Chemical Surface, Thermal and Electrical Characterization of Nafion Membranes Doped with IL-Cations
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
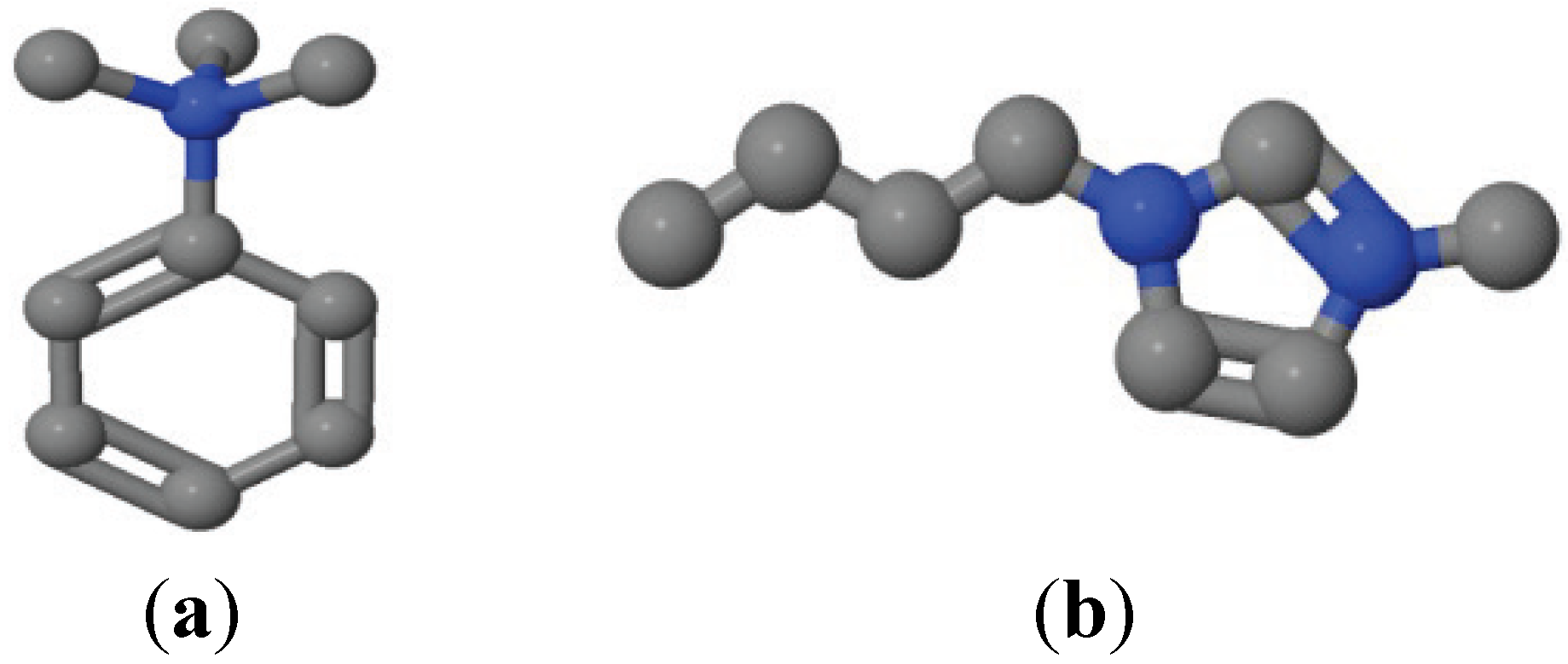
2.2. XPS Measurements
2.3. Contact Angle Measurements
2.4. Thermal Analysis
2.5. Impedance Spectroscopy Measurements
2.6. Membrane Potential Measurements
3. Results and Discussion
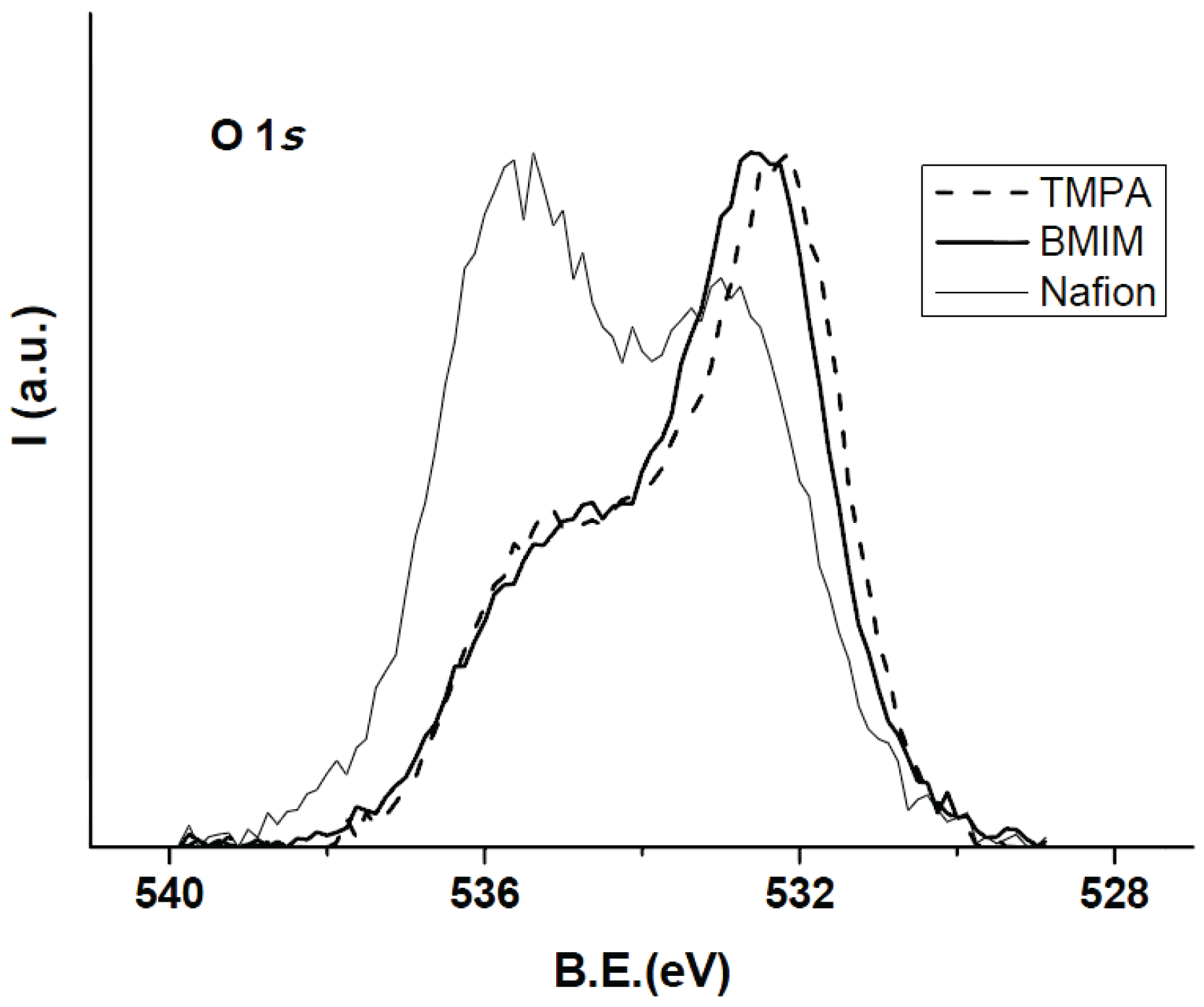
3.1. Membrane Surface Characterization
| Sample | C% | O% | F% | S% | N% |
|---|---|---|---|---|---|
| Nafion (theoretical) | 30.8 | 7.7 | 60.0 | 1.5 | - |
| Nafion112 | 39.0 | 6.6 | 53.4 | 1.0 | 0.2 |
| Nafion112/TMPA+ | 45.6 | 7.0 | 44.2 | 1.0 | 1.3 |
| Nafion112/BMIM+ | 47.9 | 4.8 | 42.3 | 0.9 | 3.6 |
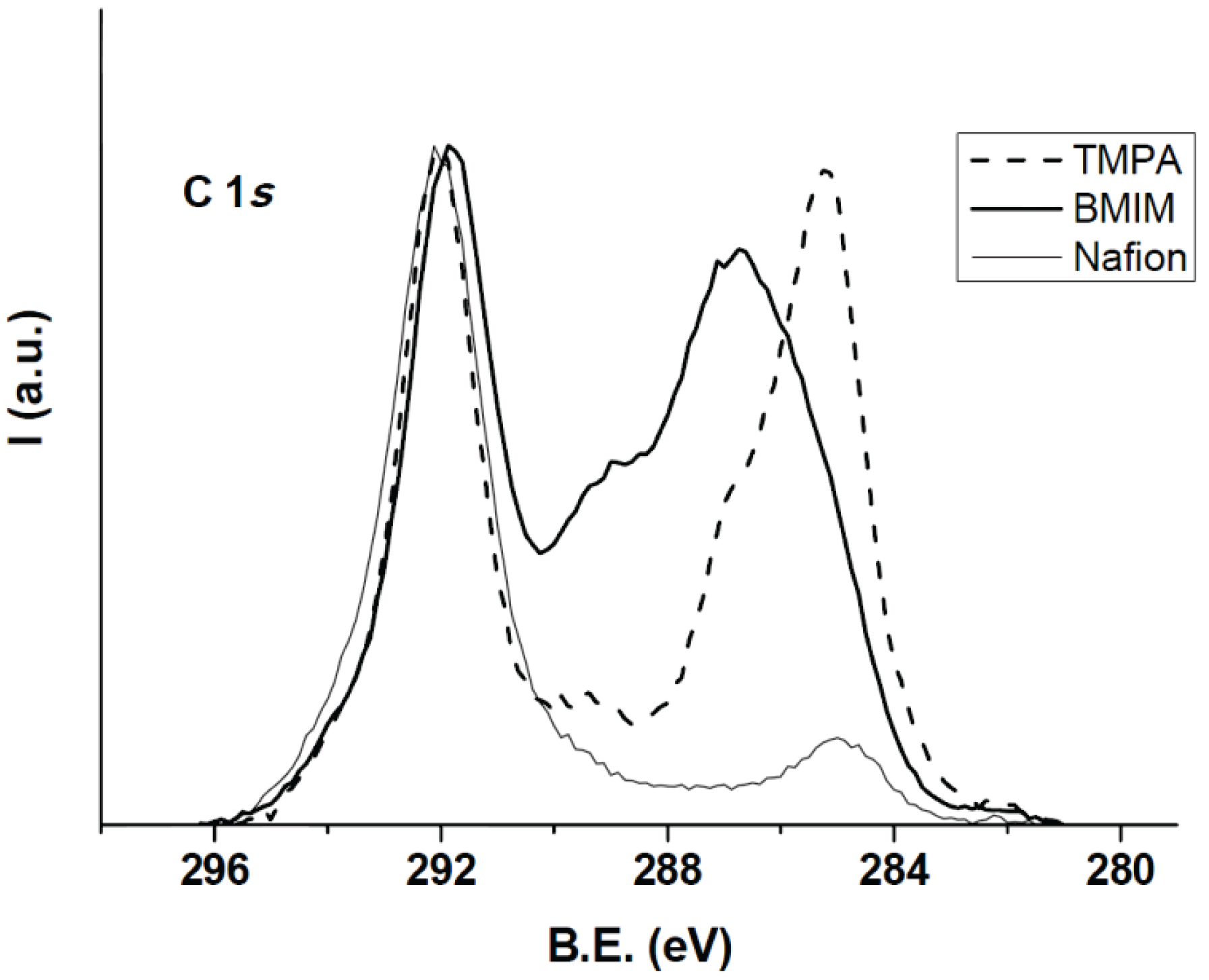
| Sample | C1 | C2 | C3 | C4 | C5 |
|---|---|---|---|---|---|
| Nafion112 | 8.05 | 3.63 | 8.37 | 69.70 | 10.20 |
| Nafion112/TMPA+ | 36.92 | 15.51 | 8.29 | 36.30 | 2.97 |
| Nafion112/BMIM+ | 14.01 | 28.05 | 22.96 | 31.56 | 3.42 |
| Membrane | Average contact angle (°) |
|---|---|
| Nafion112 | 91 ± 2 |
| Nafion112/TMPA+ | 74 ± 8 |
| Nafion112/BMIM+ | 72 ± 6 |
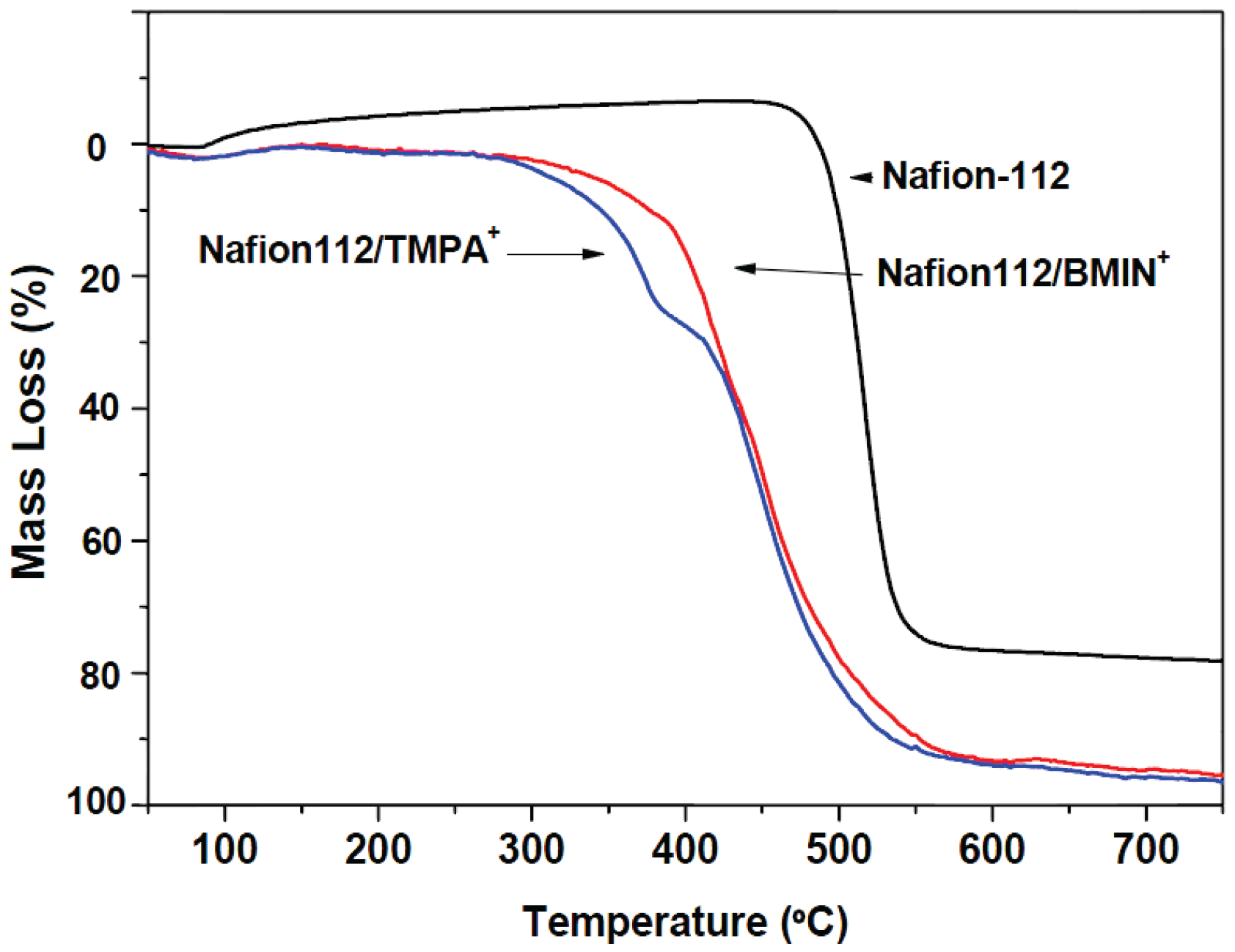
3.2. Thermal Characterization of Original and IL-Cations Doped Membranes
3.3. Electrical Characterization of Nafion112/IL-Cations Doped Membranes
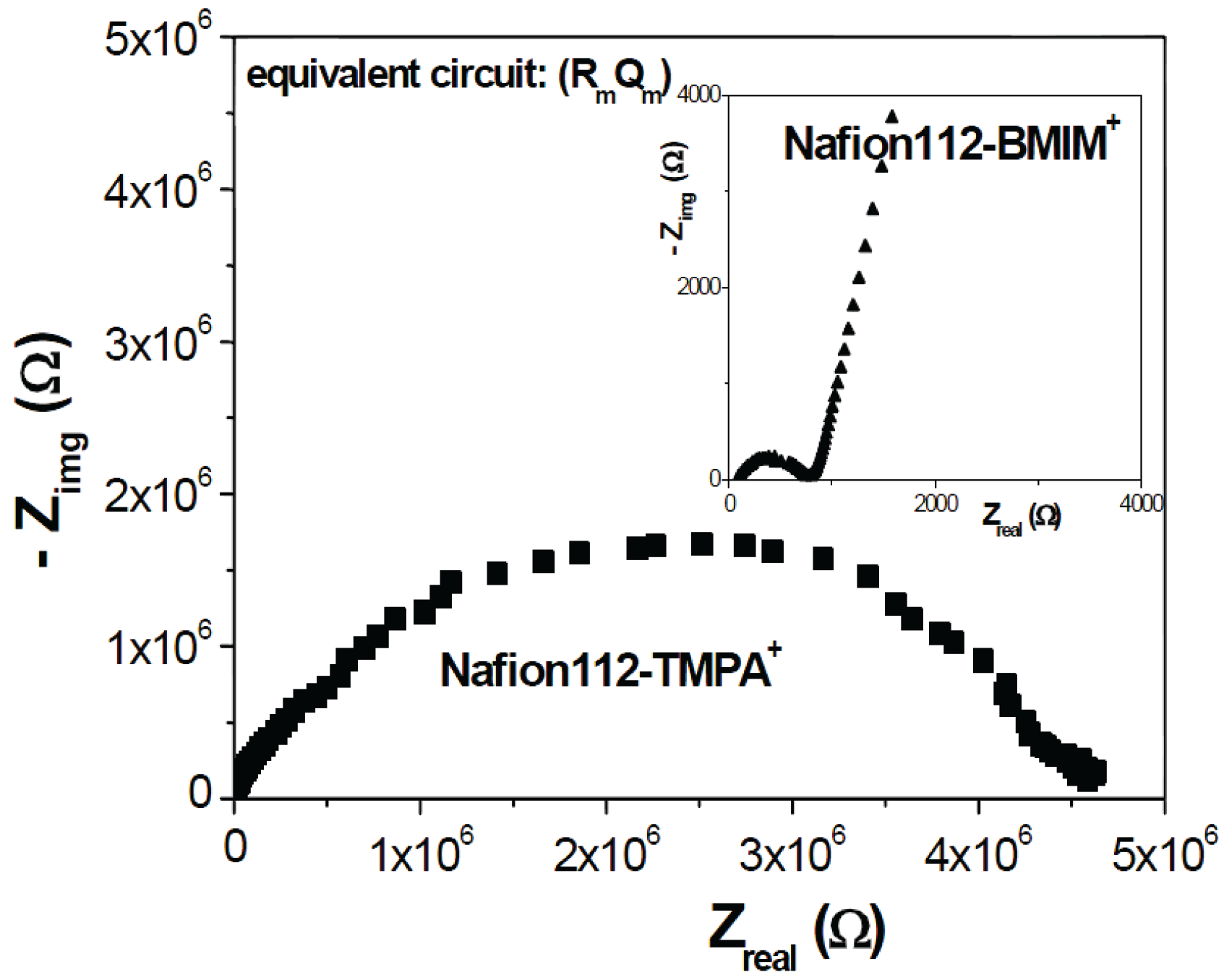
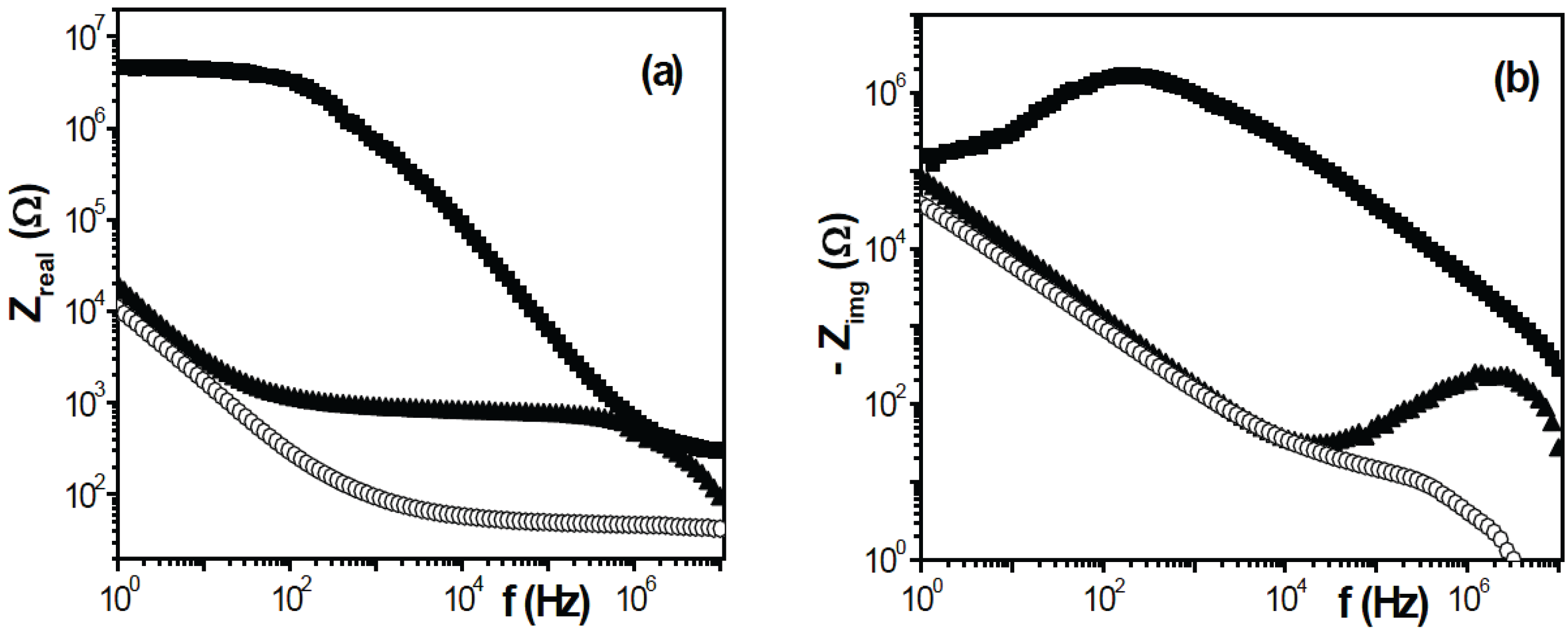
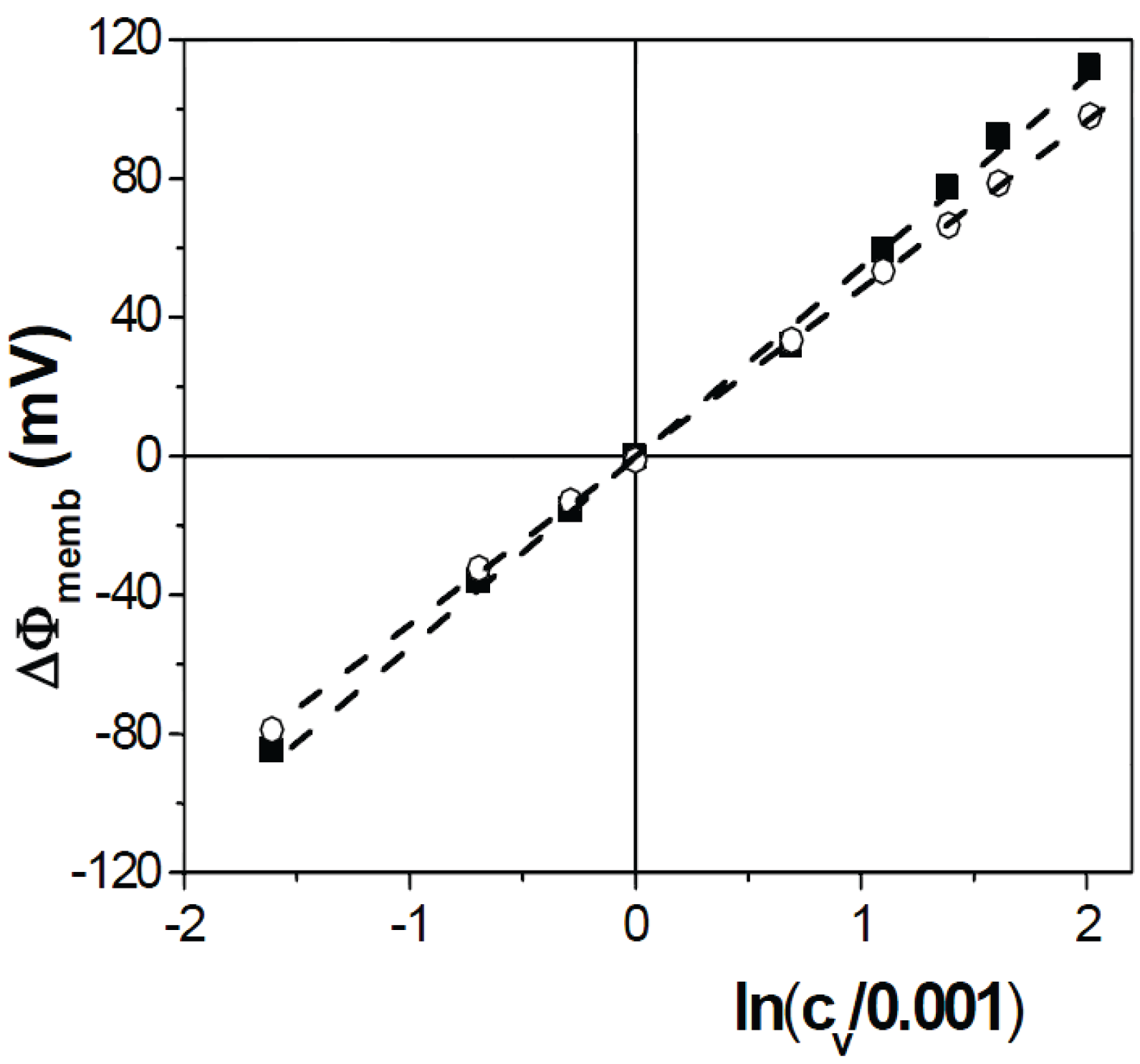
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Einsenberg, A.; Yeager, H.L. Perfluorinated Ionomer Membranes, ACS Symposium Series; American Chemical Society (ACS): Washington, DC, USA, 1982; Volume 180. [Google Scholar]
- De Yuso, M.V.M.; Neves, L.A.; Coelhoso, I.M.; Crespo, J.G.; Benavente, J.; Rodríguez-Castellón, E. A Study of Chemical Modifications of a Nafion Membrane by Incorporation of Different Room Temperature Ionic Liquids. Fuel Cells 2012, 12, 606–612. [Google Scholar] [CrossRef]
- Lojoiuo, C.; Martinez, M.; Hanna, M.; Molmeret, Y.; Cointeaux, L.; Leprêtre, J.-C.; El Kissi, N.; Guidet, J.; Judeinstein, P.; Sanchez, J.-Y. PILs-based Nafion Membranes: A Route to High-temperature PEFMCs Dedicated to Electric and Hybrid Vehicles. Polym. Adv. Technol. 2008, 19, 1406–1414. [Google Scholar] [CrossRef]
- Doyle, M.; Choi, S.; Proulx, G. High-Temperature Proton Conducting Membranes Based on Perfluorinated Ionomer Membrane-Ionic Liquid Composites. J. Electrochem. Soc. 2000, 147, 34–37. [Google Scholar]
- Schmidt, C.; Glück, T.; Schmidt-Naake, G. Modification of Nafion Membranes by Impregnation with Ionic Liquids. Chem. Eng. Technol. 2008, 31, 13–22. [Google Scholar] [CrossRef]
- Bennet, M.D.; Leo, D.J. Ionic liquids as Stable Solvents for Ionic Polymer Transducers. Sens. Actuators A 2004, 115, 79–90. [Google Scholar] [CrossRef]
- Welton, T. Room-Temperature Ionic Liquids. Solvents for Synthesis and Catalysis. Chem. Rev. 1999, 99, 2971–2984. [Google Scholar] [CrossRef]
- Armand, M.; Endres, F.; MacFarlane, D.R.; Ohno, H.; Scrosati, B. Ionic-liquid Materials for the Electrochemical Challenges of the Future. Nat. Mater. 2009, 621–629. [Google Scholar]
- Bonhôte, P.; Dias, A.P.; Papageorgiou, N.; Kalyanasundaram, K.; Grätzel, M. Hydrophobic, Highly Conductive Ambient-Temperature Molten Salts. Inorg. Chem. 1996, 35, 1168–1178. [Google Scholar] [CrossRef]
- Neves, L.; Benavente, J.; Coelhoso, I.M.; Crespo, J.G. Design and Characterisation of Nafion Membranes with Incorporated Ionic Liquids Cations. J. Membr. Sci. 2010, 347, 42–52. [Google Scholar] [CrossRef]
- Neves, L.A.; Crespo, J.G.; Coelhoso, I.M. Gas Permeation Studies in Supported Ionic Liquid Membranes. J. Membr. Sci. 2010, 357, 160–170. [Google Scholar] [CrossRef]
- Martinez, M.; Molmeret, Y.; Cointeaux, L.; Iojoiu, C.; Leprêtre, J.C.; El Kissi, N.; Judeinstein, P.; Sanchez, J.Y. Proton-conducting Ionic Liquid-based Proton Exchange Membrane Fuel Cell membranes: The Key Role of Ionomer-ionic Liquid. J. Power Sources 2010, 195, 5829–5839. [Google Scholar] [CrossRef]
- Ariza, M.J.; Benavente, J.; Rodríguez-Castellón, E. The Capability of X-ray Photoelectron Spectroscopy in the Characterization of Membranes: Correlation between Surface Chemical and Transport Properties in Polymeric Membranes. In Handbook of Membranes: Properties, Performance and Applications; Nova Science Publishers, Inc.: New York, NY, USA, 2009; pp. 257–290. [Google Scholar]
- Fortunato, R.; Afonso, C.A.M.; Benavente, J.; Rodriguez-Castellón, E.; Crespo, J.G. Stability of Supported Ionic Liquid Membranes as Studied by X-ray Photoelectron Spectroscopy. J. Membr. Sci. 2005, 256, 216–223. [Google Scholar]
- Macdonalds, J.R. Impedance Spectroscopy; Wiley: New York, NY, USA, 1987. [Google Scholar]
- Asaka, K. Dielectric Properties of Cellulose Acetate Reverse Osmosis Membranes in Aqueous Solution. J. Membr. Sci. 1990, 50, 71–84. [Google Scholar] [CrossRef]
- Mijovic, J.; Bellucci, F. Impedance Spectroscopy of Reactive Polymers. Trends Polym. Sci. 1996, 4, 74–82. [Google Scholar]
- Benavente, J. Use of impedance spectroscopy for characterization of membranes and the effect of different modifications. In Membrane Modification: Technology and Applications; Hilal, N., Khayet, M., Wright, C.J., Eds.; CRC Press: Boca Raton, FL, USA, 2012; pp. 21–40. [Google Scholar]
- Shäfer, T.; di Paolo, R.E.; Franco, R.; Crespo, J.G. Elucidating Interaction of Ionic Liquids with Polymer Films Using Confocal Raman Spectroscopy. Chem. Commun. 2005, 2594–2596. [Google Scholar]
- Ariza, M.J.; Benavente, J.; Rodríguez-Castellón, E.; Palacio, L. Effect of Hydration of Polyamide Membranes on the Surface Electrokinetic Parameters: Surface Characterization by X-ray Photoelectronic Spectroscopy and Atomic Force Microscopy. J. Colloid Interface Sci. 2002, 247, 149–158. [Google Scholar] [CrossRef]
- Moulder, J.F.; Stickle, W.F.; Sobol, P.E.; Bomben, K.D. Handbook of X-ray Photoelectron Spectroscopy; Chastain, J., Ed.; Perkin-Elmer Corporation: Minneapolis, MN, USA, 1992. [Google Scholar]
- Ramos, J.D.; Milano, C.; Romero, V.; Escalera, S.; Alba, M.C.; Vázquez, M.I.; Benavente, J. Water Effect on Physical-chemical and Elastic Parameters for a Dense Cellulose Regenerated Membrane. Transport of Different Aqueous Electrolyte Solutions. J. Membr. Sci. 2010, 352, 153–159. [Google Scholar] [CrossRef]
- Pelaez, L.; Vázquez, M.I.; Benavente, J. Interfacial and Fouling Effects on Diffusional Permeability Across a Composite Ceramic Membrane. Ceramics Int. 2010, 36, 797–801. [Google Scholar] [CrossRef]
- Mallevialle, J.; Odendaal, P.E.; Wiesner, M.R. Water Treatment Membrane Processes; McGraw-Hill: Chicago, IL, USA, 1996. [Google Scholar]
- Unemura, K.; Wang, T.; Hara, M.; Kuroda, R.; Uchida, O.; Nagai, M. Nono-characterization of a Nafion Thin Film in Air and in Water by Atomic Force Micorscopy. IOP Publ. J. Phys. 2007, 61, 1202–1206. [Google Scholar]
- De Yuso, M.V.M.; Cuberes, M.T.; Romero, V.; Neves, L.; Coelhoso, M.I.; Crespo, J.G.; Rodríguez-Castellón, E.; Benavente, J. Modification of a Nafion Membrane by n-dodecyltrimethylammonium Cation Inclusion for Potential Application in DMFC. Int. J. Hydrog. Energy 2014, 39, 4023–4029. [Google Scholar] [CrossRef]
- Romero, V.; Vega, V.; García, J.; Zierold, R.; Nielsch, K.; Prida, V.M.; Hernando, B.; Benavente, J. Changes in morphology and ionic transport induced by ALD SiO2 coating of nanoporous alumina membranes. ACS Appl. Mater. Interfaces 2013, 5, 3556–3564. [Google Scholar] [CrossRef]
- Lakshminarayanaiah, N. Transport Phenomena in Membranes; Academic Press: New York, NY, USA, 1969. [Google Scholar]
- Benavente, J. Electrochemical impedance spectroscopy as a tool for characterization of membranes in contact with electrolyte solutions in Surface Electrical Phenomena. In Membranes and Microchannels; Szymczyk, A., Ed.; Transworld Reserach Network: Rennes, France, 2008. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Del Valle Martínez de Yuso, M.; Arango-Díaz, A.; Bijani, S.; Romero, V.; Benavente, J.; Rodríguez-Castellón, E. Chemical Surface, Thermal and Electrical Characterization of Nafion Membranes Doped with IL-Cations. Appl. Sci. 2014, 4, 195-206. https://doi.org/10.3390/app4020195
Del Valle Martínez de Yuso M, Arango-Díaz A, Bijani S, Romero V, Benavente J, Rodríguez-Castellón E. Chemical Surface, Thermal and Electrical Characterization of Nafion Membranes Doped with IL-Cations. Applied Sciences. 2014; 4(2):195-206. https://doi.org/10.3390/app4020195
Chicago/Turabian StyleDel Valle Martínez de Yuso, María, Ana Arango-Díaz, Shanti Bijani, Virgina Romero, Juana Benavente, and Enrique Rodríguez-Castellón. 2014. "Chemical Surface, Thermal and Electrical Characterization of Nafion Membranes Doped with IL-Cations" Applied Sciences 4, no. 2: 195-206. https://doi.org/10.3390/app4020195
APA StyleDel Valle Martínez de Yuso, M., Arango-Díaz, A., Bijani, S., Romero, V., Benavente, J., & Rodríguez-Castellón, E. (2014). Chemical Surface, Thermal and Electrical Characterization of Nafion Membranes Doped with IL-Cations. Applied Sciences, 4(2), 195-206. https://doi.org/10.3390/app4020195






