Assessing the Impacts of Large-Scale Agro-Industrial Sugarcane Production on Biodiversity: A Case Study of Wonji Shoa Sugar Estate, Ethiopia
Abstract
:1. Introduction
2. Materials and Methods
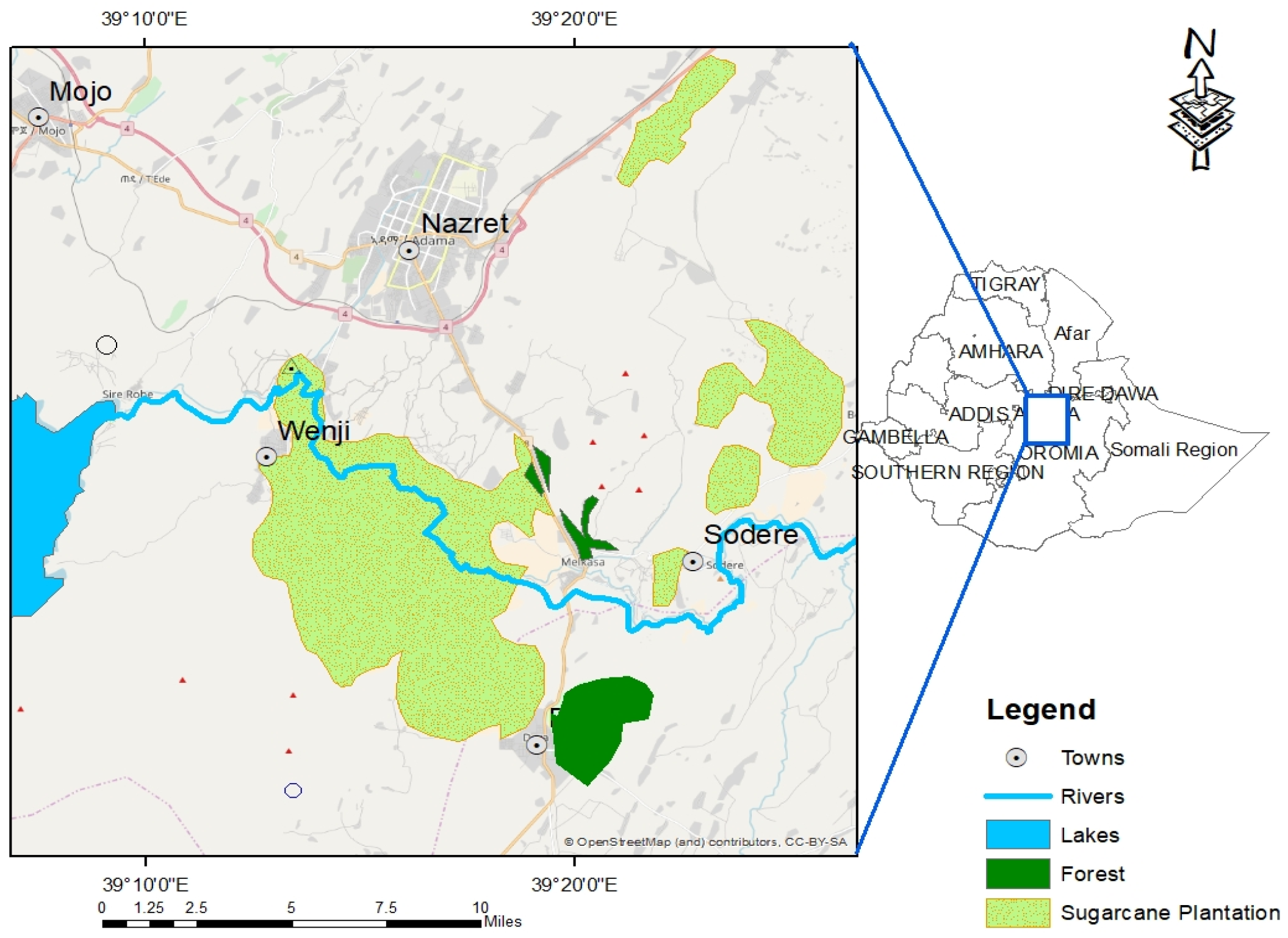
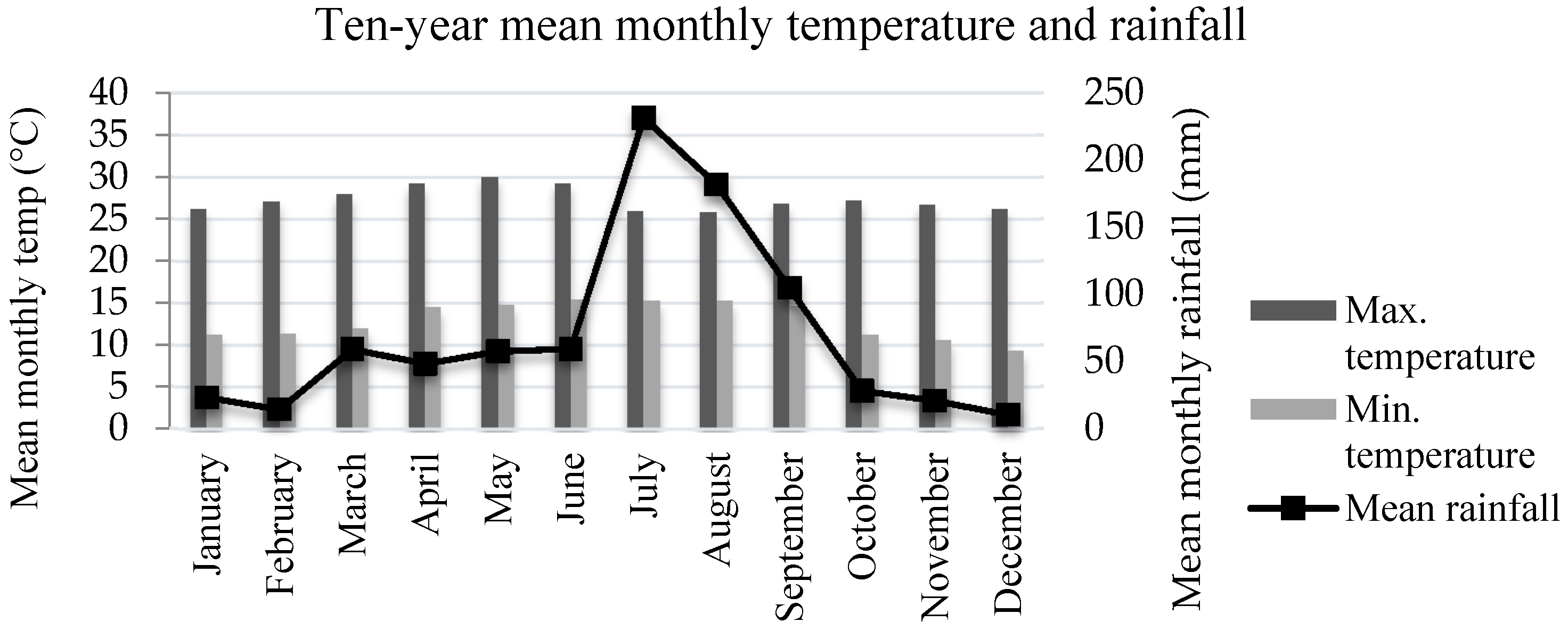
2.1. Study Area
2.2. Study Design and Data Collection
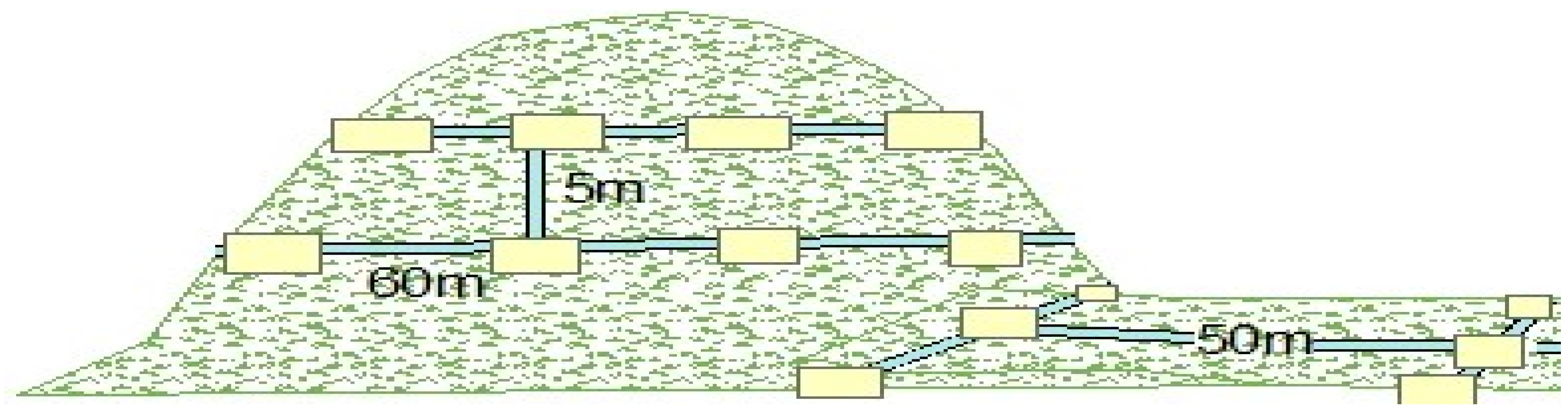
Woody Species Census Plots
2.3. Risk Score Assessment Framework
2.4. Floristic Data Analysis
3. Results
3.1. Floristic Composition and Diversity
3.1.1. Important Value Index
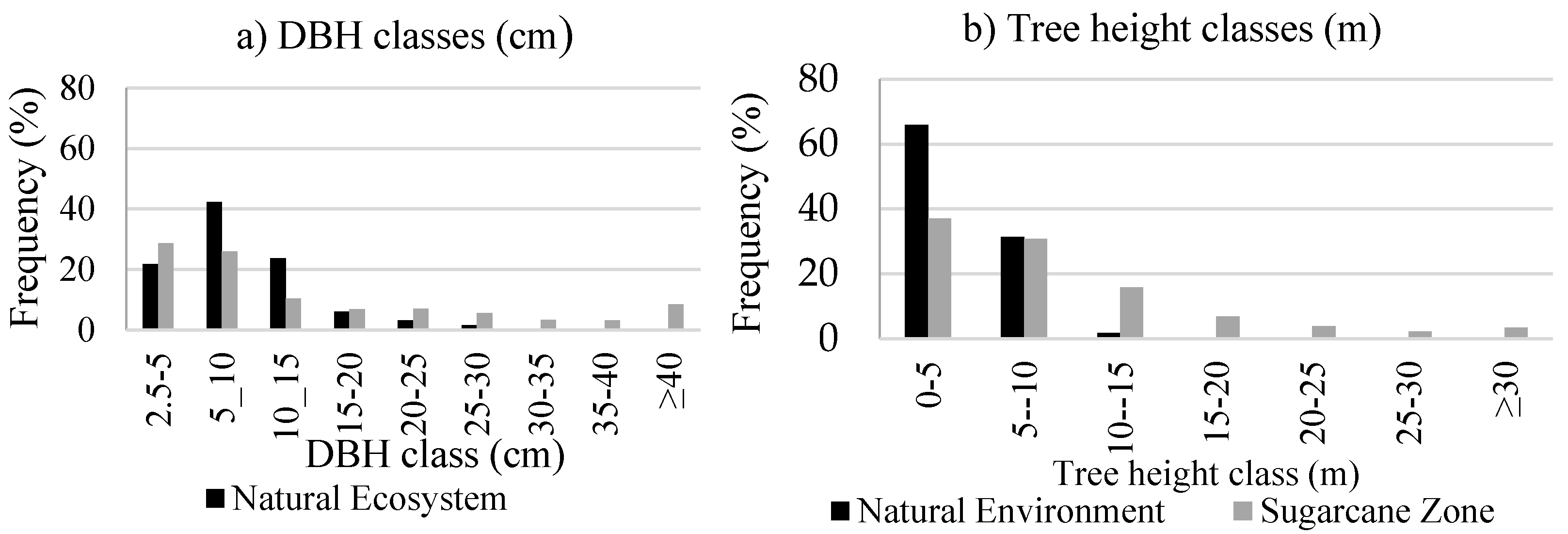
3.1.2. Vegetation Structure
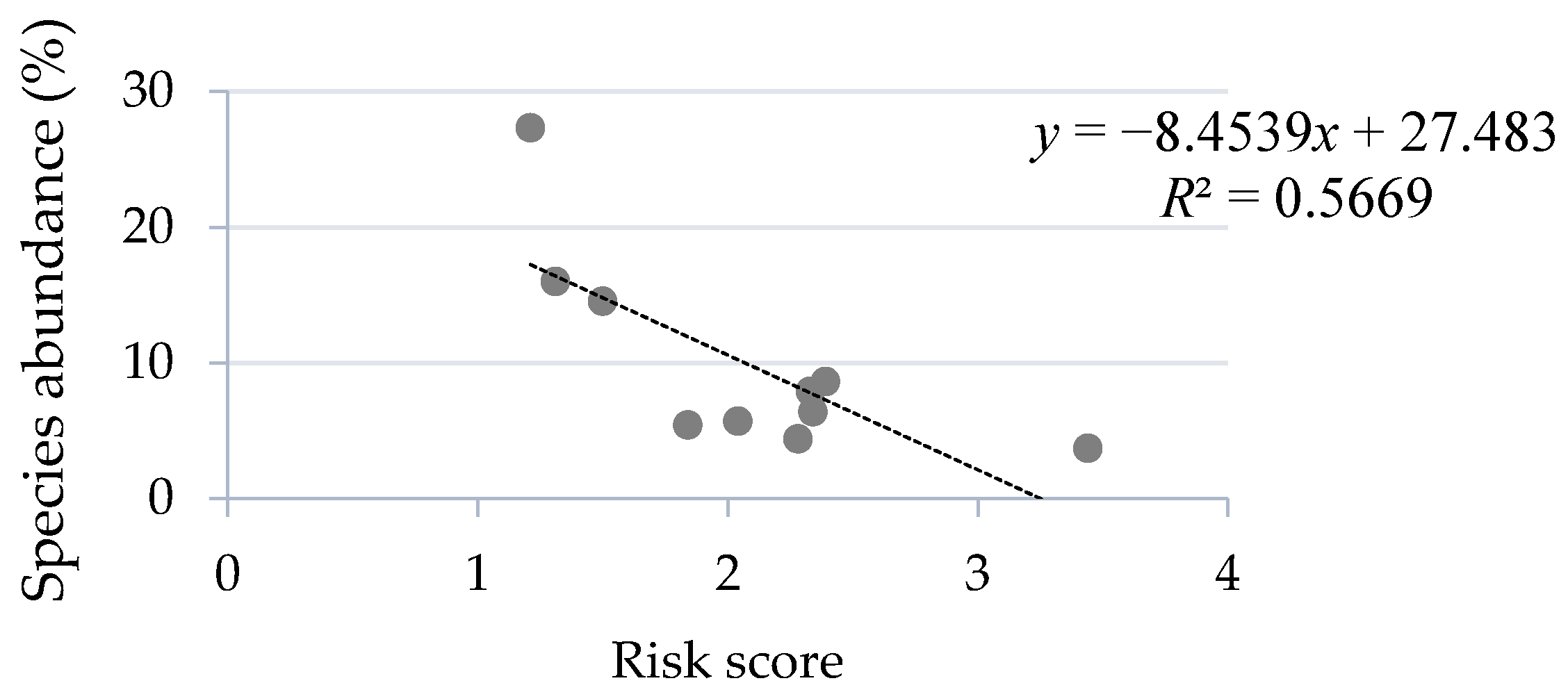
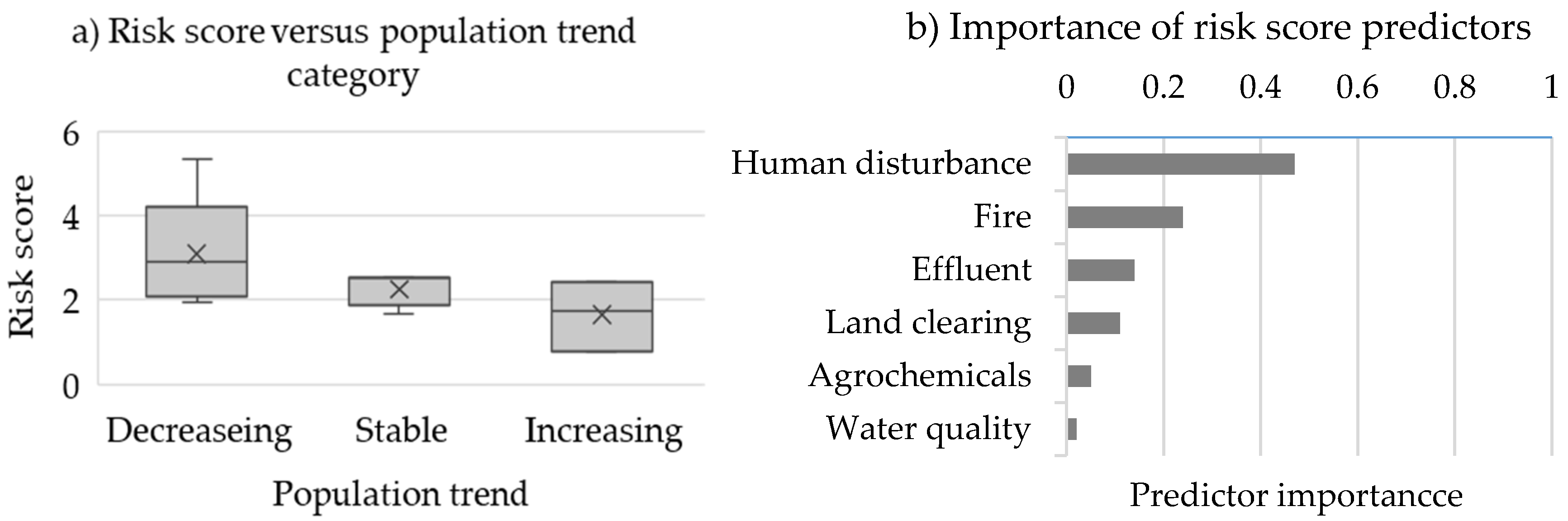
3.2. Rodent Abundance and Risk Scores
3.3. Mammal and Bird Risk Scores
4. Discussion
4.1. Floristic Composition, Richness, and Diversity
4.2. Risk Score Assessment
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Stuart, S.N.; Adams, R.J. Biodiversity in Sub-Saharan Africa and Its Islands: Conservation, Management, and Sustainable Use; No. 6; Occasional Papers of the IUCN Species Survival Commission: Gland, Switzerland, 1990. [Google Scholar]
- Board, M.E. Ecosystems and Human Well-Being, Biodiversity Synthesis: A Report of the Millennium Ecosystem Assessment; World Resources Institute: Washington, DC, USA, 2005. [Google Scholar]
- DeGeorges, P.A.; Reilly, B.K. The realities of community based natural resource management and biodiversity conservation in Sub-Saharan Africa. Sustainability 2009, 1, 734–788. [Google Scholar] [CrossRef] [Green Version]
- Darbyshire, I.; Lamb, H.; Umer, M. Forest clearance and regrowth in northern Ethiopia during the last 3000 years. Holocene 2003, 13, 537–546. [Google Scholar] [CrossRef]
- Pistorius, T.; Carodenuto, S.; Wathum, G. Implementing Forest Landscape Restoration in Ethiopia. Forests 2017, 8, 61. [Google Scholar] [CrossRef]
- Webb, A.; Coates, D. Biofuels and Biodiversity; Technical Series (No. 65); Secretariat of the Convention on Biological Diversity: Montreal, QC, Canada, 2012. [Google Scholar]
- Schoneveld, G.C. The Challenge of Governing Africa’s New Agricultural Investment Landscapes: An Analysis of Policy Arrangements and Sustainability Outcomes in Ethiopia and Nigeria. Forests 2014, 6, 88–115. [Google Scholar] [CrossRef]
- Hess, T.M.; Sumberg, J.; Biggs, T.; Georgescu, M.; Haro-Monteagudo, D.; Jewitt, G.; Ozdogan, M.; Marshall, M.; Thenkabail, P.; Daccache, A.; et al. A sweet deal? Sugarcane, water and agricultural transformation in Sub-Saharan Africa. Glob. Environ. Chang. 2016, 39, 181–194. [Google Scholar] [CrossRef] [Green Version]
- Moreda, T. Listening to their silence? The political reaction of affected communities to large-scale land acquisitions: Insights from Ethiopia. J. Peasant Stud. 2015, 42, 517–539. [Google Scholar] [CrossRef]
- Lavers, T. Patterns of agrarian transformation in Ethiopia: State-mediated commercialisation and the ‘land grab’. J. Peasant Stud. 2012, 39, 795–822. [Google Scholar] [CrossRef]
- Teklemariam, D.; Azadi, H.; Nyssen, J.; Haile, M.; Witlox, F. How Sustainable Is Transnational Farmland Acquisition in Ethiopia? Lessons Learned from the Benishangul-Gumuz Region. Sustainability 2016, 8, 213. [Google Scholar] [CrossRef] [Green Version]
- Wendimu, M.A.; Henningsen, A.; Gibbon, P. Sugarcane Out-growers in Ethiopia: “Forced” to Remain Poor? World Dev. 2016, 83, 84–97. [Google Scholar] [CrossRef]
- Hertel, T.W.; Tyner, W.E.; Birur, D.K. The global impacts of biofuel mandates. Energy J. 2010, 75–100. [Google Scholar] [CrossRef]
- Lynd, L.R.; Sow, M.; Chimphango, A.F.; Cortez, L.A.; Cruz, C.H.B.; Elmissiry, M.; Laser, M.; Mayaki, I.A.; Moraes, M.A.; Nogueira, L.A.; et al. Bioenergy and African transformation. Biotechnol. Biofuels 2015, 8, 18. [Google Scholar] [CrossRef] [PubMed]
- Muniz Kubota, A.; Dal Belo Leite, J.G.; Watanabe, M.; Cavalett, O.; Leal, M.R.; Cortez, L. The Role of Small-Scale Biofuel Production in Brazil: Lessons for Developing Countries. Agriculture 2017, 7, 61. [Google Scholar] [CrossRef]
- Fitzherbert, E.B.; Struebig, M.J.; Morel, A.; Danielsen, F.; Brühl, C.A.; Donald, P.F.; Phalan, B. How will oil palm expansion affect biodiversity? Trends Ecol. Evol. 2008, 23, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Skutsch, M.; Masera, O.; Pacheco, P. A Global Analysis of Deforestation Due to Biofuel Development; CIFOR: Bogor, Indonesia, 2011. [Google Scholar]
- Lapola, D.M.; Schaldach, R.; Alcamo, J.; Bondeau, A.; Koch, J.; Koelking, C.; Priess, J.A. Indirect land-use changes can overcome carbon savings from biofuels in Brazil. Proc. Natl. Acad. Sci. USA 2010, 107, 3388–3393. [Google Scholar] [CrossRef] [PubMed]
- Nassar, A.M.; Moreira, M. Evidences on Sugarcane Expansion and Agricultural Land Use Changes in Brazil; Institute for the International Trade Negotiation: Brighton, UK, 2013. [Google Scholar]
- Dale, V.H.; Keith, L.; Wiens, K.J.; Fargione, J. Biofuels: Implications for Land Use and Biodiversity; Ecological Society of America: Washington, DC, USA, 2010. [Google Scholar]
- Groom, M.J.; Gray, E.M.; Townsend, P.A. Biofuels and biodiversity: Principles for creating better policies for biofuel production. Conserv. Biol. 2008, 22, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Fargione, J.; Hill, J.; Tilman, D.; Polasky, S.; Hawthorne, P. Land clearing and the biofuel carbon debt. Science 2008, 319, 1235–1238. [Google Scholar] [CrossRef] [PubMed]
- Gheler-Costa, C.; Sabino-Santos, J.G.; Amorim, L.S.; Rosalino, L.M.; Figueiredo, L.T.M.; Verdade, L.M. The effect of preharvest fire on the small mammal assemblage in sugarcane fields. Agric. Ecosyst. Environ. 2013, 171, 85–89. [Google Scholar] [CrossRef]
- Gerbens-Leenes, W.; Hoekstra, A.Y.; van der Meer, T.H. The water footprint of bioenergy. Proc. Natl. Acad. Sci. USA 2009, 106, 10219–10223. [Google Scholar] [CrossRef] [PubMed]
- Scholten, W. The Water Footprint of Sugar and Sugar-Based Ethanol. Master’s Thesis, University of Twente, Enschede, The Netherlands, April 2009. [Google Scholar]
- Berndes, G. Bioenergy and water-the implications of large-scale bioenergy production for water use and supply. Glob. Environ. Chang. 2002, 12, 253–271. [Google Scholar] [CrossRef]
- Berger, M.; Finkbeiner, M. Water footprinting: How to address water use in life cycle assessment? Sustainability 2010, 2, 919–944. [Google Scholar] [CrossRef]
- Keam, S.; McCormick, N.; Howard, G.; Athanas, A. Guidelines on Biofuels and Invasive Species: IUCN 14; IUCN: Nairobi, Kenya, 2009. [Google Scholar]
- Robertson, G.P.; Hamilton, S.K.; Del Grosso, S.J.; Parton, W.J. The biogeochemistry of bioenergy landscapes: Carbon, nitrogen, and water considerations. Ecol. Appl. 2011, 21, 1055–1067. [Google Scholar] [CrossRef] [PubMed]
- Afework, D.; Tessema, T.; Yirefu, F. Efficacy of Integrated Water Hyacinth Management Strategies at Wonji-Shoa Sugar Factory. Ethiop. J. Weed Manag. 2008, 2, 57–70. [Google Scholar]
- Kloos, H. Development, drought, and famine in the Awash Valley of Ethiopia. Afr. Stud. Rev. 1982, 25, 21–48. [Google Scholar] [CrossRef]
- Barbour, M.G.; Burk, J.H.; Pitts, W.D. Terrestrial Plant Ecology, 3rd ed.; Cummings Publishing Co.: Menlo Park, CA, USA, 1987. [Google Scholar]
- Takele, S.; Bekele, A.; Belay, G.; Balakrishnan, M. A comparison of rodent and insectivore communities between sugarcane plantation and natural habitat in Ethiopia. Trop. Ecol. 2011, 52, 61–68. [Google Scholar]
- Butler, S.J.; Vickery, J.A.; Norris, K. Farmland biodiversity and the footprint of agriculture. Science 2007, 315, 381–384. [Google Scholar] [CrossRef] [PubMed]
- Butler, S.J.; Brooks, D.; Feber, R.E.; Storkey, J.; Vickery, J.A.; Norris, K. A cross-taxonomic index for quantifying the health of farmland biodiversity. J. Appl. Ecol. 2009, 46, 1154–1162. [Google Scholar] [CrossRef]
- Curtis, J.T.; McIntosh, R.P. The interrelations of certain analytic and synthetic phytosociological characters. Ecology 1950, 31, 434–455. [Google Scholar] [CrossRef]
- Mekonnen, M.; Gebrehiwot, K.; Birhane, E.; Tewoldeberhan, S. Regeneration, Density and Diversity of Woody Vegetation in Awash National Park, Ethiopia. J. Drylands 2009, 2, 101–109. [Google Scholar]
- Aynekulu, E.; Aerts, R.; Denich, M.; Negussie, A.; Friis, I.; Demissew, S.; Boehmer, H.J. Plant diversity and regeneration in a disturbed isolated dry Afromontane forest in northern Ethiopia. Folia Geobot. 2016, 51, 115–127. [Google Scholar] [CrossRef]
- Senbeta, F.; Teketay, D. Regeneration of indigenous woody species under the canopies of tree plantations in Central Ethiopia. Trop. Ecol. 2001, 42, 175–185. [Google Scholar]
- Tilman, D.; Lehman, C. Human-caused environmental change: Impacts on plant diversity and evolution. Proc. Natl. Acad. Sci. USA 2001, 98, 5433–5440. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, R.H. Evolution and measurement of species diversity. Taxon 1972, 21, 213–251. [Google Scholar] [CrossRef]
- Strong, D.R., Jr.; Simberloff, D.; Abele, L.G.; Thistle, A.B. Ecological Communities: Conceptual Issues and the Evidence; Princeton University Press: Princeton, NJ, USA, 2014. [Google Scholar]
- Mugwedi, L.F.; Rouget, M.; Egoh, B.; Ramdhani, S.; Slotow, R.; Rentería, J.L. An Assessment of a Community-Based, Forest Restoration Programme in Durban (eThekwini), South Africa. Forests 2017, 8, 255. [Google Scholar] [CrossRef]
- Shibru, S.; Balcha, G. Composition, Structure and regeneration status of woody species in Dindin Natural Forest, Southeast Ethiopia: An implication for conservation. Ethiop. J. Biol. Sci. 2004, 1, 15–35. [Google Scholar]
- Endale, Y.; Derero, A.; Argaw, M.; Muthuri, C. Farmland tree species diversity and spatial distribution pattern in semi-arid East Shewa, Ethiopia. For. Trees Livelihoods 2017, 26, 199–214. [Google Scholar] [CrossRef]
- Senbeta, F.; Denich, M. Effects of wild coffee management on species diversity in the Afromontane rainforests of Ethiopia. For. Ecol. Manag. 2006, 232, 68–74. [Google Scholar] [CrossRef]
- Yineger, H.; Kelbessa, E.; Bekele, T.; Lulekal, E. Floristic composition and structure of the dry afromontane forest at Bale Mountains National Park, Ethiopia. SINET Ethiop. J. Sci. 2008, 31, 103–120. [Google Scholar] [CrossRef]
- Takele, S.; Bekele, A.; Belay, G.; Balakrishnan, M. Pest status of rodents in Wonji sugarcane plantation. Ethiop. Int. J. Ecol. Environ. Sci. 2008, 34, 157–163. [Google Scholar]
- Takele, S. Abundance, Composition and Habitat Association of Rodents in Wonji Shoa Area, Ethiopia. Master’s Thesis, Addis Ababa University, Addis Ababa, Ethiopia, June 2006. [Google Scholar]
- Savario, C.F.; Hoy, J.W. Microbial communities in sugarcane field soils with and without a sugarcane cropping history. Plant Soil 2011, 341, 63–73. [Google Scholar] [CrossRef]
- Wallis, P.D.; Haynes, R.J.; Hunter, C.H.; Morris, C.D. Effect of land use and management on soil bacterial biodiversity as measured by PCR-DGGE. Appl. Soil Ecol. 2010, 46, 147–150. [Google Scholar] [CrossRef]
- Sajjad, A.; Ahmad, F.; Makhdoom, A.H.; Imran, A. Does trash burning harm arthropod biodiversity in sugarcane? Int. J. Agric. Biol. 2012, 14, 1021–1023. [Google Scholar]
- Srikanth, J.; Easwaramoorthy, S.; Kurup, N.K.; Santhalakshmi, G. Spider abundance in sugarcane: Impact of cultural practices, irrigation and post-harvest trash burning. Biol. Agric. Hortic. 1997, 14, 343–356. [Google Scholar] [CrossRef]
- Adeba, D.; Kansal, M.L.; Sen, S. Assessment of water scarcity and its impacts on sustainable development in Awash basin, Ethiopia. Sustain. Water Resour. Manag. 2015, 1, 71–87. [Google Scholar] [CrossRef]
- Lyra-Jorge, M.C.; Ciocheti, G.; Pivello, V.R. Carnivore mammals in a fragmented landscape in northeast of São Paulo State, Brazil. Biodivers. Conserv. 2008, 17, 1573. [Google Scholar] [CrossRef]


| Diversity Index | Sugarcane Zone | Natural Ecosystem | t-Value | p Value (p ≤ 0.05) |
|---|---|---|---|---|
| Species richness | 30 | 38 | ||
| Dominance (D) | 0.54 ± 0.24 | 0.36 ± 0.17 | 5.03 | 0.0001 |
| Simpson (1-D) | 0.45 ± 0.24 | 0.63 ± 0.17 | 5.03 | 0.0001 |
| Shannon (H) | 0.83 ± 0.50 | 1.30 ± 0.43 | 5.67 | 0.0000 |
| Evenness (e^H/S) | 0.77 ± 0.17 | 0.72 ± 0.16 | 1.25 | 0.2120 |
| Equitability (J) | 0.73 ± 0.21 | 0.77 ± 0.17 | 1.98 | 0.0900 |
| Fisher alpha | 1.42 ± 0.92 | 2.00 ± 1.10 | −3.54 | 0.0014 |
| Berger–Parker | 0.64 ± 0.22 | 0.48 ± 0.19 | 1.96 | 0.0000 |
| Whittaker’s Beta diversity | 7.68 | 6.60 | ||
| Jaccard’s Index | 0.38 | |||
| Sorensen’s Index | 0.35 | |||
| Natural Ecosystem Species | RF % | RD % | RBA % | IVI | Sugarcane-Zone Species | RF % | RD % | RBA % | IVI |
|---|---|---|---|---|---|---|---|---|---|
| Acacia nilotica | 18.52 | 27.64 | 18.89 | 65.05 | Casuarina equisetifolia * | 15.92 | 14.66 | 17.18 | 47.76 |
| Acacia etbaica | 11.63 | 27.31 | 13.41 | 52.35 | Eucalyptus globulus * | 9.95 | 20.11 | 15.53 | 45.59 |
| Acacia tortilis | 6.89 | 4.85 | 33.79 | 45.53 | Sesbania grandiflora * | 11.94 | 21.79 | 8.45 | 42.18 |
| Balanites aegyptiaca | 12.92 | 7.53 | 6.95 | 27.40 | Ficus sycomorus | 1.49 | 0.49 | 36.80 | 38.79 |
| Acacia Senegal | 10.34 | 10.15 | 6.14 | 26.63 | Grevilea robusta * | 7.46 | 5.59 | 4.67 | 17.72 |
| Dichrostachys cineera | 9.91 | 8.45 | 2.94 | 21.30 | Jacaranda mimesifolia * | 7.46 | 8.38 | 0.50 | 16.34 |
| Eucalyptus globulus * | 1.29 | 2.55 | 8.27 | 12.12 | Acacia seyal | 5.97 | 6.22 | 0.64 | 12.82 |
| Acacia oerfota | 2.58 | 1.96 | 0.60 | 5.14 | Delonix rigia * | 2.98 | 2.79 | 3.66 | 9.43 |
| Acacia mellifera | 2.58 | 0.85 | 0.52 | 3.96 | Ehretia cymosa | 4.97 | 2.72 | 0.20 | 7.90 |
| Maerua angolensis | 0.86 | 0.85 | 1.86 | 3.57 | Spathodea nilotica * | 2.49 | 2.37 | 2.99 | 7.85 |
| Rhus natalensis | 2.15 | 1.18 | 0.08 | 3.41 | Millettia ferruginea | 3.98 | 2.30 | 0.28 | 6.56 |
| Acokanthera schimperi | 1.72 | 0.85 | 0.54 | 3.12 | Acacia senegal | 2.49 | 0.77 | 2.43 | 5.69 |
| Acacia seyal | 0.86 | 1.77 | 0.44 | 3.07 | Cupress lustanica * | 1.99 | 1.82 | 1.26 | 5.07 |
| Calpurnia aurea | 2.15 | 0.79 | 0.12 | 3.06 | Cordia africana | 2.98 | 1.89 | 0.04 | 4.91 |
| Ehertia cymosa | 2.15 | 0.72 | 0.16 | 3.04 | Albizia gummifera | 2.98 | 1.19 | 0.44 | 4.61 |
| Ziziphus mauritiana | 0.86 | 1.18 | 0.92 | 2.96 | Schinus molle * | 1.49 | 0.77 | 0.64 | 2.90 |
| Pappea capensis | 0.86 | 0.52 | 1.17 | 2.55 | Mangofera indica ** | 0.99 | 0.63 | 1.16 | 2.78 |
| Terminalia brownie | 0.86 | 0.72 | 0.74 | 2.32 | Carica papaya ** | 0.50 | 1.89 | 0.20 | 2.58 |
| Commiphora rostrata | 0.86 | 1.18 | 0.21 | 2.25 | Juniperus procera | 0.99 | 0.49 | 1.08 | 2.56 |
| Cadaba farinose | 1.29 | 0.85 | 0.10 | 2.24 | Balanites aegyptiaca | 1.49 | 0.56 | 0.40 | 2.45 |
| Cordia ovalis | 0.86 | 0.79 | 0.58 | 2.23 | Podocarpus falcatus | 1.49 | 0.42 | 0.54 | 2.45 |
| Croton dichogamous | 1.29 | 0.79 | 0.07 | 2.15 | Cajanus cajan | 1.49 | 0.70 | 0.05 | 2.24 |
| Capparis fascicularis | 1.72 | 0.33 | 0.05 | 2.10 | Croton macrostachyus | 0.99 | 0.63 | 0.04 | 1.66 |
| Grewia tenax | 1.29 | 0.72 | 0.04 | 2.05 | Phonix reclanata | 0.99 | 0.14 | 0.33 | 1.46 |
| Cordia africana | 0.86 | 1.11 | 0.06 | 2.04 | Acacia nilotica | 0.99 | 0.14 | 0.32 | 1.46 |
| Syzygium guineensis | 0.86 | 0.72 | 0.44 | 2.02 | Dichrostachys cineera | 0.99 | 0.14 | 0.07 | 1.20 |
| Dodonea angustifolia | 0.86 | 0.92 | 0.07 | 1.85 | Capparis tomentosa | 0.99 | 0.14 | 0.02 | 1.15 |
| Croton macrostachyus | 0.86 | 0.79 | 0.16 | 1.81 | Psidium guajava | 0.50 | 0.14 | 0.03 | 0.67 |
| Myrsine africana | 0.86 | 0.85 | 0.04 | 1.75 | Acacia abyssinica | 0.50 | 0.07 | 0.04 | 0.61 |
| Fleuggea virosa | 0.86 | 0.79 | 0.05 | 1.69 | Caesalipinia dectaptela | 0.50 | 0.07 | 0.02 | 0.59 |
| Maytenus senegalensis | 0.86 | 0.72 | 0.08 | 1.66 | |||||
| Premna resinosa | 0.86 | 0.72 | 0.07 | 1.66 | |||||
| Capparis tomentosa | 0.86 | 0.65 | 0.12 | 1.63 | |||||
| Caesalipinia dectaptela | 0.86 | 0.65 | 0.03 | 1.55 | |||||
| Asparagus africanus | 0.86 | 0.59 | 0.00 | 1.45 | |||||
| Ficus sycomorus | 0.86 | 0.33 | 0.23 | 1.42 | |||||
| Grewia villosa | 0.86 | 0.46 | 0.02 | 1.34 | |||||
| Grewia bicolor | 0.86 | 0.39 | 0.02 | 1.28 |
| Species Name | Risk Score | Abundance (%) |
|---|---|---|
| Mastomys natalensis | 1.21 | 27.33 |
| Arvicanthis dembeensis | 1.31 | 16.00 |
| Arvicanthis niloticus | 2.28 | 4.40 |
| Stenocephalemys albipes | 1.50 | 14.53 |
| Pelomys harringtoni | 2.39 | 8.62 |
| Mus mahomet Rhoads | 2.33 | 7.90 |
| Mus musculus Linnaeus | 1.84 | 5.42 |
| Rattus rattus Linnaeus | 3.44 | 3.70 |
| Crocidura flavescens | 2.34 | 6.40 |
| Crocidura fumosa | 2.04 | 5.70 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Degefa, S.; Saito, O. Assessing the Impacts of Large-Scale Agro-Industrial Sugarcane Production on Biodiversity: A Case Study of Wonji Shoa Sugar Estate, Ethiopia. Agriculture 2017, 7, 99. https://doi.org/10.3390/agriculture7120099
Degefa S, Saito O. Assessing the Impacts of Large-Scale Agro-Industrial Sugarcane Production on Biodiversity: A Case Study of Wonji Shoa Sugar Estate, Ethiopia. Agriculture. 2017; 7(12):99. https://doi.org/10.3390/agriculture7120099
Chicago/Turabian StyleDegefa, Sileshi, and Osamu Saito. 2017. "Assessing the Impacts of Large-Scale Agro-Industrial Sugarcane Production on Biodiversity: A Case Study of Wonji Shoa Sugar Estate, Ethiopia" Agriculture 7, no. 12: 99. https://doi.org/10.3390/agriculture7120099






