3.1. Results
Five hundred and fifty 11–12 year old female patients were identified in the total population of girls seen for a well-child visit between May 2008–April 2009. Of these patients, 72.5% were nonhispanic white race, 53% had private insurance, and 48.2% were vaccinated against HPV during the study period (
Table 1).
Table 1.
Demographics of total population compared to cohort of telephone interview respondents.
Table 1.
Demographics of total population compared to cohort of telephone interview respondents.
| | Population | Cohort | p-value |
|---|
| | n = 550 | n = 242 |
|---|
| Age | 11 | 350 | 63.6% | 152 | 62.8% | 0.990 |
| 12 | 200 | 36.4% | 87 | 36.0% | |
| Race | Nonhispanic white | 396 | 72.0% | 183 | 75.6% | 0.981 |
| Black | 115 | 20.9% | 38 | 15.7% | |
| Other | 39 | 7.1% | 27 | 11.2% | |
| Insurance | Private | 292 | 53.1% | 201 | 83.1% | 0.944 |
| Public | 175 | 31.8% | 38 | 15.7% | |
| Self-pay | 22 | 4.0% | 3 | 1.2% | |
| Unknown | 61 | 11.1% | 0 | 0.0% | |
| HPV vaccination | 0 | 284 | 51.6% | 122 | 50.4% | 0.954 |
| ≥1 | 265 | 48.2% | 106 | 43.8% | |
| ≥3 | 128 | 23.3% | 42 | 17.4% | |
| Unknown | 0 | 0.0% | 14 | 5.8% | |
In this population, there were noted to be significant associations of HPV vaccination with insurance status and race. Girls with private insurance were less likely to receive the HPV vaccine compared to girls with public insurance or self-pay. Conversely, girls with public insurance were 36% more likely to be vaccinated as compared to girls with private insurance (
Table 2).
Table 2.
Human papillomavirus (HPV) vaccine uptake (≥1 dose received) by insurance and race in population of 11–12 year old girls presenting for well child visits between May 2008–April 2009.
Table 2.
Human papillomavirus (HPV) vaccine uptake (≥1 dose received) by insurance and race in population of 11–12 year old girls presenting for well child visits between May 2008–April 2009.
| | HPV vaccine uptake | % | RR | 95% CI |
|---|
| Insurance status | Private | 130/292 | 44.5% | 0.85 * | 0.72–1.01 |
| Public | 103/175 | 58.9% | 1.36 † | 1.15–1.61 |
| Self-pay § | 11/22 | 50.0% | | |
| Unknown § | 21/61 | 34.4% | | |
| Race | Nonhispanic white | 172/396 | 43.4% | 0.72 | 0.61–0.85 |
| Black | 70/115 | 60.9% | 1.36 ** | 1.13–1.63 |
| Other § | 23/39 | 59.0% | | |
| Total | | 265/550 | 48.2% | | |
Further, nonhispanic white girls presenting for well child visit were overall almost 30% less likely to initiate vaccination compared to all minority girls (RR 0.72, 95% CI 0.61–0.85). Compared to nonhispanic whites, black girls were 36% more likely to have received at least 1 dose of the HPV vaccine. There were no significant differences in HPV vaccination completion rates by race among those who received at least one dose with 84/172 (49%, 95% CI 41%–56%) of nonhispanic whites and 71/154 (46%, 95% CI 38%–54%) of minority girls completing the three injection series.
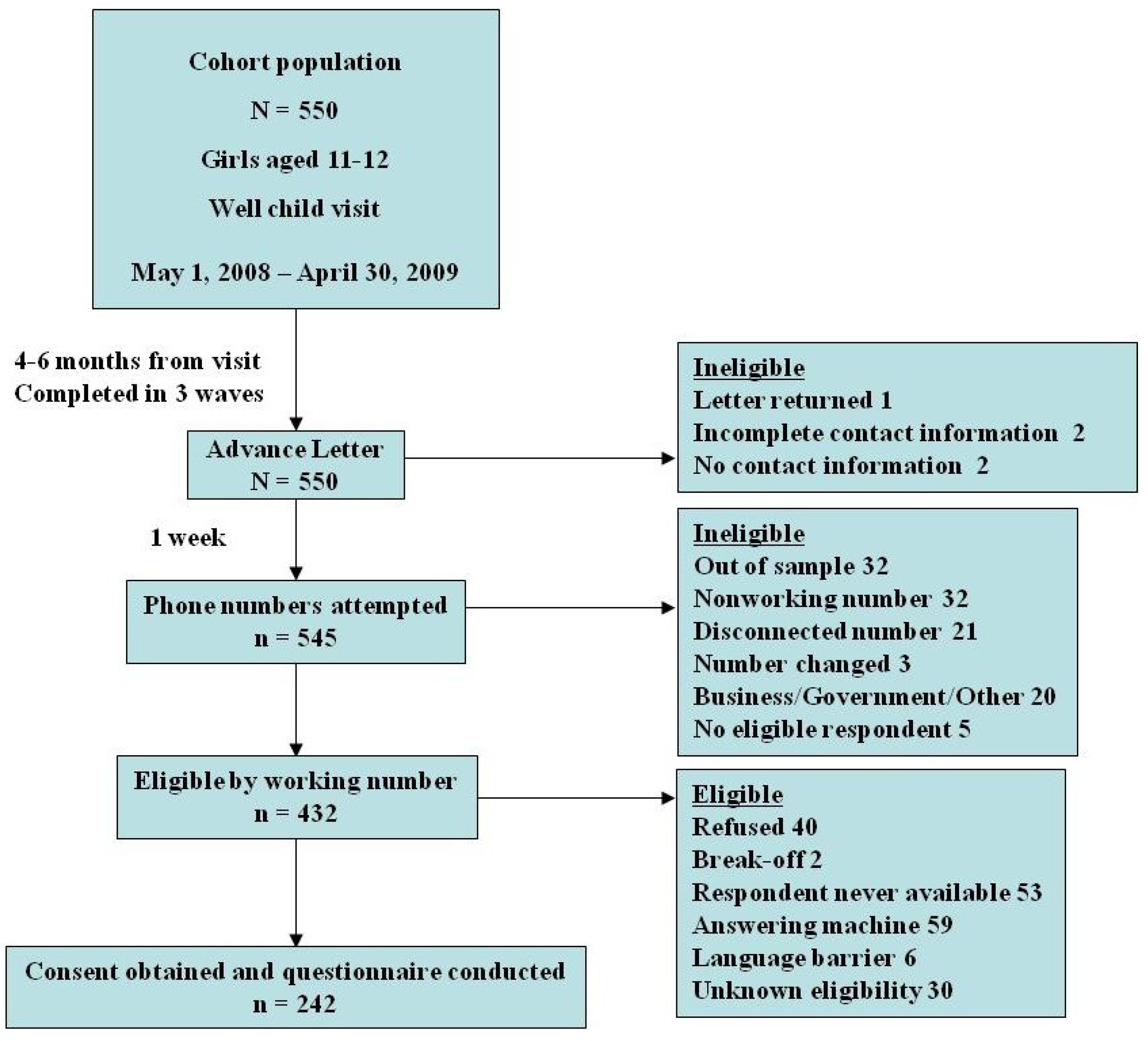
All 550 families from the original population were considered for participation in the telephone questionnaire (
Figure 1).
Figure 1.
Diagram of cohort originating from study population.
Figure 1.
Diagram of cohort originating from study population.
Four hundred and thirty-two households with a working number were approached for participation in the questionnaire and 242 parents or guardians agreed to participate in the telephone interview for a response rate of 56%. (Of note, 219 of the 242 participants were parents. Thus, the survey respondents are referred to as parents even though 23 individuals were guardians of other familial description.) See
Table 1 for demographic comparisons of the cohort participating in the telephone questionnaire compared to the entire population.
All parents confirmed the child had a recent visit to the doctor. Eighty-nine percent of participants went with the child to the documented visit, of whom 79% remember the doctor discussing the HPV vaccine. The majority of respondents confirmed that the child had been seeing this physician for 5 years or greater.
Overall, 92% of parents were aware of the HPV vaccine, and 85% reported that the HPV vaccine was recommended for their child. In regards to HPV vaccine knowledge, 69% of respondents agreed that the vaccine protects against cervical cancer, but only 20% also identified that the quadrivalent vaccine protects against genital warts. The majority of respondents (59%) agreed that the HPV vaccine is safe. Twenty percent of parents felt that their child did not need the HPV vaccine. There were no statistically significant differences in these attitudes compared by race.
After combining billing data with parental report, vaccine uptake rates were compared by race. Blacks and other races were significantly more likely than whites to have received at least one HPV vaccine injection (75.7% and 68.4%
vs. 47.5%, respectively;
p = 0.003). By comparison, whites were more likely to have received the meningococcal vaccine (74.3%
vs. 59.5% and 52.6% respectively;
p = 0.043), and rates were not significantly different for Tdap and varicella vaccines (
Figure 2).
Figure 2.
Vaccination rates of recommended adolescent vaccines by race.
Figure 2.
Vaccination rates of recommended adolescent vaccines by race.
* Error bars represent 95% confidence interval.
Further, rates of HPV vaccine uptake trended toward increased vaccination of children with public insurance or no insurance as compared to those with private insurance (66%
vs. 51%;
p = 0.06). Again, there were no significant differences for the other recommended childhood vaccines (
Figure 3).
Figure 3.
Vaccination rates of recommended adolescent vaccines by insurance status.
Figure 3.
Vaccination rates of recommended adolescent vaccines by insurance status.
* Error bars represent 95% confidence interval.
In multivariable logistic regression, patients of black and other minority races were 4.9 and 4.2 times more likely to receive the HPV vaccine compared to their white counterparts. Recommendation by the child’s physician was determined by parents answering yes, no or unsure to the question “has your doctor recommended the HPV vaccination for your daughter?” Daughters of parents who answered yes to this question were twice as likely to initiate the HPV vaccine series. Similarly daughters of parents who disagreed or strongly disagreed with the statement “My daughter does not need the HPV vaccine” (a marker of perceived susceptibility) were also significantly more likely to be vaccinated. Safety concerns were the strongest attitude barrier to vaccination (
Table 3). Insurance status was not independently associated with HPV vaccine uptake in the final model.
Table 3.
Final regression model showing factors independently associated with initiating the HPV vaccine series.
Table 3.
Final regression model showing factors independently associated with initiating the HPV vaccine series.
| | OR | 95% CI | p-value |
|---|
| Black race * | 4.9 | 1.8–13.6 | 0.0023 |
| Other race * | 4.2 | 1.1–16.6 | 0.042 |
| MD recommends | 2.1 | 0.97–4.4 | 0.06 |
| Perceived susceptibility | 1.7 | 1.1–2.6 | 0.014 |
| Safety concerns | 0.19 | 0.093–0.37 | <0.0001 |
3.2. Discussion
In this prospective cohort study, minority girls were more likely to receive the HPV vaccine compared to their nonhispanic white counterparts. In particular, race correlated more with vaccination initiation than insurance status, beliefs about safety and efficacy, recommendation by the pediatrician, and perceived susceptibility although these factors likely also played a role in the parents’ decision to have their child vaccinated. The CDC’s National Immunization Survey-Teen in 2011 documented a similar trend for higher vaccination rates among minorities [
10]. However, the NIS-Teen data specifically looks at adolescents aged 13–17 and thus does not specifically address this issue of uptake at the time of the well child visit in younger adolescents. This study demonstrates that these differences may occur at the time of the initial adolescent visit and persistent throughout adolescence. Provider recommendation remains the strongest modifiable event to promote vaccination. However, recent data suggests that provider recommendation is lower for minority races [
20]. Conversely, at least one study found that physicians with higher rates of minority patients had higher HPV vaccination rates, suggesting possible targeting of vaccine recommendation [
21]. This study cannot provide any information about provider beliefs or attitudes although the strong association between MD recommendation and racial differences in uptake warrant further study in this area. Lastly, it is possible that perceived susceptibility could also play a role in increased uptake among minorities. Racial disparities in cervical cancer incidence and mortality are well documented and have been circulated widely in the lay public.
Among girls presenting to the doctor for a well-child visit in this study, privately insured patients were less likely to receive the HPV vaccine than girls with Medicaid or no insurance. This may be related to coverage and reimbursement issues which can be heterogeneous among private payers, possibly resulting in more out-of-pocket costs for parents. By comparison, all children who qualify for Medicaid and/or are underinsured for vaccinations and meet certain financial cutoffs are covered by the Federal Vaccines for Children program for all vaccines recommended by the CDC, including the HPV vaccine. The NIS-Teen survey also noted a difference in vaccination by socioeconomic status with 46.4% of girls below the poverty level initiating vaccination compared to 35.8% of girls at or above the poverty level choosing to be vaccinated [
10]. It is possible in our study, that any role insurance status plays is mediated by the difference in race among payer groups. Future studies should assess this in more detail in the questionnaire.
This study evaluated HPV vaccination given uniform access to care in a single cohort of girls being seen for well child checks in the same health care system. This is a strength of the study as it controls for regional differences in provider counseling and issues in access to patient care. This research utilized prospective data collection and objective billing data for confirmation of vaccination status allowing for the most accurate determination of vaccination. In addition, utilization of a professional survey group for interviews, validated questionnaire, and a piloted study technique allows for high quality subjective data. This study is limited by its small sample size and evaluation of only a single health care delivery system. For the follow-up questionnaire, we had a relatively low response rate but this is comparable to similar telephone surveys conducted on HPV vaccination [
11]. In regards to nonresponder bias, we found that patients with public insurance were less likely to participate in the questionnaire. This may limit our abilities to fully characterize this group’s higher vaccination rates. Further, our research by the nature of cohort selection cannot take into account vaccination barriers related to access to care or determine causation.
This study documents HPV vaccination rates, attitudes and barriers prior to initiation of a statewide mandate requiring girls in Virginia to receive the HPV vaccine prior to their 11th birthday for school admission and begins to identify further areas that need elucidation. These data will serve as the baseline for a comparison study of HPV vaccination rates and counseling 5 years after enactment of the mandate. This study is uniquely positioned to address this question given the objective data capture through the billing database combined with a validated parental survey and uniform access to care. Further, documented differences by race and insurance status seen with HPV vaccination but not with other adolescent immunizations will serve as the baseline for post-mandate comparisons. The follow-up study is slated to be conducted starting in August 2013. These data may have implications beyond a single state given similar trends in differential rates of uptake by race seen at a national level.