Preparation and Functional Assessment of Composite Chitosan-Nano-Hydroxyapatite Scaffolds for Bone Regeneration
Abstract
:1. Introduction
2. Experimental Section
2.1. Phase I: Effects of DDA and Drying Method
2.1.1. Microsphere Fabrication
| Microsphere Type | Chitosan (g) | CaCl2●2H20 (g) | NaH2PO4●H20 (g) |
|---|---|---|---|
| 61% DDA: 3.5% CS, 1× HA | 7.00 | 2.94 | 1.66 |
| 80% DDA: 3.0% CS, 1× HA | 6.00 | 2.94 | 1.66 |
2.1.2. Scaffold Fabrication
2.2. Phase II: Effects of Hydroxyapatite Content and 2-(N-morphilino)ethanesulfonic (MES) Acid Wash
2.2.1. Microsphere and Scaffold Fabrication
| Microsphere Type | Chitosan (g) | CaCl2●2H20 (g) | NaH2PO4●H20 (g) |
|---|---|---|---|
| 3.0% CS, No HA | 6.00 | 0.00 | 0.00 |
| 3.0% CS, 1× HA* | 6.00 | 2.94 | 1.66 |
| 2.5% CS, 2× HA | 5.00 | 5.88 | 3.31 |
2.2.2. MES Acid Wash
2.3. Characterization
2.3.1. Scanning Electron Microscopy
2.3.2. Microsphere and Scaffold Degradation
2.3.3. Compression Testing
2.3.4. Swelling Ratio
2.3.5. Biocompatibility
2.4. Statistical Analysis
3. Results
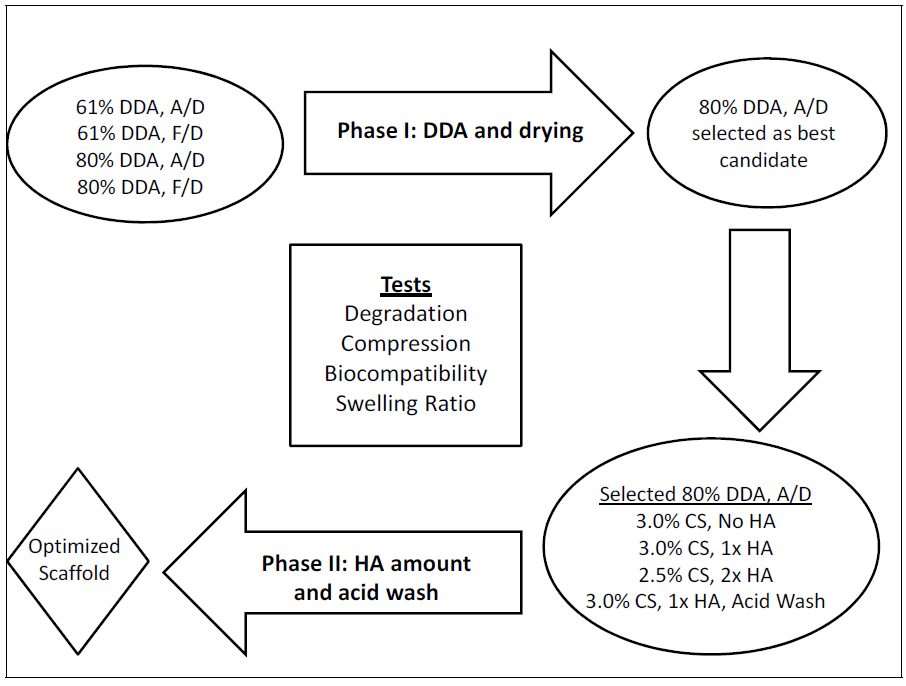
3.1. SEM Images

3.2. Microsphere Degradation
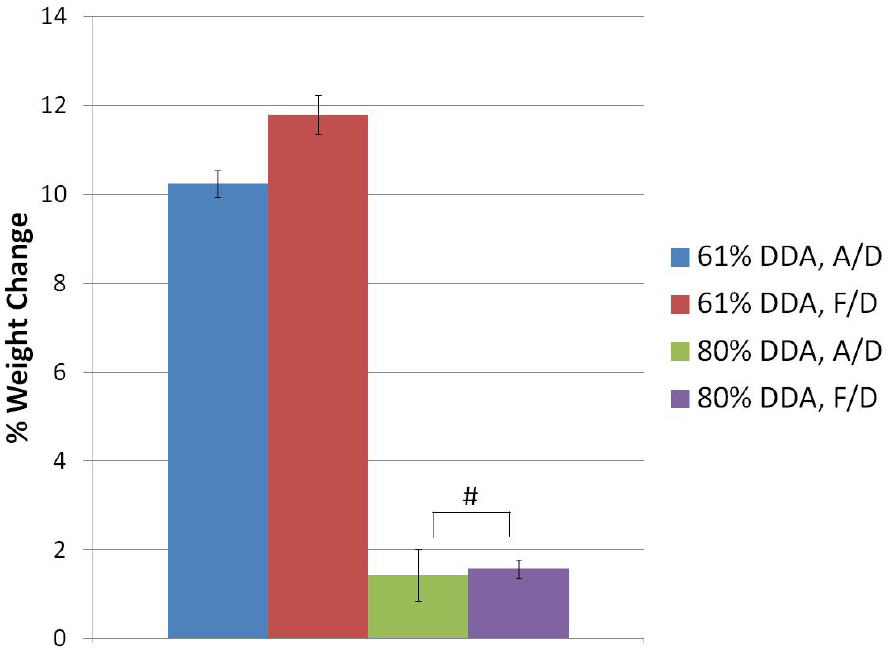
3.3. Phase I: Effects of DDA and Drying Method
| Scaffold Type | Degradation (% wt. change) (N = 4) | Compressive Modulus (MPa) (N = 3) | Swelling Ratio (%) (N = 4) |
|---|---|---|---|
| 61% DDA, A/D | 1.4 ± 0.5 | 0.67 ± 0.06 | 148.3 ± 11.7 b |
| 61% DDA, F/D | 3.5 ± 0.5 a | 0.12 ± 0.01 | 267.1 ± 15.3 b |
| 80% DDA, A/D | 1.3 ± 0.1 | 3.79 ± 0.51 a | 88.5 ± 1.9 b |
| 80% DDA, F/D | 0.8 ± 0.3 | 0.81 ± 0.14 | 116.5 ± 6.9 b |

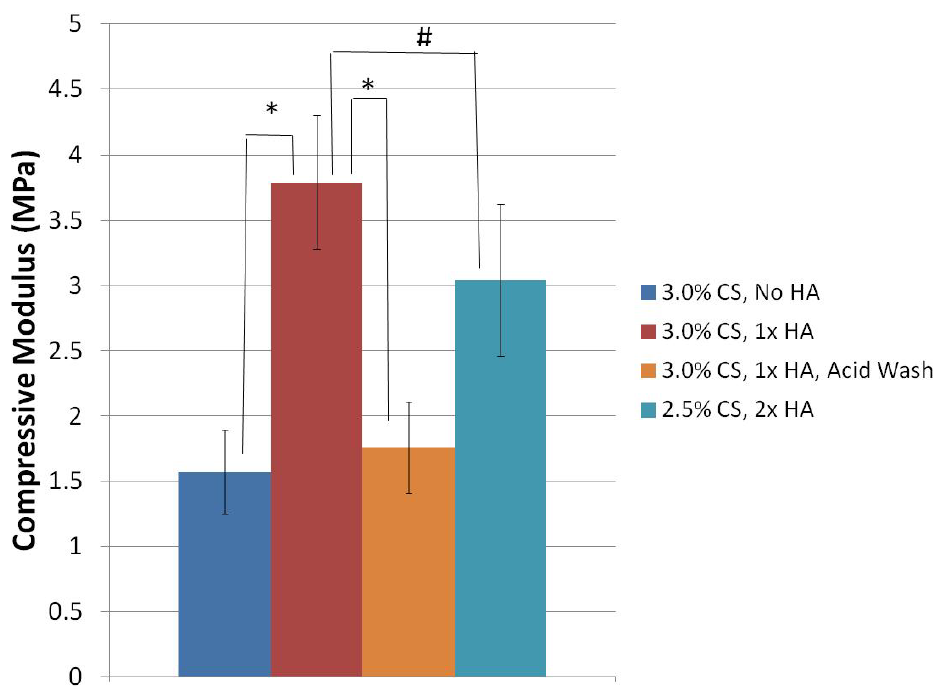
3.4. Phase II: Effects of Hydroxyapatite Content and MES Acid Wash
| Scaffold Type | Degradation (% wt. change) (N = 4) | Swelling Ratio (%) |
|---|---|---|
| (N = 4) | ||
| 3.0% CS, No HA | −0.3 ± 0.4 a | 159.1 ± 1.9 b |
| 3.0% CS, 1× HA | 1.3 ± 0.1 | 88.5 ± 1.9 b |
| 2.5% CS, 2× HA | 1.7 ± 0.2 | 66.1 ± 0.9 b |
| 3.0% CS, 1× HA, Acid Wash | 4.4 ± 0.4 a | 180.4 ± 3.3 b |

4. Discussion
4.1. Phase I: Effects of DDA and Drying Method
4.2. Phase II: Effects of Hydroxyapatite Content and MES Acid Wash
5. Conclusions
Conflict of Interest
References
- Dawson, J.I.; Oreffo, R.O. Bridging the regeneration gap: Stem cells, biomaterials and clinical translation in bone tissue engineering. Arch. Biochem. Biophys. 2008, 473, 124–131. [Google Scholar] [CrossRef]
- Schmidmaier, G.; Schwabe, P.; Strobel, C.; Wildemann, B. Carrier systems and application of growth factors in orthopaedics. Injury 2008, 39, S37–S43. [Google Scholar]
- Griffin, M.; Iqbal, S.A.; Bayat, A. Exploring the application of mesenchymal stem cells in bone repair and regeneration. J. Bone Joint Surg. Br. 2011, 93, 427–434. [Google Scholar]
- Khan, Y.; Yaszemski, M.J.; Mikos, A.G.; Laurencin, C.T. Tissue engineering of bone: Material and matrix considerations. J. Bone Joint Surg. Am. 2008, 90, 36–42. [Google Scholar] [CrossRef]
- Kretlow, J.D.; Mikos, A.G. Review: Mineralization of synthetic polymer scaffolds for bone tissue engineering. Tissue Eng. 2007, 13, 927–938. [Google Scholar] [CrossRef]
- Nandi, S.K.; Roy, S.; Mukherjee, P.; Kundu, B.; De, D.K.; Basu, D. Orthopaedic applications of bone graft & graft substitutes: A review. Indian J. Med. Res. 2010, 132, 15–30. [Google Scholar]
- Karageorgiou, V.; Kaplan, D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 2005, 26, 5474–5491. [Google Scholar] [CrossRef]
- Logeart-Avramoglou, D.; Anagnostou, F.; Bizios, R.; Petite, H. Engineering bone: Challenges and obstacles. J. Cell Mol. Med. 2005, 9, 72–84. [Google Scholar] [CrossRef]
- Muzzarelli, C.; Muzzarelli, R.A. Natural and artificial chitosan-inorganic composites. J. Inorg. Biochem. 2002, 92, 89–94. [Google Scholar] [CrossRef]
- Thein-Han, W.W.; Misra, R.D. Biomimetic chitosan-nanohydroxyapatite composite scaffolds for bone tissue engineering. Acta Biomater. 2009, 5, 1182–1197. [Google Scholar] [CrossRef]
- Kempen, D.H.; Creemers, L.B.; Alblas, J.; Lu, L.; Verbout, A.J.; Yaszemski, M.J.; Dhert, W.J. Growth factor interactions in bone regeneration. Tissue Eng. Part B 2010, 16, 551–566. [Google Scholar] [CrossRef]
- Lieberman, J.R.; Daluiski, A.; Einhorn, T.A. The role of growth factors in the repair of bone. Biology and clinical applications. J. Bone Joint Surg. Am. 2002, 84–A, 1032–1044. [Google Scholar]
- Chesnutt, B.M.; Viano, A.M.; Yuan, Y.; Yang, Y.; Guda, T.; Appleford, M.R.; Ong, J.L.; Haggard, W.O.; Bumgardner, J.D. Design and characterization of a novel chitosan/nanocrystalline calcium phosphate composite scaffold for bone regeneration. J. Biomed. Mater. Res. A 2009, 88, 491–502. [Google Scholar]
- Chesnutt, B.M.; Yuan, Y.; Buddington, K.; Haggard, W.O.; Bumgardner, J.D. Composite chitosan/nano-hydroxyapatite scaffolds induce osteocalcin production by osteoblasts in vitro and support bone formation in vivo. Tissue Eng. Part A 2009, 15, 2571–2579. [Google Scholar] [CrossRef]
- Reves, B.T.; Bumgardner, J.D.; Cole, J.A.; Yang, Y.; Haggard, W.O. Lyophilization to improve drug delivery for chitosan-calcium phosphate bone scaffold construct: A preliminary investigation. J. Biomed. Mater. Res. B 2009, 90, 1–10. [Google Scholar]
- Di Martino, A.; Sittinger, M.; Risbud, M.V. Chitosan: A versatile biopolymer for orthopaedic tissue-engineering. Biomaterials 2005, 26, 5983–5990. [Google Scholar] [CrossRef]
- Kim, I.Y.; Seo, S.J.; Moon, H.S.; Yoo, M.K.; Park, I.Y.; Kim, B.C.; Cho, C.S. Chitosan and its derivatives for tissue engineering applications. Biotechnol. Adv. 2008, 26, 1–21. [Google Scholar]
- Jayakumar, R.; Prabaharan, M.; Nair, S.V.; Tamura, H. Novel chitin and chitosan nanofibers in biomedical applications. Biotechnol. Adv. 2010, 28, 142–150. [Google Scholar]
- Shi, C.; Zhu, Y.; Ran, X.; Wang, M.; Su, Y.; Cheng, T. Therapeutic potential of chitosan and its derivatives in regenerative medicine. J. Surg. Res. 2006, 133, 185–192. [Google Scholar] [CrossRef]
- Venkatesan, J.; Kim, S.K. Chitosan composites for bone tissue engineering—An overview. Mar. Drugs 2010, 8, 2252–2266. [Google Scholar] [CrossRef]
- Coelho, P.G.; Freire, J.N.; Granato, R.; Marin, C.; Bonfante, E.A.; Gil, J.N.; Chuang, S.K.; Suzuki, M. Bone mineral apposition rates at early implantation times around differently prepared titanium surfaces: A study in beagle dogs. Int. J. Oral Maxillofac. Implants 2011, 26, 63–69. [Google Scholar]
- Hermida, J.C.; Bergula, A.; Dimaano, F.; Hawkins, M.; Colwell, C.W., Jr.; D'Lima, D.D. An in vivo evaluation of bone response to three implant surfaces using a rabbit intramedullary rod model. J. Orthop. Surg Res. 2010, 5. [Google Scholar] [CrossRef]
- Thian, E.S.; Huang, J.; Ahmad, Z.; Edirisinghe, M.J.; Jayasinghe, S.N.; Ireland, D.C.; Brooks, R.A.; Rushton, N.; Best, S.M.; Bonfield, W. Influence of nanohydroxyapatite patterns deposited by electrohydrodynamic spraying on osteoblast response. J. Biomed. Mater. Res. A 2008, 85, 188–194. [Google Scholar]
- Bagheri-Khoulenjani, S.; Taghizadeh, S.M.; Mirzadeh, H. An investigation on the short-term biodegradability of chitosan with various molecular weights and degrees of deacetylation. Carbohydr. Polym. 2009, 78, 773–778. [Google Scholar] [CrossRef]
- Madihally, S.V.; Matthew, H.W. Porous chitosan scaffolds for tissue engineering. Biomaterials 1999, 20, 1133–1142. [Google Scholar] [CrossRef]
- Seda Tigli, R.; Karakecili, A.; Gumusderelioglu, M. In vitro characterization of chitosan scaffolds: Influence of composition and deacetylation degree. J. Mater. Sci. Mater. Med. 2007, 18, 1665–1674. [Google Scholar] [CrossRef]
- Thein-Han, W.W.; Kitiyanant, Y. Chitosan scaffolds for in vitro buffalo embryonic stem-like cell culture: An approach to tissue engineering. J. Biomed. Mater. Res. B 2007, 80, 92–101. [Google Scholar]
- Lim, S.M.; Song, D.K.; Oh, S.H.; Lee-Yoon, D.S.; Bae, E.H.; Lee, J.H. In vitro and in vivo degradation behavior of acetylated chitosan porous beads. J. Biomater. Sci. Polym. Ed. 2008, 19, 453–466. [Google Scholar] [CrossRef]
- Ren, D.; Yi, H.; Wang, W.; Ma, X. The enzymatic degradation and swelling properties of chitosan matrices with different degrees of N-acetylation. Carbohydr. Res. 2005, 340, 2403–2410. [Google Scholar] [CrossRef]
- Tomihata, K.; Ikada, Y. In vitro and in vivo degradation of films of chitin and its deacetylated derivatives. Biomaterials 1997, 18, 567–575. [Google Scholar] [CrossRef]
- Liu, W.G.; Sun, S.J.; Cao, Z.Q.; Xin, Z.; Yao, K.D.; Lu, W.W.; Luk, K.D.K. An investigation on the physicochemical properties of chitosan/DNA polyelectrolyte complexes. Biomaterials 2005, 26, 2705–2711. [Google Scholar] [CrossRef]
- Jiang, T.; Kumbar, S.G.; Nair, L.S.; Laurencin, C.T. Biologically active chitosan systems for tissue engineering and regenerative medicine. Curr. Top. Med. Chem. 2008, 8, 354–364. [Google Scholar] [CrossRef]
- Jones, A.C.; Arns, C.H.; Hutmacher, D.W.; Milthorpe, B.K.; Sheppard, A.P.; Knackstedt, M.A. The correlation of pore morphology, interconnectivity and physical properties of 3D ceramic scaffolds with bone ingrowth. Biomaterials 2009, 30, 1440–1451. [Google Scholar] [CrossRef]
- Varum, K.M.; Myhr, M.M.; Hjerde, R.J.; Smidsrod, O. In vitro degradation rates of partially N-acetylated chitosans in human serum. Carbohydr. Res. 1997, 299, 99–101. [Google Scholar] [CrossRef]
- Huang, Y.; Onyeri, S.; Siewe, M.; Moshfeghian, A.; Madihally, S.V. In vitro characterization of chitosan-gelatin scaffolds for tissue engineering. Biomaterials 2005, 26, 7616–7627. [Google Scholar] [CrossRef]
- Tangsadthakun, C.; Kanokpanont, S.; Sanchavanakit, N.; Pichyangkura, R.; Banaprasert, T.; Tabata, Y.; Damrongsakkul, S. The influence of molecular weight of chitosan on the physical and biological properties of collagen/chitosan scaffolds. J. Biomater. Sci. Polym. Ed. 2007, 18, 147–163. [Google Scholar] [CrossRef]
- Peter, M.; Ganesh, N.; Selvamurugan, N.; Nair, S.V.; Furuike, T.; Tamura, H.; Jayakumar, R. Preparation and characterization of chitosan-gelatin/nanohydroxyapatite composite scaffolds for tissue engineering applications. Carbohydr. Polym. 2010, 80, 687–694. [Google Scholar] [CrossRef]
- Li, Z.; Yubao, L.; Aiping, Y.; Xuelin, P.; Xuejiang, W.; Xiang, Z. Preparation and in vitro investigation of chitosan/nano-hydroxyapatite composite used as bone substitute materials. J. Mater. Sci. Mater. Med. 2005, 16, 213–219. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Reves, B.T.; Jennings, J.A.; Bumgardner, J.D.; Haggard, W.O. Preparation and Functional Assessment of Composite Chitosan-Nano-Hydroxyapatite Scaffolds for Bone Regeneration. J. Funct. Biomater. 2012, 3, 114-130. https://doi.org/10.3390/jfb3010114
Reves BT, Jennings JA, Bumgardner JD, Haggard WO. Preparation and Functional Assessment of Composite Chitosan-Nano-Hydroxyapatite Scaffolds for Bone Regeneration. Journal of Functional Biomaterials. 2012; 3(1):114-130. https://doi.org/10.3390/jfb3010114
Chicago/Turabian StyleReves, Benjamin T., Jessica A. Jennings, Joel D. Bumgardner, and Warren O. Haggard. 2012. "Preparation and Functional Assessment of Composite Chitosan-Nano-Hydroxyapatite Scaffolds for Bone Regeneration" Journal of Functional Biomaterials 3, no. 1: 114-130. https://doi.org/10.3390/jfb3010114






