Dietary Lecithin Decreases Skeletal Muscle COL1A1 and COL3A1 Gene Expression in Finisher Gilts
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Experimental Design
2.2. RNA Extraction and cDNA Synthesis
2.3. Primer Design
2.4. Quantitative Real-Time PCR
2.5. Statistical Analysis
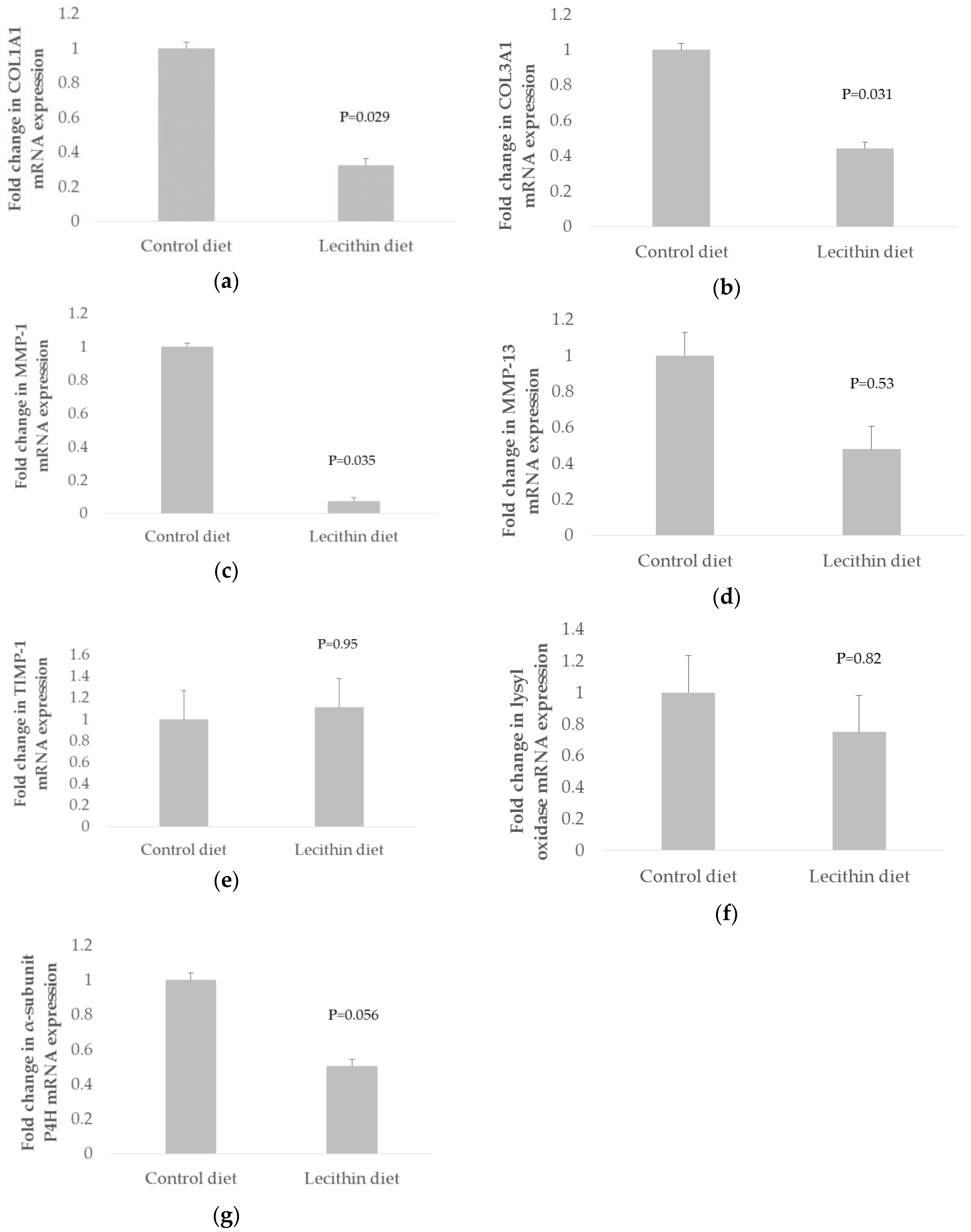
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Van Ee, J.H. Soy constituents: Modes of action in low-density lipoprotein management. Nutr. Rev. 2009, 67, 222–234. [Google Scholar] [CrossRef] [PubMed]
- Poniachik, J.; Baraona, E.; Zhao, J.B.; Lieber, C.S. Dilinoleoylphosphatidylcholine decreases hepatic stellate cell activation. J. Lab. Clin. Med. 1999, 133, 342–348. [Google Scholar] [CrossRef]
- Cao, Q.; Mak, K.M.; Lieber, C.S. Dilinoleoylphosphatidylcholine prevents transforming. Growth Factor Beta-1-mediated collagen accumulation in cultured rat hepatic stellate cells. J. Lab. Clin. Med. 2002, 139, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Cao, Q.; Mak, K.M.; Lieber, C.S. DLPC decreases TGF-beta 1-induced collagen mRNA by inhibiting p38 MAPK in hepatic stellate cells. Am. J. Physiol.-Gastrointest. Liver Physiol. 2002, 283, 1051–1061. [Google Scholar] [CrossRef] [PubMed]
- Lieber, C.S.; Robins, S.J.; Li, J.J.; Decarli, L.M.; Mak, K.M.; Fasulo, J.M.; Leo, M.A. Phosphatidylcholine protects against fibrosis and cirrhosis in the baboon. Gastroenterology 1994, 106, 152–159. [Google Scholar] [PubMed]
- Li, J.J.; Kim, C.I.; Leo, M.A.; Mak, K.M.; Rojkind, M.; Lieber, C.S. Polyunsaturated lecithin prevents acetyldehyde-mediated hepatic collagen accumulation by stimulating collagenase activity in cultured lipocytes. Hepatology 1992, 15, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Nimni, M.E. Collagen Biochemistry, 1st ed.; CRC Press Inc.: Boca Raton, FL, USA, 1988. [Google Scholar]
- Kragstrup, T.W.; Kjaer, M.; Mackey, A.L. Structural, biochemical, cellular, and functional changes in skeletal muscle extracellular matrix with aging. Scand. J. Med. Sci. Sports 2011, 21, 749–757. [Google Scholar] [CrossRef] [PubMed]
- The National Centre for Biotechnology Information Database. Available online: http://www.ncbi.nlm.nih.gov/sites/entrez (accessed on 25 May 2010).
- The Custom Primers—OligoPerfectTM Designer Software. Available online: http://www.invitrogen.com/content.cfm?pageid=9716 (accessed on 25 May 2010).
- NetPrimer. Available online: http://www.premierbiosoft.com/netprimer/netprlaunch/netprlaunch.html (accessed on 25 May 2010).
- Ahtikoski, A.M.; Koskinen, S.O.A.; Virtanen, P.; Kovanen, V.; Takala, T.E.S. Regulation of synthesis of fibrillar collagens in rat skeletal muscle during immobilization in shortened and lengthened positions. Acta Physiol. Scand. 2001, 172, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Han, X.Y.; Wang, W.; Komulainen, J.; Koskinen, S.O.A.; Kovanen, V.; Vihko, V.; Trackman, P.C.; Takala, T.E.S. Increased mRNAs for procollagens and key regulating enzymes in rat skeletal muscle following downhill running. Eur. J. Physiol. 1999, 437, 857–864. [Google Scholar] [CrossRef] [PubMed]
- Han, X.Y.; Wang, W.; Myllyla, R.; Virtanen, P.; Karpakka, J.; Takala, T.E.S. mRNA levels for alpha-subunit of prolyl 4-hydroxylase and fibrillar collagens in immobilized rat skeletal muscle. J. Appl. Physiol. 1999, 87, 90–96. [Google Scholar] [PubMed]
- Cho, E.S.; Jung, W.Y.; Kwon, E.J.; Park, D.H.; Chung, K.H.; Cho, K.K.; Kim, C.W. Cloning and expression analysis of the alpha-subunit of porcine prolyl 4-hydroxylase. Asian-Australas. J. Anim. Sci. 2007, 20, 1655–1661. [Google Scholar] [CrossRef]
- D’Souza, D.N.; Blake, B.L.; Williams, I.H.; Mullan, B.P.; Pethick, D.W.; Dunshea, F.R. Dietary lecithin supplementation can improve the quality of the M. longissimus thoracis. Animals 2015, 5, 1180–1191. [Google Scholar] [CrossRef] [PubMed]
- Akit, H.; Collins, C.L.; Fahri, F.T.; Hung, A.T.; D’Souza, D.N.; Leury, B.J.; Dunshea, F.R. Dietary lecithin improves dressing percentage and decreases chewiness in the longissimus muscle in finisher gilts. Meat Sci. 2014, 96, 1147–1151. [Google Scholar] [CrossRef] [PubMed]
- Jeremiah, L.E.; Dugan, M.E.R.; Aalhus, J.L.; Gibson, L.L. Assessment of the chemical and cooking properties of the major beef muscles and muscle groups. Meat Sci. 2003, 65, 985–992. [Google Scholar] [CrossRef]
- Riley, D.G.; Johnson, D.D.; Chase, C.C.; West, R.L.; Coleman, S.W.; Olson, T.A.; Hammond, A.C. Factors influencing tenderness in steaks from Brahman cattle. Meat Sci. 2005, 70, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Jeremiah, L.E.; Dugan, M.E.R.; Aalhus, J.L.; Gibson, L.L. Assessment of the relationship between chemical components and palatability of major beef muscles and muscle groups. Meat Sci. 2003, 65, 1013–1019. [Google Scholar] [CrossRef]
- Fang, S.H.; Nishimura, T.; Takahashi, K. Relationship between development of intramuscular connective tissue and toughness of pork during growth of pigs. J. Anim. Sci. 1999, 77, 120–130. [Google Scholar] [PubMed]
- Bao, X.; Zeng, Y.; Wei, S.; Wang, G.; Liu, C.; Sun, Y.; Chen, Q.; Li, H. Developmental changes of COL3A1 mRNA expression in muscle and their association with intramuscular collagen in pigs. J. Genet. Genomics 2007, 34, 223–228. [Google Scholar] [CrossRef]
- Burson, D.E.; Hunt, M.C.; Unruh, J.A.; Dikeman, M.E. Proportion of Type I and Type II collagen in Logissimus collagen from bulls and steers. J. Anim. Sci. 1986, 63, 453–456. [Google Scholar] [PubMed]
- Miller, E.J.; Harris, E.D.; Chung, E.; Butler, W.T.; Robertson, P.B. Cleavage of Type II and Type III collagens with mammalian collagenase site of cleavage and primary structure at NH2 terminal portion of smaller fragment released from both collagens. Biochemistry 1976, 15, 787–792. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.J.; Finch, J.E.; Chung, E.; Butler, W.T.; Robertson, P.B. Specific cleavage of native Type III collagen molecule with trypsin similarity of cleavage products to collagenase-produced fragments and primary structure at cleavage site. Arch. Biochem. Biophys. 1976, 173, 631–637. [Google Scholar] [CrossRef]
- Welgus, H.G.; Jeffrey, J.J.; Eisen, A.Z. The collagen substrate specificity of human-skin fibroblast collagenase. J. Biol. Chem. 1981, 256, 9511–9515. [Google Scholar] [PubMed]
| Gene 1 | Species | Primers | Primer Sequence (from 5′–3′) | Accession Number | Amplicon Size (bp) |
|---|---|---|---|---|---|
| COL1A1 | Mouse | Forward | GTCTGGTTTGGAGAGAGCAT | BC050014.1 | 189 |
| Reverse | CTTCTTGAGGTTGCCAGTCT | ||||
| COL3A1 | Mouse | Forward | TGATGTCAAGTCTGGAGTGG | NM_009930.2 | 223 |
| Reverse | TCCTGACTCTCCATCCTTTC | ||||
| MMP-1 | Pig | Forward | GTTCCACAAATGAGTGCTGA | EU722905.1 | 212 |
| Reverse | ATAATAACGACGGCTCATCC | ||||
| MMP-13 | Mouse | Forward | GTGACTGGCAAACTTGATGA | BC125320.1 | 211 |
| Reverse | TCACATCAGACCAGACCTTG | ||||
| TIMP-1 | Mouse | Forward | CCCAGAAATCAACGAGA | BC034260.1 | 154 |
| Reverse | TGGGACTTGTGGGCATA | ||||
| TIMP-3 | Mouse | Forward | ACACGGAAGCCTCTGAAA | BC014713.1 | 231 |
| Reverse | TGGAGGTCACAAAACAAGG | ||||
| Lysyl oxidase | Mouse | Forward | CTGCTTGATGCCAACACA | M65142.1 | 156 |
| Reverse | TGCCGCATAGGTGTCATA | ||||
| α-subunit P4H | Mouse | Forward | CCCAGTCAGGTCTGCTATTC | BC009654.1 | 204 |
| Reverse | GGAACAGTCTCTGGACAACC | ||||
| R18s | Pig | Forward | GAACGCCACTTGTCCCTCTA | AY265350.1 | 219 |
| Reverse | GACTCAACACGGGAAACCTC |
| Gene 1 | Optimized Annealing Temperature (°C) | Optimized Primer Concentration (nM) | Optimized Threshold Cycle (CT) | Coefficient of Determination (R2) | Amplification Efficiency (%) |
|---|---|---|---|---|---|
| COL1A1 | 60.9 | 50 | 310.00 | 0.903 | 96.4 |
| COL3A1 | 53.4 | 200 | 128.80 | 0.976 | 100 |
| MMP-1 | 60.9 | 100 | 335.42 | 0.963 | 99.8 |
| MMP-13 | 58.7 | 700 | 310.65 | 0.929 | 95.2 |
| TIMP-1 | 58.7 | 1100 | 189.70 | 0.924 | 99.9 |
| TIMP-3 | 60.9 | 300 | 125.02 | 0.994 | 99.9 |
| Lysyl oxidase | 58.7 | 200 | 195.72 | 0.952 | 100 |
| α-subunit P4H | 51.0 | 400 | 280.84 | 0.923 | 98.0 |
| R18s | 61.2 | 60 | 201.50 | 0.999 | 97.4 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akit, H.; Collins, C.; Fahri, F.; Hung, A.; D’Souza, D.; Leury, B.; Dunshea, F. Dietary Lecithin Decreases Skeletal Muscle COL1A1 and COL3A1 Gene Expression in Finisher Gilts. Animals 2016, 6, 38. https://doi.org/10.3390/ani6060038
Akit H, Collins C, Fahri F, Hung A, D’Souza D, Leury B, Dunshea F. Dietary Lecithin Decreases Skeletal Muscle COL1A1 and COL3A1 Gene Expression in Finisher Gilts. Animals. 2016; 6(6):38. https://doi.org/10.3390/ani6060038
Chicago/Turabian StyleAkit, Henny, Cherie Collins, Fahri Fahri, Alex Hung, Daryl D’Souza, Brian Leury, and Frank Dunshea. 2016. "Dietary Lecithin Decreases Skeletal Muscle COL1A1 and COL3A1 Gene Expression in Finisher Gilts" Animals 6, no. 6: 38. https://doi.org/10.3390/ani6060038








