Modelling Niche Differentiation of Co-Existing, Elusive and Morphologically Similar Species: A Case Study of Four Macaque Species in Nakai-Nam Theun National Protected Area, Laos
Simple Summary
Abstract
1. Introduction
2. Methods
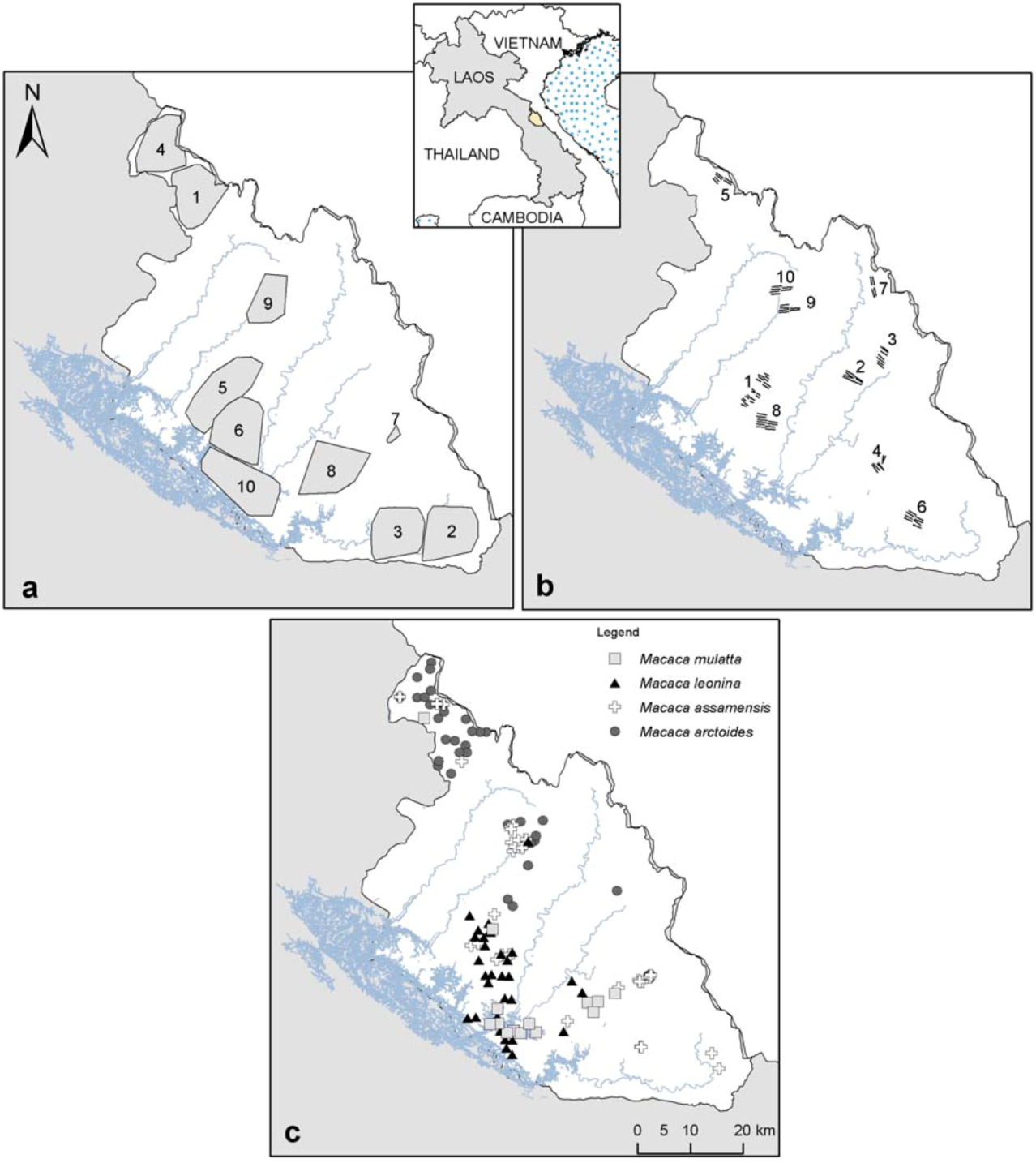
2.1. Study Area
2.2. Biological and Environmental Data
| Camera-trap surveys | Transect surveys | ||||||
|---|---|---|---|---|---|---|---|
| Site # on Figure 1 | Survey period | Total cameras a | Camera-trap-day | Site # on Figure 1 | Survey period | Nb. Transects (rep.) | km walked |
| 1 | Mar–May 06 | 49 | 2,181 | 1 | 29 Jan–2 Feb 11 | 20 (×1) | 21 |
| 2 | Oct–Nov 06 | 49 | 1,638 | 2 | 17 Feb–6 Mar 11 | 6 (×3) 1 (×1) | 36 |
| 3 | Dec 06–Feb 07 | 49 | 1,705 | 3 | 13 Mar–31 Mar 11 | 7 (×3) 1 (×2) | 40 |
| 4 | Mar–May 07 | 48 | 2,134 | 4 | 18 Jul–3 Aug 11 | 6 (×1) | 11 |
| 5 | Nov 07–Jan 08 | 50 | 2,308 | 5 | 16 Sept–28 Sept 11 | 6 (×1) | 10 |
| 6 | Jan–Mar 08 | 47 | 1,846 | 6 | 19 Oct–4 Nov 11 | 8 (×3) | 42 |
| 7 | Apr–Aug 08 | 32 | 1,687 | 7 | 11 Jan–23 Jan 12 | 4 (×3) | 22 |
| 8 | Nov 08–Jan 09 | 22 | 1,174 | 8 | 10 Feb–27 Feb 12 | 10 (×3) | 58 |
| 9 | Mar–May 09 | 3 | 183 | 9 | 12 Mar–24 Mar 12 | 6 (×3) | 34 |
| 10 | Nov–Dec 09 | 40 | 1,636 | 10 | 25 Mar–5 Apr 12 | 6 (×3) | 35 |
| 9 | Mar–May 10 | 45 | 2,405 | ||||
| 1 | Dec 10–Jan 11 | 33 | 1,319 | ||||
| TOTAL | Mar 06–Jan 11 | 467 | 20,216 | TOTAL | Jan 11–Apr 12 | 176 | 310 |
| Species | Occurrence from camera-trap | Occurrence from confirmed sighting | Total |
|---|---|---|---|
| M. mulatta | 14 | 0 | 14 |
| M. leonina | 31 | 3 | 34 |
| M. assamensis | 22 | 16 | 38 |
| M. arctoides | 45 | 3 | 48 |
2.3. Species Distribution Modelling
2.4. Model Building
2.5. Model Evaluation
2.6. Output Analysis
3. Results
3.1. Model Validation
| Species | Species-model’s test AUC | Null-model’s 95% C.I. training AUC | pValue Boyce Index (Spearman’s rho) | pValue Jackknife test (D statistic) |
|---|---|---|---|---|
| M. arctoides | 0.902 * | 0.783 | 0.000 ** | – |
| M. assamensis | 0.803 | 0.811 | 0.001 ** | – |
| M. leonina | 0.895 * | 0.820 | 0.000 ** | – |
| M. mulatta | 0.779 | 0.817 | 0.039 * | 0.001 ** |
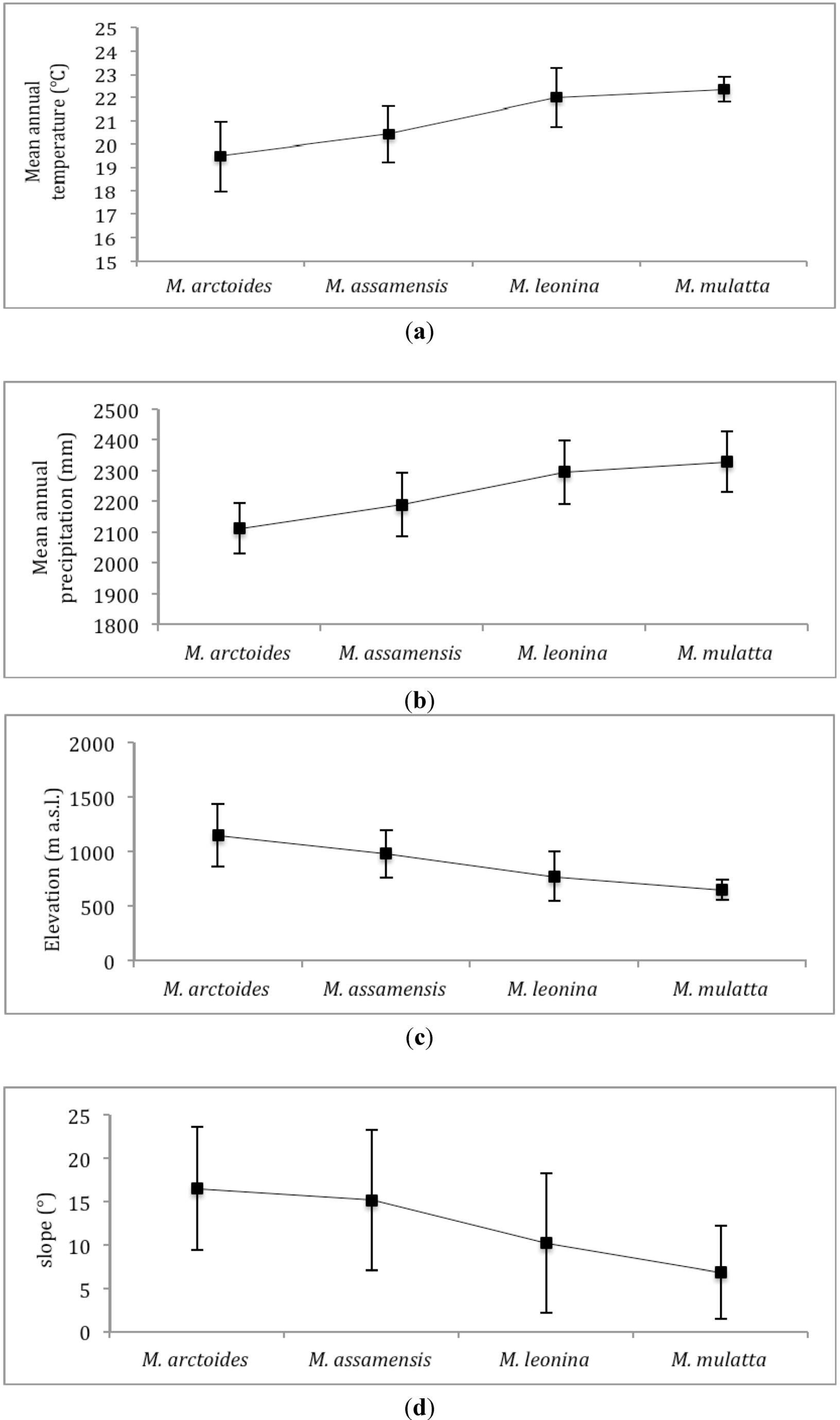
3.2. Species Distribution Models Outputs
| Species | M. arctoides | M. assamensis | M. leonina | M. mulatta |
| M. arctoides | − | 7 | 1 | 0 |
| M. assamensis | − | 0 | 0 | |
| M. leonina | − | 3 | ||
| M. mulatta | − |

| Species | M. arctoides | M. assamensis | M. leonina | M. mulatta |
| M. arctoides | − | 0.77 | 0.12 | 0.12 |
| M. assamensis | − | 0.51 | 0.35 | |
| M. leonina | − | 0.78 | ||
| M. mulatta | − | |||
| average | 0.33 | 0.54 | 0.45 | 0.42 |
| Species | test | M. artoides | M. assamensis | M. leonina | M. mulatta |
| M. artoides | D | − | 0.55 | 0.20 | 0.22 |
| I | − | 0.83 | 0.41 | 0.47 | |
| M. assamensis | D | − | 0.39 | 0.40 | |
| I | − | 0.66 | 0.70 | ||
| M. leonina | D | − | 0.52 | ||
| I | − | 0.79 | |||
| M. mulatta | D | − | |||
| I | − |
4. Discussion
4.1. Macaques’ Predicted Distribution and Niches
4.2. Model Performance
4.3. Implications
5. Conclusion
Conflict of Interest
Acknowledgements
References
- Bojórquez-Tapia, L.A.; Azuara, I.; Ezcurra, E.; Flores-Villela, O. Identifying conservation priorities in Mexico through geographic information systems and modeling. Ecol. Appl. 1995, 5, 215–231. [Google Scholar] [CrossRef]
- Raxworthy, C.J.; Ingram, C.M.; Rabibisoa, N.; Pearson, R.G. Applications of ecological niche modeling for species delimitation: A review and empirical evaluation using day geckos (Phelsuma) from Madagascar. Syst. Biol. 2007, 56, 907–923. [Google Scholar] [CrossRef]
- Buermann, W.; Saatchi, S.; Smith, T.B.; Zutta, B.R.; Chaves, J.A.; Milá, B.; Graham, C.H. Predicting species distributions across the Amazonian and Andean regions using remote sensing data. J. Biogeogr. 2008, 35, 1160–1176. [Google Scholar] [CrossRef]
- Parolo, G.; Rossi, G.; Ferrarini, A. Toward improved species niche modelling: Arnica montana in the Alps as a case study. J. Appl. Ecol. 2008, 45, 1410–1418. [Google Scholar] [CrossRef]
- Martínez-Freiría, F.; Sillero, N.; Lizana, M.; Brito, J.C. GIS-based niche models identify environmental correlates sustaining a contact zone between three species of European vipers. Divers. Distrib. 2008, 14, 452–461. [Google Scholar] [CrossRef]
- Pineda, E.; Lobo, J.M. Assessing the accuracy of species distribution models to predict amphibian species richness patterns. J. Appl. Ecol. 2009, 78, 182–190. [Google Scholar] [CrossRef]
- Timmins, R.J.; Evans, T.D.; Khounboline, K.; Sisomphone, C. Status and conservation of the giant muntjac Megamuntiacus vuquangensis, and notes on other muntjac species in Laos. Oryx 1998, 32, 59–67. [Google Scholar]
- Duckworth, J.W.; Stones, T.; Tizard, R.; Watson, S.; Wolstencroft, J. Does the fishing cat inhabit Laos? Cat News 2010, 52, 4–7. [Google Scholar]
- Timmins, R.J.; Duckworth, J.W. Distribution and habitat of Assamese Macaque Macaca assamensis in Lao PDR, including its use of low altitude karsts. Primate Conserv. 2011, 26, 1–12. [Google Scholar]
- Duckworth, J.W.; Poole, C.M.; Tizard, R.J.; Walston, J.L.; Timmins, R.J. The jungle cat Felis chaus in Indochina: A threatened population of a widespread and adaptable species. Biodivers. Conserv. 2005, 14, 1263–1280. [Google Scholar] [CrossRef]
- Tobler, M.W.; Carrillo-Percastegui, S.E.; Leite Pitman, R.; Mares, R.; Powell, G. An evaluation of camera traps for inventorying large- and medium-sized terrestrial rainforest mammals. Anim. Conserv. 2008, 11, 169–178. [Google Scholar] [CrossRef]
- Moruzzi, T.L.; Fuller, T.K.; DeGraaf, R.M.; Brooks, R.T.; Li, W. Assessing remotely triggered cameras for surveying carnivore distribution. Wildl. Soc. Bull. 2002, 30, 380–386. [Google Scholar]
- Di Bitetti, M.S.; De Angelo, C.D.; Di Blanco, Y.E.; Paviolo, A. Niche partitioning and species coexistence in a Neotropical felid assemblage. Acta Oecol. 2010, 36, 403–412. [Google Scholar] [CrossRef]
- Pereira, P.; da Silva, A.A.; Alves, J.; Matos, M.; Fonseca, C. Coexistence of carnivores in a heterogeneous landscape: Habitat selection and ecological niches. Ecol. Res. 2012, 27, 745–753. [Google Scholar] [CrossRef]
- Jenks, K.E.; Kitamura, S.; Lynam, A.J.; Ngoprasert, D.; Chutipong, W.; Steinmetz, R.; Sukmasuang, R.; Grassman, L.I.; Cutter, P.; Tantipisanuh, N.; Bhumpakphan, N.; Gale, G.A.; Reed, D.H.; Leimgruber, P.; Songsasen, N. Mapping the distribution of dholes, Cuon alpinus (Canidae, Carnivora), in Thailand. Mammalia 2012, 76, 1–10. [Google Scholar] [CrossRef]
- Timmins, R.J.; Evans, T.D. Wildlife and Habitat Survey of the Nakai-Nam Theun National Biodiversity Conservation Area; Wildlife Conservation Society: Vientiane, Lao PDR, 1996. [Google Scholar]
- Fooden, J. Ecogeographic segregation of macaque species. Primates 1982, 23, 574–579. [Google Scholar] [CrossRef]
- Robichaud, W.; Sinclair, A.; Odarkor-Lanquaye, N.; Klinkenberg, B. Stable forest cover under increasing populations of swidden cultivators in central Laos: The roles of intrinsic culture and extrinsic wildlife trade. Ecol. Soc. 2009, 14, 33–60. [Google Scholar]
- Hijmans, R.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G.; Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 2005, 25, 1965–1978. [Google Scholar] [CrossRef]
- Johnson, A.; Johnston, J. Biodiversity Monitoring and enforcement project in the Nam Theun 2 watershed (including the Nakai-Nam Theun NPA and corridors linking to the Phou Hin Poun and Hin Nam Nor PA); Wildlife Conservation Society: Vientiane, Lao PDR, 2007. [Google Scholar]
- NT2-WMPA (Nam Theun 2-Watershed Management Protection Authority), Camera-Trap Database 2006–2011. NT2-WMPA, Ban Oudomsouk: Nakai District, Lao PDR, 2011; Unpublished work.
- Coudrat, C.N.Z. Wildlife Surveys in Nakai-Nam Theun National Protected Area 2011-2012 (Final Report). NT2-WMPA, Ban Oudomsouk: Nakai District, Lao PDR, 2012; Unpublished work. [Google Scholar]
- Thierry, B. The macaques. In Primates in Perspective, 2nd ed.; Campbell, C., Fuentes, A., MacKinnon, K., Bearder, S., Stumpf, R., Eds.; Oxford University Press: New York, NY, USA, 2010; pp. 229–241. [Google Scholar]
- Phillips, S.J.; Dudík, M.; Schapire, R.E. A maximum entropy approach to species distribution modeling. In Proceedings of the Twenty-First International Conference on Machine Learning, Banff, AB, Canada, 4–8 July 2004; pp. 655–662.
- Phillips, S.J.; Anderson, R.P.; Schapire, R.E. Maximum entropy modeling of species geographic distributions. Ecol. Model. 2006, 190, 231–259. [Google Scholar] [CrossRef]
- Elith, J.; Ferrier, S.; Guisan, A.; Graham, C.H.; Anderson, R.P.; Dudı, M.; Hijmans, R.J.; Huettmann, F.; Leathwick, J.R.; Lehmann, A.; Li, J.; Lohmann, L. G.; Loiselle, B.A.; Manion, G.; Moritz, C.; Nakamura, M.; Nakazawa, Y.; Overton, J.M.; Peterson, A.T.; Phillips, S.J.; Richardson, K.; Scachetti-pereira, R.; Schapire, R.E.; Williams, S.; Wisz, M.S.; Zimmermann, N.E. Novel methods improve prediction of species distributions from occurrence data. Ecography 2006, 29, 129–151. [Google Scholar] [CrossRef]
- Kohavi, R. A study of cross-validation and bootstrap for accuracy estimation and model selection. In Proceedings of the Fourteenth International Joint Conference on Artificial Intelligence, Montréal, QC, Canada, 20–25 August 1995; pp. 1137–1143.
- Phillips, S.J.; Dudik, M. Modeling of species distributions with Maxent: New extensions and a comprehensive evaluation. Ecogeography 2008, 31, 161–175. [Google Scholar] [CrossRef]
- Raes, N.; Steege, H. A null-model for significance testing of presence-only species distribution models. Ecography 2007, 30, 727–736. [Google Scholar] [CrossRef]
- Boyce, M.S.; Vernier, P.R.; Nielsen, S.E.; Schmiegelow, F.K. Evaluating resource selection functions. Ecol. Model. 2002, 157, 281–300. [Google Scholar] [CrossRef]
- Pearson, R.G.; Raxworthy, C.J.; Nakamura, M.; Townsend Peterson, A. Predicting species distributions from small numbers of occurrence records: A test case using cryptic geckos in Madagascar. J. Biogeogr. 2007, 34, 102–117. [Google Scholar]
- Yost, A.C., Petersen; Miller, R. Predictive modeling and mapping sage grouse (Centrocercus urophasianus) nesting habitat using Maximum Entropy and a long-term dataset from Southern Oregon. Ecol. Inform. 2008, 3, 375–386. [Google Scholar]
- Hirzel, A.H.; Le Lay, G.; Helfer, V.; Randin, C.; Guisan, A. Evaluating the ability of habitat suitability models to predict species presences. Ecol. Model. 2006, 199, 142–152. [Google Scholar] [CrossRef]
- Hernandez, P.A.; Graham, C.H.; Master, L.L.; Albert, D.L. The effect of sample size and species characteristics on performance of different species distribution modeling methods. Ecogeography 2006, 29, 773–785. [Google Scholar] [CrossRef]
- Schoener, T.W. Anolis lizards of Bimini: Resource partitioning in a complex fauna. Ecology 1968, 49, 704–726. [Google Scholar] [CrossRef]
- Warren, D.L.; Glor, R.E.; Turelli, M. Environmental niche equivalency versus conservatism: Quantitative approaches to niche evolution. Evolution 2008, 62, 2868–2883. [Google Scholar] [CrossRef]
- Duckworth, J.; Salter, R.E.; Khounboline, K. Wildlife in Lao PDR 1999 Status Report; IUCN–The World Conservation Union, Wildlife Conservation Society, Centre for Protected Areas and Watershed Management: Vientiane, Lao PDR, 1999. [Google Scholar]
- Abegg, C.; Thierry, B. Macaque evolution and dispersal in insular south-east Asia. Biol. J. Linn. Soc. 2002, 75, 555–576. [Google Scholar] [CrossRef]
- Eudey, A.A. Pleistocene glacial phenomena and the evolution of Asian macaques. In The Macaques: Studies in Ecology, Behavior and Evolution; Lindburg, D.G., Ed.; Van Nostrand Reinhold: New York, NY, USA, 1980; pp. 52–83. [Google Scholar]
- Eudey, A.A. Macaques habitat preference in west-central Thailand and quaternary glacial events. In Primatology Today; Ehara, A., Kimura, T., Takenaka, O., Iwamoto, M., Eds.; Elsevier Science Publishers: Amsterdam, The Netherlands, 1991; pp. 21–24. [Google Scholar]
- Rodman, P.S. Structural Differentiation of microhabitats of sympatric Macaca fascicularis and M. nemestrina in East Kalimantan, Indonesia. Int. J. Primatol. 1991, 12, 357–375. [Google Scholar]
- Borries, C.; Larney, K.; Kreetiyutanony, K.; Koenig, A. The diurnal primate community in a dry evergreen forest in Phu Khieo Wildlife Sanctuary, Northeast Thailand. Nat. Hist. Bull. Siam Soc. 2002, 50, 75–88. [Google Scholar]
- IUCN Red List v.2011. Available online: http://www.iucnredlist.org (accessed on 16 August 2012).
- Wintle, B.A.; Elith, J.; Potts, J.M. Fauna habitat modelling and mapping: A review and case study in the Lower Hunter Central Coast region of NSW. Austral. Ecol. 2005, 30, 719–738. [Google Scholar] [CrossRef]
- Phillips, S.J.; Dudík, M.; Elith, J.; Graham, C.H.; Lehmann, A.; Leathwick, J.; Ferrier, S. Sample selection bias and presence-only distribution models: Implications for background and pseudo-absence data. Ecol. Appl. 2009, 19, 181–197. [Google Scholar] [CrossRef]
- Blanco, G.; Sergio, F.; Sanchéz-Zapata, J.A.; Pérez-García, J.M.; Botella, F.; Martínez, F.; Zuberogoitia, I.; Frías, O.; Roviralta, F.; Martínez, J.E.; Hiraldo, F. Safety in numbers? Supplanting data quality with fanciful models in wildlife monitoring and conservation. Biodivers. Conserv. 2012, 21, 3269–3276. [Google Scholar] [CrossRef]
- Thorn, J.S.; Nijman, V.; Smith, D.; Nekaris, K.A.I. Ecological niche modelling as a technique for assessing threats and setting conservation priorities for Asian slow lorises (Primates: Nycticebus). Divers. Distrib. 2009, 15, 289–298. [Google Scholar] [CrossRef]
- Chunco, A.J.; Jobe, T.; Pfennig, K.S. Why do species co-occur? A test of alternative hypotheses describing abiotic differences in sympatry versus allopatry using spadefoot toads. PLoS ONE 2012, 7, e32748. [Google Scholar]
- Wisz, M.S.; Hijmans, R.; Li, J.; Peterson, A.T.; Graham, C.H.; Guisan, A.; The Nceas Predicting Species Distributions Working Group. Effects of sample size on the performance of species distribution models. Divers. Distrib. 2008, 14, 763–773. [Google Scholar] [CrossRef]
- Graham, C.H.; Elith, J.; Hijmans, R.J.; Guisan, A.; Townsend-Peterson, A.; Loiselle, B.A.; The Nceas Predicting Species Distributions Working Group. The influence of spatial errors in species occurrence data used in distribution models. J. Appl. Ecol. 2008, 45, 239–247. [Google Scholar]
- Warren, D.; Steifert, S. Ecological niche modeling in Maxent: The importance of model complexity and the performance of model selection criteria. Ecol. Appl. 2011, 21, 335–342. [Google Scholar] [CrossRef]
- McPherson, J.M.; Jetz, W. Effects of species? Ecology on the accuracy of distribution models. Ecography 2007, 30, 135–151. [Google Scholar]
- Hardcastle, J.; Cox, S.; Dao, N.T.; Johns, A.G. Rediscovering the Saola. In Proceedings of the Rediscovering the Saola—A Status Review and Conservation Planning Workshop, Hanoi, Vietnam, 27–28 February 2004.
- Surridge, A.K.; Timmins, R.J.; Hewitt, G.M.; Bell, D.J. Striped rabbits in Southeast Asia. Nature 1999, 400, 726. [Google Scholar] [CrossRef]
- McCarthy, J.L.; Fuller, T.K.; McCarthy, K.P.; Wibisono, H.T.; Livolsi, M.C. Using camera trap photos and direct sightings to identify possible refugia for the Vulnerable Sumatran striped rabbit Nesolagus netscheri. Oryx 2012, 46, 438–441. [Google Scholar] [CrossRef]
- Johnson, A.; Vongkhamheng, C.; Hedemark, M.; Saithongdam, T. Effects of human carnivore conflict on tiger (Panthera tigris) and prey populations in Lao PDR. Anim. Conserv. 2006, 9, 421–430. [Google Scholar] [CrossRef]
- Mohamed, A.; Samejima, H.; Wilting, A. Records of five Bornean cat species from Deramakot Forest Reserve in Sabah, Malaysia. Cat News 2009, 51, 3–6. [Google Scholar]
- Wilting, A.; Mohamed, A.; Ambu, L.N.; Lagan, P.; Mannan, S.; Hofer, H.; Sollmann, R. Density of the Vulnerable Sunda clouded leopard Neofelis diardi in two commercial forest reserves in Sabah, Malaysian Borneo. Oryx 2012, 46, 423–426. [Google Scholar] [CrossRef]
- Sodhi, N.S.; Koh, L.P.; Brook, B.W.; Ng, P.K.L. Southeast Asian biodiversity: An impending disaster. Trends Ecol. Evol. 2004, 19, 654–660. [Google Scholar] [CrossRef]
- Duckworth, J.W.; Batters, G.; Belant, J.L.; Bennett, E.L.; Brunner, J.; Burton, J.; Challender, D.W.S.; Cowling, V.; Duplaix, N.; Harris, J.D.; Hedges, S.; Long, B.; Mahood, S.P.; McGowan, P.J.K.; McShea, W.J.; Oliver, W.L.R.; Perkin, S.; Rawson, B.M.; Shepherd, C.R.; Stuart, S.N.; Talukdar, B.K.; van Dijk, P.P.; Vié, J.-C.; Walston, J.L.; Whitten, T.; Wirth, R. Why South-East Asia should be the world’s priority for averting imminent species extinctions, and a call to join a developing cross-institutional programme to tackle this urgent issue. SAPIENS 2012, 5, 76–95. [Google Scholar]
- Catullo, G.; Masi, M.; Falcucci, A.; Maiorano, L.; Rondinini, C.; Boitani, L. A gap analysis of Southeast Asian mammals based on habitat suitability models. Biodivers. Conserv. 2008, 141, 2730–2744. [Google Scholar]
- Yen, P.; Ziegler, S.; Huettmann, F.; Onyeahialam, A.I. Change detection of forest and habitat resources from 1973 to 2001 in Bach Ma National Park, Vietnam, using remote sensing imagery. Int. For. Rev. 2005, 7, 1–8. [Google Scholar]
- Kelly, M.J. Design, evaluate, refine: Camera trap studies for elusive species. Anim. Conserv. 2008, 11, 182–184. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Coudrat, C.N.Z.; Nekaris, K.A.-I. Modelling Niche Differentiation of Co-Existing, Elusive and Morphologically Similar Species: A Case Study of Four Macaque Species in Nakai-Nam Theun National Protected Area, Laos. Animals 2013, 3, 45-62. https://doi.org/10.3390/ani3010045
Coudrat CNZ, Nekaris KA-I. Modelling Niche Differentiation of Co-Existing, Elusive and Morphologically Similar Species: A Case Study of Four Macaque Species in Nakai-Nam Theun National Protected Area, Laos. Animals. 2013; 3(1):45-62. https://doi.org/10.3390/ani3010045
Chicago/Turabian StyleCoudrat, Camille N. Z., and K. Anne-Isola Nekaris. 2013. "Modelling Niche Differentiation of Co-Existing, Elusive and Morphologically Similar Species: A Case Study of Four Macaque Species in Nakai-Nam Theun National Protected Area, Laos" Animals 3, no. 1: 45-62. https://doi.org/10.3390/ani3010045
APA StyleCoudrat, C. N. Z., & Nekaris, K. A.-I. (2013). Modelling Niche Differentiation of Co-Existing, Elusive and Morphologically Similar Species: A Case Study of Four Macaque Species in Nakai-Nam Theun National Protected Area, Laos. Animals, 3(1), 45-62. https://doi.org/10.3390/ani3010045




