Hippocampal Neuronal Loss in Infant Macaques Orally Infected with Virulent Simian Immunodeficiency Virus (SIV)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Brain Preservation and Histology
2.3. Design-Based Stereology
2.4. Statistical Analysis
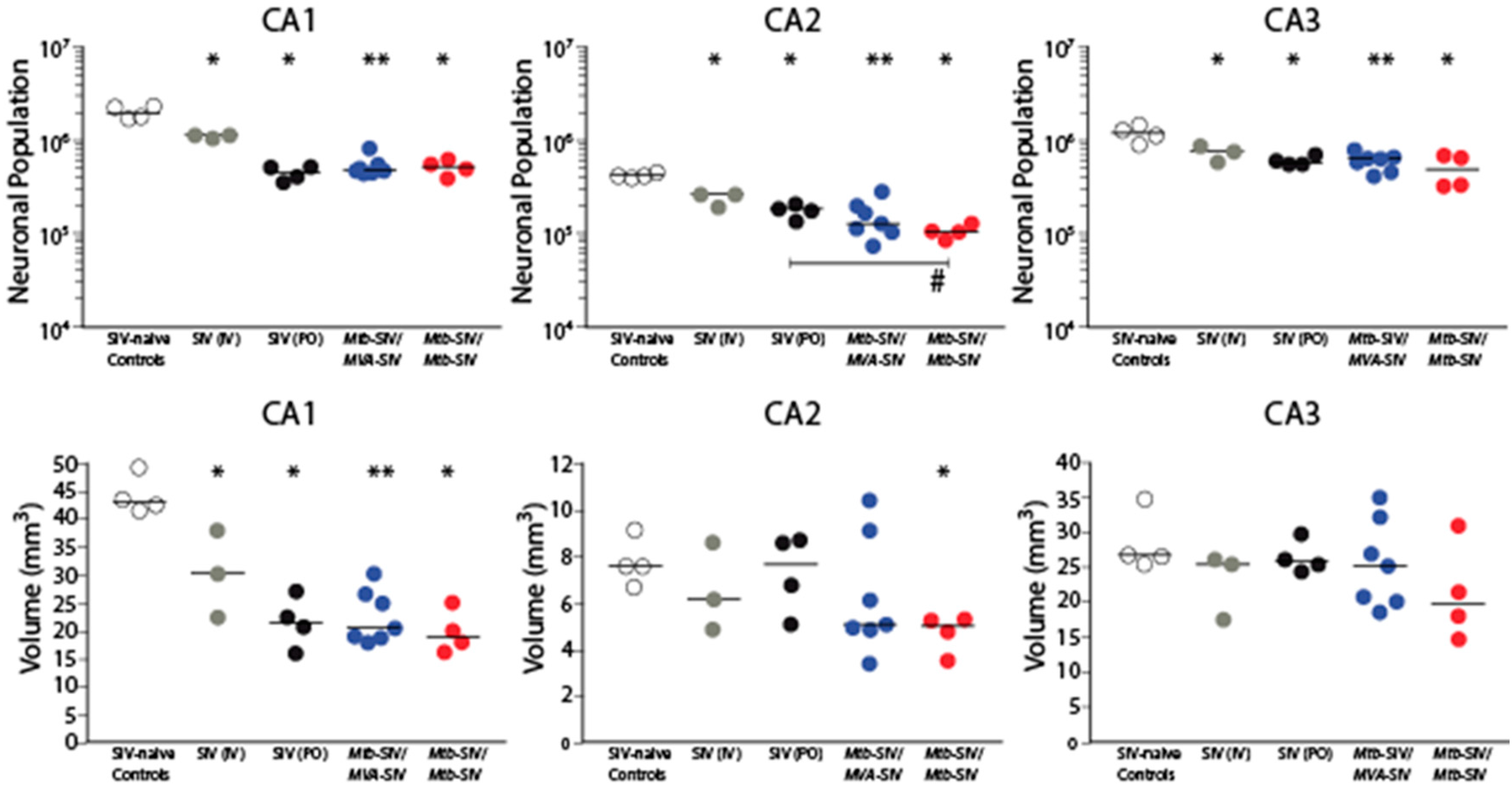
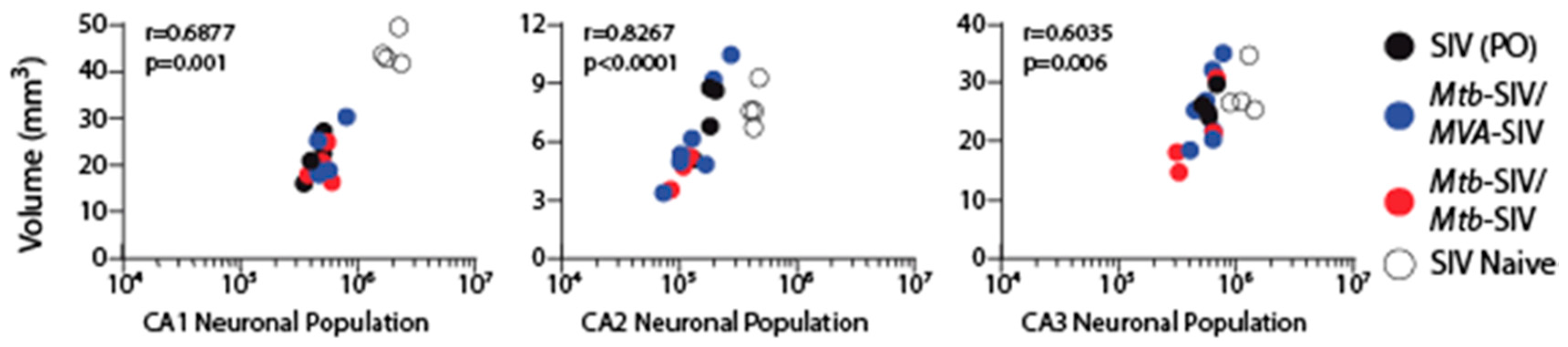
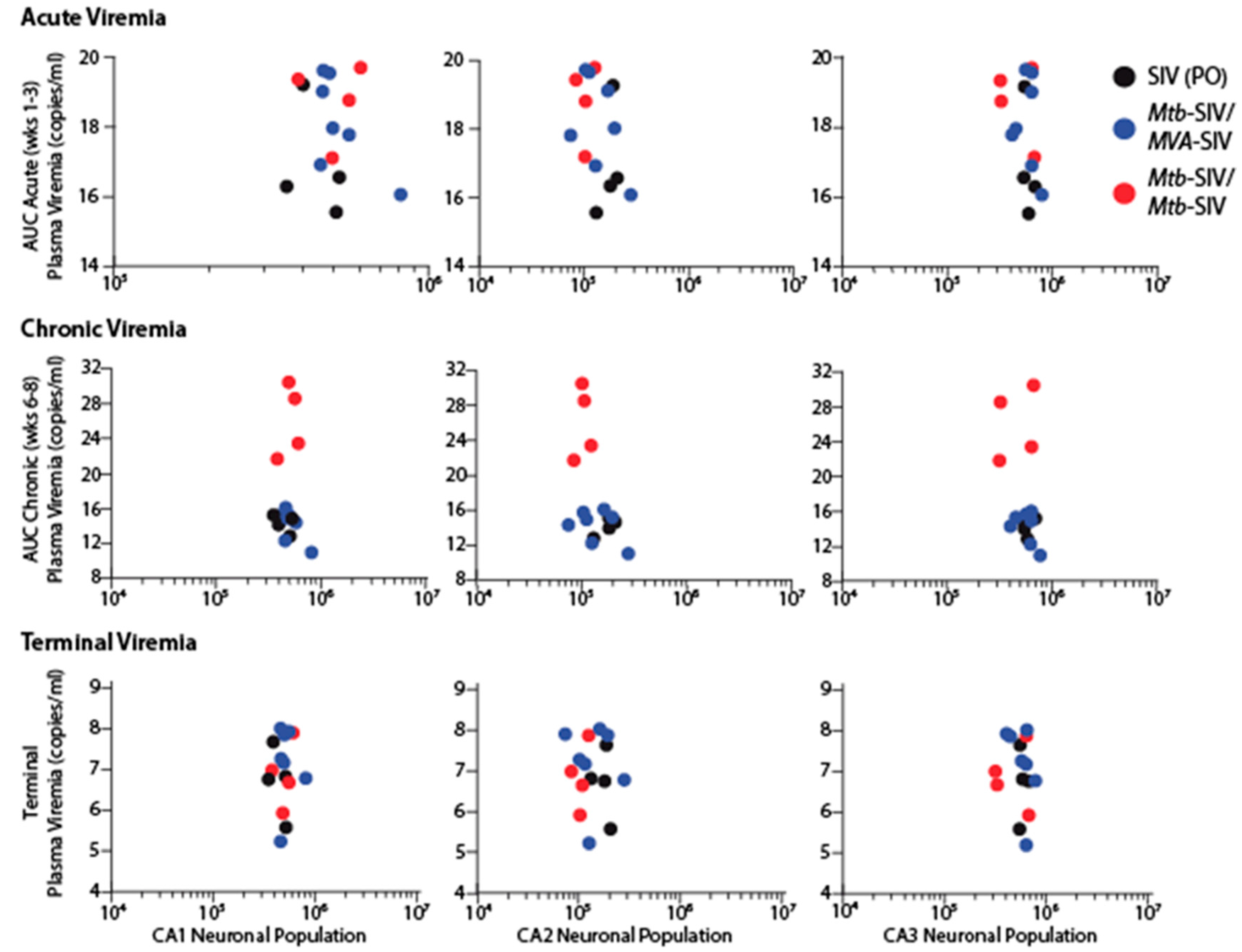
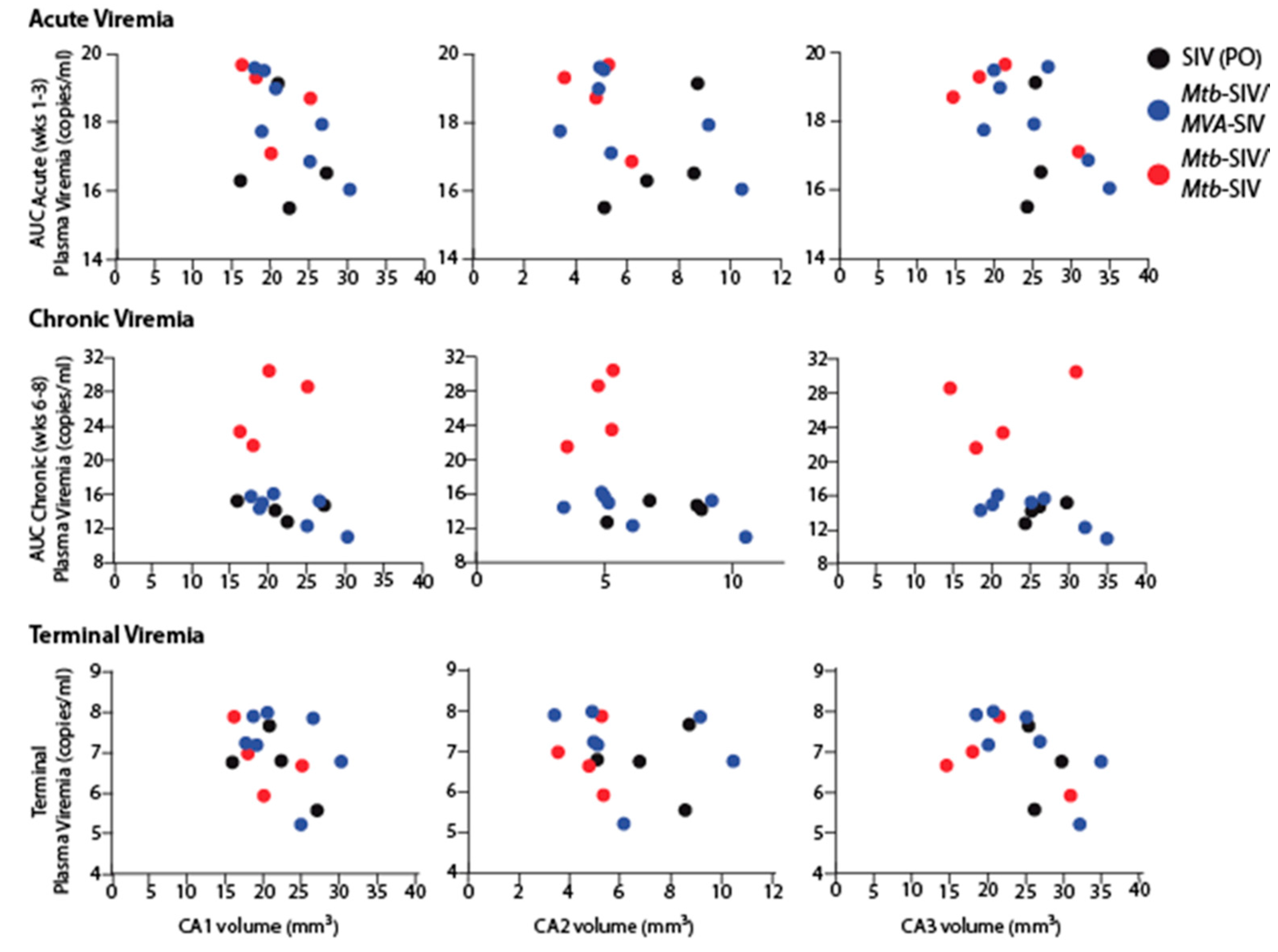
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- The Joint United Nations Programme on HIV/AIDS (UNAIDS). Unaids fact sheet 2014; The Joint United Nations Programme on HIV/AIDS: Geneva, Switzerland, 2014. [Google Scholar]
- UNAIDS/UNICEF/WHO/UNFPA. Children and Aids Fifth Stocktaking Report. Available online: http://www.unaids.org/en/targetsandcommitments/eliminatingnewhivinfectionamongchildren/ (accessed on 27 November 2016).
- UNICEF. HIV and Infant Feeding. Available online: https://www.unicef.org/programme/breastfeeding/hiv.htm (accessed on 21 December 2016).
- AIDS. HIV Prevention Preventing Mother-to-Child Transmission of HIV. Available online: https://aidsinfo.nih.gov/education-materials/fact-sheets/20/50/preventing-mother-to-child-transmission-of-hiv (accessed on 11 January 2017).
- McIntyre, J. Antiretrovirals for Reducing the Risk of Mother-to-Child Transmission of Hiv Infection: Rhl Commentary; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Zash, R.M.; Shapiro, R.L.; Leidner, J.; Wester, C.; McAdam, A.J.; Hodinka, R.L.; Thior, I.; Moffat, C.; Makhema, J.; McIntosh, K.; et al. The aetiology of diarrhoea, pneumonia and respiratory colonization of HIV-exposed infants randomized to breast- or formula-feeding. Paediatr. Int. Child Health 2016, 36, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Becquet, R.; Ekouevi, D.K.; Arrive, E.; Stringer, J.S.; Meda, N.; Chaix, M.L.; Treluyer, J.M.; Leroy, V.; Rouzioux, C.; Blanche, S.; et al. Universal antiretroviral therapy for pregnant and breast-feeding HIV-1-infected women: Towards the elimination of mother-to-child transmission of hiv-1 in resource-limited settings. Clin. Infect. Dis. 2009, 49, 1936–1945. [Google Scholar] [CrossRef] [PubMed]
- Rollins, N.C.; Ndirangu, J.; Bland, R.M.; Coutsoudis, A.; Coovadia, H.M.; Newell, M.L. Exclusive breastfeeding, diarrhoeal morbidity and all-cause mortality in infants of HIV-infected and hiv uninfected mothers: An intervention cohort study in kwazulu natal, south africa. PLoS ONE 2013, 8, e81307. [Google Scholar] [CrossRef] [PubMed]
- Van de Perre, P.; Rubbo, P.A.; Viljoen, J.; Nagot, N.; Tylleskar, T.; Lepage, P.; Vendrell, J.P.; Tuaillon, E. HIV-1 reservoirs in breast milk and challenges to elimination of breast-feeding transmission of hiv-1. Sci. Transl. Med. 2012, 4, 143sr143. [Google Scholar] [CrossRef] [PubMed]
- Ngoma, M.S.; Misir, A.; Mutale, W.; Rampakakis, E.; Sampalis, J.S.; Elong, A.; Chisele, S.; Mwale, A.; Mwansa, J.K.; Mumba, S.; et al. Efficacy of who recommendation for continued breastfeeding and maternal cart for prevention of perinatal and postnatal hiv transmission in zambia. J. Int. AIDS Soc. 2015, 18, 19352. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, I.C.; Santos, W.M.; Padoin, S.M.; Barros, S.M. A five-year review of vertical HIV transmission in a specialized service: Cross-sectional study. Sao Paulo Med. J. 2016, 134, 508–512. [Google Scholar] [CrossRef] [PubMed]
- Coovadia, H.M.; Bland, R.M. Preserving breastfeeding practice through the hiv pandemic. Trop. Med. Int. Health 2007, 12, 1116–1133. [Google Scholar] [CrossRef] [PubMed]
- Coovadia, H.M.; Rollins, N.C.; Bland, R.M.; Little, K.; Coutsoudis, A.; Bennish, M.L.; Newell, M.L. Mother-to-child transmission of HIV-1 infection during exclusive breastfeeding in the first 6 months of life: An intervention cohort study. Lancet 2007, 369, 1107–1116. [Google Scholar] [CrossRef]
- Becquet, R.; Ekouevi, D.K.; Menan, H.; Amani-Bosse, C.; Bequet, L.; Viho, I.; Dabis, F.; Timite-Konan, M.; Leroy, V. ANRS 1201/1202 Ditrame Plus Study Group. Early mixed feeding and breastfeeding beyond 6 months increase the risk of postnatal hiv transmission: Anrs 1201/1202 ditrame plus, abidjan, cote d’ivoire. Prev. Med. 2008, 47, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Manji, K.P.; Duggan, C.; Liu, E.; Bosch, R.; Kisenge, R.; Aboud, S.; Kupka, R.; Fawzi, W.W. Exclusive breast-feeding protects against mother-to-child transmission of HIV-1 through 12 months of age in tanzania. J. Trop. Pediatr. 2016, 62, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Brady, M.T.; Oleske, J.M.; Williams, P.L.; Elgie, C.; Mofenson, L.M.; Dankner, W.M.; Van Dyke, R.B. Pediatric AIDS Clinical Trials Group219/219C Team. Declines in mortality rates and changes in causes of death in HIV-1-infected children during the haart era. J. Acquir. Immune Defic. Syndr. 2010, 53, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Mofenson, L.M.; Cotton, M.F. The challenges of success: Adolescents with perinatal HIV infection. J. Int. AIDS Soc. 2013, 16, 18650. [Google Scholar] [CrossRef] [PubMed]
- Mofenson, L.M. Prevention of mother-to-child HIV transmission: Can we meet the goal of global elimination of new pediatric infections? Curr. Opin. HIV AIDS 2013, 8, 443–446. [Google Scholar] [CrossRef] [PubMed]
- Dowshen, N.; D’Angelo, L. Health care transition for youth living with HIV/AIDS. Pediatrics 2011, 128, 762–771. [Google Scholar] [CrossRef] [PubMed]
- Sohn, A.H.; Hazra, R. The changing epidemiology of the global paediatric hiv epidemic: Keeping track of perinatally HIV-infected adolescents. J. Int. AIDS Soc. 2013, 16, 18555. [Google Scholar] [CrossRef] [PubMed]
- MacDonell, K.; Naar-King, S.; Huszti, H.; Belzer, M. Barriers to medication adherence in behaviorally and perinatally infected youth living with HIV. AIDS Behav. 2013, 17, 86–93. [Google Scholar] [CrossRef] [PubMed]
- van Arnhem, L.A.; Bunders, M.J.; Scherpbier, H.J.; Majoie, C.B.; Reneman, L.; Frinking, O.; Poll-The, B.T.; Kuijpers, T.W.; Pajkrt, D. Neurologic abnormalities in HIV-1 infected children in the era of combination antiretroviral therapy. PLoS ONE 2013, 8, e64398. [Google Scholar] [CrossRef] [PubMed]
- Van Rie, A.; Harrington, P.R.; Dow, A.; Robertson, K. Neurologic and neurodevelopmental manifestations of pediatric HIV/AIDS: A global perspective. Eur. J. Paediatr. Neurol. 2007, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, W. Neurological and developmental effects of HIV and AIDS in children and adolescents. Ment. Retard. Dev. Disabil. Res. Rev. 2001, 7, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.Y.; Pierre, R.B.; Christie, C.D.; Chang, S.M. Neurocognitive function in HIV-positive children in a developing country. Int. J. Infect. Dis. 2013, 17, e862–e867. [Google Scholar] [CrossRef] [PubMed]
- Pollack, H.; Kuchuk, A.; Cowan, L.; Hacimamutoglu, S.; Glasberg, H.; David, R.; Krasinski, K.; Borkowsky, W.; Oberfield, S. Neurodevelopment, growth, and viral load in HIV-infected infants. Brain Behav. Immun. 1996, 10, 298–312. [Google Scholar] [CrossRef] [PubMed]
- Jeremy, R.J.; Kim, S.; Nozyce, M.; Nachman, S.; McIntosh, K.; Pelton, S.I.; Yogev, R.; Wiznia, A.; Johnson, G.M.; Krogstad, P.; et al. Neuropsychological functioning and viral load in stable antiretroviral therapy-experienced HIV-infected children. Pediatrics 2005, 115, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Lewis-de Los Angeles, C.P.; Alpert, K.I.; Williams, P.L.; Malee, K.; Huo, Y.; Csernansky, J.G.; Yogev, R.; Van Dyke, R.B.; Sowell, E.R.; Wang, L.; et al. Deformed subcortical structures are related to past HIV disease severity in youth with perinatally acquired hiv infection. J. Pediatr. Infect. Dis. Soc. 2016, 5, S6–S14. [Google Scholar] [CrossRef] [PubMed]
- Donald, K.A.; Hoare, J.; Eley, B.; Wilmshurst, J.M. Neurologic complications of pediatric human immunodeficiency virus: Implications for clinical practice and management challenges in the african setting. Semin. Pediatr. Neurol. 2014, 21, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Foster, S.B.; Lu, M.; Glaze, D.G.; Reuben, J.M.; Harris, L.L.; Cohen, E.N.; Lee, B.N.; Zhao, E.; Paul, M.E.; Schwarzwald, H.; et al. Associations of cytokines, sleep patterns, and neurocognitive function in youth with HIV infection. Clin. Immunol. 2012, 144, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Hoare, J.; Westgarth-Taylor, J.; Fouche, J.P.; Spottiswoode, B.; Paul, R.; Thomas, K.; Stein, D.; Joska, J. A diffusion tensor imaging and neuropsychological study of prospective memory impairment in south african HIV positive individuals. Metab. Brain Dis. 2012, 27, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Facchini, S.A.; Harding, S.A.; Waldron, R.L. Human immunodeficiency virus-1 infection and multiple sclerosis-like illness in a child. Pediatr. Neurol. 2002, 26, 231–235. [Google Scholar] [CrossRef]
- McGuire, J.L.; Gill, A.J.; Douglas, S.D.; Kolson, D.L.; group, C.H.A.-R.T.E.R. Central and peripheral markers of neurodegeneration and monocyte activation in HIV-associated neurocognitive disorders. J. Neurovirol. 2015, 21, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Norgren, N.; Rosengren, L.; Stigbrand, T. Elevated neurofilament levels in neurological diseases. Brain Res. 2003, 987, 25–31. [Google Scholar] [CrossRef]
- Jessen Krut, J.; Mellberg, T.; Price, R.W.; Hagberg, L.; Fuchs, D.; Rosengren, L.; Nilsson, S.; Zetterberg, H.; Gisslen, M. Biomarker evidence of axonal injury in neuroasymptomatic HIV-1 patients. PLoS ONE 2014, 9, e88591. [Google Scholar] [CrossRef] [PubMed]
- Peluso, M.J.; Meyerhoff, D.J.; Price, R.W.; Peterson, J.; Lee, E.; Young, A.C.; Walter, R.; Fuchs, D.; Brew, B.J.; Cinque, P.; et al. Cerebrospinal fluid and neuroimaging biomarker abnormalities suggest early neurological injury in a subset of individuals during primary HIV infection. J. Infect. Dis. 2013, 207, 1703–1712. [Google Scholar] [CrossRef] [PubMed]
- Abdulle, S.; Mellgren, A.; Brew, B.J.; Cinque, P.; Hagberg, L.; Price, R.W.; Rosengren, L.; Gisslen, M. Csf neurofilament protein (NFL)—A marker of active HIV-related neurodegeneration. J. Neurol. 2007, 254, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
- Carryl, H.; Swang, M.; Lawrence, J.; Curtis, K.; Kamboj, H.; Van Rompay, K.K.; De Paris, K.; Burke, M.W. Of mice and monkeys: Can animal models be utilized to study neurological consequences of pediatric HIV-1 infection? ACS Chem. Neurosci. 2015, 6, 1276–1289. [Google Scholar] [CrossRef] [PubMed]
- Bieniasz, P.D.; Cullen, B.R. Multiple blocks to human immunodeficiency virus type 1 replication in rodent cells. J. Virol. 2000, 74, 9868–9877. [Google Scholar] [CrossRef] [PubMed]
- Fitting, S.; Booze, R.M.; Mactutus, C.F. Neonatal intrahippocampal injection of the HIV-1 proteins GP120 and tat: Differential effects on behavior and the relationship to stereological hippocampal measures. Brain Res. 2008, 1232, 139–154. [Google Scholar] [CrossRef] [PubMed]
- Moran, L.M.; Fitting, S.; Booze, R.M.; Webb, K.M.; Mactutus, C.F. Neonatal intrahippocampal HIV-1 protein Tat(1-86) injection: Neurobehavioral alterations in the absence of increased inflammatory cytokine activation. Int. J. Dev. Neurosci. 2014, 38, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Fitting, S.; Booze, R.M.; Hasselrot, U.; Mactutus, C.F. Dose-dependent long-term effects of tat in the rat hippocampal formation: A design-based stereological study. Hippocampus 2010, 20, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.M.; Mervis, R.F.; Avidor, R.; Moody, T.W.; Brenneman, D.E. Hiv envelope protein-induced neuronal damage and retardation of behavioral development in rat neonates. Brain Res. 1993, 603, 222–233. [Google Scholar] [CrossRef]
- Fitting, S.; Booze, R.M.; Mactutus, C.F. Neonatal intrahippocampal gp120 injection: An examination early in development. Neurotoxicology 2007, 28, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Ensoli, B.; Buonaguro, L.; Barillari, G.; Fiorelli, V.; Gendelman, R.; Morgan, R.A.; Wingfield, P.; Gallo, R.C. Release, uptake, and effects of extracellular human immunodeficiency virus type 1 tat protein on cell growth and viral transactivation. J. Virol. 1993, 67, 277–287. [Google Scholar] [PubMed]
- Clements, J.E.; Mankowski, J.L.; Gama, L.; Zink, M.C. The accelerated simian immunodeficiency virus macaque model of human immunodeficiency virus-associated neurological disease: From mechanism to treatment. J. Neurovirol. 2008, 14, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Abel, K. The rhesus macaque pediatric siv infection model—A valuable tool in understanding infant hiv-1 pathogenesis and for designing pediatric HIV-1 prevention strategies. Curr. HIV Res. 2009, 7, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Worlein, J.M.; Leigh, J.; Larsen, K.; Kinman, L.; Schmidt, A.; Ochs, H.; Ho, R.J. Cognitive and motor deficits associated with HIV-2(287) infection in infant pigtailed macaques: A nonhuman primate model of pediatric neuro-aids. J. Neurovirol. 2005, 11, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Curtis, K.; Rollins, M.; Carryl, H.; Bradshaw, K.; Van Rompay, K.K.; Abel, K.; Burke, M.W. Reduction of pyramidal and immature hippocampal neurons in pediatric simian immunodeficiency virus infection. Neuroreport 2014, 25, 973–978. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.; Nabi, R.; Van Rompay, K.K.; Robichaux, S.; Lifson, J.D.; Piatak, M., Jr.; Jacobs, W.R., Jr.; Fennelly, G.; Canfield, D.; Mollan, K.R.; et al. Vaccine-elicited mucosal and systemic antibody responses are associated with reduced simian immunodeficiency viremia in infant rhesus macaques. J. Virol. 2016, 90, 7285–7302. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.; Dela Pena-Ponce, M.G.; Piatak, M., Jr.; Shoemaker, R.; Oswald, K.; Jacobs, W.R., Jr.; Fennelly, G.; Lucero, C.; Mollan, K.R.; Hudgens, M.G.; et al. Balancing trained immunity with persistent immune activation and the risk of simian immunodeficiency virus infection in infant macaques vaccinated with attenuated mycobacterium tuberculosis or mycobacterium bovis bcg vaccine. Clin. Vaccine Immunol. 2017, 24, e00360-16. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.; Ranganathan, U.D.; Van Rompay, K.K.; Canfield, D.R.; Khan, I.; Ravindran, R.; Luciw, P.A.; Jacobs, W.R., Jr.; Fennelly, G.; Larsen, M.H.; et al. A recombinant attenuated mycobacterium tuberculosis vaccine strain is safe in immunosuppressed simian immunodeficiency virus-infected infant macaques. Clin. Vaccine Immunol. 2012, 19, 1170–1181. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.; Pena, M.G.; Wilson, R.L.; Ranganathan, U.D.; Jacobs, W.R., Jr.; Fennelly, G.; Larsen, M.; Van Rompay, K.K.; Kozlowski, P.A.; Abel, K. A neonatal oral mycobacterium tuberculosis-siv prime/intramuscular mva-siv boost combination vaccine induces both siv and mtb-specific immune responses in infant macaques. Trials Vaccinol. 2013, 2, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Burke, M.W.; Ptito, M.; Ervin, F.R.; Palmour, R.M. Hippocampal neuron populations are reduced in vervet monkeys with fetal alcohol exposure. Dev. Psychobiol. 2015, 57, 470–485. [Google Scholar] [CrossRef] [PubMed]
- Jabes, A.; Lavenex, P.B.; Amaral, D.G.; Lavenex, P. Postnatal development of the hippocampal formation: A stereological study in macaque monkeys. J. Comp. Neurol. 2011, 519, 1051–1070. [Google Scholar] [CrossRef] [PubMed]
- Cline, A.N.; Bess, J.W.; Piatak, M., Jr.; Lifson, J.D. Highly sensitive siv plasma viral load assay: Practical considerations, realistic performance expectations, and application to reverse engineering of vaccines for aids. J. Med. Primatol. 2005, 34, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Holland, B.; Russo, C.; Dailey, P.J.; Marx, P.A.; Connor, R.I. Retrospective analysis of viral load and siv antibody responses in rhesus macaques infected with pathogenic siv: Predictive value for disease progression. AIDS Res. Hum. Retrovir. 1999, 15, 1691–1701. [Google Scholar] [CrossRef] [PubMed]
- Watson, A.; Ranchalis, J.; Travis, B.; McClure, J.; Sutton, W.; Johnson, P.R.; Hu, S.L.; Haigwood, N.L. Plasma viremia in macaques infected with simian immunodeficiency virus: Plasma viral load early in infection predicts survival. J. Virol. 1997, 71, 284–290. [Google Scholar] [PubMed]
- Phillips, N.; Amos, T.; Kuo, C.; Hoare, J.; Ipser, J.; Thomas, K.G.; Stein, D.J. HIV-associated cognitive impairment in perinatally infected children: A meta-analysis. Pediatrics 2016, 138. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Long, C.; Yang, L. Hippocampal-prefrontal circuit and disrupted functional connectivity in psychiatric and neurodegenerative disorders. Biomed. Res. Int. 2015, 2015, 810548. [Google Scholar] [CrossRef] [PubMed]
- Lavenex, P.; Amaral, D.G. Hippocampal-neocortical interaction: A hierarchy of associativity. Hippocampus 2000, 10, 420–430. [Google Scholar] [CrossRef]
- Friedman, H.R.; Goldman-Rakic, P.S. Activation of the hippocampus and dentate gyrus by working-memory: A 2-deoxyglucose study of behaving rhesus monkeys. J. Neurosci. 1988, 8, 4693–4706. [Google Scholar] [PubMed]
- Nagarajan, R.; Sarma, M.K.; Thomas, M.A.; Chang, L.; Natha, U.; Wright, M.; Hayes, J.; Nielsen-Saines, K.; Michalik, D.E.; Deville, J.; et al. Neuropsychological function and cerebral metabolites in hiv-infected youth. J. Neuroimmune Pharmacol. 2012, 7, 981–990. [Google Scholar] [CrossRef] [PubMed]
- Luthert, P.J.; Montgomery, M.M.; Dean, A.F.; Cook, R.W.; Baskerville, A.; Lantos, P.L. Hippocampal neuronal atrophy occurs in rhesus macaques following infection with simian immunodeficiency virus. Neuropathol. Appl. Neurobiol. 1995, 21, 529–534. [Google Scholar] [CrossRef] [PubMed]
- Spruston, N. Pyramidal neurons: Dendritic structure and synaptic integration. Nat. Rev. Neurosci. 2008, 9, 206–221. [Google Scholar] [CrossRef] [PubMed]
- Abrams, E.J.; Weedon, J.; Steketee, R.W.; Lambert, G.; Bamji, M.; Brown, T.; Kalish, M.L.; Schoenbaum, E.E.; Thomas, P.A.; Thea, D.M. Association of human immunodeficiency virus (HIV) load early in life with disease progression among hiv-infected infants. New york city perinatal hiv transmission collaborative study group. J. Infect. Dis. 1998, 178, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Shearer, W.T.; Quinn, T.C.; LaRussa, P.; Lew, J.F.; Mofenson, L.; Almy, S.; Rich, K.; Handelsman, E.; Diaz, C.; Pagano, M.; et al. Viral load and disease progression in infants infected with human immunodeficiency virus type 1. Women and infants transmission study group. N. Engl. J. Med. 1997, 336, 1337–1342. [Google Scholar] [CrossRef] [PubMed]
- Marthas, M.L.; van Rompay, K.K.; Otsyula, M.; Miller, C.J.; Canfield, D.R.; Pedersen, N.C.; McChesney, M.B. Viral factors determine progression to aids in simian immunodeficiency virus-infected newborn rhesus macaques. J. Virol. 1995, 69, 4198–4205. [Google Scholar] [PubMed]
- Van Rompay, K.K.; Singh, R.P.; Brignolo, L.L.; Lawson, J.R.; Schmidt, K.A.; Pahar, B.; Canfield, D.R.; Tarara, R.P.; Sodora, D.L.; Bischofberger, N.; et al. The clinical benefits of tenofovir for simian immunodeficiency virus-infected macaques are larger than predicted by its effects on standard viral and immunologic parameters. J. Acquir. Immune Defic. Syndr. 2004, 36, 900–914. [Google Scholar] [CrossRef] [PubMed]
- Kovalevich, J.; Langford, D. Neuronal toxicity in hiv cns disease. Future Virol. 2012, 7, 687–698. [Google Scholar] [CrossRef] [PubMed]
- Ellis, R.; Langford, D.; Masliah, E. Hiv and antiretroviral therapy in the brain: Neuronal injury and repair. Nat. Rev. Neurosci. 2007, 8, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Nath, A. Human immunodeficiency virus (HIV) proteins in neuropathogenesis of hiv dementia. J. Infect. Dis. 2002, 186, S193–S198. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Banks, W.A. Role of the immune system in hiv-associated neuroinflammation and neurocognitive implications. Brain Behav. Immun. 2015, 45, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Richards, M.H.; Narasipura, S.D.; Kim, S.; Seaton, M.S.; Lutgen, V.; Al-Harthi, L. Dynamic interaction between astrocytes and infiltrating pbmcs in context of neuroaids. Glia 2015, 63, 441–451. [Google Scholar] [CrossRef] [PubMed]
- Zayyad, Z.; Spudich, S. Neuropathogenesis of hiv: From initial neuroinvasion to HIV-associated neurocognitive disorder (hand). Curr. HIV/AIDS Rep. 2015, 12, 16–24. [Google Scholar] [CrossRef] [PubMed]

| Group | Subject ID | Gender | Vaccine | Age of SIV Infection | Age at Euthanasia | Total Infection Period | Plasma SIV Ribonucleic Acid (RNA) (Copies/mL) * |
|---|---|---|---|---|---|---|---|
| 1 | 41622 1 | M | none | 1 week | 10 weeks | 9 weeks | 160,000,000 |
| 1 | 41614 1 | F | none | 1 week | 7 weeks | 7 weeks | 240,000,000 |
| 1 | 41615 1 | F | none | 1 week | 10 weeks | 10 weeks | 650,000,000 |
| 2A | 42376 2 | F | none | 9 weeks | 21 weeks | 12 weeks | 5,800,000 |
| 2A | 42380 2 | F | none | 17 weeks | 27 weeks | 10 weeks | 6,400,000 |
| 2A | 42386 2 | F | none | 10 weeks | 22 weeks | 12 weeks | 380,000 |
| 2A | 42388 2 | F | none | 13 weeks | 25 weeks | 12 weeks | 46,000,000 |
| 2B | 42944 2 | M | Mtb-SIV/MVA-SIV | 9 weeks | 33 weeks | 24 weeks | 170,000 |
| 2B | 42958 2 | F | Mtb-SIV/MVA-SIV | 15 weeks | 33 weeks | 18 weeks | 6,000,000 |
| 2B | 42949 2 | M | Mtb-SIV/MVA-SIV | 9 weeks | 21 weeks | 12 weeks | 18,000,000 |
| 2B | 42899 2 | F | Mtb-SIV/MVA-SIV | 9 weeks | 20 weeks | 11 weeks | 84,000,000 |
| 2B | 42929 2 | F | Mtb-SIV/MVA-SIV | 10 weeks | 20 weeks | 10 weeks | 99,000,000 |
| 2B | 42906 2 | F | Mtb-SIV/MVA-SIV | 9 weeks | 22 weeks | 13 weeks | 73,000,000 |
| 2B | 42937 2 | M | Mtb-SIV/ MVA-SIV | 9 weeks | 21 weeks | 12 weeks | 15,000,000 |
| 2C | 42925 3 | F | Mtb-SIV/Mtb –SIV | 10 weeks | 22 weeks | 12 weeks | 4,700,000 |
| 2C | 42943 3 | M | Mtb-SIV/Mtb –SIV | 10 weeks | 21 weeks | 11 weeks | 9,900,000 |
| 2C | 42950 3 | M | Mtb-SIV/Mtb –SIV | 9 weeks | 19 weeks | 10 weeks | 77,000,000 |
| 2C | 42918 3 | M | Mtb-SIV/Mtb –SIV | 19 weeks | 34 weeks | 15 weeks | 860,000 |
| 3 | 40967 4 | F | None | N/A | 15 wks | N/A | N/A |
| 3 | 40929 4 | M | None | N/A | 16 wks | N/A | N/A |
| 3 | 41656 4 | F | None | N/A | 16 wks | N/A | N/A |
| 3 | 41660 4 | F | None | N/A | 16 wks | N/A | N/A |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carryl, H.; Van Rompay, K.K.A.; De Paris, K.; Burke, M.W. Hippocampal Neuronal Loss in Infant Macaques Orally Infected with Virulent Simian Immunodeficiency Virus (SIV). Brain Sci. 2017, 7, 40. https://doi.org/10.3390/brainsci7040040
Carryl H, Van Rompay KKA, De Paris K, Burke MW. Hippocampal Neuronal Loss in Infant Macaques Orally Infected with Virulent Simian Immunodeficiency Virus (SIV). Brain Sciences. 2017; 7(4):40. https://doi.org/10.3390/brainsci7040040
Chicago/Turabian StyleCarryl, Heather, Koen K. A. Van Rompay, Kristina De Paris, and Mark W. Burke. 2017. "Hippocampal Neuronal Loss in Infant Macaques Orally Infected with Virulent Simian Immunodeficiency Virus (SIV)" Brain Sciences 7, no. 4: 40. https://doi.org/10.3390/brainsci7040040
APA StyleCarryl, H., Van Rompay, K. K. A., De Paris, K., & Burke, M. W. (2017). Hippocampal Neuronal Loss in Infant Macaques Orally Infected with Virulent Simian Immunodeficiency Virus (SIV). Brain Sciences, 7(4), 40. https://doi.org/10.3390/brainsci7040040






