Scholar]
- Chrzanowski, W.; Neel, E.A.A.; Armitage, D.A.; Knowles, J.C. Effect of surface treatment on the bioactivity of nickel-titanium. Acta Biomater. 2008, 4, 1969–1984. [Google Scholar]
- Brier-Russell, D.; Salzman, E.W.; Lindon, J.; Merrill, E.W.; Dincer, A.K.; Wu, J.S. In vitro assessment of interaction of blood with model surfaces: Acrylates and methacrylates. J. Colloid Interface Sci. 1981, 81, 311–318. [Google Scholar]
- Haynes, C.A.; Norde, W. Globular proteins at solid/liquid interface. Colloid Surface B 1994, 2, 517–566. [Google Scholar]
- Wang, G.X.; Shen, Y.; Zhang, H.; Quan, X.J.; Yu, Q.S. Influence of surface microroughness by plasma deposition and chemical erosion followed by TiO2 coating upon anticoagulation, hydrophilicity, and corrosion resistance of NiTi alloy stent. J. Biomed. Mater. Res. 2008, 85A, 1096–1102. [Google Scholar]
- Yang, Z.; Wang, J.; Luo, R.; Li, X.; Chen, S.; Sun, H.; Huang, N. Improved hemocompatibility guided by pulsed plasma tailoring the surface amino functionalities of TiO2 coating for covalent immobilization of heparin. Plasma Process. Polym. 2011, 8, 850–858. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huan, Z.; Fratila-Apachitei, L.E.; Apachitei, I.; Duszczyk, J. Characterization of Porous TiO2 Surfaces Formed on 316L Stainless Steel by Plasma Electrolytic Oxidation for Stent Applications. J. Funct. Biomater. 2012, 3, 349-360. https://doi.org/10.3390/jfb3020349
Huan Z, Fratila-Apachitei LE, Apachitei I, Duszczyk J. Characterization of Porous TiO2 Surfaces Formed on 316L Stainless Steel by Plasma Electrolytic Oxidation for Stent Applications. Journal of Functional Biomaterials. 2012; 3(2):349-360. https://doi.org/10.3390/jfb3020349
Chicago/Turabian StyleHuan, Zhiguang, Lidy E. Fratila-Apachitei, Iulian Apachitei, and Jurek Duszczyk. 2012. "Characterization of Porous TiO2 Surfaces Formed on 316L Stainless Steel by Plasma Electrolytic Oxidation for Stent Applications" Journal of Functional Biomaterials 3, no. 2: 349-360. https://doi.org/10.3390/jfb3020349





