Aging Impairs the Proliferative Capacity of Cardiospheres, Cardiac Progenitor Cells and Cardiac Fibroblasts: Implications for Cell Therapy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cardiosphere Culture
2.2. Flow Cytometry
2.3. Myocardial Infarction
2.4. Cardiac Fibroblast Isolation and Primary Culture
2.5. Co-Culture of CPCs and Cardiac Fibroblasts
3. Results
3.1. Reduction in Cardiosphere Growth and Cardiac Progenitor Cells with Aging
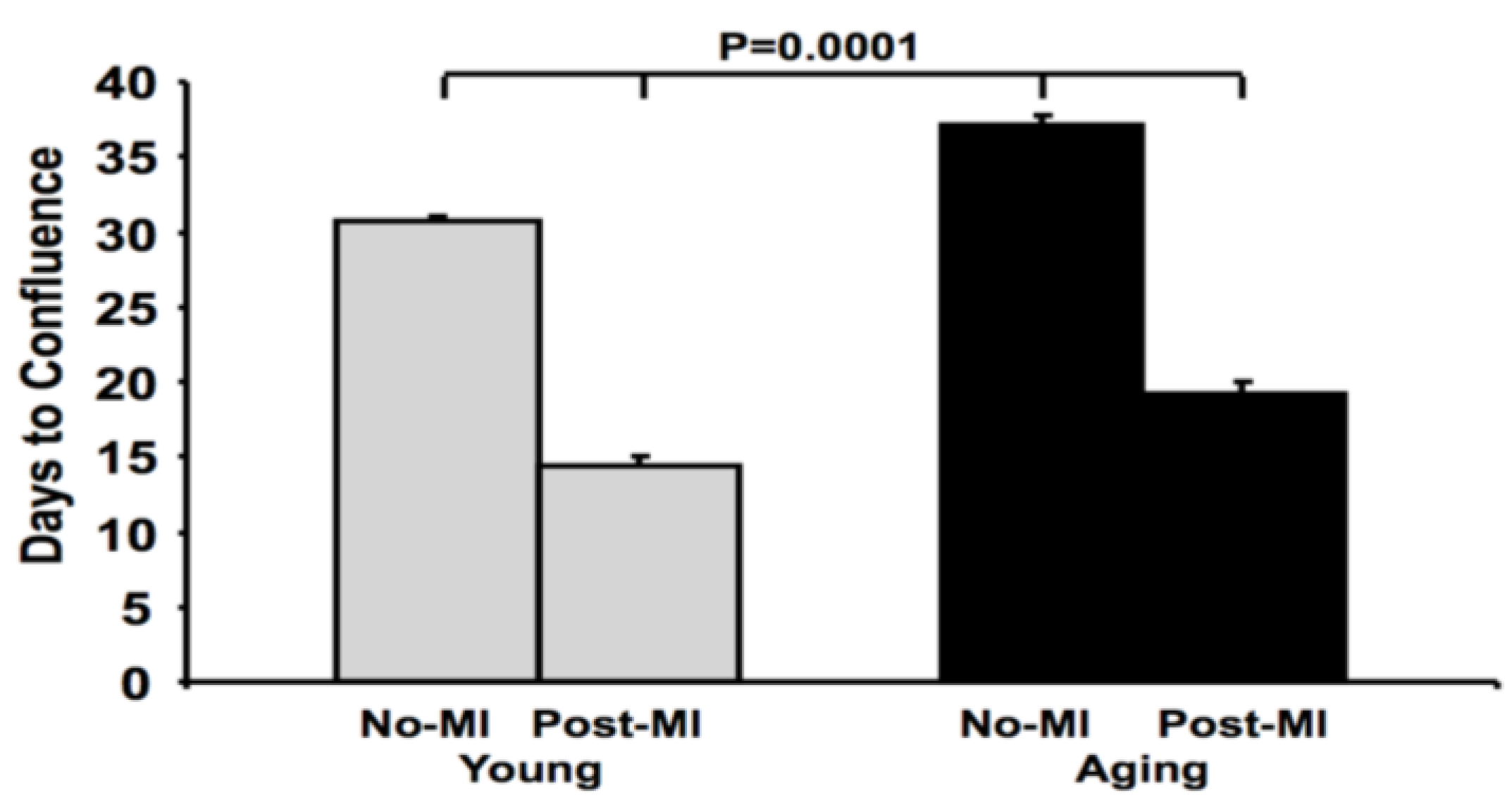
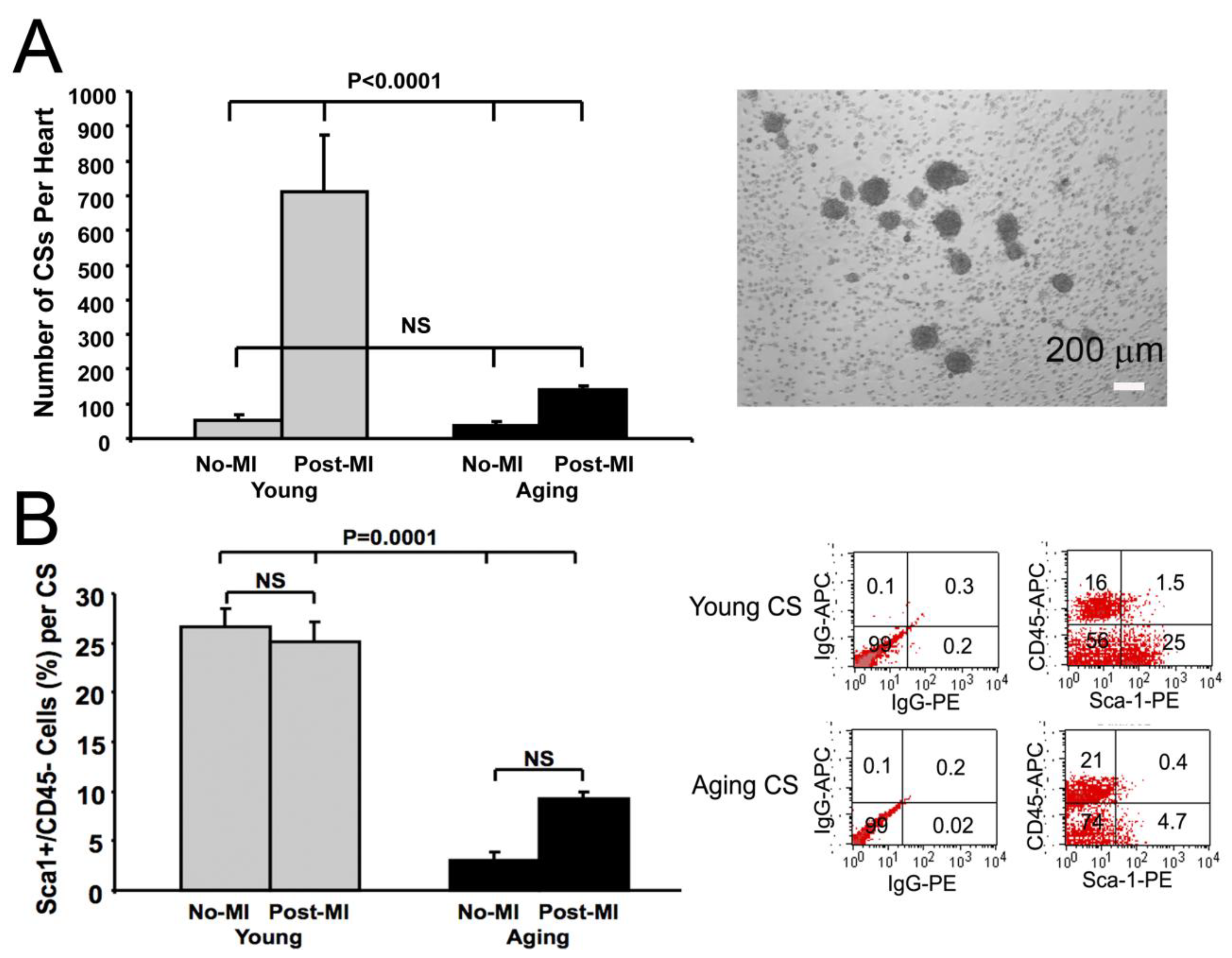
3.2. Impaired Cardiac Fibroblast Proliferation with Age
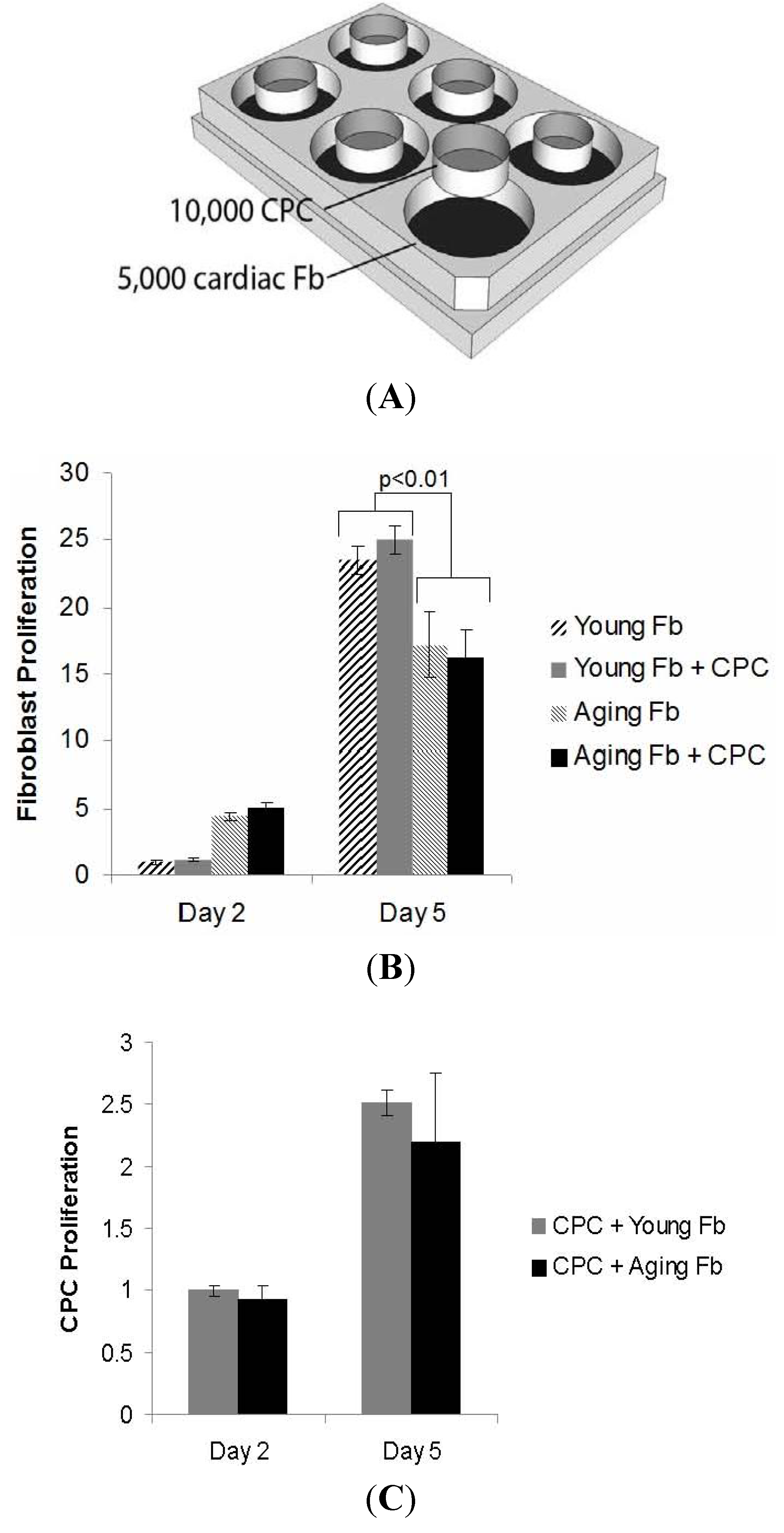
3.3. Aging Fibroblasts Have No Paracrine Signaling Effect on CPC Proliferation
4. Discussion

5. Conclusions
Acknowledgements
Conflicts of Interest
References
- National Heart Lung and Blood Institute, Incidence and Prevalence. In 2006 Chart Book on Cardiovascular and Lung Diseases; National Institutes of Health: Bethesday, MD, USA, 2006; pp. 30–31.
- Maggioni, A.A.; Maseri, A.; Fresco, C.; Franzosi, M.G.; Mauri, F.; Santoro, E.; Tognoni, G. The investigators of the gruppo italiano por lo studio della sopravvivenza nell’infarto M: Age-related increase in mortality among patients with first myocardial infarctions treated with thrombolysis. N. Engl. J. Med. 1993, 329, 1442–1448. [Google Scholar] [CrossRef]
- Shih, H.; Lee, B.; Lee, R.J.; Boyle, A.J. The aging heart and post-infarction left ventricular remodeling. J. Am. Coll. Cardiol. 2011, 57, 9–17. [Google Scholar] [CrossRef]
- Anversa, P.; Rota, M.; Urbanek, K.; Hosoda, T.; Sonnenblick, E.; Leri, A.; Kajstura, J.; Bolli, R. Myocardial aging—A stem cell problem. Bas. Res. Cardiol. 2005, 100, 482–493. [Google Scholar] [CrossRef]
- Ye, J.; Boyle, A.J.; Shih, H.; Sievers, R.E.; Zhang, Y.; Prasad, M.; Su, H.; Zhou, Y.; Grossman, W.; Bernstein, H.S.; et al. Sca-1+ cardiosphere-derived cells are enriched for isl1-expressing cardiac precursors and improve cardiac function after myocardial injury. PLoS One 2012, 7, e30329. [Google Scholar] [CrossRef]
- Ye, J.; Boyle, A.; Shih, H.; Sievers, R.; Wang, Z.-E.; Gormley, M.; Yeghiazarians, Y. CD45 positive cells are not an essential component in cardiosphere formation. Cell Tissue Res. 2013, 351, 201–205. [Google Scholar] [CrossRef]
- Li, T.-S.; Cheng, K.; Lee, S.-T.; Matsushita, S.; Davis, D.; Malliaras, K.; Zhang, Y.; Matsushita, N.; Smith, R.R.; Marbán, E. Cardiospheres recapitulate a niche-like microenvironment rich in stemness and cell-matrix interactions, rationalizing their enhanced functional potency for myocardial repair. Stem Cells 2010, 28, 2088–2098. [Google Scholar] [CrossRef]
- Makkar, R.R.; Smith, R.R.; Cheng, K.; Malliaras, K.; Thomson, L.E.J.; Berman, D.; Czer, L.S.C.; Marban, L.; Mendizabal, A.; Johnston, P.V.; et al. Intracoronary cardiosphere-derived cells for heart regeneration after myocardial infarction (CADUCEUS): A prospective, randomised phase 1 trial. Lancet 2012, 379, 895–904. [Google Scholar] [CrossRef]
- Banerjee, I.; Fuseler, J.W.; Price, R.L.; Borg, T.K.; Baudino, T.A. Determination of cell types and numbers during cardiac development in the neonatal and adult rat and mouse. Am. J. Physiol. Heart Circ. Physiol. 2007, 293, 1883–1891. [Google Scholar] [CrossRef]
- Smith, R.R.; Barile, L.; Cho, H.C.; Leppo, M.K.; Hare, J.M.; Messina, E.; Giacomello, A.; Abraham, M.R.; Marban, E. Regenerative potential of cardiosphere-derived cells expanded from percutaneous endomyocardial biopsy specimens. Circulation 2007, 115, 896–908. [Google Scholar] [CrossRef]
- Lindsey, M.L.; Goshorn, D.K.; Squires, C.E.; Escobar, G.P.; Hendrick, J.W.; Mingoia, J.T.; Sweterlitsch, S.E.; Spinale, F.G. Age-dependent changes in myocardial matrix metalloproteinase/tissue inhibitor of metalloproteinase profiles and fibroblast function. Cardiovasc. Res. 2005, 66, 410–419. [Google Scholar] [CrossRef]
- Messina, E.; de Angelis, L.; Frati, G.; Morrone, S.; Chimenti, S.; Fiordaliso, F.; Salio, M.; Battaglia, M.; Latronico, M.V.G.; Coletta, M.; et al. Isolation and expansion of adult cardiac stem cells from human and murine heart. Circ. Res. 2004, 95, 911–921. [Google Scholar] [CrossRef]
- Yeghiazarians, Y.; Zhang, Y.; Prasad, M.; Shih, H.; Saini, S.A.; Takagawa, J.; Sievers, R.E.; Wong, M.L.; Kapasi, N.K.; Mirsky, R.; et al. Injection of bone marrow cell extract into infarcted hearts results in functional improvement comparable to intact cell therapy. Mol. Ther. 2009, 17, 1250–1256. [Google Scholar] [CrossRef]
- Roger, V.L.; Go, A.S.; Lloyd-Jones, D.M.; Benjamin, E.J.; Berry, J.D.; Borden, W.B.; Bravata, D.M.; Dai, S.; Ford, E.S.; Fox, C.S.; et al. Heart disease and stroke statistics—2012 update. A report from the American Heart Association. Circulation 2012, 125. [Google Scholar] [CrossRef]
- Wang, X.; Takagawa, J.; Haddad, D.J.; Pinnamaneni, K.; Zhang, Y.; Sievers, R.E.; Grossman, W.; Yeghiazarians, Y.; Springer, M.L. Advanced donor age impairs bone marrow cell therapeutic efficacy for cardiac disease. J. Tissue Sci. Eng. 2011, 3, 2. [Google Scholar]
- Fan, M.; Chen, W.; Liu, W.; Du, G.-Q.; Jiang, S.-L.; Tian, W.-C.; Sun, L.; Li, R.-K.; Tian, H. The effect of age on the efficacy of human mesenchymal stem cell transplantation after a myocardial infarction. Rejuv. Res. 2010, 13, 429–438. [Google Scholar] [CrossRef]
- Khan, M.; Mohsin, S.; Khan, S.N.; Riazuddin, S. Repair of senescent myocardium by mesenchymal stem cells is dependent on the age of donor mice. J. Cell. Mol. Med. 2011, 15, 1515–1527. [Google Scholar] [CrossRef]
- Pallante, B.A.; Duignan, I.; Okin, D.; Chin, A.; Bressan, M.C.; Mikawa, T.; Edelberg, J.M. Bone marrow Oct3/4+ cells differentiate into cardiac myocytes via age-dependent paracrine mechanisms. Circ. Res. 2007, 100, 1–11. [Google Scholar] [CrossRef]
- Kan, C.-D.; Li, S.-H.; Weisel, R.D.; Zhang, S.; Li, R.-K. Recipient age determines the cardiac functional improvement achieved by skeletal myoblast transplantation. J. Am. Coll. Cardiol. 2007, 50, 1086–1092. [Google Scholar] [CrossRef]
- Boyle, A.J.; Shih, H.; Hwang, J.; Ye, J.; Lee, B.; Zhang, Y.; Kwon, D.; Jun, K.; Zheng, D.; Sievers, R.; et al. Cardiomyopathy of aging in the mammalian heart is characterized by myocardial hypertrophy, fibrosis and a predisposition towards cardiomyocyte apoptosis and autophagy. Exp. Gerontol. 2011, 46, 549–559. [Google Scholar] [CrossRef]
- Yuan, R.; Tsaih, S.-W.; Petkova, S.B.; Evsikova, C.M.D.; Xing, S.; Marion, M.A.; Bogue, M.A.; Mills, K.D.; Peters, L.L.; Bult, C.J.; et al. Aging in inbred strains of mice: Study design and interim report on median lifespans and circulating IGF1 levels. Aging Cell 2009, 8, 277–287. [Google Scholar] [CrossRef]
- Cieslik, K.A.; Trial, J.; Carlson, S.; Taffet, G.E.; Entman, M.L. Aberrant differentiation of fibroblast progenitors contributes to fibrosis in the aged murine heart: Role of elevated circulating insulin levels. FASEB J. 2013, 27, 1761–1771. [Google Scholar] [CrossRef]
- Forte, E.; Miraldi, F.; Chimenti, I.; Angelini, F.; Zeuner, A.; Giacomello, A.; Mercola, M.; Messina, E. TGFβ-dependent epithelial-to-mesenchymal transition is required to generate cardiospheres from human adult heart biopsies. Stem Cells Dev. 2012, 21, 3081–3090. [Google Scholar] [CrossRef]
- Leri, A.; Kajstura, J.; Anversa, P. Cardiac stem cells and mechanisms of myocardial regeneration. Physiol. Rev. 2005, 85, 1373–1416. [Google Scholar] [CrossRef]
- Urbanek, K.; Cesselli, D.; Rota, M.; Nascimbene, A.; de Angelis, A.; Hosoda, T.; Bearzi, C.; Boni, A.; Bolli, R.; Kajstura, J.; et al. Stem cell niches in the adult mouse heart. PNAS 2006, 103, 9226–9231. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ye, J.; Hom, D.S.; Hwang, J.; Yeghiazarians, Y.; Lee, R.J.; Boyle, A.J. Aging Impairs the Proliferative Capacity of Cardiospheres, Cardiac Progenitor Cells and Cardiac Fibroblasts: Implications for Cell Therapy. J. Clin. Med. 2013, 2, 103-114. https://doi.org/10.3390/jcm2030103
Ye J, Hom DS, Hwang J, Yeghiazarians Y, Lee RJ, Boyle AJ. Aging Impairs the Proliferative Capacity of Cardiospheres, Cardiac Progenitor Cells and Cardiac Fibroblasts: Implications for Cell Therapy. Journal of Clinical Medicine. 2013; 2(3):103-114. https://doi.org/10.3390/jcm2030103
Chicago/Turabian StyleYe, Jianqin, Douglas S. Hom, Joy Hwang, Yerem Yeghiazarians, Randall J. Lee, and Andrew J. Boyle. 2013. "Aging Impairs the Proliferative Capacity of Cardiospheres, Cardiac Progenitor Cells and Cardiac Fibroblasts: Implications for Cell Therapy" Journal of Clinical Medicine 2, no. 3: 103-114. https://doi.org/10.3390/jcm2030103
APA StyleYe, J., Hom, D. S., Hwang, J., Yeghiazarians, Y., Lee, R. J., & Boyle, A. J. (2013). Aging Impairs the Proliferative Capacity of Cardiospheres, Cardiac Progenitor Cells and Cardiac Fibroblasts: Implications for Cell Therapy. Journal of Clinical Medicine, 2(3), 103-114. https://doi.org/10.3390/jcm2030103



