Addition of Fish Oil to Cream Cheese Affects Lipid Oxidation, Sensory Stability and Microstructure
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of 70% Fish Oil-in-Water Emulsions
2.3. Production of Fish Oil Enriched Cream Cheese
2.4. Storage and Sampling
2.5. Characterization of the Delivery Emulsions and Cream Cheeses
2.5.1. Droplet Size and pH
2.5.2. Confocal Laser Scanning Microscopy
2.6. Lipid Extraction, and Analyses of Fatty Acid Compositions and Tocopherols
2.7. Measurements of Lipid Oxidation
2.7.1. Primary Oxidation Products—Peroxide Values
2.7.2. Secondary Oxidation Products—Dynamic Headspace GC-MS
2.8. Sensory Evaluation
2.9. Statistical Analyses
3. Results
3.1. Characterization of the Neat Oil and Delivery Emulsions
| SFA | MUFA | PUFA | |||
|---|---|---|---|---|---|
| Fatty acid | Av [%] | Fatty acid | Av [%] | Fatty acid | Av [%] |
| 14:0 | 3.0 | 16:1 (n-6) | 8.2 | 18:2 (n-6) | 1.8 |
| 16:0 | 8.9 | 18:1 (n-9) | 16.0 | 18:3 (n-3) | 0.8 |
| 18:0 | 1.9 | 18:1 (n-7) | 5.2 | 18:4 (n-3) | 3.5 |
| 20:1 (n-9+n-11) | 11.6 | 20:4 (n-3) | 0.7 | ||
| 22:1 (n-11) | 6.1 | 20:5 (n-3) | 9.3 | ||
| 22:1 (n-9) | 0.8 | 22:5 (n-3) | 1.1 | ||
| 22:6 (n-3) | 11.6 | ||||
| Total | 13.8 | Total | 48.3 | Total | 29.4 |
| Total n-3 | 26.6 |
| Fish oil | Em_CAS | Em_WPI | Em_MPL20 | |
|---|---|---|---|---|
| Emulsifier type | -- | CAS | WPI | MPL20 |
| pH | -- | 7.1 | 6.8 | 7.3 |
| PV [meq peroxides/kg oil] | 0.1 ± 0.0 | 0.2 ± 0.0 | 0.1 ± 0.0 | 0.3 ± 0.0 |
| Droplet size, D3,2 [µm] | -- | 8.3 | 21.2 | 12.6 |
| α-tocopherol [µg/g oil] | 207 ± 16 | -- | -- | -- |
| γ-tocopherol [µg/ g oil] | 100 ± 1 | -- | -- | -- |
3.2. Characterization of the Cream Cheeses
| Cream cheese sample | pH | α-tocopherol [µg/g oil] | γ-tocopherol [µg/g oil] | |||
|---|---|---|---|---|---|---|
| Week 0 | Week 20 | Week 0 | Week 20 | Week 0 | Week 20 | |
| CAS | 4.8 | 4.7 | 26 ± 5b | 12 ± 0ab | 8 ± 1b | 3 ± 0b |
| WPI | 4.8 | 4.7 | 35 ± 1c | 11 ± 0a | 10 ± 0d | 4 ± 0c |
| MPL20 | 4.8 | 4.7 | 30 ± 3bc | 8 ± 0a | 9 ± 0c | 4 ± 0c |
| FO | 4.8 | 4.7 | 31 ± 1bc | 8 ± 1a | 9 ± 0c | 4 ± 0c |
| REF | 4.8 | 4.7 | 16 ± 1a | 17 ± 0b | 1 ± 0a | 0 ± 0a |

3.2.1. Contents of Tocopherol
3.3. Lipid Oxidation in Emulsions
3.3.1. Peroxide Values
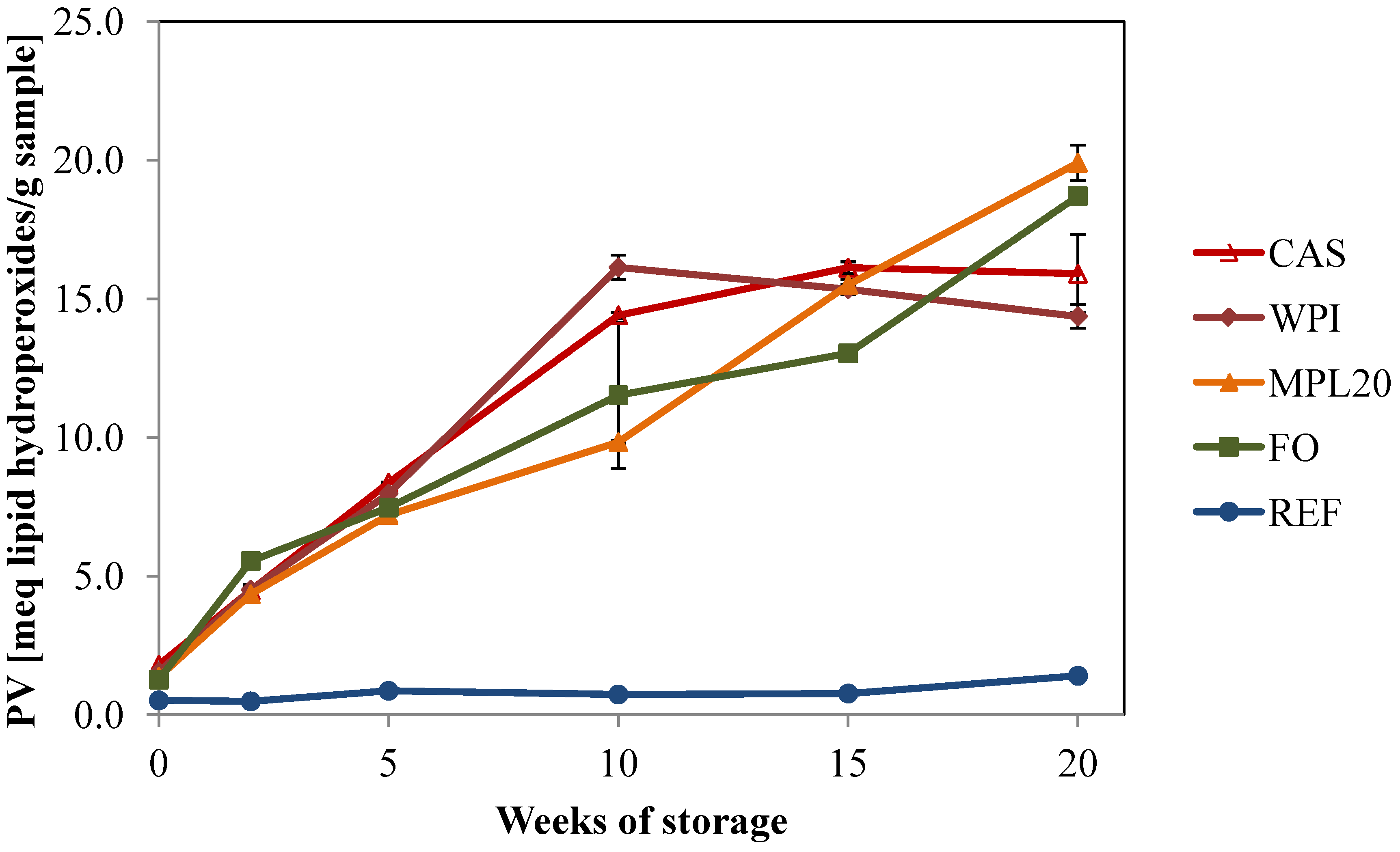
3.3.2. Secondary Volatile Oxidation Products

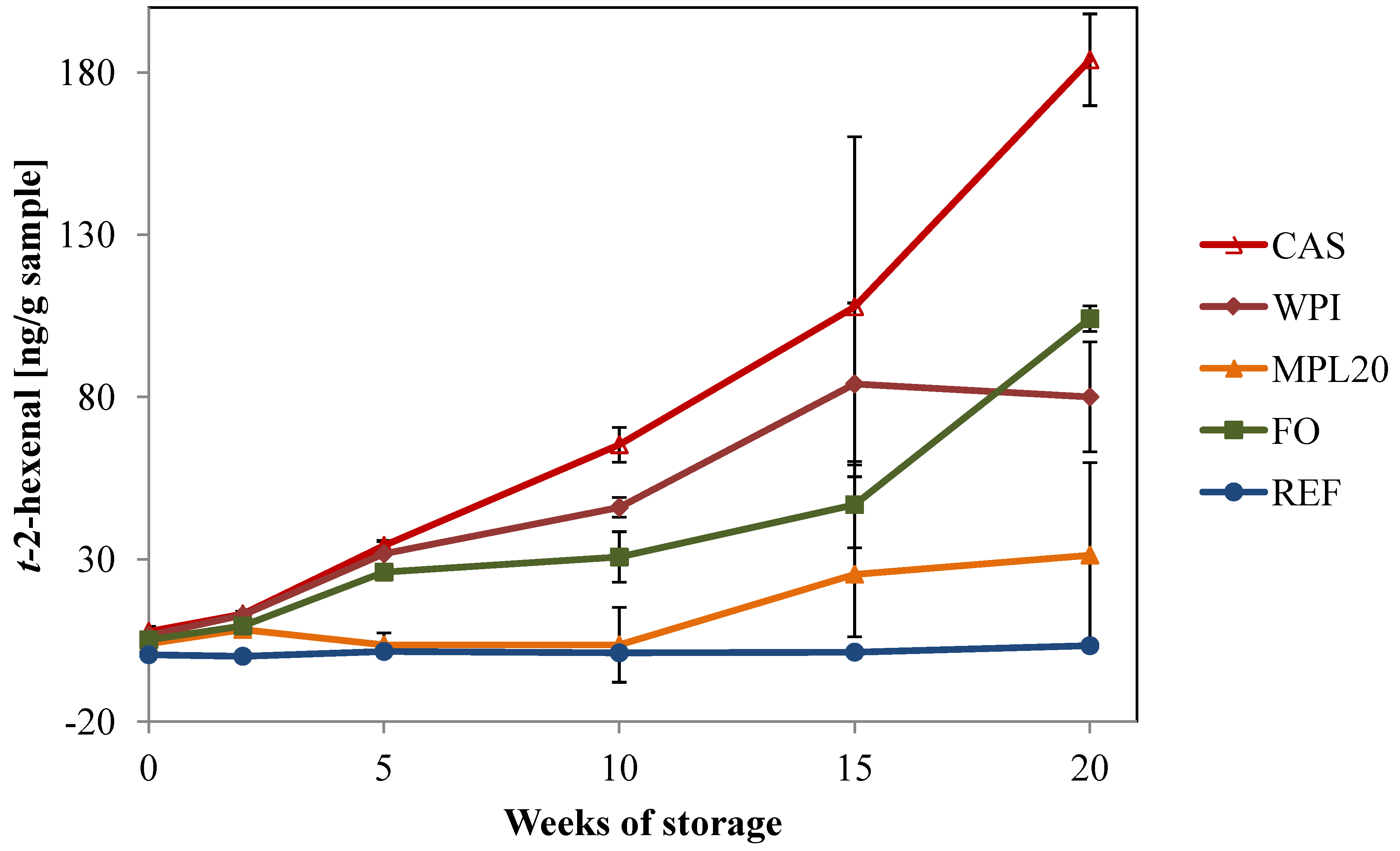
3.3.3. Sensory Profiling of the Cream Cheeses
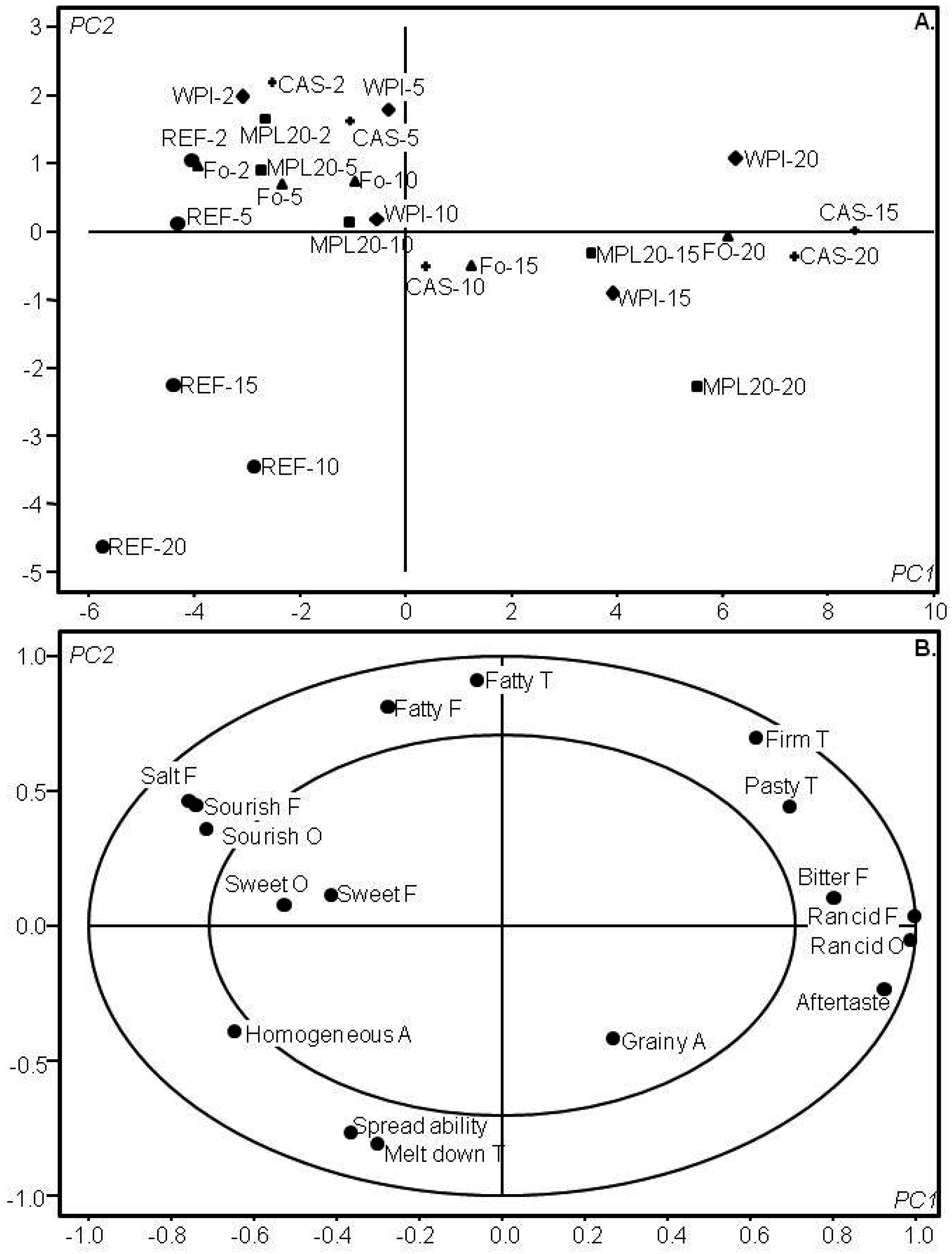
4. Discussion
5. Conclusions and Perspectives
Supplementary Material
Acknowledgments
References
- Riediger, N.D.; Othman, R.A.; Suh, M.; Moghadasian, M.H. A systemic review of the roles of n-3 fatty acids in health and disease. J. Am. Diet. Assoc. 2009, 109, 668–679. [Google Scholar] [CrossRef]
- Let, M.B.; Jacobsen, C.; Meyer, A.S. Lipid oxidation in milk, yoghurt, and salad dressing enriched with neat fish oil or pre-emulsified fish oil. J. Agric. Food Chem. 2007, 55, 7802–7809. [Google Scholar] [CrossRef]
- Ye, A.; Cui, J.; Taneja, A.; Zhu, X.; Singh, H. Evaluation of processed cheese fortified with fish oil emulsion. Food Res. Int. 2009, 42, 1093–1098. [Google Scholar] [CrossRef]
- Kolanowski, W.; Weißrodt, J. Sensory quality of dairy products fortified with fish oil. Int. Dairy J. 2007, 17, 1248–1253. [Google Scholar] [CrossRef]
- EU Commision Regulation No 116/2010. Off. J. Eur. Union 2010, 53, L37:16–L37:18.
- Horn, A.F.; Nielsen, N.S.; Andersen, U.; Søgaard, L.H.; Horsewell, A.; Jacobsen, C. Oxidative stability of 70% fish oil-in-water emulsions: Impact of emulsifiers and pH. Eur. J. Lipid Sci. Technol. 2011, 113, 1243–1257. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- AOCS Official Method Ca 2-66. In Preparation of Methyl Esters of Long Chain Fatty Acids; AOCS: Champaign, IL, USA, 1998.
- AOCS Official Method Ce 1b-89. In Fatty Acid Composition by GLC, Marine Oils; AOCS: Champaign, IL, USA, 1998.
- AOCS Official Method Ce 8-89. In Determination of Tocopherols and Tocotrienols in Vegetable Oils and Fats by HPLC; AOCS: Champaign, IL, USA, 1998.
- Shantha, N.C.; Decker, E.A. Rapid, sensitive, iron-based spectrophotometric methods for determination of peroxide values of food lipids. J. AOAC Int. 1994, 77, 421–424. [Google Scholar]
- ISO 1993. In ISO Standards 8586-1. Sensory Analysis-General Guidance for the Selection, Training and Monitoring of Assessors. Part 1. Selected Assessors, 1st ed; International Standard Organization: Geneva, Switzerland, 1993.
- ISO 1994. In ISO Standards 11035. Sensory Analysis-Identification and Selection of Descriptors for Establishing a Sensory Profile by a Multidimensional Approach, 1st ed; International Standard Organization: Geneva, Switzerland; p. 1994.
- ISO 1988. In ISO Standards 8589. Sensory Analysis-General Guidance for Design of Test Rooms, 1st ed; International Standard Organization: Geneva, Switzerland, 1988.
- Thybo, A.K.; Martens, M. Analysis of sensory assessors in texture profiling of potatoes by multivariate modelling. Food Qual. Prefer. 2000, 11, 283–288. [Google Scholar] [CrossRef]
- Horn, A.F.; Nielsen, N.S.; Jacobsen, C. Iron-mediated lipid oxidation in 70% fish oil-in-water emulsions: Effect of emulsifier type and pH. Int. J. Food Sci. Technol. 2012, 47, 1097–1108. [Google Scholar] [CrossRef]
- Faraji, H.; McClements, D.J.; Decker, E.A. Role of continuous phase protein on the oxidative stability of fish oil-in-water emulsions. J. Agric. Food Chem. 2004, 52, 4558–4564. [Google Scholar]
- Ries, D.; Ye, A.; Haisman, D.; Singh, H. Antioxidant properties of caseins and whey proteins in model oil-in-water emulsions. Int. Dairy J. 2010, 20, 72–78. [Google Scholar] [CrossRef]
- Nielsen, N.S.; Jacobsen, C. Methods for reducing lipid oxidation in fish-oil-enriched energy bars. Int. J. Food Sci. Technol. 2009, 44, 1536–1546. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Horn, A.F.; Green-Petersen, D.; Nielsen, N.S.; Andersen, U.; Hyldig, G.; Jensen, L.H.S.; Horsewell, A.; Jacobsen, C. Addition of Fish Oil to Cream Cheese Affects Lipid Oxidation, Sensory Stability and Microstructure. Agriculture 2012, 2, 359-375. https://doi.org/10.3390/agriculture2040359
Horn AF, Green-Petersen D, Nielsen NS, Andersen U, Hyldig G, Jensen LHS, Horsewell A, Jacobsen C. Addition of Fish Oil to Cream Cheese Affects Lipid Oxidation, Sensory Stability and Microstructure. Agriculture. 2012; 2(4):359-375. https://doi.org/10.3390/agriculture2040359
Chicago/Turabian StyleHorn, Anna Frisenfeldt, Ditte Green-Petersen, Nina Skall Nielsen, Ulf Andersen, Grethe Hyldig, Louise Helene Søgaard Jensen, Andy Horsewell, and Charlotte Jacobsen. 2012. "Addition of Fish Oil to Cream Cheese Affects Lipid Oxidation, Sensory Stability and Microstructure" Agriculture 2, no. 4: 359-375. https://doi.org/10.3390/agriculture2040359






