Oxygen Selective Membranes for Li-Air (O2) Batteries
Abstract
:1. Introduction
| Metal-air system | OCV (V) | Specific energy (Wh/kg) | Specific capacity (mAh/g) |
|---|---|---|---|
| 2Li + ½O2→ Li2O (aprotic organic sln) | 2.913 | 11,248 * | 3,862 |
| Li + ½O2 ⇌ ½Li2O2 (aprotic organic sln) | 2.959 | 11,425 * | 3,862 |
| 2Li + ½O2 + H2SO4 ⇌ Li2SO4 + H2O | 4.274 | 2,046 * | 479 |
| 2Li + ½O2 + H2O ⇌ 2LiOH | 3.446 | 5,789 * | 1,681 |
| 2Li + H2O (seawater) → 2LiOH + ½H2 | 2.512 | 9,701 ** | 3,862 |
| Al + 0.75O2 + 1.5H2O ⇌ Al(OH)3 | 2.701 | 4,021 * | 1,489 |
| Mg + ½O2 + H2O ⇌ Mg(OH)2 | 2.756 | 3,491 * | 1,267 |
| Zn + ½O2 ⇌ ZnO | 1.650 | 1,353 * | 820 |
| x6C + LiCoO ⇌ xLiC6 + Li1-xCoO2 (organic) | ~4.2 | 420 *** | 139 *** |
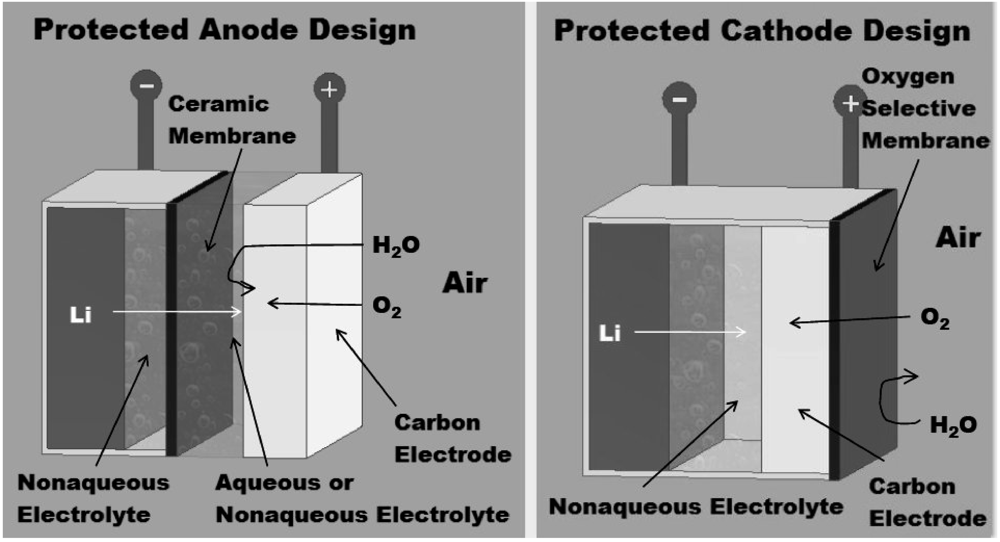
2. Properties of Oxygen Selective Membranes
- High O2 permeability;
- No water vapor permeability;
- No electrolyte solvent permeability;
- No carbon dioxide (CO2) permeability for secondary applications.
2.1. High O2 Permeability
 , or transmission rate of O2 (OTR) through a barrier is dependent upon the solubility of O2 in the membrane and the rate of diffusion of O2 through the membrane, and is defined by
, or transmission rate of O2 (OTR) through a barrier is dependent upon the solubility of O2 in the membrane and the rate of diffusion of O2 through the membrane, and is defined by
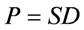
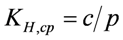
 , can be directly related to maximum (limiting) current density, imax, assuming the oxygen reduction reaction (ORR) is mass transfer limited by O2 transport through the OSM. The maximum (limiting) current density due to O2 diffusion for an air cathode protected by an OSM is highly dependent on the magnitude of P, and below we show that membranes where P < 100 Barrer are not practical for a commercial Li-air cell. In view of the limited amount of data required to calculate permeability values from Equations (1–3), values of P can easily be determined experimentally using a barrier testing instrument such as the one available from MOCON, Inc.
, can be directly related to maximum (limiting) current density, imax, assuming the oxygen reduction reaction (ORR) is mass transfer limited by O2 transport through the OSM. The maximum (limiting) current density due to O2 diffusion for an air cathode protected by an OSM is highly dependent on the magnitude of P, and below we show that membranes where P < 100 Barrer are not practical for a commercial Li-air cell. In view of the limited amount of data required to calculate permeability values from Equations (1–3), values of P can easily be determined experimentally using a barrier testing instrument such as the one available from MOCON, Inc.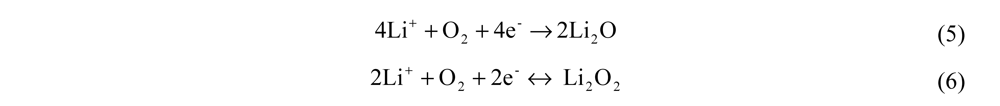
 , which can be related to permeability by the following equation.
, which can be related to permeability by the following equation. 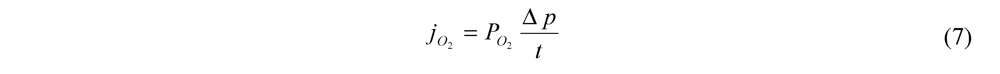
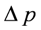 is the partial pressure gradient of O2 across the membrane and t is the membrane thickness. The partial pressure of O2 in air is ~160 mm Hg and will be 0 in the air electrode assuming O2 is the limiting reactant so
is the partial pressure gradient of O2 across the membrane and t is the membrane thickness. The partial pressure of O2 in air is ~160 mm Hg and will be 0 in the air electrode assuming O2 is the limiting reactant so 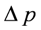 = 160 mm Hg. The flux of O2 through the membrane can also be improved by decreasing the thickness of the membrane. However, membrane fabrication techniques typically limit the thickness to 10–25 µm before pinhole defects are present. Also, decreasing the thickness of the membrane will also increase the permeation of water vapor and electrolyte solvent. The maximum current density is given by
= 160 mm Hg. The flux of O2 through the membrane can also be improved by decreasing the thickness of the membrane. However, membrane fabrication techniques typically limit the thickness to 10–25 µm before pinhole defects are present. Also, decreasing the thickness of the membrane will also increase the permeation of water vapor and electrolyte solvent. The maximum current density is given by
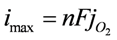
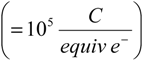 . From Equations (5) and (6), n is equal to 4 for Li2O formation and 2 for Li2O2 formation. McCloskey et al. [17] experimentally determined n = 2.6 for the ORR in alkyl carbonates due to the reaction of the electrolyte solvent with Li+ and O2 to form lithium carbonate and lithium alkyl carbonates. Figure 2 shows the effect of O2 permeability in Barrer on maximum current density for a 1 µm thick membrane. The data were derived using the Equations (3, 4, 7, 8). This assumes the air window and air electrode have equal geometric areas. The maximum current density for a Li-air battery limited by O2 transport through an OSM of any thickness can be determined by dividing the imax for a given permeability obtained from Figure 2 by the actual thickness in microns of the OSM. For example, a membrane that is 1 µm thick with a permeability of 500 Barrer would have a limiting current density of approximately 68 mA cm−2 based on Li2O2 formation. The limiting current density is 1.4 mA cm−2 for the same membrane if it is 50 µm thick.
. From Equations (5) and (6), n is equal to 4 for Li2O formation and 2 for Li2O2 formation. McCloskey et al. [17] experimentally determined n = 2.6 for the ORR in alkyl carbonates due to the reaction of the electrolyte solvent with Li+ and O2 to form lithium carbonate and lithium alkyl carbonates. Figure 2 shows the effect of O2 permeability in Barrer on maximum current density for a 1 µm thick membrane. The data were derived using the Equations (3, 4, 7, 8). This assumes the air window and air electrode have equal geometric areas. The maximum current density for a Li-air battery limited by O2 transport through an OSM of any thickness can be determined by dividing the imax for a given permeability obtained from Figure 2 by the actual thickness in microns of the OSM. For example, a membrane that is 1 µm thick with a permeability of 500 Barrer would have a limiting current density of approximately 68 mA cm−2 based on Li2O2 formation. The limiting current density is 1.4 mA cm−2 for the same membrane if it is 50 µm thick.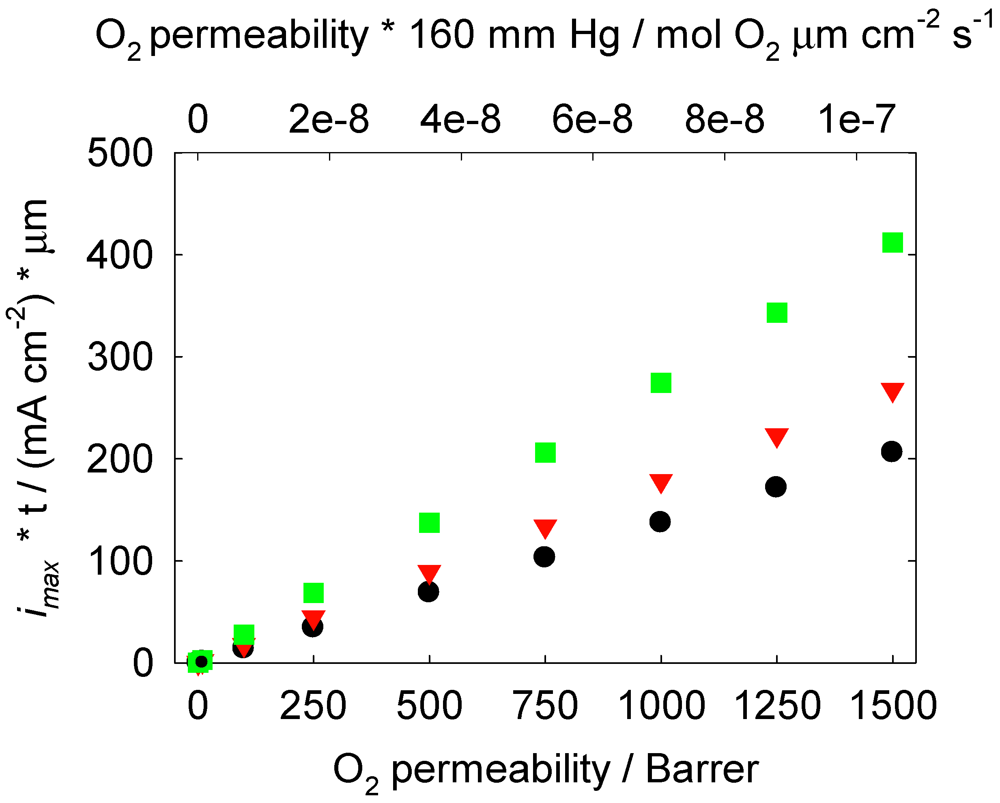
2.2. No Water Vapor Permeability

2.3. No Electrolyte Solvent Permeability
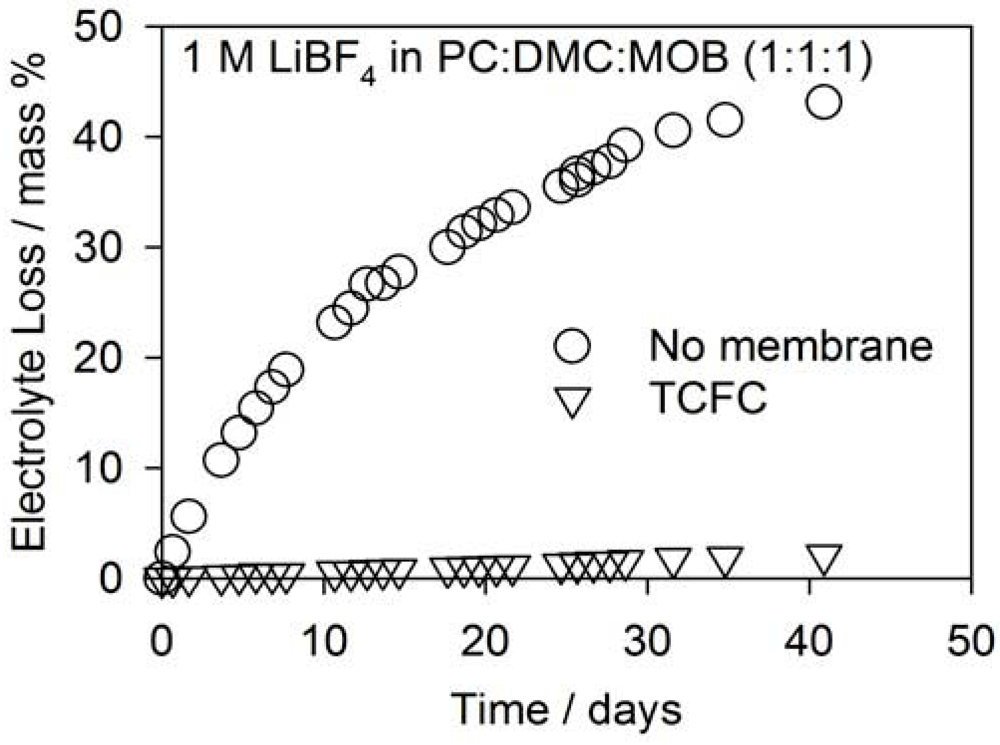
2.4. No CO2 Permeability for Secondary Applications
3. Basic Materials for Oxygen Selective Membranes
4. Review of Oxygen Selective Membrane Literature
5. Conclusions
Acknowledgments
References
- Atwater, T.G.; Dobley, A. Metal/air batteries. In Linden’s Handbook of Batteries, 4th; Reddy, T.B., Ed.; McGraw-Hill Professional: New York, NY, USA, 2010. [Google Scholar]
- Abraham, K.M.; Jiang, Z. A polymer electrolyte-based rechargeable lithium/oxygen battery. J. Electrochem. Soc. 1996, 143, 1–5. [Google Scholar]
- Kowalczk, I.; Read, J.; Salomon, M. Li-air batteries: A classic example of limitations owing to solubilities. Pure Appl. Chem 2007, 79, 851–860. [Google Scholar]
- Crowther, O.; Meyer, B.; Morgan, M.; Salomon, M. Primary Li-air cell development. J. Power Sources 2011, 196, 1498–1502. [Google Scholar]
- Crowther, O.; Meyer, B.; Morgan, M.; Salomon, M. Erratum to “primary Li-air cell development”. J. Power Sources 2011, 196, 1498–1502. [Google Scholar]
- Bard, A.J.; Parsons, R.; Jordan, J. Standard Potentials in Aqueous Solution; Marcel Dekker: New York, NY, USA, 1995. [Google Scholar]
- Christensen, J.; Albertus, P.; Sanchez-Carrera, R.S.; Lohmann, T.; Kozinsky, B.; Liedtke, R.; Ahmed, J. A critical review of Li/air batteries. J. Electrochem. Soc. 2012, 159, R1–R30. [Google Scholar]
- Crowther, O.; Salomon, M. Aqueous Li-air (O2) systems. In Advanced Lithium Batteries; Scrosati, B., Abraham, K.M., van Shalkwijk, W., Eds.; John Wiley: Hoboken, NJ, USA, 2012; in press. [Google Scholar]
- Fu, J. Superionic conductivity of glass-ceramics in the system Li2O-Al2O3-TiO2-P2O5. Solid State Ionics 1997, 96, 195–200. [Google Scholar]
- Fu, J. Fast Li+ ion conducting glass-ceramics in the system Li2O-Al2O3-GeO2-P2O5. Solid State Ionics 1997, 104, 191–194. [Google Scholar]
- Fu, J. Lithium Ion Conductive Glass-Ceramics and Electric Cells And Gas Sensors Using the Same. U.S. Patent 6,030,909, 24 October 1997. [Google Scholar]
- Visco, S.J.; Katz, B.D.; Nimon, Y.S.; de Jonghe, L.C. Protected Active Metal Electrode and Battery Cell Structure with Non-Aqueous Interlayer Architecture. US Patent 7,282,295, 16 October 2007. [Google Scholar]
- Scrosati, B.; Garche, J. Lithium batteries: Status, prospects, and future. J. Power Sources 2010, 195, 2419–2430. [Google Scholar] [CrossRef]
- Battino, R.; Rettich, T.R.; Tominaga, T. The solubility of oxygen and ozone in liquids. J. Phys. Chem. Ref. Data 1983, 12, 163–179. [Google Scholar]
- Battino, R. IUPAC Solubility Data Ser. 1984, 7, 1–519.
- Dias, A.M.A.; Freire, M.; Coutinho, J.A.P.; Marrucho, I.M. Solubility of oxygen in liquid perfluorocarbons. Fluid Phase Equilib. 2004, 222–223, 325–330. [Google Scholar] [CrossRef]
- McCloskey, B.D.; Bethune, D.S.; Shelby, R.M.; Girishkumar, G.; Luntz, A.C. Solvent’ critical role in nonaqueous lithium-oxygen battery electrochemistry. J. Phys. Chem. Lett. 2011, 2, 1161–1166. [Google Scholar]
- Crowther, O.; Keeny, D.; Moureau, D.M.; Meyer, B.; Salomon, M.; Hendrickson, M. Electrolyte optimization for the primary lithium metal air battery using an oxygen selective membrane. J. Power Sources 2012, 202, 347–351. [Google Scholar]
- Zaban, A.; Zinigrad, E.; Aurbarch, D. Impedance spectroscopy of li electrodes. 4. A general simple model of the li-solution interphase in polar aprotic systems. J. Phys. Chem. 1996, 100, 3089–3101. [Google Scholar] [CrossRef]
- Read, J.; Mutolo, K.; Ervin, M.; Behl, W.; Wolfenstine, J.; Driedger, A.; Foster, D. Oxygen transport properties of organic electrolytes and performance of lithium/oxygen battery. J. Electrochem. Soc. 2003, 150, A1351–A1356. [Google Scholar]
- Xu, W.; Xiao, J.; Zhang, J.; Wang, D.; Zhang, J.-G. Optimization of nonaqueous electrolyte for primary lithium/air batteries operated in ambient environments. J. Electrochem. Soc. 2009, 156, A773–A779. [Google Scholar]
- Freunberger, S.A.; Chen, Y.; Peng, Z.; Griffin, J.M.; Hardwick, L.J.; Bardé, F.; Novák, P.; Bruce, P.G. Reactions in rechargeable lithium-o2 battery with alkyl carbonate electrolytes. J. Am. Chem. Soc. 2011, 133, 8040–8047. [Google Scholar]
- McCloskey, B.D.; Speidel, A.; Scheffler, R.; Miller, D.C.; Viswanathan, V.; Hummelshøj, J.S.; Nørskov, J.K.; Luntz, A.C. Twin problems of interfacial carbonate formation in nonaqueous Li-O2 batteries. J. Phys. Chem. Lett. 2012, 3, 997–1001. [Google Scholar]
- Goldstein, J.; Korall, M.; Brokman, A. Electrodes for Metal/Air Batteries and Bipolar Metal/Air Batteries Incorporating the Same. US Patent 5,145,752, 8 September 1992. [Google Scholar]
- Reynolds, T.A.; Brose, D.J.; Golovin, M.N. Membrane for Selective Transport of Oxygen Over Water Vapor and Metal-Air Electrochemical Cell Including Said Membrane. US Patent 5,985,475, 16 November 1999. [Google Scholar]
- Chua, D.; Driedger, A.; Meyer, B.; Morgan, M.; Salomon, M. Protected Lithium-Air Cells by Oxygen-Selective Permeable Cathode Membranes. US Patent 0,177,400, 21 January 2011. [Google Scholar]
- Clarke, R. Fluorosiloxane Optical Cladding. US Patent 4,317,616, 2 March 1982. [Google Scholar]
- Blume, I.; Schwering, P.J.F.; Mulder, M.H.V.; Smolders, C.A. Vapor sorption and permeation properties of poly(dimethylsiloxane) films. J. Membr. Sci. 1991, 61, 85–97. [Google Scholar]
- Zhang, H.; Cloud, A. The permeability characteristics of silicone rubber. Dallas, TX, USA, November 2006; Pilato, L.A., Kliger, H.S., Beckwith, S.W., Eds.; Destech Pubns Inc.: Lancaster, PA, USA, 2006. [Google Scholar]
- Lai, Y.-C.; Lang, W.; Quinn, E.T.; Ruscio, D.V. Hydrogel Copolymers for Biomedical Devices. US Patent 2006/0142525 A1, 29 June 2006. [Google Scholar]
- Zhang, J.-G.; Wang, D.; Xu, W.; Xiao, J.; Williford, R.E. Ambient operation of Li/air batteries. J. Power Sources 2010, 195, 4332–4337. [Google Scholar]
- Wang, D.; Xiao, J.; Xu, W.; Zhang, J.-G. High capacity pouch-type Li-air batteries. J. Electrochem. Soc. 2010, 157, A760–A764. [Google Scholar]
- Xiao, J.; Mei, D.; Li, X.; Xu, W.; Wang, D.; Graff, G.L.; Bennet, W.D.; Ne, Z.; Saraf, L.V.; Aksay, I.A.; Liu, J.; Zhang, J.-Z. Hierarchically porous graphene as lithium-air battery electrode. Nano Lett. 2011, 11, 5071–5078. [Google Scholar]
- Zhang, J.; Xu, W.; Li, X.; Liu, W. Air dehydration membranes for nonaqueous lithium-air batteries. J. Electrochem. Soc. 2010, 157, A940–A946. [Google Scholar]
- Zhang, J.; Xu, W.; Liu, W. Oxygen-selective immobilized liquid membranes for operation of lithium-air batteries in ambient air. J. Power Sources 2010, 195, 7438–7444. [Google Scholar]
- Crowther, O.; Chua, D.; Eppley, W.; Meyer, B.; Salomon, M.; Driedger, A.; Morgan, M. Lithium-air cell protective membranes comprising polytetrafluroethylene coated fiberglass cloth. US Patent 2011/0177401 A1, 21 July 2011. [Google Scholar]
- Liu, R.Y.F.; Hu, Y.S.; Schiraldi, D.A.; Hiltner, A.; Baer, E. Crystallinity and oxygen transport properties of PET bottle walls. J. Appl. Polymer Sci. 2004, 94, 671–677. [Google Scholar]
- Kraytsberg, A.; Ein-Eli, Y. Review on Li-air batteries-opportunities, limitations, and perspective. J. Power Sources 2011, 196, 886–893. [Google Scholar] [CrossRef]
- Aiba, S.; Ohashi, M.; Huang, S.-Y. Rapid determination of oxygen permeability of polymer membranes. Ind. Eng. Chem. Fundamen. 1968, 7, 497–502. [Google Scholar]
- Muthiah, P.; Hsu, S.H.; Sigmund, W. Coaxially electrospun PVDF-Teflon AF and Teflon AF-PVDF core-sheath nanofiber mats with superhydrophobic properties. Langmuir 2010, 26, 12483–12487. [Google Scholar]
- Scheffler, R.; Bell, N.S.; Sigmund, W. Electrospun Teflon AF fibers for superhydrophobic membranes. J. Mater. Res. 2010, 25, 1595–1600. [Google Scholar]
- DuPont™ Teflon® Afamorphous Fluoropolymers; DuPont Fluoroproducts: Wilmington, DE, USA, 2012. Available online: http://www2.dupont.com/Teflon_Industrial/en_US/assets/downloads/h44587.pdf (accessed on 12 April 2012).
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Crowther, O.; Salomon, M. Oxygen Selective Membranes for Li-Air (O2) Batteries. Membranes 2012, 2, 216-227. https://doi.org/10.3390/membranes2020216
Crowther O, Salomon M. Oxygen Selective Membranes for Li-Air (O2) Batteries. Membranes. 2012; 2(2):216-227. https://doi.org/10.3390/membranes2020216
Chicago/Turabian StyleCrowther, Owen, and Mark Salomon. 2012. "Oxygen Selective Membranes for Li-Air (O2) Batteries" Membranes 2, no. 2: 216-227. https://doi.org/10.3390/membranes2020216
APA StyleCrowther, O., & Salomon, M. (2012). Oxygen Selective Membranes for Li-Air (O2) Batteries. Membranes, 2(2), 216-227. https://doi.org/10.3390/membranes2020216




