Global Change and Helminth Infections in Grazing Ruminants in Europe: Impacts, Trends and Sustainable Solutions
Abstract
:1. Impact of Helminth Infection on the Sustainability and Efficiency of Livestock Farming
1.1. Livestock Farming As a Cornerstone of Society
1.2. Costs to Economies and Animals
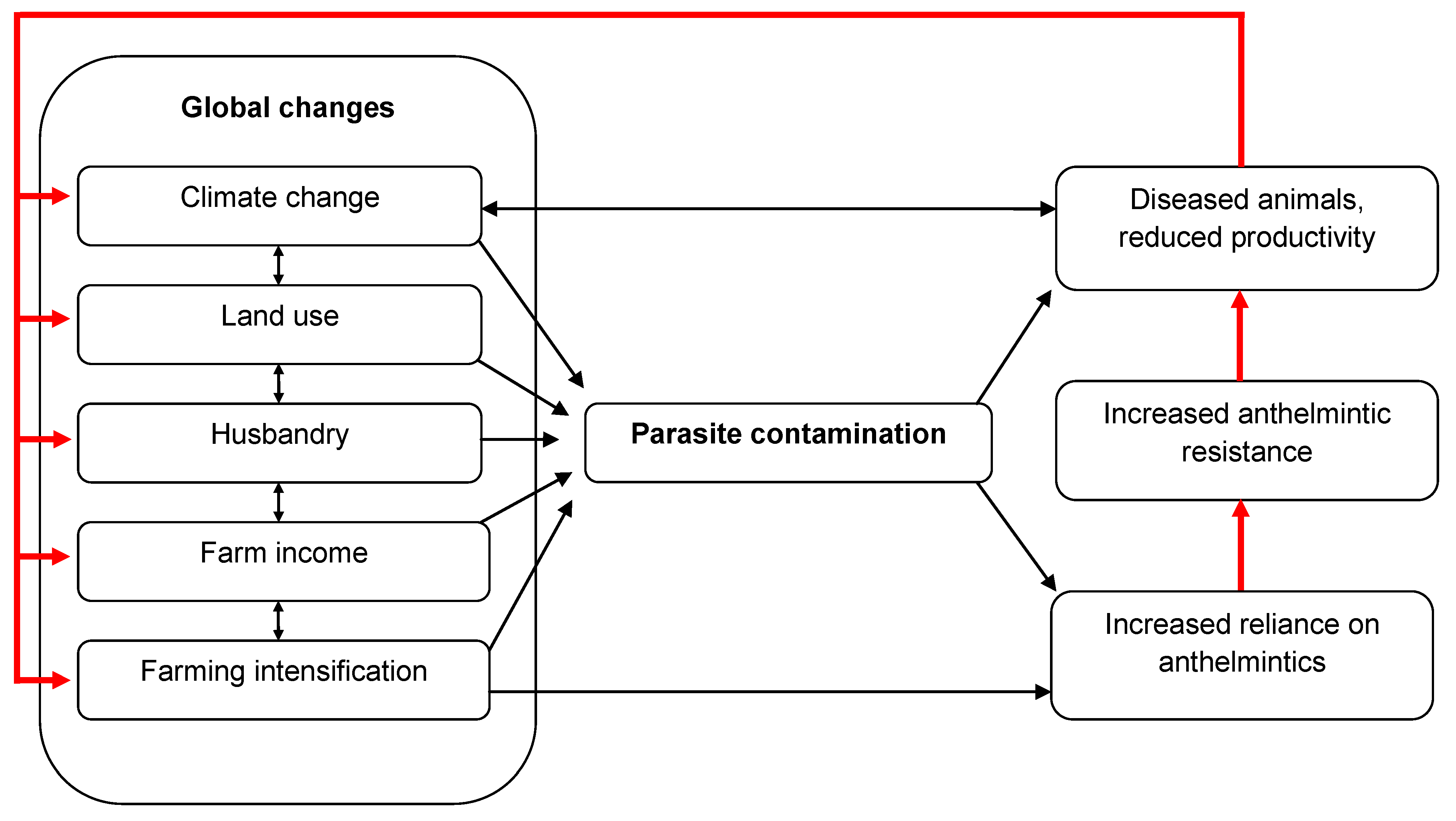
2. Increased Risks of Helminth Infection in Livestock Due to Global Change
2.1. The Changing Environment
2.2. The Importance of Anthelmintic Resistance
2.3. The Need for Increased Carbon Efficiency in Ruminant Farming
3. Scientific Priorities to Support Sustainable Helminth Control
3.1. Diagnosis
3.2. Spatial Epidemiology and Forecasting of Helminth Disease
3.3. Predicting the Timing of Parasite Risk
3.4. Sustainable Parasite Control
4. Conclusions
Acknowledgements
Conflicts of Interest
References
- FAOSTAT (2009). Available online: http://faostat.fao.org/ (accessed on 15 July 2013).
- Baulcombe, D.; Crute, I.; Dunwell, J.; Gale, M.; Jones, J.; Pretty, J.; Sutherland, W.; Toulmin, C. Reaping the Benefits—Science and the Sustainable Intensification of Global Agriculture; Royal Society Policy Document: London, UK, 2009. [Google Scholar]
- Chiotti, Q.P.; Johnston, T. Extending the boundaries of climate change research—A discussion on agriculture. J. Rural Stud. 1995, 11, 335–350. [Google Scholar] [CrossRef]
- Nieuwhof, G.J.; Bishop, S.C. Costs of the major endemic diseases in Great Britain and the potential benefits of reduction in disease impact. Anim. Sci. 2005, 81, 23–29. [Google Scholar]
- Schweizer, G.; Braun, W.; Deplazes, D.; Torgerson, P.R. The economic effects of bovine fasciolosis in Switzerland. Vet. Rec. 2005, 157, 188–193. [Google Scholar]
- Selzer, P.M. Preface. In Antiparasitic and Antibacterial Drug Discovery. From Molecular Targets to Drug Candidates; Wiley-Blackwell: Hoboken, USA, 2009; pp. 11–12. [Google Scholar]
- Charlier, J.; Höglund, J.; von Samson-Himmelstjerna, G.; Dorny, P.; Vercruysse, J. Gastrointestinal nematode infections in adult dairy cattle: Impact on production, diagnosis and control. Vet. Parasitol. 2009, 164, 70–79. [Google Scholar] [CrossRef]
- Rehbein, S.; Baggott, D.G.; Johnson, E.G.; Kunkle, B.N.; Yazwinski, T.A.; Yoon, S.; Cramer, L.G.; Soll, M.D. Nematode burdens of pastured cattle treated once at turnout with eprinomectin extended-release injection. Vet. Parasitol. 2013, 192, 321–331. [Google Scholar] [CrossRef]
- Van Dijk, J.; Morgan, E.R. The influence of temperature on the development, hatching and survival of Nematodirus battus larvae. Parasitology 2008, 135, 269–283. [Google Scholar]
- Mejia, M.E.; Perri, A.F.; Licoff, N.; Miglierina, M.M.; Cseh, S.; Ornstein, A.M.; Becu-Villalobos, D.; Lacau-Mengido, I.M. Comparison of three methods for gastrointestinal nematode diagnosis determination in grazing dairy cattle in relation to milk production. Vet. Parasitol. 2011, 183, 174–177. [Google Scholar] [CrossRef]
- Charlier, J.; van der Voort, M.; Hogeveen, H.; Vercruysse, J. ParaCalc®—A novel tool to evaluate the economic importance of worm infections on the dairy farm. Vet. Parasitol. 2012, 184, 204–211. [Google Scholar] [CrossRef]
- Charlier, J.; Levecke, B.; Devleesschauwer, B.; Vercruysse, J.; Hogeveen, H. The economic effects of whole-herd versus selective anthelmintic treatment strategies in dairy cows. J. Dairy Sci. 2012, 95, 2977–2987. [Google Scholar] [CrossRef]
- Mason, W.A.; Pomroy, W.E.; Lawrence, K.E.; Scott, I. The effect of repeated, four-weekly eprinomectin treatment on milk production in pasture-based, seasonally-calving dairy cattle. Vet. Parasitol. 2012, 189, 250–259. [Google Scholar] [CrossRef]
- Van Dijk, J.; Sargison, N.D.; Kenyon, F.; Skuce, P. Climate change and infectious disease: Helminthological challenge to farmed ruminants in temperate regions. Invited Review. Animal 2010, 4, 377–392. [Google Scholar] [CrossRef]
- Sargison, N.D.; Wilson, D.J.; Bartley, D.J.; Penny, C.D.; Jackson, F. Haemonchosis and teladorsagiosis in a Scottish sheep flock putatively associated with the over-wintering of hypobiotic fourth stage larvae. Vet. Parasitol. 2007, 147, 326–331. [Google Scholar] [CrossRef]
- Kenyon, F.; Sargison, N.D.; Skuce, P.J.; Jackson, F. Sheep helminth parasitic disease in South-Eastern Scotland arising as a possible consequence of climate change. Vet. Parasitol. 2009, 163, 293–297. [Google Scholar] [CrossRef]
- Lindqvist, A.; Ljungström, B.L.; Nilsson, O.; Waller, P.J. The dynamics, prevalence and impact of nematode infections in organically raised sheep in Sweden. Acta Vet. Scand. 2001, 42, 377–389. [Google Scholar] [CrossRef]
- De la Rocque, S.; Rioux, J.A.; Slingenbergh, J. Climate change: Effects on animal disease systems and implications for surveillance and control. Rev. Sci. Tech. Int. Off. of Epizoot. 2008, 27, 339–354. [Google Scholar]
- Morgan, E.R.; Wall, R. Climate change and parasitic disease: Farmer mitigation? Trends Parasitol. 2009, 7, 308–313. [Google Scholar] [CrossRef]
- Leathwick, D.M.; Barlow, N.D.; Vlassoff, A. A model for nematodiasis in New Zealand lambs. Int. J. Parasitol. 1992, 22, 789–799. [Google Scholar] [CrossRef]
- Reynecke, D.P.; Waghorn, T.S.; Oliver, A.-M.B.; Miller, C.M.; Vlassoff, A.; Leathwick, D.M. Dynamics of the free-living stages of sheep intestinal parasites on pasture in the North Island of New Zealand. 2. Weather variables associated with development. Vet. J. 2011, 59, 287–292. [Google Scholar]
- Leathwick, D.M.; Waghorn, T.S.; Miller, C.M.; Candy, P.M.; Oliver, A.-M.B. Managing anthelmintic resistance—Use of a combination anthelmintic and leaving some lambs untreated to slow the development of resistance to ivermectin. Parasitology 2012, 187, 285–294. [Google Scholar]
- Dobson, R.J.; Barnes, E.H.; Tyrrell, K.L.; Hosking, B.C.; Larsen, J.W.A.; Besier, R.B.; Love, S.; Rolfe, P.F.; Bailey, J.N. A multi-species modelto assess the effect of refugia on worm control and anthelmintic resistance insheep grazing systems. Aust. Vet. J. 2011, 89, 200–208. [Google Scholar] [CrossRef]
- Laurenson, Y.C.S.M.; Bishop, S.C.; Forbes, A.B.; Kyriazakis, I. Modelling the short- and long-term impacts of drenching frequency and targeted selective treatment on the performance of grazing lambs and the emergence of anthelmintic resistance. Parasitology 2013, 140, 780–791. [Google Scholar] [CrossRef]
- Fox, N.J.; White, P.C.L.; McClean, C.J.; Marion, G.; Evans, A.; Hutchings, M.R. Predicting impacts of climate change on Fasciola hepatica risk. PLoS One 2011, 6, e16126. [Google Scholar]
- Jackson, F.; Miller, J. Alternative approaches to control—Quo vadit? Vet. Parasitol. 2006, 139, 371–384. [Google Scholar] [CrossRef]
- Kaplan, R.M. Drug resistance in nematodes of veterinary importance: A status report. Trends Parasitol. 2004, 20, 477–481. [Google Scholar] [CrossRef]
- Familton, A.S.; Mason, P.; Coles, G.C. Anthelmintic-resistant Cooperia species in cattle. Vet. Rec. 2001, 149, 719–720. [Google Scholar]
- Sangster, N.C.; Dobson, R.J. Anthelmintic Resistance. In The Biology of Nematodes; Lee, D.L., Ed.; Taylor and Francis: London, UK, 2002; pp. 531–567. [Google Scholar]
- Sutherland, I.A.; Leathwick, D.M. Anthelmintic resistance in nematode parasites of cattle—A global issue? Trends Parasitol 2010, 27, 176–181. [Google Scholar] [CrossRef]
- Demeler, J.; van Zeveren, A.M.; Kleinschmidt, N.; Vercruysse, J.; Höglund, J.; Koopmann, R.; Cabaret, J.; Claerebout, E.; Areskog, M.; von Samson-Himmelstjerna, G. Monitoring the efficacy of ivermectin and albendazole against gastro intestinal nematodes of cattle in Northern Europe. Vet. Parasitol. 2009, 160, 109–115. [Google Scholar] [CrossRef]
- El-Abdellati, A.; Charlier, J.; Geldhof, P.; Levecke, B.; Demeler, J.; von Samson-Himmelstjerna, G.; Claerebout, E.; Vercruysse, J. The use of a simplified faecal egg count reduction test for assessing anthelmintic efficacy on Belgian and German cattle farms. Vet. Parasitol. 2010, 169, 352–357. [Google Scholar] [CrossRef]
- Bartley, D.J.; Jackson, F.; Jackson, E.; Sargison, N. Characterisation of two triple resistant field isolates of Teladorsagia from Scottish lowland sheep farms. Vet. Parasitol. 2004, 123, 189–199. [Google Scholar] [CrossRef]
- Sargison, N.D.; Jackson, F.; Bartley, D.J.; Wilson, D.J.; Stenhouse, L.J.; Penny, C.D. Observations on the emergence of multiple anthelmintic resistance in sheep flocks in the south-east of Scotland. Vet. Parasitol. 2007, 45, 65–76. [Google Scholar]
- Wolstenholme, A.J.; Fairweather, I.; Prichard, R.; von Samson-Himmelstjerna, G.; Sangster, N.; Nicholas, C. Drug resistance in veterinary helminths. Trends Parasitol. 2004, 20, 469–476. [Google Scholar] [CrossRef]
- Kaminsky, R.; Ducray, P.; Jung, M.; Clover, R.; Rufener, L.; Bouvier, J.; Weber, S.S.; Wenger, A.; Wieland-Berghausen, S.; Goebel, T.; et al. A new class of anthelmintics effective against drug-resistant nematodes. Nature 2008, 452, 176–180. [Google Scholar] [CrossRef]
- Little, P.R.; Hodges, A.; Watson, T.G.; Seed, J.A.; Maeder, S.J. Field efficacy and safety of an oral formulation of the novel combination anthelmintic, derquantel-abamectin, in sheep in New Zealand. N. Z. Vet. J. 2010, 58, 121–129. [Google Scholar] [CrossRef]
- Steinfeld, H.; Gerber, P.; Wassenaar, T.; Castel, V.; Rosales, M.; de Haan, C. Livestock’s Long Shadow: Environmental Issues and Options; Report presented to the Food and Agricultural Organisation of the United Nations (FAO): Rome, Italy, 2006; p. 284. [Google Scholar]
- Gill, M.; Smith, P.; Wilkinson, J.M. Mitigating climate change: The role of domestic livestock. Animal 2010, 4, 323–333. [Google Scholar] [CrossRef]
- Thornton, P.K. Livestock production: Recent trends, future prospects. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 2853–2867. [Google Scholar]
- Taylor, M.A.; Hunt, K.R.; Goodyear, K.L. Anthelmintic resistance detection methods. Vet. Parasitol. 2002, 103, 183–194. [Google Scholar] [CrossRef]
- Gasser, R.B.; Bott, N.J.; Chilton, N.B.; Hunt, P.; Beveridge, I. Toward practical, DNA-based diagnostic methods for parasitic nematodes of livestock—Bionomic and biotechnical implications. Biotechnol. Adv. 2008, 26, 325–334. [Google Scholar]
- Zarlenga, D.S.; Chute, M.; Gasbarre, L.C.; Boyd, P.C. A multiplex PCR assay for differentiating economically important gastrointestinal nematodes of cattle. Vet. Parasitol. 2001, 97, 199–209. [Google Scholar]
- Wimmer, B.; Craig, B.H.; Pilkington, J.G.; Pemberton, J.M. Non-invasive assessment of parasitic nematode species diversity in wild Soay sheep using molecular markers. Int. J. Parasitol. 2004, 34, 625–631. [Google Scholar] [CrossRef]
- Learmount, J.; Conyers, C.; Hird, H.; Morgan, C.; Craig, B.H.; von Samson-Himmelstjerna, G.; Taylor, M. Development and validation of real-time PCR methods for diagnosis of Teladorsagia circumcincta and Haemonchus contortus in sheep. Vet. Parasitol. 2009, 166, 268–274. [Google Scholar] [CrossRef]
- Dobson, R.J.; Sangster, N.C.; Besier, R.B.; Woodgate, R.G. Geometric means provide a biased efficacy result when conducting a faecal egg count reduction test (FECRT). Vet. Parasitol. 2009, 161, 162–167. [Google Scholar] [CrossRef]
- Coles, G.C.; Jackson, F.; Pomroy, W.E.; Prichard, R.K.; von Samson-Himmelstjerna, G.; Silvestre, A.; Taylor, M.A.; Vercruysse, J. The detection of anthelmintic resistance in nematodes of veterinary importance. Vet. Parasitol. 2006, 136, 167–185. [Google Scholar] [CrossRef]
- Von Samson-Himmelstjerna, G.; Blackhall, W.J.; McCarthy, J.S.; Skuce, P.J. Single nucleotide polymorphism (SNP) markers for benzimidazole resistance in veterinary nematodes. Parasitology 2007, 134, 1077–1086. [Google Scholar] [CrossRef]
- Von Samson-Himmelstjerna, G.; Walsh, T.K.; Donnan, A.A.; Carriere, S.; Jackson, F.; Skuce, P.J.; Rohn, K.; Wolstenholme, A.J. Molecular detection of benzimidazole resistance in Haemonchus contortus using real-time PCR and pyrosequencing. Parasitology 2009, 136, 349–358. [Google Scholar] [CrossRef]
- Skuce, P.J.; Stenhouse, L.; Jackson, F.; Hypsa, V.; Gilleard, J.S. Benzimidazole resistance allele haplotype diversity in United Kingdom isolates of Teladorsagia circumcincta supports a hypothesis of multiple origins of resistance by recurrent mutation. Int. J. Parasitol. 2010, 40, 1247–1255. [Google Scholar] [CrossRef]
- Höglund, J.; Gustaffson, K.; Ljungstrom, B.L.; Engstrom, A.; Donnan, A.A.; Skuce, P.J. Anthelmintic resistance in Swedish sheep flocks based on a comparison of the results from the faecal egg count reduction test and resistant allele frequencies of the beta-tubulin gene. Vet. Parasitol. 2009, 161, 60–68. [Google Scholar] [CrossRef]
- Beech, R.N.; Skuce, P.; Bartley, D.J.; Martin, R.J.; Prichard, R.K.; Gilleard, J.S. Anthelmintic resistance: Markers for resistance, or susceptibility? Parasitology 2010, 138, 160–174. [Google Scholar]
- Gilleard, J.S. Understanding anthelmintic resistance: The need for genomics and genetics. Int. J. Parasitol. 2006, 36, 1227–1239. [Google Scholar] [CrossRef]
- Vignali, D.A.A. Multiplexed particle-based flow cytometric assays. J. Immunol. Methods 2000, 243, 243–255. [Google Scholar] [CrossRef]
- Pickering, J.W.; Martin, T.B.; Schroder, M.C.; Hill, H.R. Comparison of a multiplex flow cytometric assay with Enzyme-Linked Immunosorbent Assay for quantification of antibodies to Tetanus, Diphtheria and Haemophilus influenza Type b. Clin. Diagn. Lab. Immunol. 2002, 9, 872–876. [Google Scholar]
- Dunbar, S.A.; Vander Zee, C.A.; Oliver, K.G.; Karem, K.L.; Jacobson, J.W. Quantitative, multiplexed detection of bacterial pathogens: DNA and protein applications of the Luminex LabMAPTM system. J. Microbiol. Methods 2003, 53, 245–252. [Google Scholar] [CrossRef]
- Morgan, E.R.; Varro, R.; Sepulveda, H.; Ember, J.A.; Apgar, J.; Wislon, J.; Lowe, L.; Chen, R.; Shivraj, L.; Agadir, A.; et al. Cytometric bead array: A multiplexed assay platform with applications in various areas of biology. Clin. Immunol. 2004, 110, 252–266. [Google Scholar] [CrossRef]
- Dunbar, S.A. Application of Luminex xMAPTM technology for rapid, high-throughput multiplexed nucleic acid detection. Clinia Chim. Acta 2006, 363, 71–82. [Google Scholar] [CrossRef]
- Charlier, J.; Vercruysse, J.; Smith, J.; Vanderstichel, R.; Stryhn, H.; Claerebout, E.; Dohoo, I. Evaluation of anti-Ostertagia ostertagi antibodies in individual milk samples as decision parameter for selective anthelmintic treatment in dairy cows. Prev. Vet. Med. 2009, 93, 147–152. [Google Scholar]
- McCann, C.M.; Baylis, M.; Williams, D.J.L. Seroprevalence and spatial distribution of Fasciola hepatica-infected dairy herds in England and Wales. Vet. Rec. 2010, 166, 612–617. [Google Scholar] [CrossRef]
- Andersen, U.V.; Howe, D.K.; Dangoudoubiyam, S.; Toft, N.; Reinemeyer, C.R.; Lyons, E.T.; Olsen, S.N.; Monrad, J.; Nejsum, P.; Nielsen, M.K. SvSXP: A Strongylus vulgaris antigen with potential for prepatent diagnosis. Parasites Vectors 2013, 6, 84. [Google Scholar] [CrossRef]
- Ganheim, C.; Höglund, J.; Waller, P. Acute phase proteins in response to Dictyocaulus viviparus infection in calves. Acta Vet. Scand. 2004, 45, 79–86. [Google Scholar] [CrossRef]
- Charlier, J.; Dorny, P.; Levecke, B.; Demeler, J.; von Samson-Himmelstjerna, G.; Höglund, J.; Vercruysse, J. Serum pepsinogen levels to monitor gastrointestinal nematode infections in cattle revisited. Res. Vet. Sci. 2011, 90, 451–456. [Google Scholar] [CrossRef]
- Bandyopadhyay, K.; Kellar, K.L.; Moura, I.; Casaqui Carollo, M.C.; Graczyk, T.K.; Slemenda, S.; Johnston, S.P.; da Silva, A.J. Rapid microsphere assay for identification of Cryptosporidium hominis and Cryptosporidium parvum in stool and environmental samples. J. Clin. Microbiol. 2007, 45, 2835–2840. [Google Scholar] [CrossRef]
- Li, W.; Zhang, N.; Gong, P.; Cao, L.; Li, J.; Su, L.; Li, S.; Diao, Y.; Wu, K.; Li, L.; et al. A novel multiplex PCR coupled with Luminex assay for the simultaneous detection of Cryptosporidium spp., Cryptosporidium parvum and Giardia duodenalis. Vet. Parasitol 2010, 173, 11–18. [Google Scholar]
- Stark, D.; Al-Qassab, S.E.; Barratt, J.L.; Stanley, K.; Roberts, T.; Marriott, D.; Harkness, J.; Ellis, J. An evaluation of a multiplex tandem real-time PCR for the detection of Cryptosporidium spp, Dientamoeba fragilis, Entamoeba histolytica, and Giardia intestinalis from clinical stool samples. J. Clin. Microbiol. 2010, 49, 257–262. [Google Scholar]
- Iseki, H.; Alhassan, A.; Ohta, N.; Thekisoe, O.M.M.; Yokoyama, N.; Inoue, N.; Nambota, A.; Yasuda, J.; Igarashi, I. Development of a multiplex loop-mediated isothermal amplification (mLAMP) method for the simultaneous detection of bovine Babesia parasites. J. Microbiol. Methods 2007, 71, 281–287. [Google Scholar] [CrossRef]
- Parida, M.; Sannarangaiah, S.; Dash, P.K.; Rao, P.V.; Morita, K. Loop mediated isothermal amplification (LAMP): A new generation of innovative gene amplification technique; perspectives in clinical diagnosis of infectious diseases. Rev. Med. Virol. 2008, 18, 407–421. [Google Scholar] [CrossRef]
- Aonuma, H.; Yoshimura, A.; Perera, N.; Shinzawa, N.; Bando, H.; Oshiro, S.; Nelson, B.; Fukumoto, S.; Kanuka, H. Loop-mediated isothermal amplification applied to filarial parasites detection in the mosquito vectors: Dirofilaria immitis as a study model. Parasites Vectors 2009, 2, 15–21. [Google Scholar] [CrossRef]
- Mori, Y.; Notomi, T. Loop-mediated isothermal amplification (LAMP): A rapid, accurate and cost-effective diagnostic method for infectious diseases. J. Infect. Chemother. 2009, 15, 62–69. [Google Scholar] [CrossRef]
- Ollerenshaw, C.B.; Rowlands, W.T. A method of forecasting the incidence of fascioliasis in Anglesey. Vet. Rec. 1959, 71, 591–598. [Google Scholar]
- Cornell, S. Modelling nematode populations: 20 years of progress. Trends Parasitol. 2005, 21, 542–545. [Google Scholar] [CrossRef]
- Fuentes, M.V. Remote sensing and climate data as a key for understanding fasciolosis transmission in the Andes: Review and update of an ongoing interdisciplinary project. Geospat. Health 2006, 1, 59–70. [Google Scholar]
- Rose, H.; Wall, R. Modelling the impact of climate change on spatial patterns of disease risk: Sheep blowfly strike by Lucilia sericata in Great Britain. Int. J. Parasitol. 2011, 41, 739–746. [Google Scholar] [CrossRef]
- Biggeri, A.; Catelan, D.; Dreassi, E.; Rinaldi, L.; Musella, V.; Veneziano, V.; Cringoli, G. Multivariate spatially-structured variability of ovine helminth infections. Geospat. Health 2007, 2, 97–104. [Google Scholar]
- Hendrickx, G.; Biesemans, J.; de Deken, R. The Use of GIS in Veterinary Parasitology. In GIS and Spatial Analysis in Vet. Science; Durr, P., Gatrell, A., Eds.; CABI Publishing: Wallingford, UK, 2004; pp. 145–176. [Google Scholar]
- Rinaldi, L.; Musella, V.; Biggeri, A.; Cringoli, G. New insights into the application of geographical information systems and remote sensing in veterinary parasitology. Geospat. Health 2006, 1, 33–47. [Google Scholar]
- Bergquist, R.; Rinaldi, L. Health research based on geospatial tools: A timely approach in a changing environment. J. Helminthol. 2010, 84, 1–11. [Google Scholar] [CrossRef]
- Geospatial health: Health applications in geospatial science. Available online: http://www.geospatialhealth.unina.it (accessed on 11 August 2013).
- Morgan, E.R.; Milner-Gulland, E.J.; Torgerson, P.R.; Medley, G.F. Ruminating on complexity: Macroparasites of wildlife and livestock. Trends Ecol. Evol. 2004, 19, 181–188. [Google Scholar] [CrossRef]
- Morgan, E.R.; van Dijk, J. Climate and the epidemiology of gastrointestinal nematode infections of sheep in Europe. Vet. Parasitol. 2012, 189, 8–14. [Google Scholar] [CrossRef]
- Van Dijk, J.; David, G.P.; Baird, G.; Morgan, E.R. Back to the future: Developing hypotheses on the effects of climate change on ovine parasitic gastroenteritis from historical data. Vet. Parasitol. 2008, 158, 73–84. [Google Scholar] [CrossRef]
- Smith, G. Modeling of parasite populations—Gastrointestinal nematode models. Vet. Parasitol. 1994, 54, 127–143. [Google Scholar] [CrossRef]
- Van Dijk, J.; Morgan, E.R. Variation in the hatching behaviour of Nematodirus battus: Polymorphic bet hedging? Int. J. Parasitol. 2010, 40, 675–681. [Google Scholar] [CrossRef]
- Kenyon, F.; Greer, A.W.; Coles, G.C.; Cringoli, G.; Papadopoulos, E.; Cabaret, J.; Berrag, B.; Varady, M.; van Wyk, J.A.; Thomas, E.; et al. The role of targeted selective treatments in the development of refugia-based approaches to the control of gastrointestinal nematodes of small ruminants. Vet. Parasitol. 2009, 164, 3–11. [Google Scholar] [CrossRef]
- Cooper, K.M.; Whelan, M.; Kennedy, D.G.; Trigueros, G.; Cannavan, A.; Boon, P.E.; Wapperom, D.; Danaher, M. ProSafeBeef and anthelmintic drug residues-a case study in collaborative application of multi-analyte mass spectrometry to enhance consumer safety. Anal. Bioanal. Chem. 2012, 404, 1623–1630. [Google Scholar] [CrossRef]
- Imperiale, F.; Ortiz, P.; Cabrera, M.; Farias, C.; Sallovitz, J.M.; Iezzi, S.; Perez, J.; Alvarez, L.; Lanusse, C. Residual concentrations of the flukicidal compound triclabendazole in dairy cows’ milk and cheese. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2011, 28, 438–445. [Google Scholar] [CrossRef]
- Greer, A.W.; Kenyon, F.; Bartley, D.J.; Jackson, E.B.; Gordon, Y.; Donnan, A.A.; McBean, D.W.; Jackson, F. Development and field evaluation of a decision support model for anthelmintic treatments as part of a targeted selective treatment (TST) regime in lambs. Vet. Parasitol. 2009, 164, 12–20. [Google Scholar] [CrossRef]
- Cringoli, G.; Rinaldi, L.; Veneziano, V.; Mezzino, L.; Vercruysse, J.; Jackson, F. Evaluation of targeted selective treatments in sheep in Italy: Effects on faecal worm egg count and milk production in four case studies. Vet. Parasitol. 2009, 164, 36–43. [Google Scholar] [CrossRef]
- Gallidis, E.; Papadopoulos, E.; Ptoches, S.; Arsenos, G. The use of targeted selective treatments against gastrointestinal nematodes in milking sheep and goats in Greece based on parasitological and performance criteria. Vet. Parasitol. 2009, 164, 53–58. [Google Scholar] [CrossRef]
- Höglund, J.; Morrison, D.A.; Charlier, J.; Dimander, S.; Larrson, A. Targeted selective treatments for gastrointestinal nematodes in first-season grazing cattle based on mid-season daily weight gains. Vet. Parasitol. 2009, 164, 80–88. [Google Scholar] [CrossRef]
- Leask, R.; van Wyk, J.A.; Thompson, P.N.; Bath, G.F. The effect of application of the FAMACHA© system on selected production parameters in sheep. Small Rumin. Res. 2013, 110, 1–8. [Google Scholar] [CrossRef]
- Gaba, S.; Cabaret, J.; Chylinski, C.; Sauve, C.; Cortet, J.; Silvestre, A. Can efficient management of sheep gastro-intestinal nematodes be based on random treatment? Vet. Parasitol. 2012, 190, 178–184. [Google Scholar] [CrossRef]
- Terrill, T.H.; Miller, J.E.; Burke, J.M.; Mosjidis, J.A.; Kaplan, R.M. Experiences with integrated concepts for the control of Haemonchus contortus in sheep and goats in the United States. Vet. Parasitol. 2012, 186, 28–37. [Google Scholar] [CrossRef]
- Van Wyk, J.A.; Reynecke, D.P. Blueprint for an automated specific decision support system for countering anthelmintic resistance in Haemonchus spp. at farm level. Vet. Parasitol. 2011, 177, 212–223. [Google Scholar] [CrossRef]
- Gauly, M.; Bollwein, H.; Breves, G.; Bruegemann, K.; Daenicke, S.; Das, G.; Demeler, J.; Hansen, H.; Isselstein, J.; Koenig, S.; et al. Future consequences and challenges for dairy cow production systems arising from climate change in Central Europe—A review. Animal 2013, 7, 843–859. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Morgan, E.R.; Charlier, J.; Hendrickx, G.; Biggeri, A.; Catalan, D.; Von Samson-Himmelstjerna, G.; Demeler, J.; Müller, E.; Van Dijk, J.; Kenyon, F.; et al. Global Change and Helminth Infections in Grazing Ruminants in Europe: Impacts, Trends and Sustainable Solutions. Agriculture 2013, 3, 484-502. https://doi.org/10.3390/agriculture3030484
Morgan ER, Charlier J, Hendrickx G, Biggeri A, Catalan D, Von Samson-Himmelstjerna G, Demeler J, Müller E, Van Dijk J, Kenyon F, et al. Global Change and Helminth Infections in Grazing Ruminants in Europe: Impacts, Trends and Sustainable Solutions. Agriculture. 2013; 3(3):484-502. https://doi.org/10.3390/agriculture3030484
Chicago/Turabian StyleMorgan, Eric R., Johannes Charlier, Guy Hendrickx, Annibale Biggeri, Dolores Catalan, Georg Von Samson-Himmelstjerna, Janina Demeler, Elizabeth Müller, Jan Van Dijk, Fiona Kenyon, and et al. 2013. "Global Change and Helminth Infections in Grazing Ruminants in Europe: Impacts, Trends and Sustainable Solutions" Agriculture 3, no. 3: 484-502. https://doi.org/10.3390/agriculture3030484
APA StyleMorgan, E. R., Charlier, J., Hendrickx, G., Biggeri, A., Catalan, D., Von Samson-Himmelstjerna, G., Demeler, J., Müller, E., Van Dijk, J., Kenyon, F., Skuce, P., Höglund, J., O'Kiely, P., Van Ranst, B., De Waal, T., Rinaldi, L., Cringoli, G., Hertzberg, H., Torgerson, P., ... Vercruysse, J. (2013). Global Change and Helminth Infections in Grazing Ruminants in Europe: Impacts, Trends and Sustainable Solutions. Agriculture, 3(3), 484-502. https://doi.org/10.3390/agriculture3030484




