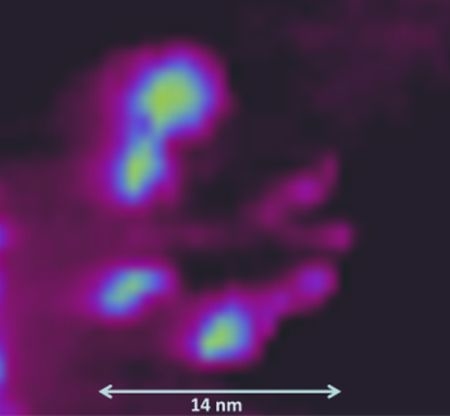
Microscopic Analysis of Current and Mechanical Properties of Nafion® Studied by Atomic Force Microscopy
Abstract
:1. Introduction
2. Experimental Section
2.1. Sample Preparation
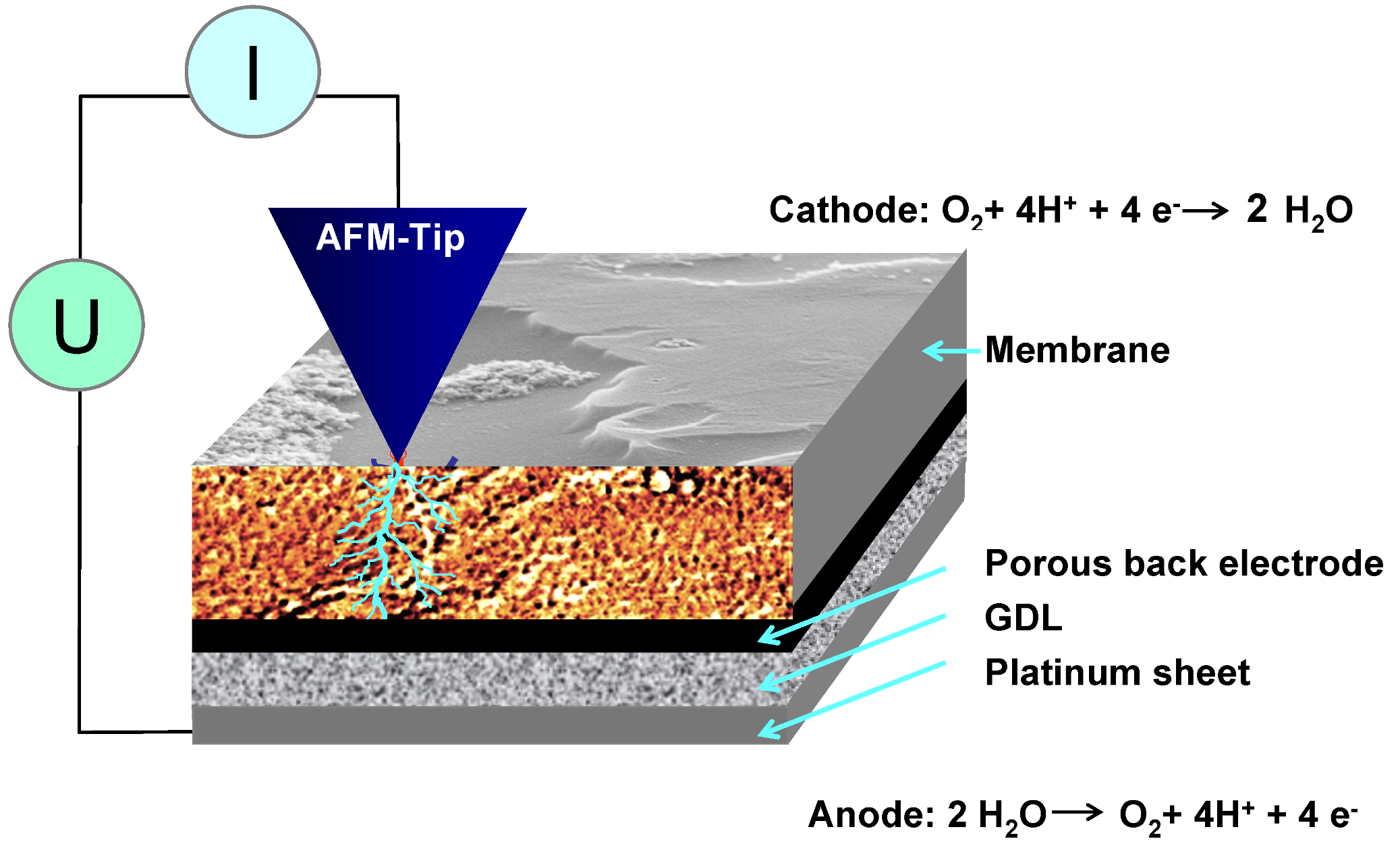
2.2. AFM Measurements

2.3. Calibration of Adhesion Force and Energy Dissipation
3. Results and Discussion
3.1. Activation of Protonated Nafion®
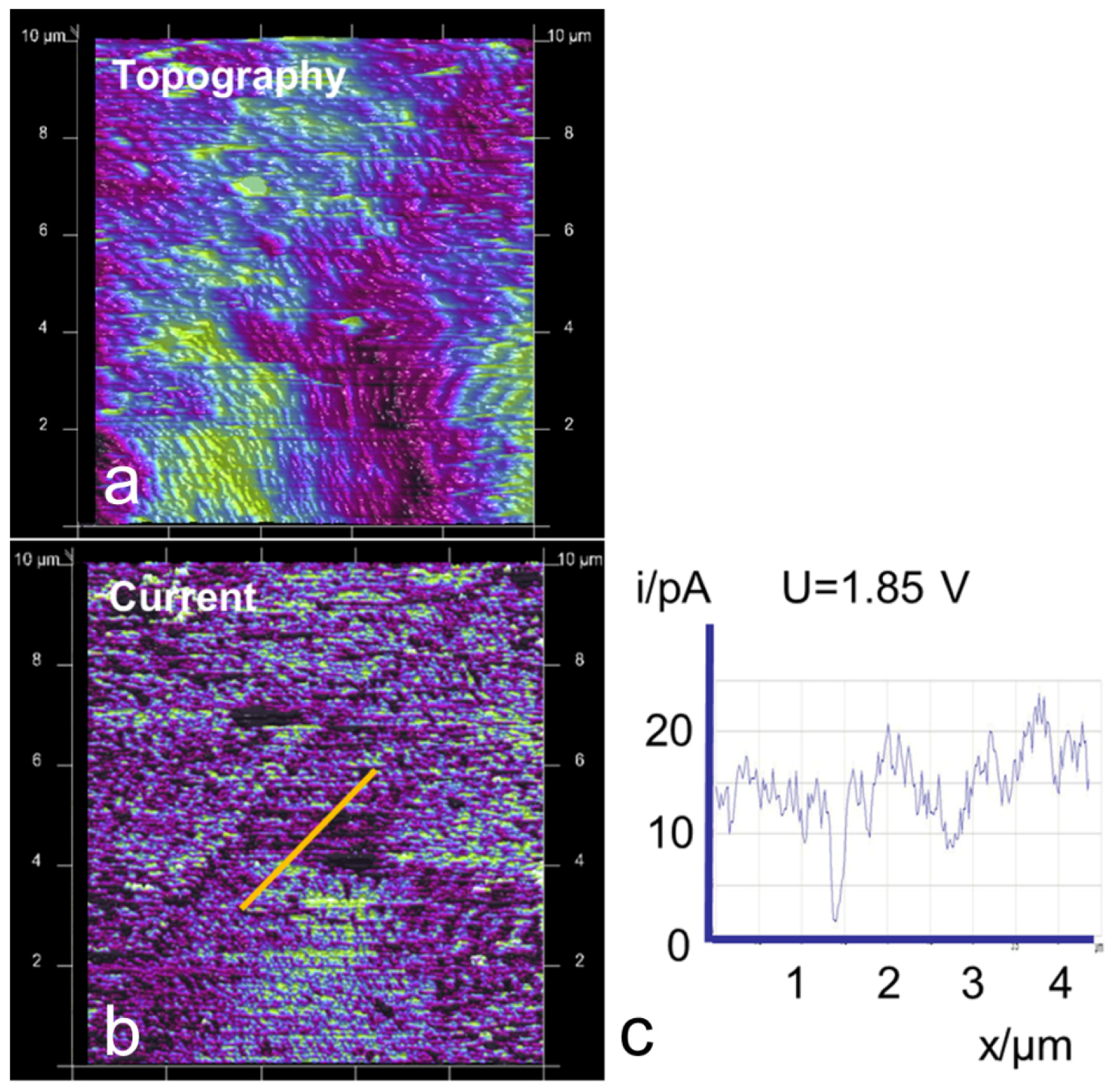
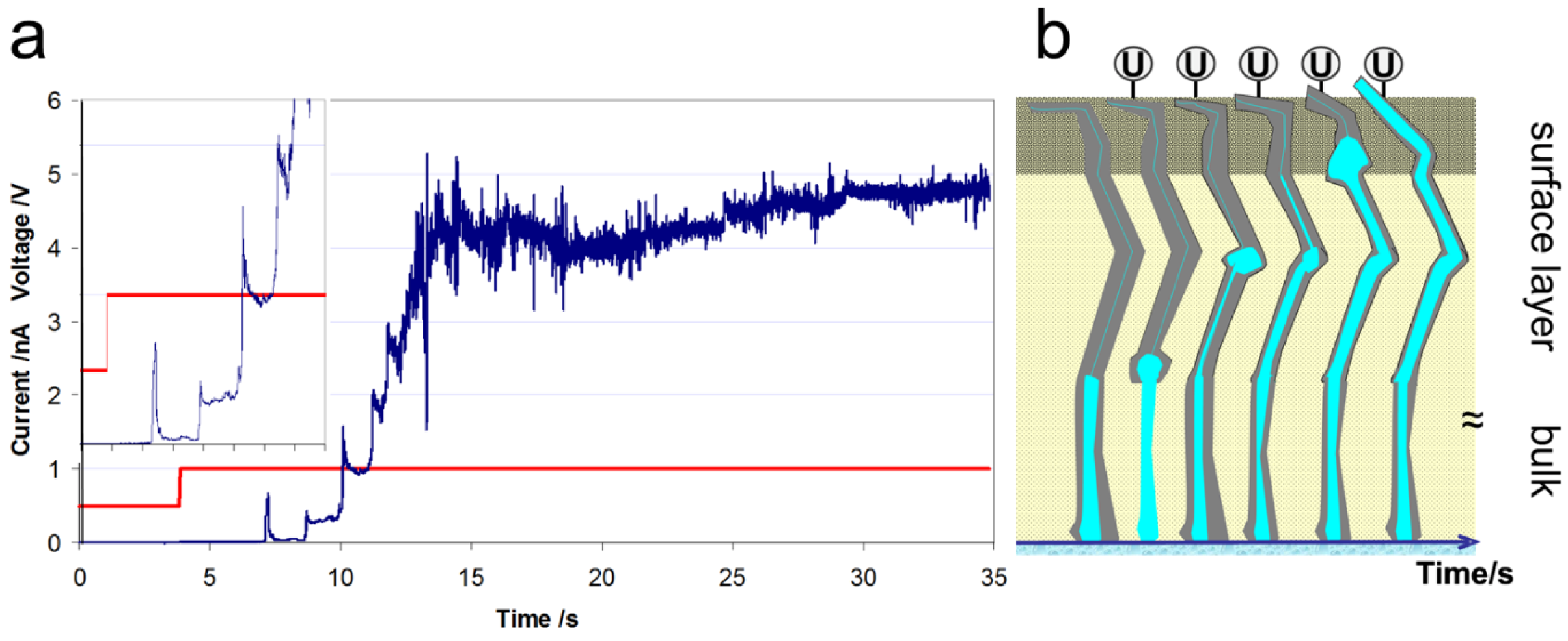
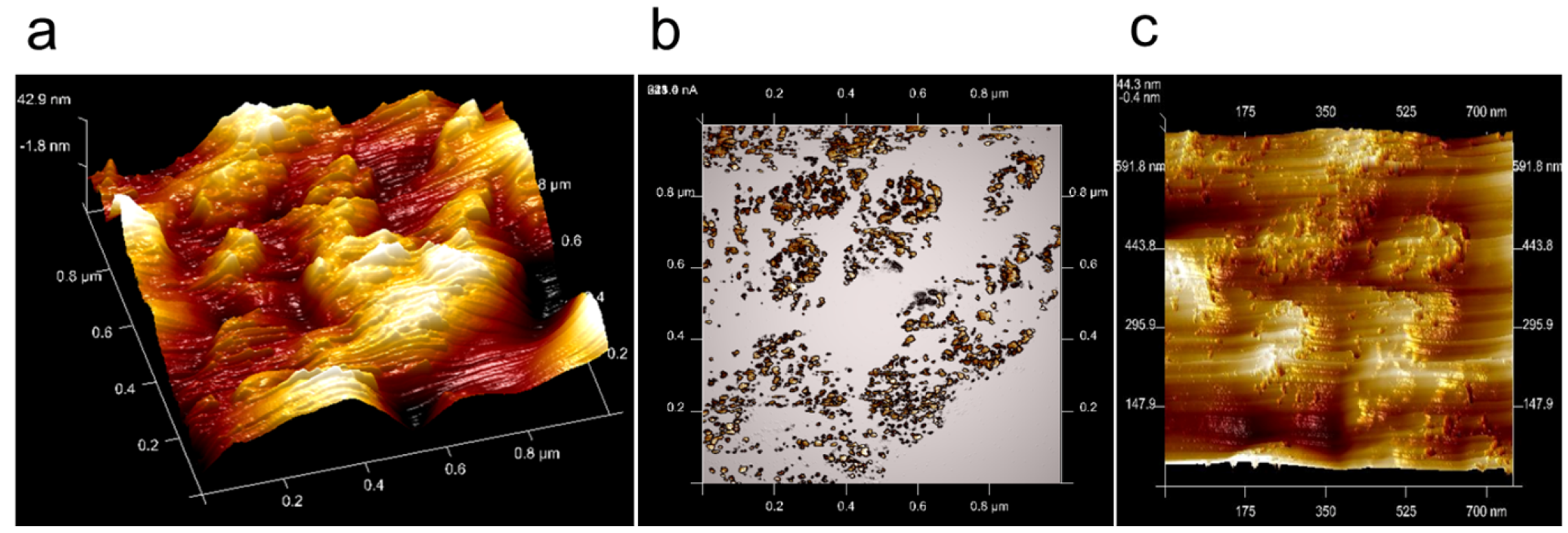
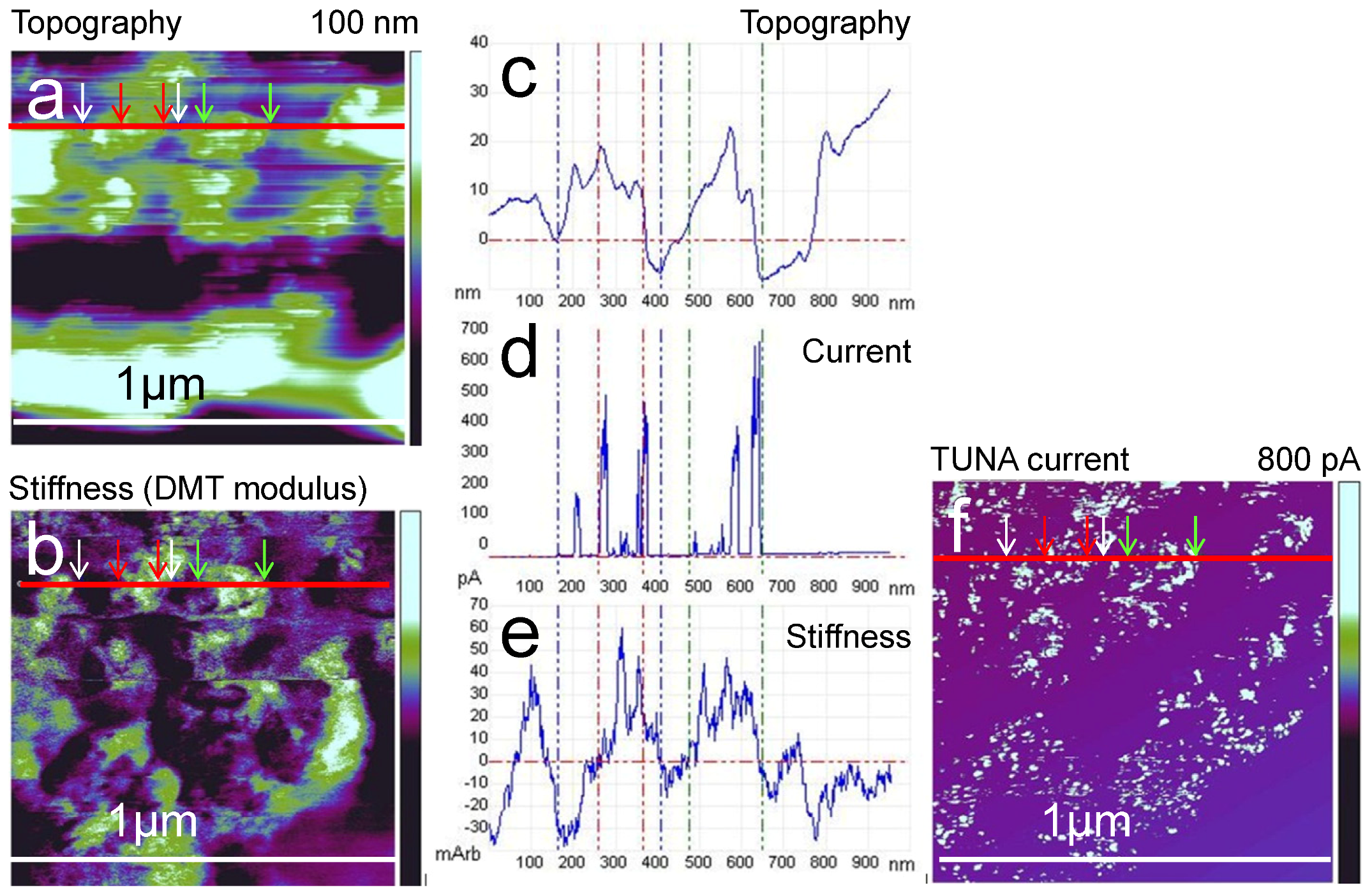
3.2. AFM Measurements of Topography and Current
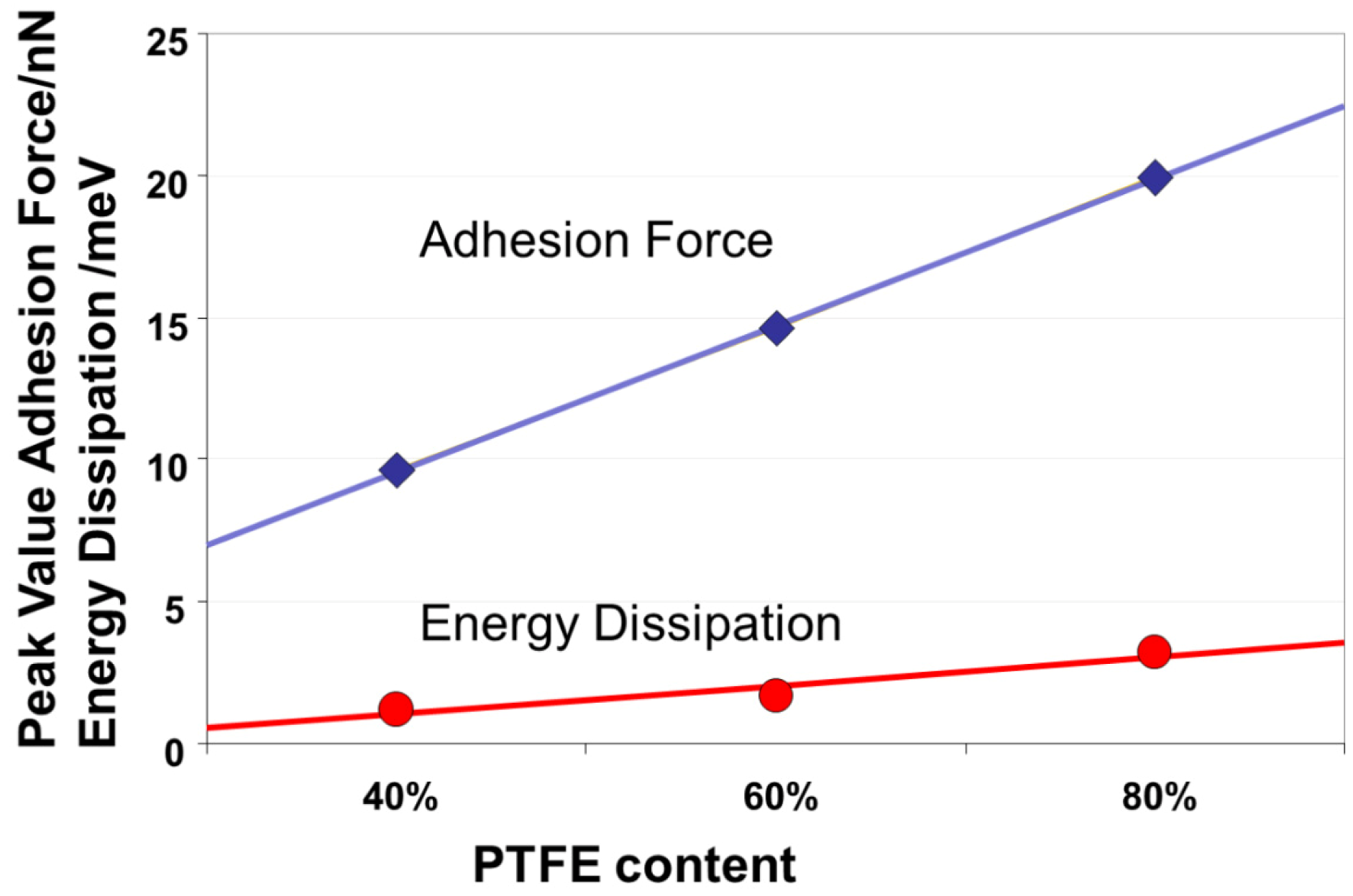
3.3. Mechanical Properties


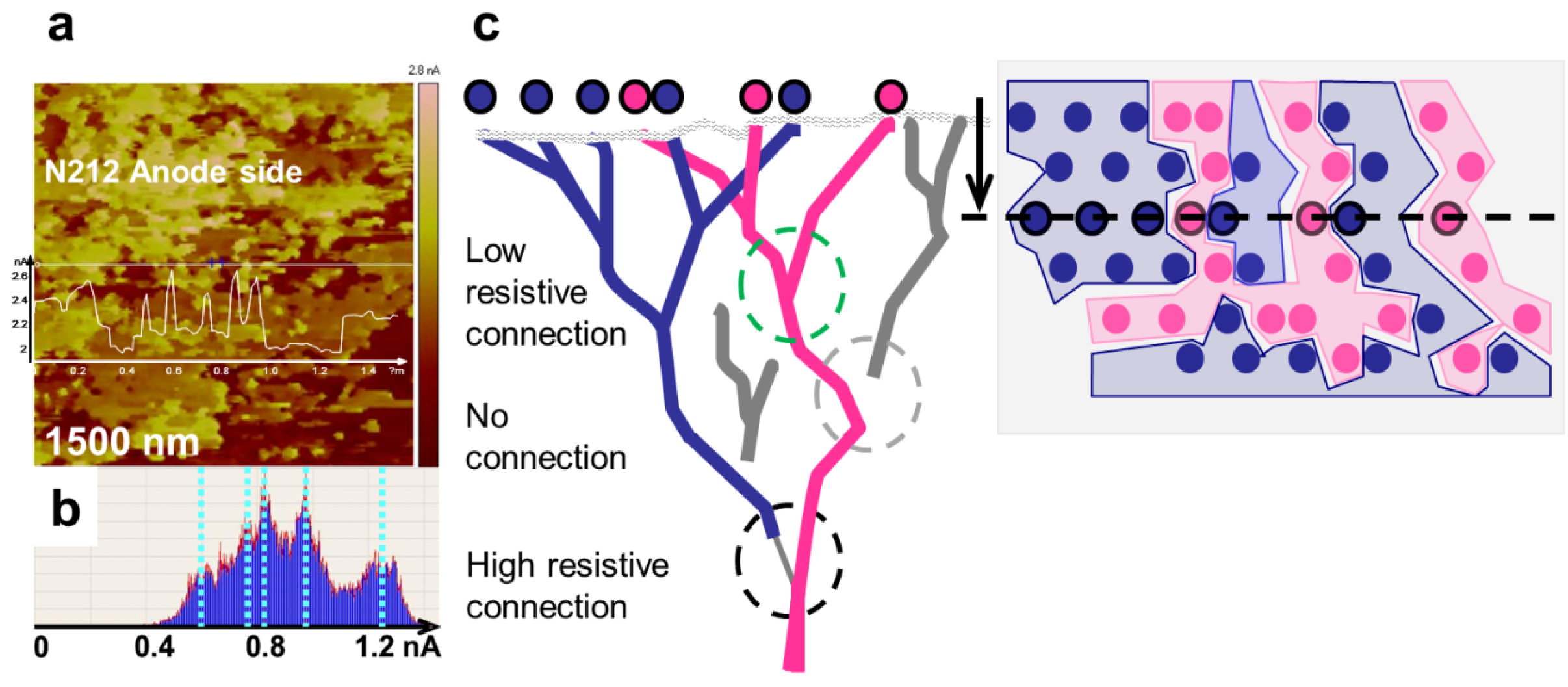
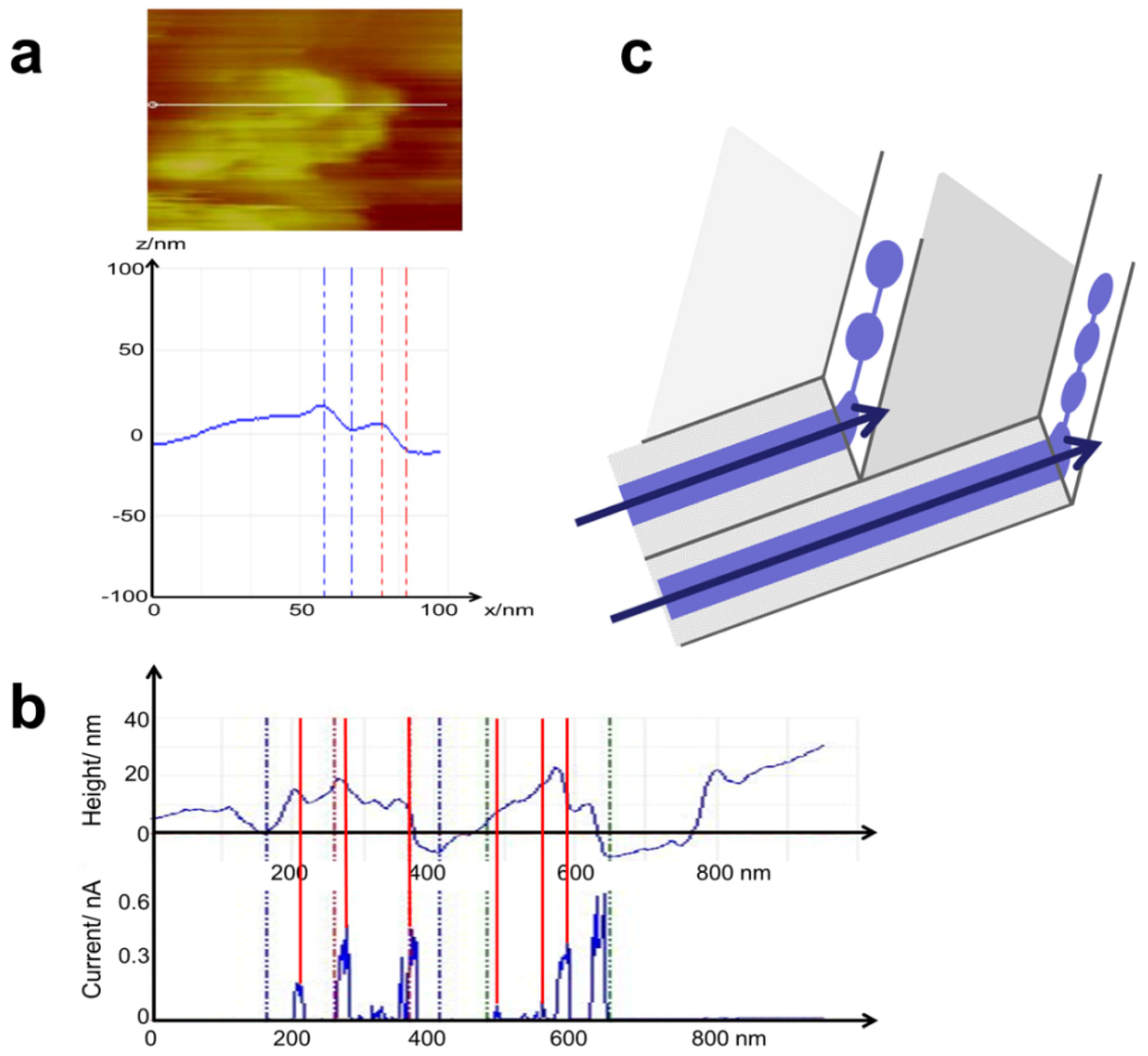
3.4. Time Dependence of Conductive Channels

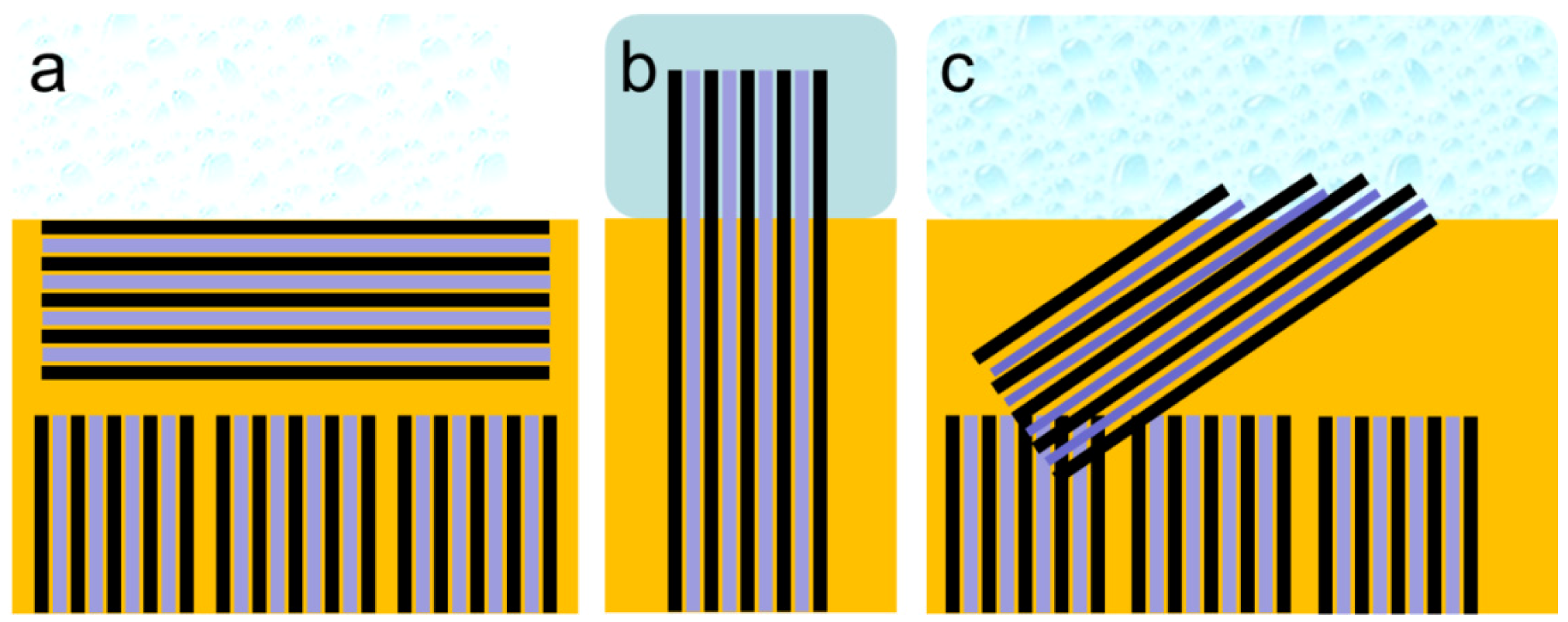
3.5. Comparison of Nanostructure Observations with Literature Models
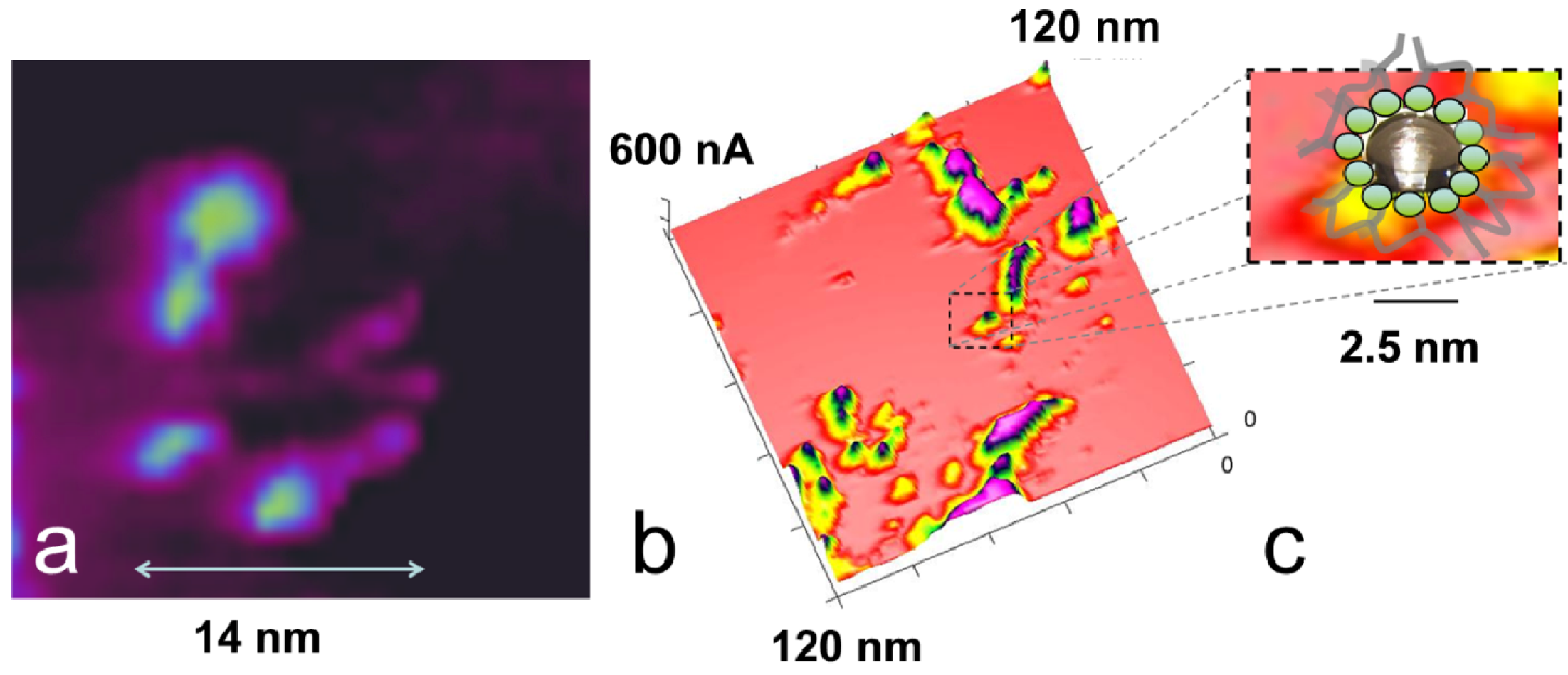
4. Conclusions
Acknowledgments
References
- Alberti, G.; Narducci, R.; Sganappa, M. Effects of hydrothermal/thermal treatments on the water-uptake of Nafion membranes and relations with changes of conformation, counter-elastic force and tensile modulus of the matrix. J. Power Sources 2008, 178, 575–583. [Google Scholar] [CrossRef]
- Eisenberg, A. Clustering of ions in organic polymers. A theoretical approach. Macromolecules 1970, 3, 147–154. [Google Scholar] [CrossRef]
- Zhao, Q.; Majsztrik, P.; Benziger, J. Diffusion and interfacial transport of water in Nafion®. J. Phys. Chem. B 2011, 115, 2717–2727. [Google Scholar] [CrossRef]
- Satterfield, M.B.; Benziger, J.B. Non-Fickian water vapor sorption dynamics by Nafion membranes. J. Phys. Chem. B 2008, 112, 3693–3704. [Google Scholar] [CrossRef]
- Bass, M.; Berman, A.; Singh, A.; Konovalov, O.; Freger, V. Surface structure of Nafion® in vapor and liquid. J. Phys. Chem. B 2010, 114, 3784–3790. [Google Scholar]
- Bass, M.; Berman, A.; Singh, A.; Konovalov, O.; Freger, V. Surface-induced micelle orientation in Nafion films. Macromolecules 2011, 44, 2893–2899. [Google Scholar] [CrossRef]
- Gierke, T.D.; Munn, G.E.; Wilson, F.C. The morphology in nafion perfluorinated membrane products, as determined by wide- and small-angle X-ray studies. J. Polym. Sci. Polym. Phys. 1981, 19, 1687–1704. [Google Scholar]
- Hsu, W.Y.; Gierke, T.D. Elastic theory for ionic clustering in perfluorinated ionomers. Macromolecules 1982, 15, 101–105. [Google Scholar] [CrossRef]
- Hsu, W.Y.; Gierke, T.D. Ion transport and clustering in nation perfluorinated membranes. J. Membr. Sci. 1983, 13, 307–326. [Google Scholar] [CrossRef]
- Schmidt-Rohr, K.; Chen, Q. Parallel cylindrical water nanochannels in Nafion fuel-cell membranes. Nature Mater. 2008, 7, 75–83. [Google Scholar] [CrossRef]
- Gebel, G. Structural evolution of water swollen perfluorosulfonated ionomers from dry membrane to solution. Polymer 2000, 41, 5829–5838. [Google Scholar] [CrossRef]
- Rubatat, L.; Rollet, A.L.; Gebel, G.; Diat, O. Evidence of elongated polymeric aggregates in Nafion. Macromolecules 2002, 35, 4050–4055. [Google Scholar] [CrossRef]
- Rollet, A.L.; Gebel, G.; Diat, O. A new insight into Nafion structure. J. Phys. Chem. B 2002, 106, 3033–3036. [Google Scholar] [CrossRef]
- Rubatat, L.; Gebel, G.; Diat, O. Orientation of Drawn Nafion at Molecular and Mesoscopic Scales. Macromolecules 2004, 37, 7772–7783. [Google Scholar] [CrossRef]
- Rubatat, L.; Gebel, G.; Diat, O. Stretching effect on Nafion fibrillar nanostructure. Macromolecules 2007, 40, 9455–9462. [Google Scholar] [CrossRef]
- Mauritz, K.A.; Moore, R.B. State of Understanding of Nafion. Chem. Rev. 2004, 104, 4535–4586. [Google Scholar] [CrossRef]
- Rao, V.; Friedrich, K.A.; Stimming, U. Part III: Membrane Applications in Industrial Waste Management (including nuclear Nuclear), Environmenatal Engineering and Future Trends in membrane Science Applications in Industrial Waste Management (Including Nuclear), Environmental Engineering and Future Trends in Membrane Science. In Handbook of Membrane Separations: Chemical, Pharmaceutical, Food, and Biotechnological Applications; Pabby, A.K., Rizvi, S.S.H., Sastre, A.M., Eds.; CRC Press: New York, NY, USA, 2008. [Google Scholar]
- Smitha, B.; Sridhar, S.; Khan, A.A. Solid polymer electrolyte membranes for fuel cell applications-A review. J. Membr.Sci. 2005, 259, 10–26. [Google Scholar] [CrossRef]
- Anantaraman, A.V.; Gardner, C.L. Studies on ion-exchange membranes. Part 1. Effect of humidity on the conductivity of Nafion. J. Electroanal. Chem. 1996, 414, 115–120. [Google Scholar] [CrossRef]
- Vol’fkovich, Yu.M.; Sosenkin, V.E.; Nikol’skaya, N.F. Hydrophilic-hydrophobic and sorption properties of the catalyst layers of electrodes in a proton exchange membrane fuel cell: A stage by stage study. Russ. J. Electrochem. 2010, 46, 438–449. [Google Scholar] [CrossRef]
- Vol’fkovich, Yu.M.; Sosenkinz, V.E.; Nikol’skaya, N.F. Hydrophilic-hydrophobic and sorption properties of the catalyst layers of electrodes in a proton-exchange membrane fuel cell: A stage-by-stage study. Elektrokhimiya 2010, 46, 462–474. [Google Scholar]
- Derjaguin, B.V.; Muller, V.M.; Toporov, Y.P. Effect of contact deformations on the adhesion of particles. J. Colloid Interface Sci. 1975, 53, 314–326. [Google Scholar] [CrossRef]
- Hiesgen, R.; Wehl, I.; Aleksandrova, E.; Roduner, E.; Bauder, A.; Friedrich, K.A. Nanoscale properties of polymer fuel cell materials-A selected review. Int. J. Energy Res. 2010, 34, 1223–1238. [Google Scholar]
- Hink, S.; Wagner, N.; Bessler, W.; Roduner, E. Impedance spectroscopic investigation of proton conductivity in Nafion using transient electrochemical atomic force microscopy (AFM). Membranes 2012, 2, 237–252. [Google Scholar] [CrossRef]
- Roduner, E.; Hiesgen, R. Spatially resolved Measurements. In Encyclopedia of Electrochemical Power Sources; Garche, J., Dyer, C., Moseley, P., Ogumi, Z., Rand, D., Scrosatti, B., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 775–786. [Google Scholar]
- Hiesgen, R.; Haiber, J. Structural properties: Atomic force microscopy. In Encyclopedia of Electrochemical Power Sources; Garche, J., Dyer, C., Moseley, P., Ogumi, Z., Rand, D., Scrosatti, B., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 696–717. [Google Scholar]
- O'Dea, J.R.; Buratto, S.K. Phase Imaging of Proton Exchange Membranes under Attractive and Repulsive Tip Sample Interaction Forces. J. Phys. Chem. B 2011, 115, 1014–1020. [Google Scholar]
- Sanchez, D.G.; Diaz, D.G.; Hiesgen, R.; Wehl, I.; Friedrich, K.A. Oscillations of PEM fuel cells at low cathode humidification. J. Electroanal. Chem. 2010, 649, 219–231. [Google Scholar] [CrossRef]
- Gebel, G.; Lyonnard, S.; Mendil-Jakani, H.; Morin, A. The kinetics of water sorption kinetics in Nafion® membranes: A small-angle neutron scattering study. J. Phys. Conds. Matter 2011, 23, 234107:–1-7. [Google Scholar]
- Choi, P.; Jalani, N.H.; Datta, R. Thermodynamics and proton transport in Nafion®. II. Proton diffusion mechanisms and conductivity. J. Electrochem. Soc. 2005, 152, E123–E130. [Google Scholar] [CrossRef]
- Haubold, H.G.; Vad, T.; Jungbluth, H.; Hiller, P. Nano structure of Nafion: A SAXS study. Electrochim. Acta 2001, 46, 1559–1563. [Google Scholar] [CrossRef]
- Zhang, S.; Yuan, X.Z.; Hiesgen, R.; Friedrich, K.A.; Wang, H. Effect of open circuit voltage on degradation of a short proton exchange membrane fuel cell stack with bilayer membrane configurations. J. Power Sources 2012, 205, 290–300. [Google Scholar] [CrossRef]
- Yuan, X.Z.; Zhang, S.; Ban, S.; Huang, C.; Wang, H.; Singara, V.; Fowler, M.; Hiesgen, R.; Schulze, M.; Haug, A.; Friedrich, K.A. Degradation of a PEM fuel cell stack with Nafion® membranes of different thicknesses. Part II: Ex situ diagnosis. J. Power Sources 2012, 205, 324–334. [Google Scholar] [CrossRef]
- Aleksandrova, E.; Hiesgen, R.; Eberhard, D.; Friedrich, K.A.; Kaz, T.; Roduner, E. Nanometer scale visualization of ionic channels at the surface of a proton exchange membrane. ChemPhysChem 2007, 8, 519–522. [Google Scholar] [CrossRef]
- Aleksandrova, E.; Hiesgen, R.; Eberhard, D.; Friedrich, K.A.; Roduner, E. Electrochemical atomic force microscopy study of proton conductivity in a Nafion® Membrane. Phys. Chem. Chem. Phys. 2007, 9, 2735–2743. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hiesgen, R.; Helmly, S.; Galm, I.; Morawietz, T.; Handl, M.; Friedrich, K.A. Microscopic Analysis of Current and Mechanical Properties of Nafion® Studied by Atomic Force Microscopy. Membranes 2012, 2, 783-803. https://doi.org/10.3390/membranes2040783
Hiesgen R, Helmly S, Galm I, Morawietz T, Handl M, Friedrich KA. Microscopic Analysis of Current and Mechanical Properties of Nafion® Studied by Atomic Force Microscopy. Membranes. 2012; 2(4):783-803. https://doi.org/10.3390/membranes2040783
Chicago/Turabian StyleHiesgen, Renate, Stefan Helmly, Ines Galm, Tobias Morawietz, Michael Handl, and K. Andreas Friedrich. 2012. "Microscopic Analysis of Current and Mechanical Properties of Nafion® Studied by Atomic Force Microscopy" Membranes 2, no. 4: 783-803. https://doi.org/10.3390/membranes2040783