A Double-Edged Sword: Volatile Anesthetic Effects on the Neonatal Brain
Abstract
:1. Introduction
2. Methods
3. Results
4. Physiology of Brain Development, Synaptogenesis and Neuroapoptosis
5. Volatile Anesthetics—Mechanisms of Action
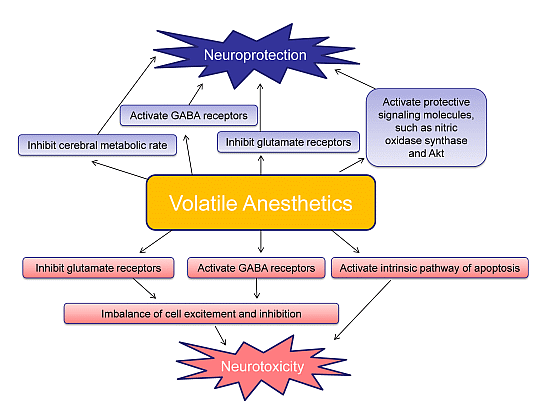
6. Volatile Anesthetic-Induced Neuroprotection
6.1. Animal Studies

6.2. Clinical Studies
7. Volatile Anesthetic-Induced Neurotoxicity
7.1. Animal Studies
7.2. Suggested Mechanisms of Neuroapoptosis Induced by General Anesthesia
7.3. Clinical Studies
8. Looking Forward on Anesthetic-Induced Neurotoxicity Research
9. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- DeFrances, C.J.; Cullen, K.A.; Kozak, L.J. National Hospital Discharge Survey: 2005 annual summary with detailed diagnosis and procedure data. Vital Health Stat. 13 2007, 165, 1–209. [Google Scholar]
- Cao, L.; Li, L.; Lin, D.; Zuo, Z. Isoflurane induces learning impairment that is mediated by interleukin 1β in rodents. PLoS One 2012, 7, e51431. [Google Scholar]
- Jevtovic-Todorovic, V.; Hartman, R.E.; Izumi, Y.; Benshoff, N.D.; Dikranian, K.; Zorumski, C.F.; Olney, J.W.; Wozniak, D.F. Early exposure to common anesthetic agents causes widespread neurodegeneration in the developing rat brain and persistent learning deficits. J. Neurosci. 2003, 23, 876–882. [Google Scholar]
- Ikonomidou, C.; Bosch, F.; Miksa, M.; Bittigau, P.; Vöckler, J.; Dikranian, K.; Tenkova, T.I.; Stefovska, V.; Turski, L.; Olney, J.W. Blockade of NMDA receptors and apoptotic neurodegeneration in the developing brain. Science 1999, 283, 70–74. [Google Scholar] [CrossRef]
- Head, B.P.; Patel, H.H.; Niesman, I.R.; Drummond, J.C.; Roth, D.M.; Patel, P.M. Inhibition of p75 neurotrophin receptor attenuates isoflurane-mediated neuronal apoptosis in the neonatal central nervous system. Anesthesiology 2009, 110, 813–825. [Google Scholar] [CrossRef]
- Wang, S.Q.; Fang, F.; Xue, Z.G.; Cang, J.; Zhang, X.G. Neonatal sevoflurane anesthesia induced long-term memory impairment and decreases hippocampal PSD-95 expression without neuronal loss. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 941–950. [Google Scholar]
- Brambrink, A.M.; Evers, A.S.; Avidan, M.S.; Farber, N.B.; Smith, D.J.; Zhang, X.; Dissen, G.A.; Creeley, C.E.; Olney, J.W. Isoflurane-induced neuroapoptosis in the neonatal rhesus macaque brain. Anesthesiology 2010, 112, 834–841. [Google Scholar] [CrossRef]
- Fredriksson, A.; Pontén, E.; Gordh, T.; Eriksson, P. Neonatal exposure to a combination of N-methyl-d-aspartate and γ-aminobutyric acid type A receptor anesthetic agents potentiates apoptotic neurodegeneration and persistent behavioral deficits. Anesthesiology 2007, 107, 427–436. [Google Scholar] [CrossRef]
- Zhu, C.; Gao, J.; Karlsson, N.; Li, Q.; Zhang, Y.; Huang, Z.; Li, H.; Kuhn, H.G.; Blomgren, K. Isoflurane anesthesia induced persistent, progressive memory impairment, caused a loss of neural stem cells, and reduced neurogenesis in young, but not adult, rodents. J. Cereb. Blood Flow Metab. 2010, 30, 1017–1030. [Google Scholar] [CrossRef]
- Lunardi, N.; Ori, C.; Erisir, A.; Jevtovic-Todorovic, V. General anesthesia causes long-lasting disturbances in the ultrastructural properties of developing synapses in young rats. Neurotox. Res. 2010, 17, 179–188. [Google Scholar] [CrossRef]
- Yon, J.H.; Daniel-Johnson, J.; Carter, L.B.; Jevtovic-Todorovic, V. Anesthesia induces neuronal cell death in the developing rat brain via the intrinsic and extrinsic apoptotic pathways. Neuroscience 2005, 135, 815–827. [Google Scholar] [CrossRef]
- Uemura, E.; Levin, E.D.; Bowman, R.E. Effects of halothane on synaptogenesis and learning behavior in rats. Exp. Neurol. 1985, 89, 520–529. [Google Scholar] [CrossRef]
- Stratmann, G.; Sall, J.W.; May, L.D.V.; Bell, J.S.; Magnusson, K.R.; Rau, V.; Visrodia, K.H.; Alvi, R.S.; Ku, B.; Lee, M.T.; et al. Isoflurane differentially affects neurogenesis and long-term neurocognitive Ffnction in 60-day-old and 7-day-old rats. Anesthesiology 2009, 110, 834–848. [Google Scholar] [CrossRef]
- Chalon, J.; Tang, C.K.; Ramanathan, S.; Eisner, M.; Katz, R.; Turndorf, H. Exposure to halothane and enflurane affects learning function of murine progeny. Anesth. Analg. 1981, 60, 794–797. [Google Scholar]
- Sprung, J.; Flick, R.P.; Katusic, S.K.; Colligan, R.C.; Barbaresi, W.J.; Bojanic, K.; Welch, T.L.; Olson, M.D.; Hanson, A.C.; Schroeder, D.R.; et al. Attention deficit/hyperactivity disorder after early exposure to procedures requiring general anesthesia. Mayo Clin. Proc. 2012, 87, 120–129. [Google Scholar] [CrossRef]
- Lee, J.J.; Li, L.; Jung, H.H.; Zuo, Z. Postconditioning with isoflurane reduced ischemia-induced brain injury in rats. Anesthesiology 2008, 108, 1055–1062. [Google Scholar] [CrossRef]
- Zhao, P.; Peng, L.; Li, L.; Xu, X.; Zuo, Z. Isoflurane preconditioning improves long-term neurologic outcome after hypoxic-ischemic brain injury in neonatal rats. Anesthesiology 2007, 107, 963–970. [Google Scholar] [CrossRef]
- Zhao, P.; Zuo, Z. Isoflurane preconditioning induces neuroprotection that is inducible nitric oxide synthase-dependent in neonatal rats. Anesthesiology 2004, 101, 695–702. [Google Scholar] [CrossRef]
- McMurtrey, R.J.; Zuo, Z. Isoflurane preconditioning and postconditioning in rat hippocampal neurons. Brain Res. 2010, 1358, 184–190. [Google Scholar] [CrossRef]
- Jevtovic-Todorovic, V.; Todorovic, S.M.; Mennerick, S.; Powell, S.; Dikranian, K.; Benshoff, N.; Zorumski, C.F.; Olney, J.W. Nitrous oxide (laughing gas) is an NMDA antagonist, neuroprotectant and neurotoxin. Nat. Med. 1998, 4, 460–463. [Google Scholar] [CrossRef]
- Dobbing, J.; Sands, J. Comparative aspects of the brain growth spurt. Early Hum. Dev. 1979, 3, 79–83. [Google Scholar] [CrossRef]
- Rodier, P.M. Behavioral Effects of Antimitotic Agents Administered During Neurogenesis. In Handbook of Behavioral Teratology; Riley, E.P., Vorhees, C.V., Eds.; Springer: Rochester, NY, USA, 1986; pp. 185–209. [Google Scholar]
- Rodier, P.M.; Webster, W.S.; Langman, J. Morphological and Behavioral Consequences of Chemically-Induced Lesions of the CNS. In Aberrant Development in Human Infancy: Human and Animal Studies; Ellis, N., Ed.; John Wiley & Sons: Hillsdale, NJ, USA, 1975; pp. 169–176. [Google Scholar]
- Wiggins, R.C. Myelin development and nutritional insufficiency. Brain Res. 1982, 257, 151–175. [Google Scholar] [CrossRef]
- Chi, J.G.; Dooling, E.C.; Gilles, F.H. Gyral development of the human brain. Ann. Neurol. 1977, 1, 86–93. [Google Scholar] [CrossRef]
- Buss, R.R.; Oppenheim, R.W. Role of programmed cell death in normal neuronal development and function. Anat. Sci. Int. 2004, 79, 191–197. [Google Scholar] [CrossRef]
- Braun, S.; Gaza, N.; Werdehausen, R.; Hermanns, H.; Bauer, K.; Durieux, M.E.; Hollman, M.W.; Stevens, M.F. Ketamine induces apoptosis via the mitochondrial pathway in human lymphocytes and neuronal cells. Br. J. Anaesth. 2010, 105, 347–354. [Google Scholar] [CrossRef]
- Kim, J.M.; Lee, J.H.; Lee, H.J.; Koo, B.N. Comparison of emergence time in children undergoing minor surgery according to anesthetic: Desflurane and sevoflurane. Yonsei Med. J. 2013, 54, 732–738. [Google Scholar] [CrossRef]
- Van Hemelrijck, J.; Smith, I.; White, P.F. Use of desflurane for outpatient anesthesia: A comparison with propofol and nitrous oxide. Anesthesiology 1991, 75, 197–203. [Google Scholar] [CrossRef]
- Zwass, M.S.; Fisher, D.M.; Welborn, L.G.; Coté, C.J.; Davis, P.J.; Dinner, M.; Hannallah, R.S.; Liu, L.M.; Sarner, J.; McGill, W.A.; et al. Induction and maintenance characteristics of anesthesia with desflurane and nitrous oxide in infants and children. Anesthesiology 1992, 76, 373–378. [Google Scholar] [CrossRef]
- Yurino, M.; Kimura, H. Vital capacity breath technique for rapid anaesthetic induction: Comparison of sevoflurane and isoflurane. Anaesthesia 1992, 47, 946–949. [Google Scholar] [CrossRef]
- Smith, I.; Ding, Y.; White, P.F. Comparison of induction, maintenance and recovery characteristics of sevoflurane-N2O and propofol-sevoflurane-N2O with propofol-isoflurane-N2O. Anesth. Analg. 1992, 74, 253–259. [Google Scholar] [CrossRef]
- Frink, E.J., Jr.; Malan, T.P.; Atlas, M.; Dominguez, L.M.; DiNardo, J.A.; Brown, B.R., Jr. Clinical comparison of sevoflurane and isoflurane in healthy patients. Anesth. Analg. 1992, 74, 241–245. [Google Scholar] [CrossRef]
- Eger, E.I., II; Johnson, B.H. Rates of awakening from anesthesia with I-653, halothane, isoflurane, and sevoflurane: A test of the effect of anesthetic concentration and duration in rats. Anesth. Analg. 1987, 66, 977–982. [Google Scholar]
- Overton, C.E. Studien über die Narkose zugleich ein Beitrag zur Allgemeinen Pharmakologie; (in German). Fischer: Berlin, German, 1901. [Google Scholar]
- Meyer, H. Zur Theorie der Alkoholnarkose. Arch. Exp. Pathol. Pharmakol. 1899, 42, 109–118. (in German). [Google Scholar] [CrossRef]
- Franks, N.P.; Lieb, W.R. Do general anaesthetics act by competitive binding to specific receptors? Nature 1984, 310, 599–601. [Google Scholar] [CrossRef]
- Koblin, D.D.; Chortkoff, B.S.; Laster, M.J.; Eger, E.I., II; Halsey, M.J.; Ionescu, P. Polyhalogenated and perfluorinated compounds that disobey the Meyer-Overton hypothesis. Anesth. Analg. 1994, 79, 1043–1048. [Google Scholar]
- Franks, N.P.; Lieb, W.R. Stereospecific effects of inhalational general anesthetic optical isomers on nerve ion channels. Science 1991, 254, 427–430. [Google Scholar]
- Olsen, R.W.; Li, G.D. GABAA receptors as molecular targets of general anesthetics: Identification of binding sites provides clues to allosteric modulation. Can. J. Anaesth. 2011, 58, 206–215. [Google Scholar] [CrossRef]
- Cechova, S.; Zuo, Z. Inhibition of glutamate transporters increases the minimum alveolar concentration for isoflurane in rats. Br. J. Anaesth. 2006, 97, 192–195. [Google Scholar] [CrossRef]
- Nishikawa, K.; Harrison, N.L. The actions of sevoflurane and desflurane on the gamma-aminobutyric acid receptor type A: Effects of TM2 mutations in the alpha and beta subunits. Anesthesiology 2003, 99, 678–684. [Google Scholar] [CrossRef]
- Mihic, S.J.; Ye, Q.; Wick, M.J.; Koltchine, V.V.; Krasowski, M.D.; Finn, S.E.; Mascia, M.P.; Valenzuela, C.F.; Hanson, K.K.; Greenblatt, E.P.; et al. Sites of alcohol and volatile anaesthetic action on GABAA and glycine receptors. Nature 1997, 389, 385–389. [Google Scholar] [CrossRef]
- Harrison, N.L.; Simmonds, M.A. Quantitative studies on some antagonists of N-methyl d-aspartate in slices of rat cerebral cortex. Br. J. Pharmacol. 1985, 84, 381–391. [Google Scholar] [CrossRef]
- Wachtel, R.E.; Wegrzynowicz, E.S. Mechanism of volatile anesthetic action on ion channels. Ann. N. Y. Acad. Sci. 1991, 625, 116–128. [Google Scholar] [CrossRef]
- Horishita, T.; Eger, E.I., II; Harris, R.A. The effects of volatile aromatic anesthetics on voltage-gated Na+ channels expressed in Xenopus oocytes. Anesth. Analg. 2008, 107, 1579–1586. [Google Scholar] [CrossRef]
- Sonner, J.M.; Antognini, J.F.; Dutton, R.C.; Flood, P.; Gray, A.T.; Harris, R.A.; Homanics, G.E.; Kendig, J.; Orser, B.; Raines, D.E.; et al. Inhaled anesthetics and immobility: Mechanisms, mysteries, and minimum alveolar anesthetic concentration. Anesth. Analg. 2003, 97, 718–740. [Google Scholar]
- Wells, B.A.; Keats, A.S.; Cooley, D.A. Increased tolerance to cerebral ischemia produced by general anesthesia during temporary carotid occlusion. Surgery 1963, 54, 216–223. [Google Scholar]
- Schifilliti, D.; Grasso, G.; Conti, A.; Fodale, V. Anaesthetic-related neuroprotection: Intravenous or inhalational agents? CNS Drugs 2010, 24, 893–907. [Google Scholar]
- Burchell, S.R.; Dixon, B.J.; Tang, J.; Zhang, J.H. Isoflurane provides neuroprotection in neonatal hypoxic ischemic brain injury. J. Investig. Med. 2013, 61, 1078–1083. [Google Scholar]
- Yan, J.; Jiang, H. Dual effects of ketamine: Neurotoxicity versus neuroprotection in anesthesia for the developing brain. J. Neurosurg. Anesthesiol. 2014, 26, 155–160. [Google Scholar] [CrossRef]
- Wang, L.; Traystman, R.J.; Murphy, S.J. Inhalational anesthetics as preconditioning agents in ischemic brain. Curr. Opin. Pharmacol. 2008, 8, 104–110. [Google Scholar] [CrossRef]
- Dezfulian, C.; Garrett, M.; Gonzalez, N.R. Clinical application of preconditioning and postconditioning to achieve neuroprotection. Transl. Stroke Res. 2013, 4, 19–24. [Google Scholar] [CrossRef]
- Narayanan, S.V.; Dave, K.R.; Perez-Pinzon, M.A. Ischemic preconditioning and clinical scenarios. Curr. Opin. Neurol. 2013, 26, 1–7. [Google Scholar] [CrossRef]
- Gidday, J.M. Cerebral preconditioning and ischaemic tolerance. Nat. Rev. Neurosci. 2006, 7, 437–448. [Google Scholar] [CrossRef]
- Cason, B.; Gamperl, A.; Slocum, R.; Kickey, R. Anesthetic-induced preconditioning: Previous administration of isoflurane decreases myocardial infarct size in rabbits. Anesthesiology 1997, 87, 1182–1190. [Google Scholar] [CrossRef]
- Zaugg, M.; Lucchinetti, E.; Uecker, M.; Pasch, T.; Schaub, M.C. Anaesthetics and cardiac preconditioning. Part I. Signalling and cytoprotective mechanisms. Br. J. Anaesth. 2003, 94, 551–565. [Google Scholar]
- Cope, D.K.; Impastato, W.K.; Cohen, M.V.; Downey, J.M. Volatile anesthetics protect the ischemic rabbit myocardium from infarction. Anesthesiology 1997, 86, 699–709. [Google Scholar] [CrossRef]
- Altay, O.; Suzuki, H.; Hasegawa, J.; Caner, B.; Krafft, P.R.; Fujii, M.; Tang, J.; Zhang, J.H. Isoflurane attenuates blood-brain barrier disruption in ipsilateral hemisphere after subarachnoid hemorrhage in mice. Stroke 2012, 43, 2513–2516. [Google Scholar] [CrossRef]
- Sakai, H.; Sheng, H.; Yates, R.B.; Ishida, K.; Pearlstein, R.D.; Warner, D.S. Isoflurane provides long-term protection against focal cerebral ischemia in the rat. Anesthesiology 2007, 106, 92–99. [Google Scholar] [CrossRef]
- Bedirli, N.; Bagriacik, E.U.; Emmez, H.; Yilmaz, G.; Unal, Y.; Ozkose, Z. Sevoflurane and isoflurane preconditioning provides neuroprotection by inhibition of apoptosis-related mRNA expression in a rat model of focal cerebral ischemia. J. Neurosurg. Anesthesiol. 2012, 24, 336–344. [Google Scholar] [CrossRef]
- Warner, D.S. Isoflurane neuroprotection: A passing fantasy, again? Anesthesiology 2000, 92, 1226–1228. [Google Scholar] [CrossRef]
- Kitano, H.; Kirsch, J.R.; Hurn, P.D.; Murphy, S.J. Inhalational anesthetics as neuroprotectants or chemical preconditioning agents in ischemic brain. J. Cereb. Blood Flow Metab. 2007, 27, 1108–1128. [Google Scholar] [CrossRef]
- Li, L.; Zuo, Z. Isoflurane preconditioning improves short-term and long-term neurological outcome after focal brain ischemia in adult rats. Neuroscience 2009, 164, 497–506. [Google Scholar] [CrossRef]
- Pape, M.; Engelhard, K.; Eberspächer, E.; Hollweck, R.; Kellermann, K.; Zintner, S.; Hutzler, P.; Werner, C. The long-term effect of sevoflurane on neuronal cell damage and expression of apoptotic factors after cerebral ischemia and reperfusion in rats. Anesth. Analg. 2006, 103, 173–179. [Google Scholar] [CrossRef]
- Kurth, C.D.; Priestley, M.A.; Watzmann, H.M.; McCann, J.C.; Golden, J. Desflurane confers neurologic protection for deep hypothermic circulatory arrest in newborn pigs. Anesthesiology 2001, 95, 959–964. [Google Scholar] [CrossRef]
- Loepke, A.W.; Priestley, M.A.; Schultz, S.E.; McCann, J.C.; Golden, J.; Kurth, C.D. Desflurane improves neurologic outcome after low-flow cardiopulmonary bypass in newborn pigs. Anesthesiology 2002, 97, 1521–1527. [Google Scholar] [CrossRef]
- Li, H.; Yin, J.; Li, L.; Deng, J.; Feng, C.; Zuo, Z. Isoflurane postconditioning reduces ischemia-induced nuclear factor-κB activation and interleukin 1β production to provide neuroprotection in rats and mice. Neurobiol. Dis. 2013, 54, 216–224. [Google Scholar] [CrossRef]
- Lee, H.T.; Kim, M.; Jan, M.; Emala, C.W. Anti-inflammatory and antinecrotic effects of the volatile anesthetic sevoflurane in kidney proximal tubule cells. Am. J. Physiol. Renal Physiol. 2006, 291, F67–F78. [Google Scholar] [CrossRef]
- Dobbing, J.; Sands, J. Quantitative growth and development of human brain. Arch. Dis. Child. 1973, 48, 757–767. [Google Scholar] [CrossRef]
- Zheng, S.; Zuo, Z. Isoflurane preconditioning decreases glutamate receptor overaction-induced Purkinje neuronal injury in rat cerebellar slices. Brain Res. 2005, 1054, 143–151. [Google Scholar] [CrossRef]
- Patel, P.M.; Drummond, J.C.; Cole, D.J.; Kelly, P.J.; Watson, M. Isoflurane and pentobarbital reduce the frequency of transient ischemic depolarizations during focal ischemia in rats. Anesth. Analg. 1998, 86, 773–780. [Google Scholar]
- Zhou, Y.; Lekic, T.; Fathali, N.; Ostrowski, R.P.; Martin, R.D.; Tang, J.; Zhang, J.H. Isoflurane posttreatment reduces neonatal hypoxic-ischemic brain injury in rats by the sphingosine-1-phosphate/phosphatidylinositol-3-kinase/Akt pathway. Stroke 2010, 41, 1521–1527. [Google Scholar] [CrossRef]
- Khatibi, N.H.; Ma, Q.; Rolland, W.; Ostrowski, R.; Fathali, N.; Martin, R.; Applegate, R.; Stier, G.; Tang, J.; Zhang, J.H. Isoflurane posttreatment reduced brain injury after an intracerebral hemorrhagic stroke in mice. Anesth. Analg. 2011, 113, 343–348. [Google Scholar] [CrossRef]
- Chen, H.; Burris, M.; Fajilan, A.; Spagnoli, F.; Tang, J.; Zhang, J.H. Prolonged exposure to isoflurane ameliorates infarction severity in the rat pup model of neonatal hypoxia-ischemia. Transl. Stroke Res. 2011, 2, 382–390. [Google Scholar] [CrossRef]
- Hoffman, W.E.; Charbel, F.T.; Edelman, G.; Ausman, J.I. Thiopental and desflurane treatment for brain protection. Neurosurgery 1998, 43, 1050–1053. [Google Scholar] [CrossRef]
- Michenfelder, J.D.; Sundt, T.M.; Fode, N.; Sharbrough, F.W. Isoflurane when compared to enflurane and halothane decreases the frequency of cerebral ischemia during carotid endarterectomy. Anesthesiology 1987, 67, 336–340. [Google Scholar] [CrossRef]
- Lavine, S.D.; Masri, L.S.; Levy, M.L.; Giannotta, S.L. Temporary occlusion of the middle cerebral artery in intracranial aneurysm surgery: Time limitation and advantage of brain protection. J. Neurosurg. 1997, 87, 817–824. [Google Scholar] [CrossRef]
- Singh, P.; Chauhan, S.; Jain, G.; Talwar, S.; Makhija, N.; Kiran, U. Comparison of cardioprotective effects of volatile anesthetics in children undergoing ventricular septal defect closure. World J. Pediatr. Congenit. Heart Surg. 2013, 4, 24–29. [Google Scholar] [CrossRef]
- Swyers, T.; Redford, D.; Larson, D. Volatile anesthetic-induced preconditioning. Perfusion 2014, 29, 10–15. [Google Scholar] [CrossRef]
- Ikonomidou, C.; Bittigau, P.; Ishimaru, M.J.; Wozniak, D.F.; Koch, C.; Genz, K.; Price, M.T.; Stefovska, V.; Hörster, F.; Tenkova, T.; et al. Ethanol-induced apoptotic neurodegeneration and fetal alcohol syndrome. Science 2000, 287, 1056–1060. [Google Scholar] [CrossRef]
- Satomoto, M.; Satoh, Y.; Terui, K.; Miyao, H.; Takishima, K.; Ito, M.; Imaki, J. Neonatal exposure to sevoflurane induces abnormal social behaviors and deficits in mice. Anesthesiology 2009, 110, 628–637. [Google Scholar] [CrossRef]
- Shih, J.; May, L.D.V.; Gonzalez, H.E.; Lee, E.W.; Alvi, R.S.; Sall, J.W.; Rau, V.; Bickler, P.E.; Lalchandani, G.R.; Yusupova, M.; et al. Delayed environmental enrichment reverses sevoflurane-induced memory impairment in rats. Anesthesiology 2012, 116, 586–602. [Google Scholar] [CrossRef]
- Istaphanous, G.K.; Howard, J.; Nan, X.; Hughes, E.A.; McCann, J.C.; McAuliffe, J.J.; Danzer, S.C.; Loepke, A.W. Comparison of the neuroapoptotic properties of equipotent anesthetic concentrations of desflurane, isoflurane, or sevoflurane in neonatal mice. Anesthesiology 2011, 114, 578–587. [Google Scholar] [CrossRef]
- Kodama, M.; Satoh, Y.; Otsubo, Y.; Araki, Y.; Yonamine, R.; Masui, K.; Kazama, T. Neonatal desflurane exposure induces more robust neuroapoptosis than do isoflurane and sevoflurane and impairs working memory. Anesthesiology 2011, 115, 979–991. [Google Scholar] [CrossRef]
- Liang, G.; Ward, C.; Peng, J.; Zhao, Y.; Huang, B.; Wei, H. Isoflurane causes greater neurodegeneration than an equivalent exposure of sevoflurane in the developing brain of neonatal mice. Anesthesiology 2010, 112, 1325–1334. [Google Scholar] [CrossRef]
- SmartTots Related Research Projects. 2013. Available online: http://www.smarttots.org/research/relatedstudies.html (accessed on 14 November 2013).
- McDonald, J.W.; Silverstein, F.S.; Johnston, M.V. Neurotoxicity of N-methyl-d-aspartate is markedly enhanced in developing rat nervous sytem. Brain Res. 1988, 459, 200–203. [Google Scholar] [CrossRef]
- Ikonomidou, C.; Mosinger, J.L.; Salles, K.S.; Labruyere, J.; Olney, J.W. Sensitivity of the developing rat brain to hypobaric/ischemic damage parallels sensitivity to N-methyl-aspartate neurotoxicity. J. Neurosci. 1989, 9, 2809–2818. [Google Scholar]
- Gao, X.B.; Chen, G.; van den Pol, A.N. GABA-dependent firing of glutamate-evoked action potentials at AMPA/kainate receptors in developing hypothalamic neurons. J. Neurophysiol. 1998, 79, 716–726. [Google Scholar]
- Bano, D.; Nicotera, P. Ca2+ signals and neuronal death in brain ischemia. Stroke 2007, 38, 674–676. [Google Scholar] [CrossRef]
- Lipton, S.A.; Nicotera, P. Calcium, free radicals and excitotoxins in neuronal apoptosis. Cell Calcium 1998, 23, 165–171. [Google Scholar] [CrossRef]
- Famy, C.; Streissguth, A.P.; Unis, A.S. Mental illness in adults with fetal alcohol syndrome or fetal alcohol effects. Am. J. Psychiatry 1998, 155, 552–554. [Google Scholar]
- Murphy, K.R.; Barkley, R.A.; Bush, T. Executive functioning and olfactory identification in young adults with attention deficit hyperactivity disorder. Neuropsychology 2001, 15, 211–220. [Google Scholar]
- Wilder, R.T.; Flick, R.P.; Sprung, J.; Katusic, S.; Barbaresi, W.J.; Mickelson, C.; Gleich, S.J.; Schroeder, D.R.; Weaver, A.L.; Warner, D.O. Early exposure to anesthesia and learning disabilities in a population-based birth cohort. Anesthesiology 2009, 110, 796–804. [Google Scholar] [CrossRef]
- DiMaggio, C.; Sun, L.S.; Kakavouli, A.; Byrne, M.W.; Li, G. A retrospective cohort study of the association of anesthesia and hernia repair surgery with behavioral and developmental disorders in young children. J. Neurosurg. Anesthesiol. 2009, 21, 286–291. [Google Scholar] [CrossRef]
- DiMaggio, C.; Sun, L.S.; Li, G. Early childhood exposure to anesthesia and risk of developmental and behavioral disorders in a sibling birth cohort. Anesth. Analg. 2011, 113, 1143–1151. [Google Scholar] [CrossRef]
- Flick, R.P.; Katusic, S.K.; Colligan, R.C.; Wilder, R.T.; Voigt, R.G.; Olson, M.D.; Sprung, J.; Weaver, A.L.; Schroeder, D.R.; Warner, D.O. Cognitive and behavioral outcomes after early exposure to anesthesia and surgery. Pediatrics 2011, 128, e1053–e1061. [Google Scholar] [CrossRef]
- Kalkman, C.J.; Peelen, L.; Moons, K.G.; Veenhuizen, M.; Bruens, M.; Sinnema, G.; de Jong, T.P. Behavior and development in children and age at the time of first anesthetic exposure. Anesthesiology 2009, 110, 805–812. [Google Scholar] [CrossRef]
- Bartels, M.; Althoff, R.R.; Boomsma, D.I. Anesthesia and cognitive performance in children: No evidence for a causal relationship. Twin Res. Hum. Genet. 2009, 12, 246–253. [Google Scholar] [CrossRef]
- Hansen, T.G.; Pedersen, J.K.; Henneberg, S.W.; Morton, N.S.; Christensen, K. Educational outcome in adolescence following pyloric stenosis repair before 3 months of age: A nationwide cohort study. Pediatr. Anesth. 2013, 23, 883–890. [Google Scholar] [CrossRef]
- Hansen, T.G.; Pedersen, J.K.; Henneberg, S.W.; Pedersen, D.A.; Murray, J.C.; Morton, N.S.; Christensen, K. Academic performance in adolescence after inguinal hernia repair in infancy: A nationwide cohort study. Anesthesiology 2011, 114, 1076–1085. [Google Scholar] [CrossRef]
- Ing, C.; DiMaggio, C.; Whitehouse, A.; Hegarty, M.K.; Brady, J.; von Ungern-Sternberg, B.S.; Davidson, A.; Wood, A.J.J.; Li, G.; Sun, L.S. Long-term differences in language and cognitive function after childhood exposure to anesthesia. Pediatrics 2012, 130, 476–485. [Google Scholar] [CrossRef]
- Jevtovic-Todorovic, V. Pediatric anesthesia neurotoxicity: An overview of the 2011 SmartTots panel. Anesth. Analg. 2011, 113, 965–968. [Google Scholar] [CrossRef]
- Sun, L.S.; Li, G.; DiMaggio, C.J.; Byrne, M.W.; Ing, C.; Miller, T.L.; Bellinger, D.C.; Han, S.; McGowan, F.X. Feasibility and pilot study of the Pediatric Anesthesia NeuroDevelopment Assessment (PANDA) project. J. Neurosurg. Anesthesiol. 2012, 24, 382–388. [Google Scholar] [CrossRef]
- PANDA Study. Available online: http://www.kidspandastudy.org/index.html (accessed on 2 December 2013).
- Children’s Hospital Boston A Multi-site Randomized Controlled Trial Comparing Regional and General Anesthesia for Effects on Neurodevelopmental Outcome and Apnea in Infants (GAS). 2013. Available online: http://clinicaltrials.gov/show/NCT00756600 (accessed on 9 December 2013).
- De Graaff, J.C.; van der Werff, D.B.; Kalkman, C.J. Is anesthesia detrimental to brain development? Ned. Tijdschr. Geneeskd. 2013, 157, A5198. [Google Scholar]
- Davidson, A.J. Neurotoxicity and the need for anesthesia in the newborn: Does the emperor have no clothes? Anesthesiology 2012, 116, 507–509. [Google Scholar] [CrossRef]
- Anand, K.J.; Sippel, W.G.; Aynsley-Green, A. Randomised trial of fentanyl anaesthesia in preterm babies undergoing surgery: Effects on the stress response. Lancet 1987, 1, 243–248. [Google Scholar]
- Anand, K.J.; Sippel, W.G.; Schofield, N.M.; Aynsley-Green, A. Does halothane anaesthesia decrease the metabolic and endocrine stress responses of newborn infants undergoing operation? Br. Med. J. 1988, 296, 668–672. [Google Scholar] [CrossRef]
- Anand, K.J.; Hickey, P.R. Halothane-morphine compared with high-dose sufentanil for anesthesia and postoperative analgesia in neonatal cardiac surgery. N. Engl. J. Med. 1992, 326, 1–9. [Google Scholar] [CrossRef]
- Anand, K.J.; Hansen, D.D.; Hickey, P.R. Hormonal-metabolic stress responses in neonates undergoing cardiac surgery. Anesthesiology 1990, 73, 661–670. [Google Scholar] [CrossRef]
- Anand, K.J.; Aynsley-Green, A. Measuring the severity of surgical stress in newborn infants. J. Pediatr. Surg. 1988, 23, 297–305. [Google Scholar] [CrossRef]
- Rokyta, R.; Yamamotová, A.; Slamberová, R.; Franek, M.; Vaculín, S.; Hrubá, L.; Schutová, B.; Pometlová, M. Prenatal and perinatal factors influencing nociception, addiction and behavior during ontogenetic development. Physiol. Res. 2008, 57, S79–S88. [Google Scholar]
- Anand, K.J.; Coskun, V.; Thrivikraman, K.V.; Nemeroff, C.B.; Plotsky, P.M. Long-term behavioral effects of repetitive pain in neonatal rat pups. Physiol. Behav. 1999, 66, 627–637. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chiao, S.; Zuo, Z. A Double-Edged Sword: Volatile Anesthetic Effects on the Neonatal Brain. Brain Sci. 2014, 4, 273-294. https://doi.org/10.3390/brainsci4020273
Chiao S, Zuo Z. A Double-Edged Sword: Volatile Anesthetic Effects on the Neonatal Brain. Brain Sciences. 2014; 4(2):273-294. https://doi.org/10.3390/brainsci4020273
Chicago/Turabian StyleChiao, Sunny, and Zhiyi Zuo. 2014. "A Double-Edged Sword: Volatile Anesthetic Effects on the Neonatal Brain" Brain Sciences 4, no. 2: 273-294. https://doi.org/10.3390/brainsci4020273