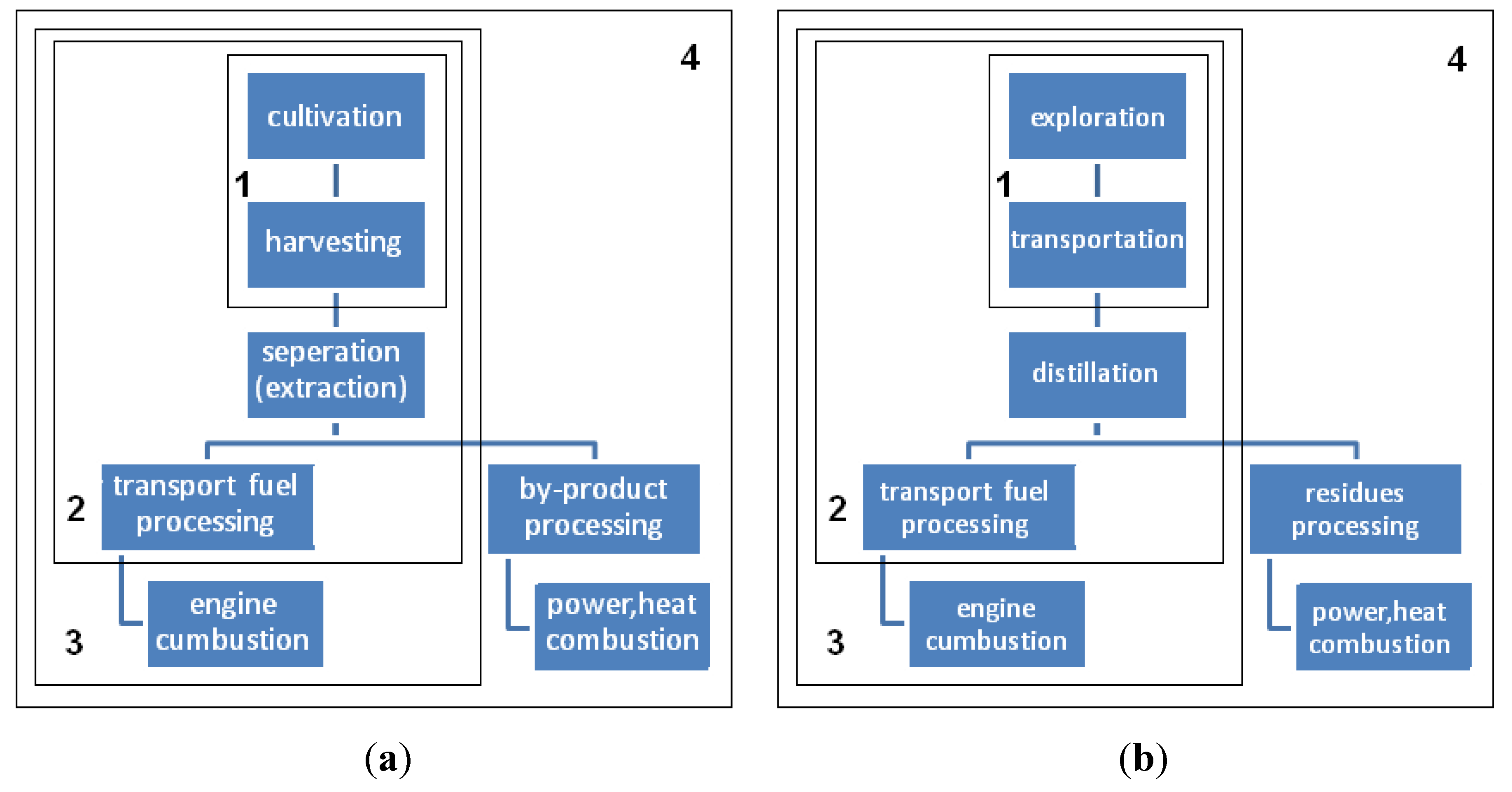
The industrial production of
JCL is a fairly recent development. Almost 0.9 M·ha, 0.765 M·ha (0.32 M·ha in Senegal), and 0.12 M·ha of
JCL farm have been established to date in Asia, Africa, and Latin America, respectively, but it is still far from the targets of 5 M·ha by 2010 and 13 M·ha to be achieved by 2015 [
5]. Meanwhile, the prices of
JCL seed have increased from 0.10 $/kg in 2005 to 0.34 $/kg in 2011 [
4].
3.1. Fertilizer and Watering in JCL Plantation
In Thailand, a demonstration of
JCL plantation was conducted by Kasetsart University, in which annual crop cutting was set within an area of 1 ha (hectare) and a crop density of 2 m × 1 m or two trees per m
2 was utilized. Land preparation was comprised of plowing, harrowing, and a furrowing process using a tractor with an engine of 75 hp to adjust the soil condition for the new cutting set; ternary (N–P–K: 15–15–15) fertilizer was applied with a rate of 650 kg/ha per year; weedicides and insecticides were also used for the general maintenance of the plantation; the pumping rate was 4.5 m
3/m
2 per year for watering and manual harvesting [
6].
In India, the yield of
JCL increased from 1.5 t/ha (rain-fed) to 5.9 t/ha (irrigated) when double fertilizers and an additional 105 kg/ha diesel were used in the irrigation mode as compared with the rain-fed mode [
7].
In West Africa (Mali and Ivory Coast),
JCL planting was up to 1500 ha in 2007, and at least 2000 ha more in 2008. Two 5-ha experimental fields with plantation densities of 1111 plants·ha
−1 were selected with contrasting soil conditions. Ternary fertilizer was only applied during the first three years: 100 kg/ha in the 1st year, 150 kg/ha in the 2nd year, 200 kg/ha in the 3rd year, whereas both 248 kg/ha ternary fertilizer and 201 kg/ha of ammonium nitrate were applied in the 4th year [
6].
3.2. Productivities of Biomass, JME and By-Products
The yield hypotheses had a significant impact on the GHG and energy balances of JME (Jatropha Methyl Ester). The weight of each fresh fruit and seed was around 10–15 g and 2–4 g, respectively. The annual yield of
JCL fresh fruit was about 16 t/ha. The yield of seed was widely spread from 0.1 to10 t/ha [
8] and an increase of 1 t/ha of seeds resulted in a 10% reduction in fossil energy use compared to the baseline value of 4 t/ha. An increase of 1 t/ha of seed production from the baseline of 4 t/ha resulted in a 10% reduction in fossil energy usage. Thus, it appears critical to pursue large-scale field cultivation experiments of JCL.
The yield of co-products such as wood, leaves, and seed shells, are 4 t, 2 t, and 0.8 t (dry weight), respectively from the process of
JCL plantation. Press cake (91.5 kg, dry weight) is obtained when 1 FU (1 GJ of JME) is produced [
9]. All of them can be used as combustion fuels to produce heat or power to replace part of fossil fuel energy used in the JME production while the biodiesel production is developed for commercialization.
Sunil K
et al. [
7] reported details of inventory requirements for the farming, oil extraction, biodiesel production, and transportation stages for the entire JME production process in India. The oil percentage of
JCL seed ranged from 21.0%–48.2%, and oil seeds were assumed to be sun-dried. The energy required for harvesting, handling, and storing of oil seeds, oil, and biodiesel, and the separation of husk from the seeds were neglected due to the cheap and abundant labor force available.
3.3. Energy Balance (Net Energy Use)
The selected JME projects of energy balance expressed in NER (energy yield) and environmental balance expressed in biofuel CO
2 emission factor per GJ energy are shown in
Table 1.
Table 1.
Energy balance and yield, net energy ratio (NER), and CO2 emission saving (CES).
Table 1.
Energy balance and yield, net energy ratio (NER), and CO2 emission saving (CES).
| Author | Energy Output/MJ | Energy Input/MJ | NER 1 | CES 2/% | Plantation | Reference |
|---|
| JME | By-Product |
|---|
| Ndong (2009) | 1 | | 0.21 | 4.7 | 72 | Mali/baseline | [5] |
| Ndong (2009) | 1 | | | 1.8 | 11 | irrigation/motorized | [5] |
| Achten (2008) | 1 | | 0.886 | 1.1 | 77 | 50%Faming E | [10] |
| Achten (2008) | 1 | | 0.16 | 6.3 | 93 | 17%Faming E | [10] |
| Prueksakorn (2006) | 1 | | 0.88 | 1.1 | 72 | Thailand | [9] |
| Prueksakorn (2006) | 1 | 17.88 | 0.88 | 21.5 | 72 | Thailand | [9] |
| Kumar (2012) 2 | 37.27 | | 21.83 | 1.7 | 54 | Irrigated | [7] |
| Kumar (2012) 2 | 37.27 | | 27.6 | 1.4 | 40 | Rain-fed | [7] |
| Kumar (2012) 3 | 37.27 | 107.8 | | 1.5–8.6 | 50–107 | Irrigated | [7] |
| Kumar (2012) 3 | 37.27 | 107.8 | | 1.2–7.0 | 40–93 | Rain-fed | [7] |
| Pandey (2011) | 1 | | 0.578 | 1.73 | 23 | Five years 4 | [6] |
Robert
et al. [
5] proposed a very detailed LCI analysis on the JEM project in West Africa in which an allocation analysis was also included. The energy yield (the ratio of biodiesel energy output to fossil energy input) was 4.7. In other words, 4.7 MJ of energy was produced from 1 MJ of fossil fuel consumed to produce JME (bondary 2). Biofuel production requires direct (electricity, fuels, natural gas) and indirect (manufacturing of agricultural inputs, methanol
etc.) energy consumption. In the allocation of energy expense, cultivation accounted for only 12%, which is even less than the 15% in the transport of seeds, oilcake, and unrefined
JCL oil. Transesterification requires 61% of the energy expense. In an alternative scenario, all co-products and JME are considered as output energy sources. The motorization and irrigation for the first three years were also included in
JCL production with a high consumption of fuel and irrigation water, which resulted in the energy yield being lowered down to only 1.8 compared to 4.7 in the baseline scenario. Meanwhile, GHG savings compared to fossil diesel was only marginal (11%), only one-sixth of the baseline scenario.
In a Thailand demonstration test [
9], the allocation for energy expenses of transesterification, irrigation, and fertilization processes was approximately 40% (0.197 GJ for producing steam), 23% (0.205 GJ) and 22% (0.198 GJ), respectively. The highest energy consumption was in the process of transesterification (61%) and other energy expenses were from using diesel, electricity, and producing fertilizer. Others are from 100 kg of fertilizer with the chemical formula 15–15–15 (energy consumption for transportation of fertilizer is excluded). The consumption of diesel for water pumping is a main contributor to energy expense in the irrigation process.
JCL plantation requires the process of land preparation once every 5 years as the stems are cut every year but a new plantation is made every five years. The residue (by-product)-produced energy is 17.883 GJ, whereas JME-produced energy is only 1 GJ. The highest energy by-product is wood, which produces an amount of energy of 10.289 GJ.
3.4. Environmental Balance (GHG Emission)
Achten [
10] deducted GHG emissions from the production phase of JME and considered the energy content of the co-products in calculating the CES (GHG emissions from JME). The result was about 77% compared to the GHG emission using fossil diesel. In total, the production and combustion of JME emitted 23.5 g CO
2e/MJ JME. The allocation of emitted CO
2 accounted for 52% in cultivation, while the shares of the transesterification and final combustion steps were 17% and 16%, respectively. Large shares of the emissions occurring during the agricultural step were due to fertilizers.
It is assumed that the energy consumed in the production stage is from 100% fossil fuel fired power plants. In India, fossil fuel contributes to 64% of electricity generation, whereby 52% of electricity is generated in coal-fired power plants, and the other 12% stem from natural gas (11%) and oil (1%). The rest is derived from from hydro (23%), nuclear (3%) and renewable materials (10%) [
6]. In China, coal is the major component of fossil fuel in electricity generation (80%), resulting in more CO
2 emissions.
Sunil [
7] compared energy and environmental balances of irrigated and rain-fed scenarios. Seed annual yields of irrigated (5.9 t/ha, Farming energy: 9333 MJ/t JME; Farming emission: 680 kg CO
2/t JME) and rain-fed (1.5 t/ha, Farming energy: 15098 MJ/t JME; Farming emission: 1114 kg CO
2/t JME) scenarios are closely related.
It was found that the utilization of JME saved more energy and emitted less CO
2 (saving 1.2 GJ/ha; emitting 80 kg CO
2/ha per year) than direct use of
JCL oil (saving 1.0 GJ/ha; 67 kg·CO
2/ha per year) based on the comparison of energy and environmental balance in Central India [
11].
3.5. Life Cycle Costs
It has been reported that the total cost of JME without externalities is higher than the current market retail selling price of diesel in Thailand [
12]. The cost allocations are 62.62%, 25.27% and 12.12% for agricultural, bio-diesel production, and environmental processes, respectively. The highest expenditure are the operation costs at the agricultural stage such as fertilizers, insecticide, and electricity for water pumping systems, especially in the dry season.
Generally, production of JME costs less than palm oil, soybean diesel costs more, and the cost of rapeseed diesel is the highest [
13]. As a commercially viable option, microalgae will require further improvements in genetic and metabolic engineering to obtain promising strains and produce higher yields of oil. Moreover, development of equipment and methodologies for cost-effective culturing, harvesting, and processing are also required, as year-round production of biofuels requires constant, reliable feedstock supply [
14]. While microalgae are projected as a future feedstock of biodiesel, production costs are much higher than for terrestrial crops, in the range of US$2 to $22 per liter [
1]. In addition, the entire microalgae biomass has to be used to produce biofuel, otherwise the yield of biofuel is too low to be accepted [
15].
3.6. Situation in China
It is widely hoped that JME production will offer a newer, more sustainable energy source. That, however, has yet to be proven. In 2007, the global output of biodiesel (FAME) was 8.82 Mt, but only 0.1 Mt in China, and there has been no obvious change since then in suffering from arable land limitation [
1]. Therefore, there has been more effort to search for energy crops in China than in the EU, US, and even Asia countries, such as India and Thailand. However, the climate is not suitable to plant
JCL on most of the land in China. Although Southwest China is considered as a prospective area to plant
JCL, it has been discovered that both prospective planting areas and yields of seed have been overestimated after comprehensive consideration using the Agro Ecological Zone method. Based on the remote sensing data on land use, meteorological, soil and land slope, and suitable environment for
JCL plantation in Southwest China, the potential land to expand
JCL areas was only 0.15 M·ha up until 2008, which is far from the government goal (1.667 M·ha by 2020). Although the moderately suitable land was increased to 1.433 M·ha after softening the terms, the poor yield of seed on the moderate land would most likely destroy the balance of both energy and environment (NER and CES become minus value) [
16].
In China, the yield of fresh fruit, dried seed, and extracted oil are be less than in countries in the tropic zone, and there is little data from farming sites. The prospective yield of
JCL seeds and JME are often exaggerated and questionable. For instance, the JME yield (5 t/ha) and farming (planting) energy (160 MJ/t JME) were accepted in a published LCA research. Nevertheless, the energy of soybean farming is as high 2497 MJ/t FAME as usual and the average yield of
JCL seed in the tropic zone is only 4 t/ha (the JME content is less than 40% of
JCL seed). A misleading NER of JME (2.004) even higher than soybean oil (0.981) was proposed based on an unconfirmed
JCL annual yield (5 t/ha) in the Hainan province of China, which was greatly overestimated and even over the yield obtained in the tropical zone [
17]. The reasonable annual yield of JME is 1–2 t/ha based on demonstration tests in Thailand [
9], Malaysia [
4], and West Africa [
5] where a JME content of 30%–50% is usually accepted.
3.7. Comparison with Microalgae
It is known that microalgae have attracted the spotlight around the world during the past years and have been considered to be very hopeful competitors to replace terrestrial plants as feedstock in the next generation of biofuels. One reason for the superiority of microalgae is that fresh water and arable land are not necessary for culturing. Waste, saline, or brackish water, and land resources, such as mudflats or deserts, are all usable for microalgae culturing so that there is no interference in food production as was the case for the first generation of biofuels [
13]. Another benefit of microalgae is the high expected yield of biomass, which can be higher than 100 t/ha, and the oil content, which can be higher than 70% of dry weight in the form of triglycerides [
18]. However, these are only speculations based on the excessively optimistic assumptions or laboratory data using minimalistic culturing volumes of several milliliters to liters, which largely deviate from the larger scale culturing results either in ponds or PBRs. Although
Chlorella,
Diatom,
Scenedesmus,
Tetraselmis,
Nannochloropsis, and
Haematococcus pluvialis were preferred as hopeful candidates, the real potential of their productivity was not clear until their production was realized at a large scale. The resulting differences between published biomass and oil (lipid) yield potentials are up to 16-fold [
19].
The NER of JME oil and algal oil were selected to quantify the energy balance and to associate it with both the boundaries of LCI and the yield of algal biomass and lipid. Meanwhile, CES was selected to quantify the GHG balance. Some of the calculated NER and CES based on published LCA results are summarized in
Table 2, and some are laboratory data and extremely exaggerated. The results are dependent on the manner of culturing (pond or PBR), harvesting, biomass yield, lipid content, and the boundary. NER for
Nannochloropsis cultivation process is 4.33 for flat-plate PBR but 7.01 for raceway ponds, indicating that both processes are energetically favorable for biomass production (boundary 1) [
20]. NER decreases to below one when the harvesting stage was included (boundary 2) (except for HRJ or wet harvesting) indicating that dewatering is the most energy consuming process in the upstream (shown in
Table 2). The NER is strongly dependent on the yield of biomass or lipid and varies as much as six-fold depending on the source [
21]. In order to attain the energy benefit (NER > 1, overestimated LCI data (biomass yield or lipid fraction) were often accepted in the published papers [
18,
22].
Table 2.
Energy input (expense) and greenhouse gas (GHG) emission for producing 1 kg algal oil and energy yield (NER).
Table 2.
Energy input (expense) and greenhouse gas (GHG) emission for producing 1 kg algal oil and energy yield (NER).
| Author | Yield 1 t/ha | Lipid %(D W) | Output MJ | Input MJ | NER | CES 2 % | Cultivation | Reference |
|---|
| Harvesting | Algal Species |
|---|
| Kalnes (2012) | | 25.0 | 83.6 | 85.8 | 0.97 | −2 | Dewatering | Nannochloropsis | [23] |
| Kalnes (2012) | | 25.0 | 83.6 | 59.0 | 1.42 | 57 | HRJ | Nannochloropsis | [23] |
| Batan (2010) | 91 | 42.5 | 1 | 0.93 | 0.93 | 29 | PBR bags | Nannochloropsis | [24] |
| Lardon (2009) | 62 | 17.5 | 103.8 | 106.4 | 0.98 | | Dry | Chlorella vulgaris | [25] |
| Lardon (2009) | 62 | 17.5 | 146.8 | 41.4 | 3.55 | | Wet | Chlorella vulgaris | [25] |
| Xu(2011) | 128 | 40.0 | 37.2 | 27.5 | 1.35 | 41 | Tubular | P. tricornutum | [21] |
| Vasudevan (2012) | 76 | 25.0 | 9.2 | 32 | 0.3 | −232 | Dry Extract | Algae | [26] |
| Vasudevan (2012) | 76 | 25.0 | 9.2 | 3.7 | 2.5 | 37 | Wet Extract | Algae | [26] |
If FAME is considered as the only product of algal fuel (Boundary 3), it is almost impossible for NER and CES to be >1 and >0, respectively (net energy and CO
2 emission reduction are positive) as large amounts of energy are consumed in the dehydration and extraction processes as shown in
Table 2 [
21].
In fact, current commercial microalgae production is only focused on a few high-value products used mainly for human nutritional supplements, including entire algal biomasses, such as those of
Spirulina (
Arthrospira) (3000 t/a) and
Chlorella (2000 t/a), and extracted products, including β-carotene, Astaxanthin, and docosahexanoic acid (DHA). The total annual yield of microalgal biomass (dry matter basis) around the world is only about 10 Kt. However, the total revenue of the microalgae-containing products is up to several billion dollars per year, with a typical selling price of $5000 to $100,000 per dry ton of biomass or extracted products [
27]. The biomass of microalgae is also used as live feeds in aquaculture, and in wastewater treatment systems with a lower price in the culturing stage [
28]. Over 90% of the world’s commercial microalgae production uses shallow, open, paddle wheel mixed raceway type ponds, in addition to open circular ponds for
Chlorella production in Japan.
In China, the productions of
Spirulina (
Arthrospira) and
Chlorella are sold as nutrients, and high nutrition feeds have increased rapidly recently. The annual yields of
Spirulina (
Arthrospira) and
Chlorella have attained up to 3000 t and 1000 t, respectively. The protein content and lipid content of
Spirulina sold as a nutrient are as high as 60% and less than 10%, respectively.
Chlorella, with a lower protein content and a higher lipid content than
Spirulina, ia recommended to be used as feedstock for biodiesel, although the price makes it too expensive to be used nowadays. A several-fold increase in algal biomass or lipid production is not feasible by cultivation in either low-nitrogen nutrient or CO
2-rich environments. When low-N nutrients are used in algal cultivation, lipid content increases but the yield of biomass usually decreases [
29]. The improvement of biomass yield with additional (5%–14%) CO
2 aeration is only 1.5 times and difficult to double compared with air aeration without additional CO
2. In the lipid fraction, there are not only triglyceride (TAG) and free fatty acids (FFA) but also sterols, terpenes and hydrocarbons, which are unable to be transesterified or esterified to form FAME so that the yield of FAME would be lower than the lipid content of
Chlorella [
22].
Besides FAME (biodiesel), in order to increase the yield of algal biofuels, other hydrocarbon fuels including methane and FT-fuel have to be considered as algal biofuels produced from the lipid extracted fraction (Lipid Extracted Algae, LEA) which are difficult to esterify to form FAME [
23]. Even now, the algal biofuel has not been produced in China due to the low algal biomass productivity and the complexity of the algal biomass. This is also the reason why the species of microalgae are often not specified in the literature of LCA, easily misleading the reader, as the productivity and lipid composition of various species of microalgae are very different and dependent on the area and time of culturing. Handler summarized the worldwide LCA results of the prior literature and investigated the wide variance in predicted environmental impacts from microalgae cultivation in open-air raceway ponds and deduced a very wide range of CO
2 emission (0.1–4.4 g CO
2e/g microalgae) [
30].
3.8. The Difficulties of Commercialization
3.8.1. Jatropha
JCL has been successfully planted on a large scale, especially in Asia and Africa where planting areas are near to 1 M·ha. The seed yield ranges from 4 to 5 t/ha in higher areas but is relatively area and climate dependent. The oil content ranges from 20% to 50% and typically lies between 30% and 40%, depending on the culturing area and conditions. The energy and environmental balance of culturing, harvesting, and processing show JCL to be a preferable sustainable feedstock for biofuel. The use of JME shows more favorable results in energy saving and CO2 emission than direct use of JCL oil.
In the subtropical zone and even in the south of China, the feasibility of JCL cultivation must be carefully considered. The yield of JCL oil is dependent on the climate and planting area. The published data from tropical zones such as south Asia, south-east Asia and Africa are not suitable for the LCI in China. The real, domestic and large-scale planting data strongly suggests the acceptance as LCI instead of published data from different zones of the world.
In the environmental balance assessment of JME, the CO2 emission due to LUC was not considered in the paper. Based on the accomplished production scale in South Asia, Southeast Asia, and West Africa, Jatropha may be more easily accessible as the next generation feedstock of biodiesel provided there is enough land with proper climate, and the CO2 emission resulting from LUC can be avoided at the same time.
3.8.2. Microalgae
The cultivation of microalgae, even that of Chlorella and Spirulina, is still in the beginning stages. The biggest farm for culturing Spirulina is only 0.1 M·ha scale, which is much less than big JCL farms up to 1 M·ha scale. Therefore, more demonstration farms or projects need to be created to verify the feasibility for microalgae to become the feedstock for biofuel in the future.
The triglyceride and FFA (free fatty acid) contents in the lipid fraction of microalgae are much smaller than in the lipid fraction of JCL. Therefore, the biodiesel yield cannot be directly deduced from the lipid content of microalgae.
The lipid in microalgae should be referred to as bio-crude other than biodiesel. All bio-crude or biomass has to be processed to produce sustainable bio-energy instead of biodiesel (FAME), so that biohydrogenated diesel (BHD), FT-fuel, bio-gas, bio-power, and bio-heat can be produced and used together. The NER and NEB are hardly larger than one and they are positive. CES is also rarely positive if the energy prepared from LEA is not accounted for at the present stage.