Antioxidant Constituents of Cotoneaster melanocarpus Lodd.
Abstract
:1. Introduction
2. Experimental Section
2.1. Plant Material and Extraction
2.2. DPPH Radical Scavenging Assay
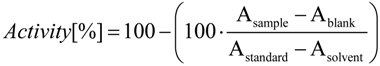
2.3. TLC Profiles of the Extracts
2.4. Analytical HPLC Method
| Time (min) | Solvent A a [%] | Solvent B b [%] |
|---|---|---|
| Initial | 90 | 10 |
| 20 | 80 | 20 |
| 30 | 60 | 40 |
| 40 | 10 | 90 |
| 50 | 0 | 100 |
| 55 | 0 | 100 |
2.5. LC-MS Analysis
3. Results and Discussion
3.1. DPPH Radical Scavenging Assay
| Extraction medium | Sterile shoots | Old stems | Leaves |
|---|---|---|---|
| H2O | 53.53 ± 6.21 | 86.72 ± 7.50 | 67.74 ± 6.14 |
| MeOH | 30.91 ± 2.97 | 48.21 ± 4.38 | 106.41 ± 12.26 |
| EtAc | 124.07 ± 15.80 | 74.88 ± 14.59 | >200 |
| DCM | >200 | 134.60 ± 32.77 | >200 |
| Hexane | no activity | no activity | no activity |
| Reference rutin | 13.26 ± 0.58 |
3.2. TLC Profiling
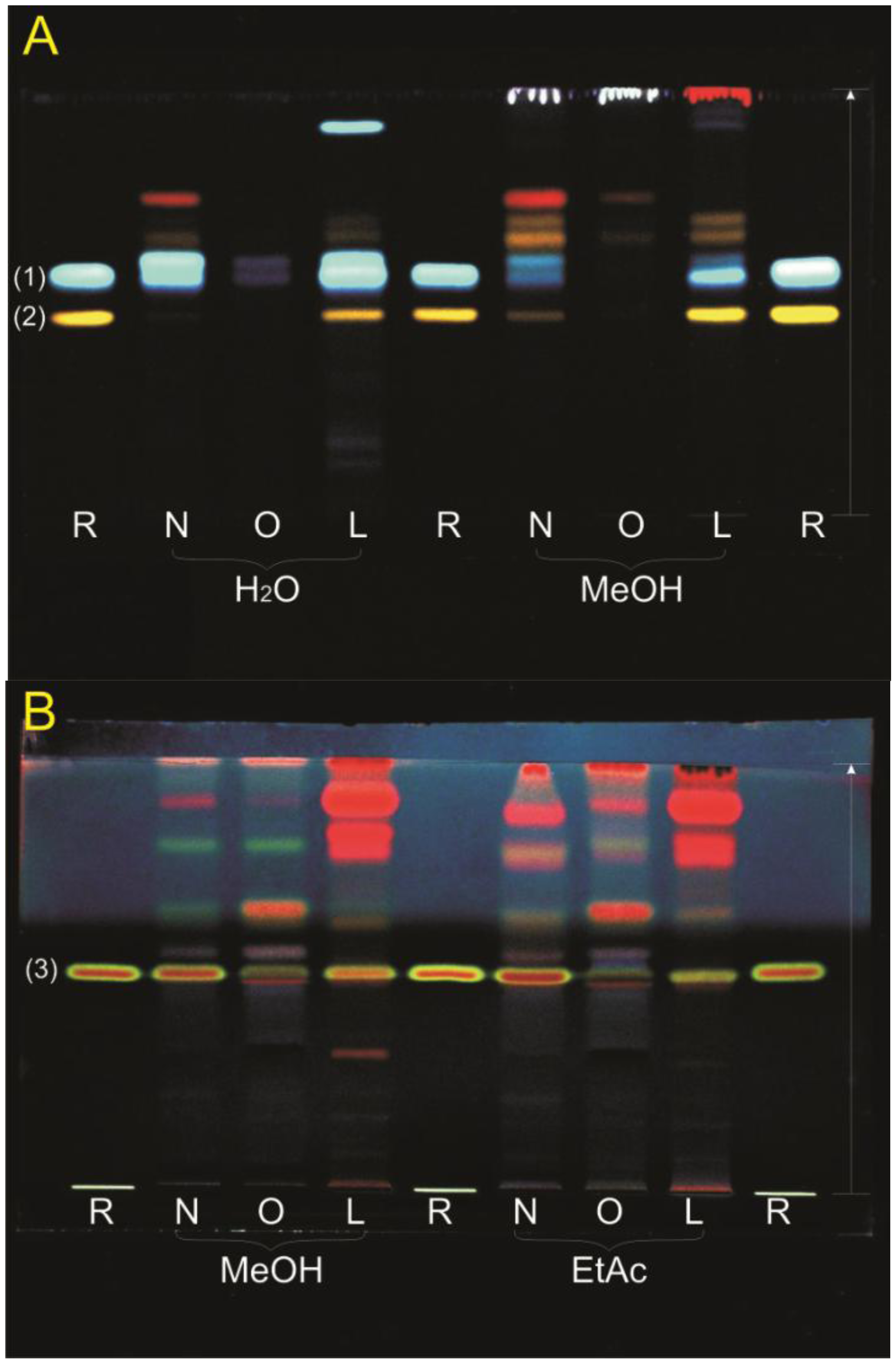
3.3. HPLC and LC-MS Analysis
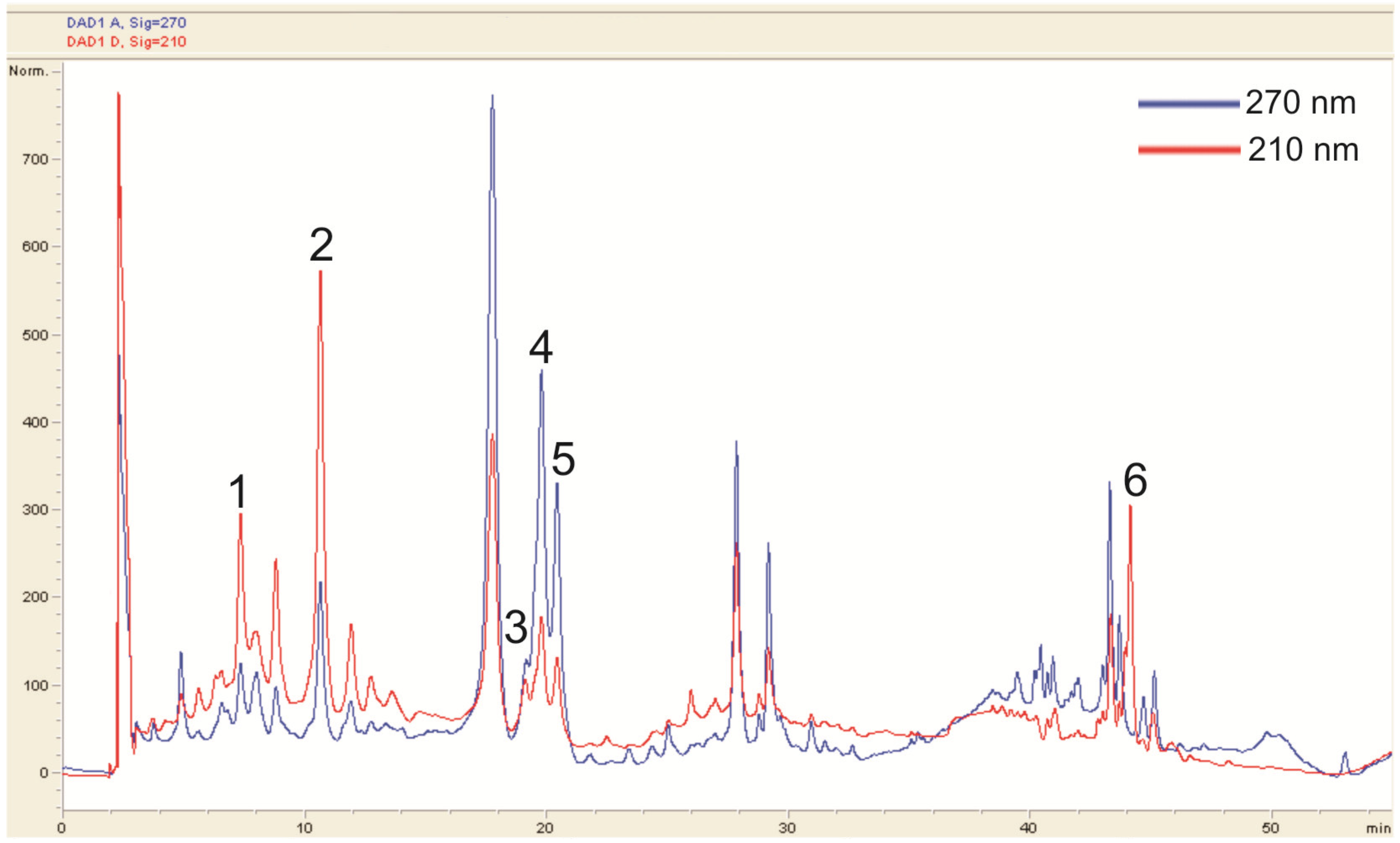
| Peak No. | Compound | Rt [min] |
|---|---|---|
| 1 | Neochlorogenic acid | 4.74 |
| 2 | Chlorogenic acid | 7.77 |
| 3 | Rutin | 19.04 |
| 4 | Hyperoside | 19.36 |
| 5 | Isoquercetin | 20.03 |
| 6 | Ursolic acid | 44.12 |
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Harman, D. The aging process. Proc. Natl. Acad. Sci.USA 1981, 78, 7124–7128. [Google Scholar] [CrossRef]
- Allen, R.G.; Lafuse, W.P.; Powell, N.D.; Marketon, J.I.W.; Stiner-Jones, L.M.; Sheridan, J.F.; Bailey, M.T. Stressor-induced increase in microbicidal activity of splenic macrophages is dependent upon peroxynitrite production. Infect. Immun. 2012, 80, 3429–3437. [Google Scholar] [CrossRef]
- Knight, J.A. Review: Free radicals, antioxidants, and the immune system. Ann. Clin. Lab. Sci. 2000, 30, 145–158. [Google Scholar] [PubMed]
- Kuka, S.; Tatarkova, Z.; Kaplan, P. Oxidative damage to proteins and lipids during ageing. Acta Medica Martiniana 2012, 12, 5–11. [Google Scholar]
- Berlett, B.S.; Stadtman, E.R. Protein oxidation in aging, disease, and oxidative stress. J. Biol. Chem. 1997, 272, 20313–20316. [Google Scholar] [CrossRef]
- Tiwari, A. Imbalance in antioxidant defence and human diseases: Multiple approach of natural antioxidants therapy. Curr. Sci. 2001, 81, 1179–1187. [Google Scholar]
- Pratt, D.E. Natural Antioxidants from Plant Material. In Phenolic Compounds in Food and Their Effects on Health II; (ACS Symposium Series;); American Chemical Society: Washington, DC, USA, 1992; Volume 507, pp. 54–71. [Google Scholar]
- Pandey, M.R. Use of medicinal plants in Traditional Tibetan therapy system in Upper Mustang, Nepal. Our Nat. 2007, 4, 69–82. [Google Scholar]
- Pala, N.; Negi, A.; Todaria, N. Traditional uses of medicinal plants of Pauri Garhwal, Uttrakhand. N. Y. Sci. J. 2010, 8, 57–61. [Google Scholar]
- Dulamsuren, C.; Hauck, M.; Muhlenberg, M. Vegetation at the taiga forest-steppe borderline in the western Khentey Mountains, northern Mongolia. Ann. Bot. Fenn. 2005, 42, 411–426. [Google Scholar]
- Ligaa, U. Medicinal Plants of Mongolia Used in Traditional Mongolian Medicine; KCA Press: Seoul, South Korea, 1996; p. 222. [Google Scholar]
- Selenge, E.; Odontuya, G.; Batkhuu, J. Screening of Mongolian medicinal plants for their antioxidant activity. Proc. Mong. Acad. Sci. 2010, 195, 48–56. [Google Scholar]
- Mensor, L.L.; Menezes, F.S.; Leitão, G.G.; Reis, A.S.; dos Santos, T.C.; Coube, C.S.; Leitão, S.G. Screening of Brazilian plant extracts for antioxidant activity by the use of DPPH free radical method. Phytother. Res. 2001, 15, 127–130. [Google Scholar] [CrossRef]
- Wagner, H.; Bladt, S. Plant Drug Analysis: A Thin Layer Chromatography Atlas, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 1996. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Clifford, M.N.; Johnston, K.L.; Knight, S.; Kuhnert, N. Hierarchical scheme for LC-MSn identification of chlorogenic acids. J. Agric. Food Chem. 2003, 51, 2900–2911. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Holzer, V.M.D.; Lower-Nedza, A.D.; Nandintsetseg, M.; Batkhuu, J.; Brantner, A.H. Antioxidant Constituents of Cotoneaster melanocarpus Lodd. Antioxidants 2013, 2, 265-272. https://doi.org/10.3390/antiox2040265
Holzer VMD, Lower-Nedza AD, Nandintsetseg M, Batkhuu J, Brantner AH. Antioxidant Constituents of Cotoneaster melanocarpus Lodd. Antioxidants. 2013; 2(4):265-272. https://doi.org/10.3390/antiox2040265
Chicago/Turabian StyleHolzer, Veronika M. D., Agnieszka D. Lower-Nedza, Myagmar Nandintsetseg, Javzan Batkhuu, and Adelheid H. Brantner. 2013. "Antioxidant Constituents of Cotoneaster melanocarpus Lodd." Antioxidants 2, no. 4: 265-272. https://doi.org/10.3390/antiox2040265
APA StyleHolzer, V. M. D., Lower-Nedza, A. D., Nandintsetseg, M., Batkhuu, J., & Brantner, A. H. (2013). Antioxidant Constituents of Cotoneaster melanocarpus Lodd. Antioxidants, 2(4), 265-272. https://doi.org/10.3390/antiox2040265




