Changes in the Concentration of Leaf Nitrogen over the Season Affect the Diagnosis of Deficiency or Sufficiency in Strawberries in the Subtropics
Abstract
:1. Introduction
2. Materials and Methods
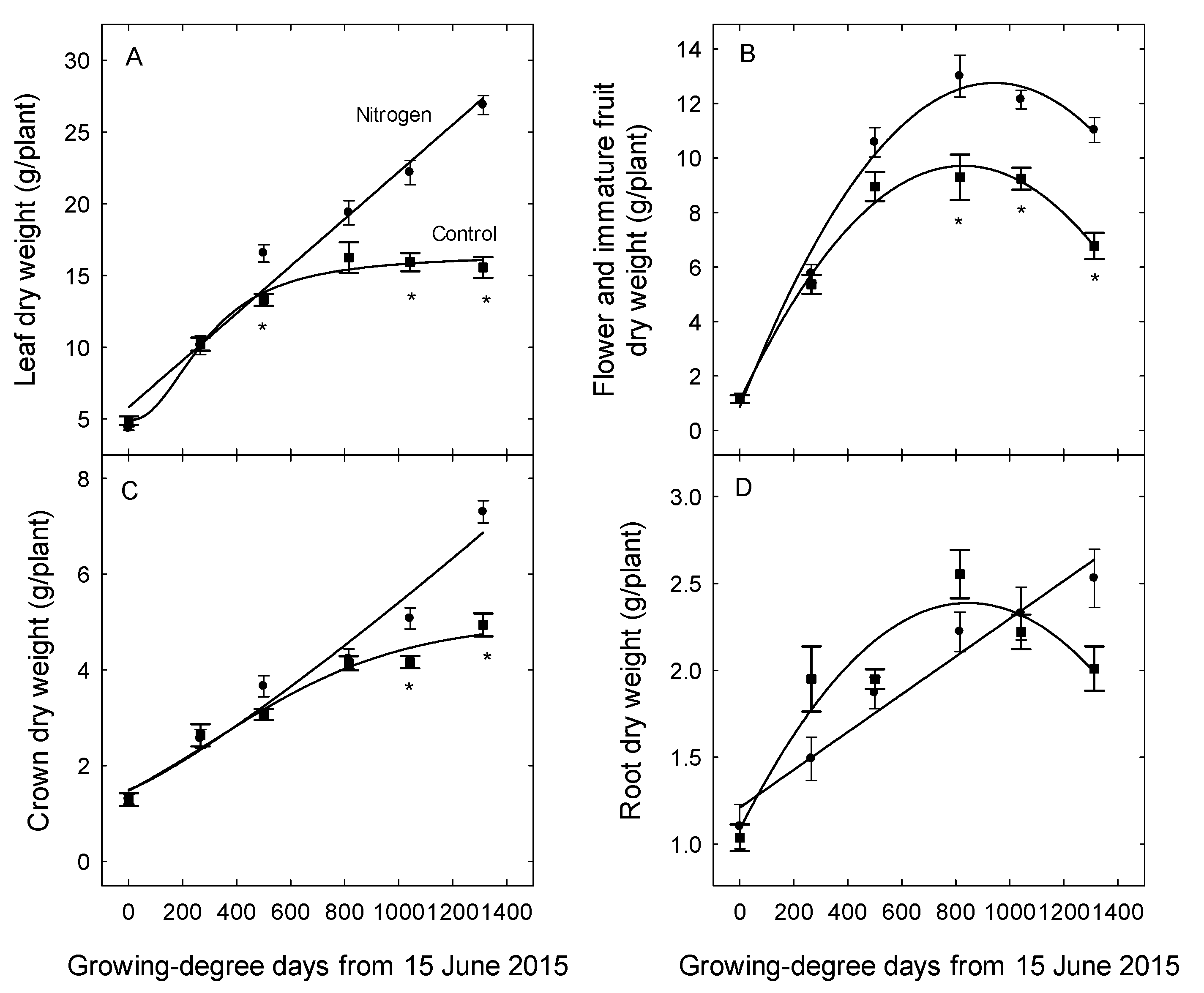
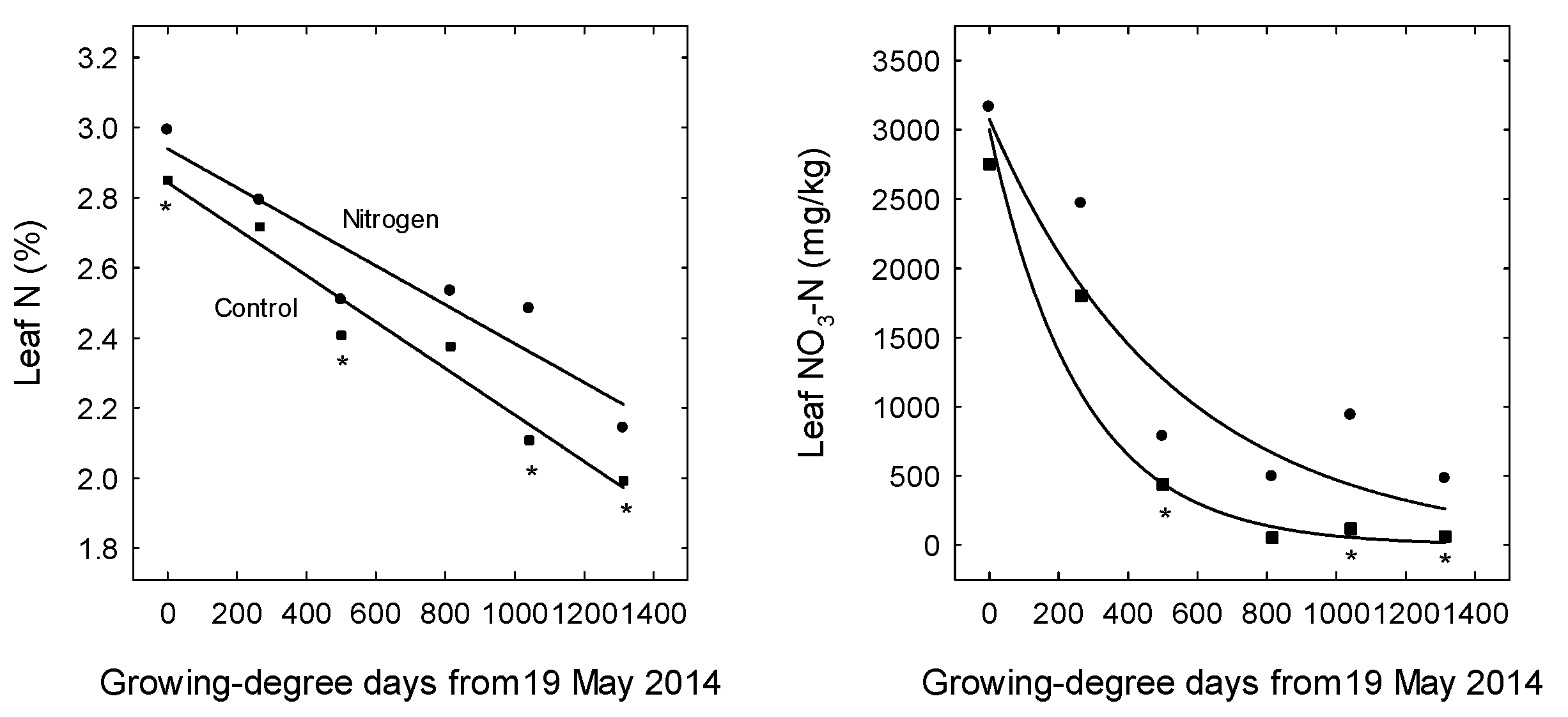
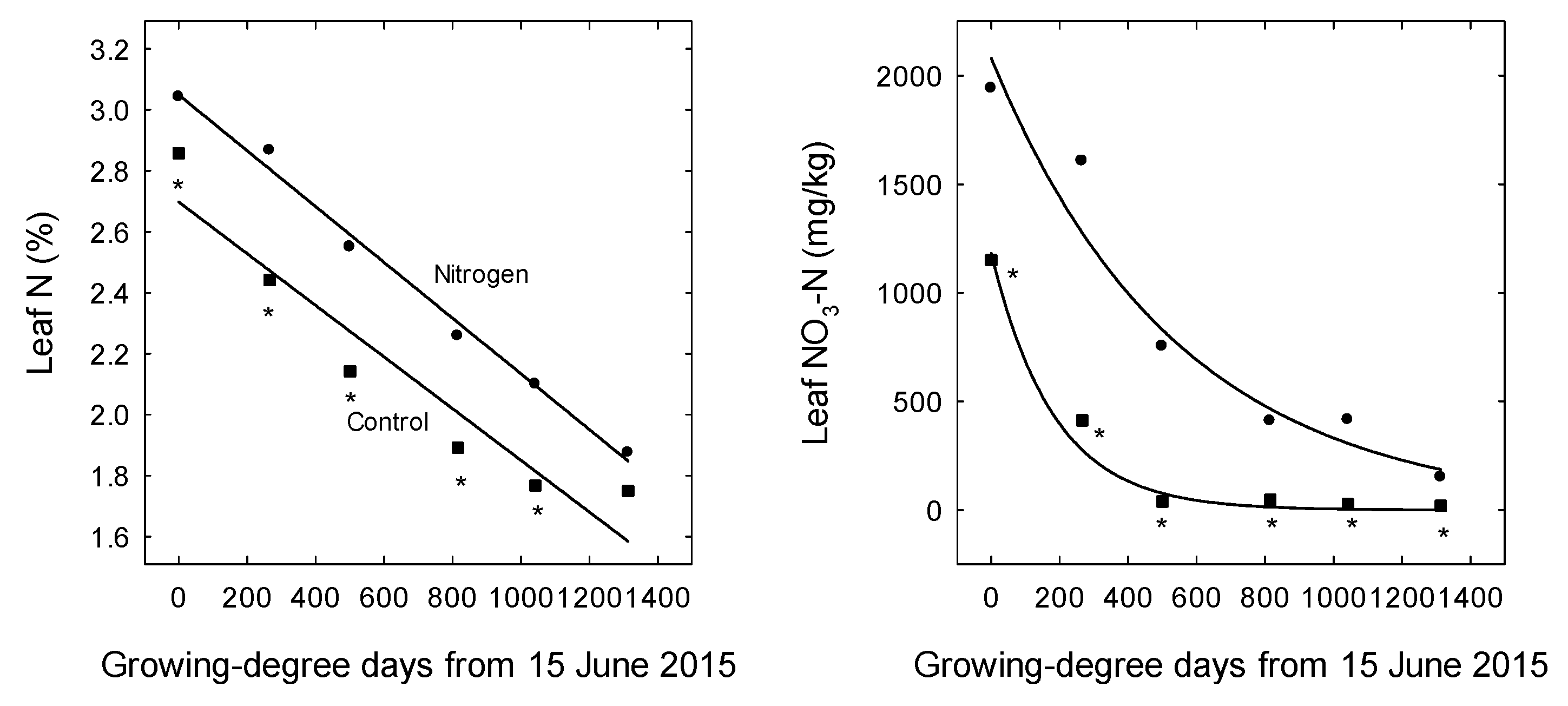
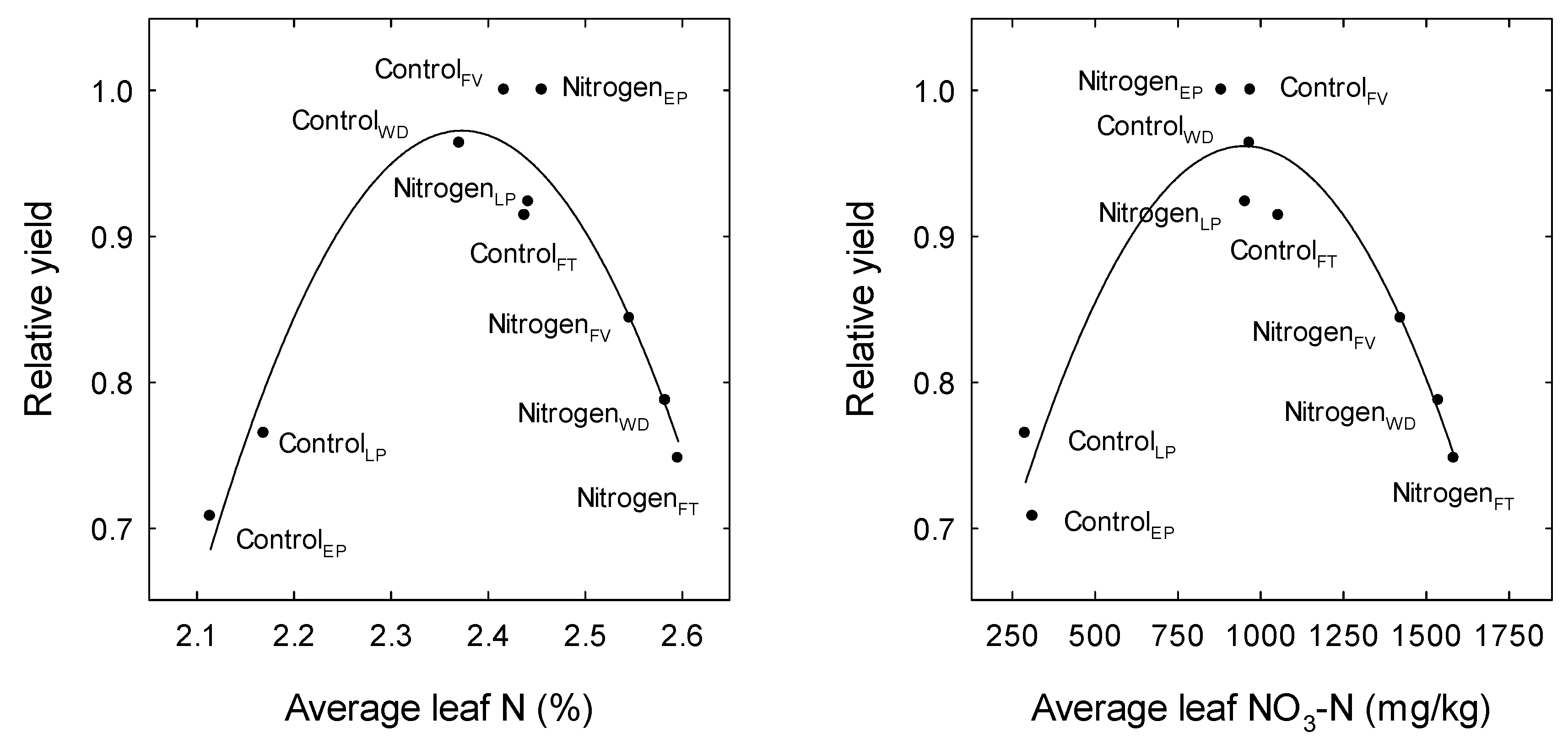
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Husaini, A.M.; Zaki, F.A. Strawberries: A general account. In Strawberry: Growth, Development and Diseases; Husaini, A.M., Neri, D., Eds.; CABI: Wallingford, UK, 2016; pp. 1–9. [Google Scholar]
- Hancock, J.F. Fragaria × ananassa. In The Encyclopedia of Fruit & Nuts; Janick, J., Paull, R.E., Eds.; CABI: Cambridge, MA, USA, 2008; pp. 651–661. [Google Scholar]
- Whitaker, V.M.; Price, J.F.; Peres, N.A.; Folta, K.M.; Noling, J.W. Current strawberry research at the University of Florida. Acta Hortic. 2014, 1049, 161–166. [Google Scholar] [CrossRef]
- Menzel, C.M.; Gomez, A.; Smith, L.A. Control of grey mould and stem-end rot in strawberry plants in a subtropical environment. Australas. Plant Pathol. 2016, 45, 489–498. [Google Scholar] [CrossRef]
- Whitaker, V.M.; Osorio, L.F.; Peres, N.A.; Fan, Z.; Herrington, M.; Nunes, M.C.N.; Plotto, A.; Sims, C.A. ‘Florida Beauty’ strawberry. HortScience 2017, 52, 1443–1447. [Google Scholar] [CrossRef]
- Albregts, E.E.; Howard, C.M.; Chandler, C.K. Effect of high N rates on fruiting strawberry. Soil Crop Sci. Soc. Fla. Proc. 1991, 50, 134–136. [Google Scholar]
- Albregts, E.E.; Howard, C.M.; Chandler, C.K.; Martin, F.G. Fruiting response of strawberry as affected by rates and sources of controlled release N fertilizer, and irrigation method. Soil Crop Sci. Soc. Fla. Proc. 1990, 49, 46–49. [Google Scholar]
- Hochmuth, G.J.; Albregts, E.E.; Chandler, C.K.; Cornell, J.; Harrison, J. Nitrogen fertigation requirements of drip-irrigated strawberries. J. Am. Soc. Hortic. Sci. 1996, 121, 660–665. [Google Scholar]
- Santos, B.M.; Chandler, C.K. Influence of nitrogen fertilization rates on the performance of strawberry cultivars. Int. J. Fruit Sci. 2009, 9, 126–135. [Google Scholar] [CrossRef]
- Miner, G.S.; Poling, E.B.; Carroll, D.E.; Nelson, L.A.; Campbell, C.R. Influence of fall nitrogen and spring nitrogen-potassium applications on yield and fruit quality of ‘Chandler’ strawberry. J. Am. Soc. Hortic. Sci. 1997, 122, 290–295. [Google Scholar]
- Kirschbaum, D.S.; Borquez, A.M.; Quipildor, S.L.; Correa, M.; Magen, H.; Imas, P. Nitrogen requirements of drip irrigated strawberries grown in subtropical environments. Acta Hortic. 2006, 567, 93–96. [Google Scholar] [CrossRef]
- Simonne, E.; Studstill, D.; Hochmuth, R.C. Understanding water movement in mulched beds on sandy soils: An approach to ecologically sound fertigation in vegetable production. Acta Hortic. 2006, 700, 173–178. [Google Scholar] [CrossRef]
- Hochmuth, G.; Maynard, D.; Vavrina, C.; Hanlon, E.; Simonne, E. Plant Tissue Analysis and Interpretation for Vegetable Crops in Florida; HS964; Institute of Food Agricultural Sciences, University of Florida Publishing: Gainesville, FL, USA, 1991; p. 48. [Google Scholar]
- Kwong, S.S.; Boynton, D. Time of sampling, leaf age and leaf fraction as factors influencing the concentrations of nutrient elements in strawberry leaves. Proc. Am. Soc. Hortic. Sci. 1959, 73, 168–173. [Google Scholar]
- Bottoms, T.G.; Bolda, M.P.; Gaskell, M.L.; Hartz, T.K. Determination of strawberry optimum ranges through diagnosis and recommendation integrated system analysis. HortTechnology 2013, 23, 312–318. [Google Scholar]
- Domínquez, A.; Martínez, E.; Trigo, A.; Alonso, D.; García, R. Seasonal changes in leaf mineral content may affect foliar diagnostic in strawberry. Acta Hortic. 2009, 842, 147–150. [Google Scholar]
- Locascio, S.J.; Martin, F.G. Nitrogen source and application timing for trickle irrigated strawberries. J. Am. Soc. Hortic. Sci. 1985, 110, 820–823. [Google Scholar]
- Jones, J.B., Jr.; Wolf, B.; Mill, H.A. Plant Analysis Handbook; Micro-Macro Publishing: Athens, OH, USA, 1991; p. 164. [Google Scholar]
- Ulrich, A.; Mostafa, M.A.E.; Allen, W.W. Strawberry Deficiency Symptoms. A Visual and Plant Analysis Guide to Fertilization; Agriculture Experiment Station University California Bulletin No. 1917; University of California, Division of Agricultural Sciences: Oakland, CA, USA, 1992; p. 42. [Google Scholar]
- Campbell, C.R.; Miner, G.S. Strawberry—Annual hill culture. In Reference Sufficiency Ranges for Plant Analysis in the Southern Region of the United States; Campbell, C.R., Ed.; North Carolina Department Agriculture Consumer Services South Co-operative Services Bulletin No. 394; N.C. Department of Agriculture and Consumer Services: Raleigh, NC, USA, 2000; pp. 111–112. [Google Scholar]
- Dale, A.; Walker, G.; Fisher, P. Growing Strawberries in Ontario; Ministry of Agriculture, Food and Rural Affairs: Toronto, ON, Canada, 2000; p. 17.
- Carroll, J.; Pritts, M.; Heidenreich, C. Production Guide for Organic Strawberries; No. 226; Co-operative Extension Cornell University New York State Integrated Pest Management Publishing: Geneva, NY, USA, 2010; p. 9. [Google Scholar]
- Bellomonte, G.; Costantini, A.; Giammarioli, S. Comparison of modified automatic Dumas method and the traditional Kjeldahl method for nitrogen determination in infant food. J. Assoc. Off. Anal. Chem. 1987, 70, 227–229. [Google Scholar] [PubMed]
- Sweeney, R.A.; Rexroad, P.R. Comparison of LECO FP-228 “nitrogen determinator” with AOAC copper catalyst Kjeldahl method for crude protein. J. Assoc. Off. Anal. Chem. 1987, 70, 1028–1030. [Google Scholar] [PubMed]
- Ott-Borrelli, K.A.; Koenig, R.T.; Miles, C.A. A comparison of rapid potentiometric and colormetric methods for measuring tissue nitrate concentrations in leafy green vegetables. HortTechnology 2009, 19, 439–444. [Google Scholar]
- Diel, M.I.; Pinheiro, M.V.M.; Cocco, C.; Fontana, D.C.; Caron, B.O.; de Paula, G.M.; Pretto, M.M.; Thiesen, L.A.; Schmidt, D. Phyllochron and phenology of strawberry cultivars from different origins cultivated in organic substrates. Sci. Hortic. 2017, 220, 226–232. [Google Scholar] [CrossRef]
- Gu, S. Growing degree hours—A simple, accurate, and precise protocol to approximate growing heat summation for grapevines. Int. J. Biometerol. 2016, 60, 1123–1134. [Google Scholar] [CrossRef] [PubMed]
- McMaster, G.S.; Wilhelm, W.W. Growing degree-days: One equation, two interpretations. Agric. For. Meteorol. 1997, 87, 291–300. [Google Scholar] [CrossRef]
- Rayment, G.E.; Lyons, D.J. Soil Chemical Methods—Australasia; CSIRO Publishing: Collingwood, Australia, 2011; pp. 93–144. [Google Scholar]
- MacKenzie, A.J.; Spencer, W.F.; Stockinger, K.R.; Krantz, B.A. Seasonal nitrate-nitrogen content of cotton petioles as affected by nitrogen application and its relationship to yield. Agron. J. 1963, 55, 55–59. [Google Scholar] [CrossRef]
- Christensen, P. Seasonal changes and distribution of nutritional elements in Thompson Seedless grapevines. Am. J. Vitic. 1969, 20, 176–190. [Google Scholar]
- Vos, J.; Bom, M. Hand-held chlorophyll meter: A promising tool to assess the nitrogen status of potato foliage. Potato Res. 1993, 36, 301–308. [Google Scholar] [CrossRef]
- Opstad, N. Mineral concentrations in leaf dry matter and leaf and petiole sap in strawberry depend on leaf age and plant development stage. Acta Hortic. 2010, 868, 143–148. [Google Scholar] [CrossRef]
- Campbell, W.H. Nitrate reductase and its role in nitrate assimilation in plants. Physiol. Plant. 1988, 74, 214–219. [Google Scholar] [CrossRef]
- Oosterhuis, D.M.; Bate, G.C. Nitrogen uptake of field-grown cotton. II. Nitrate reductase activity and petiole nitrate concentration as indicators of plant nitrogen status. Exp. Agric. 1983, 19, 103–109. [Google Scholar] [CrossRef]
- Taghavi, T.; Folta, K.M. A comparison of wild and cultivated strawberries for nitrogen uptake and reduction. Hortic. Environ. Biotechnol. 2014, 55, 196–206. [Google Scholar] [CrossRef]
- Bottoms, T.G.; Hartz, T.K.; Cahn, M.D.; Farrara, B.F. Crop and soil nitrogen dynamics in annual strawberry production in California. HortScience 2013, 48, 1034–1039. [Google Scholar]
- Strong, W.M.; Mason, M.G. Nitrogen. In Soil Analysis—An Interpretation Manual; Peverill, K.I., Sparrow, L.A., Reuter, D.J., Eds.; CSIRO: Collingwood, Australia, 1999; pp. 171–185. [Google Scholar]
- Glendinning, J.S. Australian Soil Fertility Manual; CSIRO Publishing: Collingwood, Australia, 1999; pp. 19–28. [Google Scholar]
- Niskanen, R.; Dris, R. Nutritional status of strawberry fields. Acta Hortic. 2002, 567, 439–442. [Google Scholar] [CrossRef]
- Bould, C. Soil and leaf analysis in relation to fruit nutrition. J. Sci. Food Agric. 1963, 14, 710–718. [Google Scholar] [CrossRef]
| Yield (g Fresh wt per Plant) | |||
|---|---|---|---|
| Cultivar | Nitrogen | Control | Avg |
| Festival | 766 | 908 | 837 |
| Fortuna | 679 | 830 | 754 |
| Winter Dawn | 715 | 875 | 795 |
| Avg | 720 | 871 | |
| Significance | Nitrogen * | Cultivar ns | N × cultivar ns |
| Yield (g Fresh wt per Plant) | |||
|---|---|---|---|
| Planting Date | Nitrogen | Control | Difference |
| Mid-April | 1123 | 795 | 328 * |
| Late-April | 1037 | 859 | 221 * |
| Difference | 86 * | −64 ns | |
| Significance | Nitrogen * | Planting date ns | N × date * |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Menzel, C.M. Changes in the Concentration of Leaf Nitrogen over the Season Affect the Diagnosis of Deficiency or Sufficiency in Strawberries in the Subtropics. Agriculture 2018, 8, 126. https://doi.org/10.3390/agriculture8080126
Menzel CM. Changes in the Concentration of Leaf Nitrogen over the Season Affect the Diagnosis of Deficiency or Sufficiency in Strawberries in the Subtropics. Agriculture. 2018; 8(8):126. https://doi.org/10.3390/agriculture8080126
Chicago/Turabian StyleMenzel, Christopher M. 2018. "Changes in the Concentration of Leaf Nitrogen over the Season Affect the Diagnosis of Deficiency or Sufficiency in Strawberries in the Subtropics" Agriculture 8, no. 8: 126. https://doi.org/10.3390/agriculture8080126




